
Preface: Embracing a New Era of “Super Minimally Invasive Surgery”
The ultimate challenge in medicine lies in eradicating diseases while preserving the perfect masterpiece sculpted by millions of years of human evolution. This sacred balance is being redefined by Super Minimally Invasive Surgery (SMIS). As you open Endoscopic Therapeutics: Super Minimally Invasive Surgery, meticulously compiled by Professor Enqiang Linghu, you are presented with the core tenets and practical blueprint of this quiet revolution.
The publication of this book is no accident. It emerges at the forefront of a profound evolution in medical philosophy—shifting from a trauma-centric paradigm of “resection and reconstruction” to a curative philosophy of “preserving organ structure and function.” Through profound insight and exceptional clinical practice, Professor Linghu has systematized this philosophy into a rigorous theoretical framework and actionable technical pathways. The book’s structure is both concise and profound:
Foundational Principles (chapters 1–7): The opening chapters establish SMIS as a paradigm shift of epochal significance. Tracing its origins, the text clearly outlines the innovative trajectory of SMIS in the field of digestive endoscopy, elaborating on its four access pathways and the hundreds of sophisticated techniques derived from them. Notably, it establishes a rigorous nomenclature system, instrumentation standards and operational guidelines for this emerging field. Its core philosophy places “disease eradication” and “organ preservation” on equal footing, with particular emphasis on safeguarding the complex interconnected systems endowed by human evolution—this constitutes the essential leap that distinguishes SMIS from conventional organ-resectional surgical models.
Clinical Applications (chapters 8–14): Here, theory translates into therapeutic power. The chapters provide detailed analyses of SMIS solutions for over 30 diseases involving critical organs such as the esophagus, stomach, duodenum, and pancreas. From organ-preserving resection of early-stage cancers to precise interventions for obesity and minimally invasive management of complex pancreatic cysts, SMIS demonstrates unprecedented clinical breadth and depth. The book highlights cutting-edge technologies such as organ-preserving tumor resection, functional repair for motility disorders, and precise energy therapies like radiofrequency ablation. Each technique is illustrated with detailed surgical images and dynamic videos, offering surgeons a precise navigational guide.
However, the greatness of SMIS extends beyond technical refinement. It reshapes medical ethics and practice guidelines centered on patient welfare: striving for fewer complications, transparent doctor-patient communication and informed consent, and stringent infection control standards. The book itself embodies this philosophy—its content is organized in strict adherence to surgical protocols and case documentation rules, serving both as a forward-looking technical manifesto and an immediately applicable clinical manual.
It is crucial to recognize that SMIS remains a dynamic and rapidly evolving discipline. As Professor Linghu explicitly states in the book, the SMIS framework “is still in its preliminary formation” and its refinement urgently requires the collaborative efforts and sustained exploration of global colleagues. It calls on medical elites to redefine “success” as uncompromising cure—eradicating disease while devoutly preserving the body’s inherent evolutionary wisdom and natural architecture. Every successful SMIS procedure is a vivid interpretation of the supreme therapeutic ideal of “restoration to original state” and a profound tribute to the human body as a “masterpiece of evolution.”
At this critical juncture as super-minimally invasive medicine enters a new era, the publication of Endoscopic Therapeutics: Super Minimally Invasive Surgery is a milestone. It is not merely a compendium of technical procedures but a sublimation of therapeutic philosophy. In my capacity as president of the World Endoscopy Organization, I wholeheartedly recommend this book to all pioneers dedicated to shaping the next medical era. It will undoubtedly become an essential classic illuminating the path ahead, guiding us to jointly write a new chapter of preservation and cure.


Lars Aabakken
President of the World Endoscopy Organization
June 2025
Preface: Pioneering the SMIS Philosophy
In the cosmic expanse of medical exploration, every conceptual revolution shines as a luminous new star, illuminating uncharted courses for human health. In 2016, Professor Linghu Enqiang, driven by profound reflection on the limitations of traditional surgical paradigms, pioneered the Super Minimally Invasive Surgery (SMIS) philosophy. Little did the world anticipate that this vision would catalyze a transformative wave across global medicine, ultimately crystallizing into this seminal work—Super Minimally Invasive Surgery – Gastrointestinal Endoscopy Section—a testament to collective wisdom and rigorous practice.
The journey of SMIS—from conceptual germination to theoretical maturity—epitomizes groundbreaking innovation. Conventional surgical paradigms of “partial or total organ resection followed by anatomical reconstruction” have saved countless lives, yet often at the cost of organ loss and functional compromise. The genesis of SMIS directly confronts this dilemma: it advocates for disease eradication while preserving anatomical integrity and physiological function. Rooted in reverence for the human body’s innate physiology, SMIS embodies an unwavering pursuit of the ideal: functional restoration to pristine status. Today, this philosophy has matured into a robust therapeutic framework. Originating in digestive endoscopy, it now extends across multidisciplinary clinical practice, demonstrating profound efficacy in managing complex pathologies.
This volume, forged by Professor Linghu’s team and global proponents of SMIS, meticulously documents the evolution and clinical achievements of SMIS within digestive endoscopy. Its rigorous structure systematically elucidates the academic significance and clinical applicability of SMIS. Theoretical Foundation: Traces the conceptual origins, dissects four innovative technical pathways and hundreds of derivative procedures, and establishes standardized nomenclature protocols, instrumentation criteria, and operational guidelines—fortifying the discipline’s intellectual bedrock. Practical Mastery: Serves as a definitive clinical manual, detailing SMIS solutions for over 30 diseases spanning the esophagus, stomach, intestines, and colorectum. From early-stage tumor resection to complex pathology management, it provides exhaustive technical guidance, case analyses, and high-definition procedural visuals with dynamic video support—ensuring tangible, reproducible skill acquisition. These elements distill clinical experience into standardized, replicable methodologies for the field.
SMIS transcends technical innovation. When surgery prioritizes functional preservation and therapies are designed around quality of life, physician-patient communication evolves beyond curative expectations into a covenant safeguarding human dignity. This advancement elevates medical ethics: in balancing radical cure against quality-of-life preservation, and in choosing between lesion eradication and functional retention, each clinical decision becomes an ethical act—transforming abstract principles into tangible enhancements in patients’ lived experiences.
As a witness to SMIS’s evolution, I attest that this field remains dynamically evolving, brimming with unexplored potential. May this book, Super Minimally Invasive Surgery, bridge global collaborations, inviting broader engagement to refine SMIS theory and practice. Let us embark with technological innovation as our vessel and patient welfare as our sail, navigating the vast currents of human health to inscribe yet more brilliant chapters in medical history.


Jean-Francois Rey
Past President of the World Endoscopy Organization
June 2025
Super Minimally Invasive Surgery: An Inevitable Choice for Humanity
Millions of years of human evolution have shaped our intricate and complete physical structure. From skeletal support to muscular coordination, from neural transmission to the specialized functions of internal organs, the integrity of the human body is not merely a simple physical combination but a highly unified and coordinated living system. This integrity not only serves as the foundation of physiological functions but also embodies the lived experience and dignity of an individual as a “complete person.” However, when disease strikes, especially those requiring surgical intervention, how can we eradicate the affliction while preserving this inherent “integrity” to the greatest extent possible? This has become the ultimate question medicine must answer.
I. The Necessity of Treatment and the Limitations of the Model
It must be acknowledged that for many solid tumors, severe trauma, or structural deformities, surgical resection remains the core curative approach. The traditional surgical model—“organ removal + anatomical reconstruction”—has saved countless lives over the past century. However, this model inherently contains a profound paradox: to cure disease, we inevitably destroy the body’s original, evolutionarily perfected structure.
The drawbacks are evident: subtotal gastrectomy can lead to diarrhea, gastroparesis, and dumping syndrome; low rectal surgery may necessitate the removal of the anus, permanently altering the mode of excretion; and patients undergoing total gastrectomy may even be unable to lie flat due to digestive fluid reflux, suffering severely impaired quality of life. These are not merely “side effects”; they constitute structural damage to the integrity of the life system. Their impact, akin to the “Butterfly Effect,” triggers a cascade of physiological and psychological chain reactions in the patient’s long-term postoperative life. While surgical intervention may be but a brief moment in an individual’s lifespan, the resulting alterations in bodily structure can linger for a lifetime.
II. Super Minimally Invasive Surgery: A Revolution in Philosophy
It is against this backdrop that “Super Minimally Invasive Surgery” (SMIS) has emerged. It is not merely a technical advancement towards being “less invasive,” but a fundamental philosophical revolution. Its core definition is: achieving radical cure of disease based on absolutely preserving the anatomical integrity and physiological function of the organ.
This philosophy aligns perfectly with the pursuit of maintaining human structural integrity. The goal of SMIS is to restore the patient’s post-treatment state “to the natural anatomical state of the human body evolved over millions of years,” truly realizing “curing the disease and restoring the original condition.” It signifies an upgrade in medicine’s goal from “resecting the lesion” to “repairing life,” shifting from “cure priority” to “balance between cure and functional preservation.”
Achieving this grand goal relies on four ingenious “pathways of life”:
1. Natural Orifice Pathway: Utilizing the body’s natural openings such as the mouth, nasal cavity, and anus to perform surgery, for example, Transoral Endoscopic Resection of Esophageal Tumors.
2. Tunnel Pathway: Creating a micro-tunnel within tissues to reach the lesion, for example, Submucosal Tunneling Endoscopic Resection of Gastric Wall Tumors.
3. Percutaneous Puncture Pathway: Delivering instruments precisely to deep-seated organs through extremely small puncture sites on the body surface, for example, Percutaneous Radiofrequency Ablation of Renal Tumors.
4. Multi-Space Combined Pathway: Skillfully combining multiple natural cavities or micro-incisions to perform complex procedures.
Each pathway has spawned dozens or even hundreds of surgical techniques and continues to expand. In the future, with advancements in robotics, AI navigation, and biomaterials, the feasibility of developing newer, more precise, and intelligent techniques via these four pathways is immense. The philosophy of SMIS has also expanded from the field of digestive endoscopy to urology, thoracic surgery, thyroid surgery, and other disciplines, demonstrating its universal vitality.
III. The Crucial Role in the Long River of Cancer Treatment
In the prolonged campaign against solid tumors, Super Minimally Invasive Surgery is becoming an indispensable core component, its role spanning different stages of the disease:
1. Early-stage Tumors: For early-stage cancers and precancerous lesions in areas such as the digestive tract and urinary tract, SMIS enables complete resection directly through endoscopy or micro-puncture, achieving radical cure while preserving organ integrity. This has become a first-line treatment option for conditions like early gastric cancer and colorectal cancer.
2. Locally Advanced Tumors: For locally advanced tumors, SMIS does not fight alone. Following successful tumor shrinkage and downstaging through neoadjuvant chemotherapy, radiotherapy, targeted therapy, or immunotherapy, SMIS can play the crucial role of “precise finishing.” It can resect the residual primary focus—which may harbor heterogeneity and drug resistance—after systemic treatment, avoiding excessive trauma and preserving vital organ function for the patient.
3. Future Trends: As cancer treatment enters the era of “chronic disease management,” radiotherapy, chemotherapy, and immunotargeted therapies are becoming increasingly precise and low-toxicity. In synergy with these advancements, the role of SMIS will become even more prominent—serving as a “precision scalpel” for local control. Within the comprehensive treatment framework, SMIS addresses localized issues with minimal trauma, maximizing patients’ quality of life.
IV. Returning to the Irreplaceability of Organs
We emphasize the preservation of bodily structures so profoundly because every organ in the human body is a unique masterpiece forged through the long river of evolution—each one irreplaceable. The liver, a comprehensive biochemical factory for nearly a thousand chemical reactions, is responsible for detoxification, synthesis, and metabolism. The heart, an unceasing life-pump, possesses an intricate electrophysiological and mechanical structure that cannot be replicated. The brain, the vessel of consciousness and thought, harbors neural networks whose complexity surpasses that of any supercomputer. Even the stomach, with its unique acidic environment, peristaltic rhythms, and hormonal secretions, serves as a critical link in systemic nutrient absorption and immune balance.
Removing an organ is never a simple “part replacement.” While breakthroughs have been made in xenotransplantation (such as pig liver transplants), it still falls far short of fully replicating the complex functions and long-term compatibility of human organs. Each organ is intricately interconnected with all other systems of the body, and a change in one part inevitably affects the whole. Surgical treatment is merely a brief moment of medical intervention in the long journey of life. Yet, the path chosen—whether it is destructive removal or preservative repair—determines the quality of life for the patient in the many years to come.
Conclusion
From viewing the body as a detachable “machine,” to recognizing it as an “organic whole” where a move in one part affects the whole, and further to respecting it as a “complete life experience” connected to the world, the progress of medicine has always been accompanied by a deepening understanding of bodily integrity. Super Minimally Invasive Surgery is the brilliant embodiment of this understanding in clinical practice.
It is not only a technological upgrade but also a return to the humanistic spirit of medicine. It responds to humanity’s most fundamental attachment to and guardianship of its own integrity. In moments when surgical intervention is unavoidable, choosing super minimally invasive surgery means choosing to eliminate illness while respecting—as much as possible—the original design of life and preserving the patient’s right and dignity to return to a normal life. Thus, on the path toward the future of medicine, super minimally invasive surgery is undoubtedly humanity’s inevitable and optimal choice.
Enqiang Linghu
Director, Department of Gastroenterology, First Medical Center, Chinese PLA General Hospital
President, 8th of the Chinese Society of Digestive Endoscopy
President, Digestive Physicians Branch, Chinese Medical Doctor Association
June 2025
Part One General Introduction to Super Minimally Invasive Surgery
Super minimally invasive theory is a brand-new surgical treatment concept proposed by Professor Linghu Enqiang in 2016. This concept aims to remedy the shortcomings of the traditional surgical mode of “partial or total excision of organs, anatomical reconstruction,” thus proposing the super minimally invasive surgical mode for “preserving the overall structure and function of organs and achieving the goal of radical cure of diseases.” Thanks to its unique theoretical advantages as well as its achievable and visible operation results and prognosis state, this concept, once proposed, has won the recognition of official organizations like the Chinese Medical Association, the World Endoscopy Organization.
Super minimally invasive theory has been developed to achieve a set of perfect treatment concepts so far, and its core objective lies in removing diseases by a super minimally invasive surgical mode on the basis of maintaining the anatomical integrity that evolved over millions of years. The main difference with traditional surgery lies in the fact it keeps intact the patient’s original organ while resecting the tissues with lesions and treating the disease. Therefore, its goal is not merely “curing disease,” but also reaching the further result of “recovering as before.” This conforms to the requirement of prolonging the human lifespan and of improving the quality of life, but also of making creative efforts on the surgical paradigm in the medical equipment and apparatus innovation by the medical community, also the revolution of the surgical concept in the future.
The emergence of super minimally invasive surgery (SMIS) is the inevitable result of the development of therapeutic gastrointestinal endoscopy. After it experienced the vascular cavity treatment stage to the digestive endoscopic tunnel technique stage, it transitioned smoothly to the super minimally invasive theory guiding stage. During the development of these three stages, the endoscopic treatment of diseases experienced the breakthrough from inside the digestive lumen to outside the digestive lumen, and gradually developed from treating internal-medicine diseases to surgical diseases, and finally tends to be consistent with the scope of surgical diagnosis and treatment. Meanwhile, to better put the super minimally invasive theory into practice, a multi-disciplinary deep integration is underway between the field of gastrointestinal endoscopy and the surgical field, in order to develop super minimally invasive surgery without limiting it to the field of endoscopy.
Gastrointestinal endoscopic super minimally invasive surgery (eSMIS) is a branch of SMIS operations. After multiple years of development and clinical practice, presently eSMIS has a perfect endoscopic technique, a nomenclature of procedures and implementation principles, as well as specific development perspectives. Over the recent 10 years, endoscopic scholars in China have been constantly innovating and developing a series of SMIS surgical methods according to four directions, namely, natural, tunnel, puncture, and multi-cavity channels. For example, in the SMIS operation of early gastric cancer, full-thickness resection of the lesion by natural channel, full-thickness resection by multiple-cavity + lymphadenectomy approaches, the technique of sealing large wounds after non-full-thickness resection, and the traction technique in non-full-thickness resection; in the treatment of large-area early esophageal cancer by tunnel channel, autologous skin graft was used to prevent esophageal stenosis after large esophageal defect, and for the diagnosis and treatment through the muscularis propria to the outside of the digestive lumen; some breakthroughs in SMIS operations of the bile pancreatic disease, including laser lithotripsy under direct choledochoscope with sphincter preserved, exploratory pancreatic duct with cholangiopancreatic vision system, super minimally invasive photodynamic therapy under pancreatoscope, radiofrequency ablation of bile duct under direct choledochoscope, direct choledochoscopy with a duodenal papillary support for the extraction of gallbladder silt stones and of biliary calculus, etc., have been achieved.
Super minimally invasive theory is a concept, and it guides the innovation and development path of future surgical therapeutics. As Hayek once said, “in the long run it is ideas, and therefore the men who give currency to new ideas that govern evolution.” In the future, there will be also more new endoscopic technologies and surgical evolutions and improvements that will make it possible to cure diseases while preserving the organ and its function, without altering the anatomical structure.
Chapter 1 The Birth and Significance of Super Minimally Invasive Theory
1.1 The Rapid Development of Gastrointestinal Surgery, the Emergence of the Super Minimally Invasive Theory
With the rapid development of gastrointestinal surgery, the super minimally invasive theory came forth. The constant development of medicine is due to the concept of continuous innovative treatment in order to prolong human lifespan and improve the quality of life, to the upgrading of medical equipment, and to the unceasing advances in surgical techniques. Looking back over its 100-year development history, gastrointestinal surgery has evolved from traditional surgery to laparoscopic surgery, and then to minimally invasive surgery, to finally usher in the period of “super minimally invasive surgery (SMIS),” focused on “returning to the human body itself.”
Traditional surgery (figure 1.1), also called operation, commonly known as cuts, refers to the therapeutic process whereby surgeons or other professionals enter the human body or other biological tissues using surgical equipment or surgical instruments to excise lesions, alter the composition or implanting foreign substances, etc., using techniques involving external force. When it first came out is hard to define exactly, but there have been some important development nodes: in the 17th century BC, eye surgery regulations, recorded in the Ninevitic Code of Hammurabi, constitute the earliest surgical operation recorded in human civilization. Superficial mass excision and drainage of abscess are known since the time of Hippocrates, in about 400 BC; anatomy as well as debridement and suturing began to emerge at the time of the Renaissance. In 1809, an American doctor performed the first cavity operation without antibiotics. In the 1840s, the subsequent emergence of anesthesia with diethyl ether and asepsis made the rapid development of surgical operation possible and allowed one to enter the organ resection time. At this time, operations were usually conducted by skin incision, and needed to excise the affected parts or all organs, which was followed by the reconstruction of the human anatomical structure. Such methods can cure diseases, but they change the normal structure of the human body and sacrifice part of the normal physiological function. In addition, traditional surgery also has drawbacks such as big wounds, slow recovery, high cost, and the relatively high cost of subsequent complications.
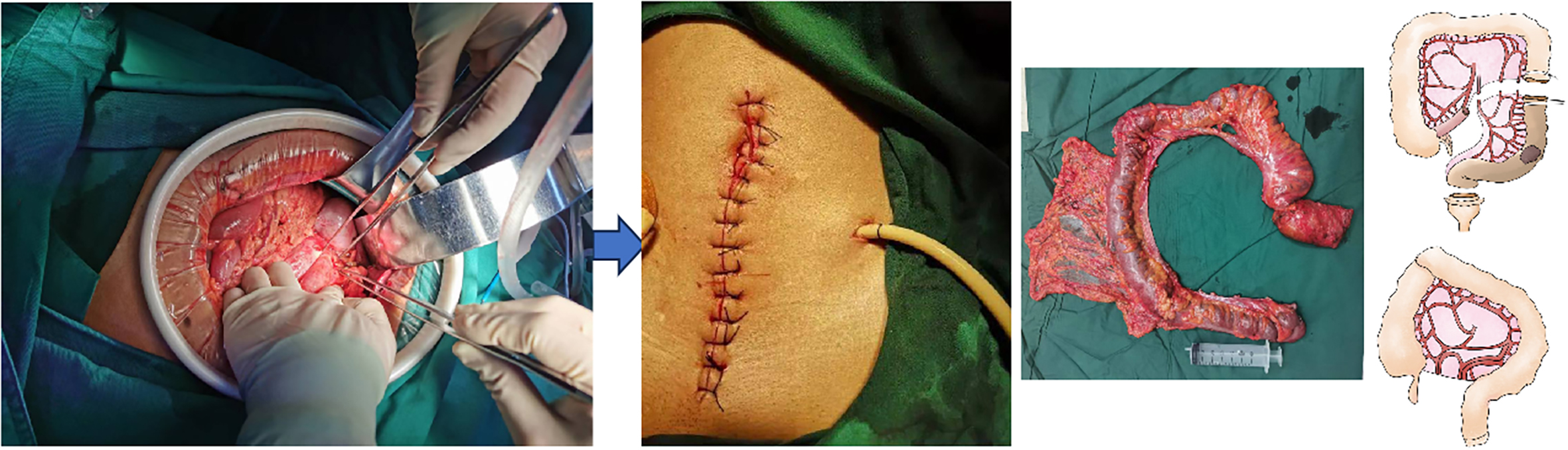

FIG. 1.1 — Traditional ‘organ resection + anatomical reconstruction’ surgical technique. Note: Traditional surgery involves skin approach, lesion and organ excision + anatomical reconstruction, with big trauma, slow recovery.
Based on the limitations of traditional surgery, endoscopic surgery began to develop (figure 1.2). Endoscopic surgery introduces surgical instruments, physical energy or chemical drugs into the body by a minimal incision or by the natural cavities of the human body, and is used to carry out surgical operations to inactivate, excise, repair the body’s internal lesions, abnormalities and trauma, or to reconstruct organs to reach the purpose of treatment. The history of the development of endoscopic surgery can be dated back to 1901: the Russian gynecologist Ott made small incisions in the front abdominal wall to insert a vaginal speculum into the abdomen, and penetrated the abdomen to carry out examinations with the aid of a reflected beam of light from the front, meanwhile the German surgeon Kelling introduced a cystoscope in the abdomen of a dog to make examinations, and called such operation the endoscopic laparoscope examination; all were animal experiments; at the same year, Jacobaeus in Stockholm, Sweden, used the word ‘laparoscopy’ for the first time, and created a pneumoperitoneum using a trocar, and named it ‘laparothorakoskopie;’ this is the first such experiment on a human body ever recorded. In 1933, Ferver reported having used biopsy devices and inustion to loosen intraabdominal adhesion by laparoscopy, which marks the beginning of the use of the endoscope in the treatment of diseases. In 1987, the French gynecologist Mouret successfully conducted the first laparoscopic cholecystectomy known to the general public, opening a new era in the history of endoscopic surgery therapy. In April 1989, at the annual meeting of the American Society for Gastrointestinal Endoscopy, Professor Dubois presented a video on laparoscopic cholecystectomy, which generated a great deal of interest, and launched a craze for laparoscopic cholecystectomy. However, endoscopic surgery essentially does not change traditional surgery techniques, still involving the excision of lesions while partial organs are sometimes removed, changing the integrity of the anatomical structure, which may affect the postoperative quality of life.
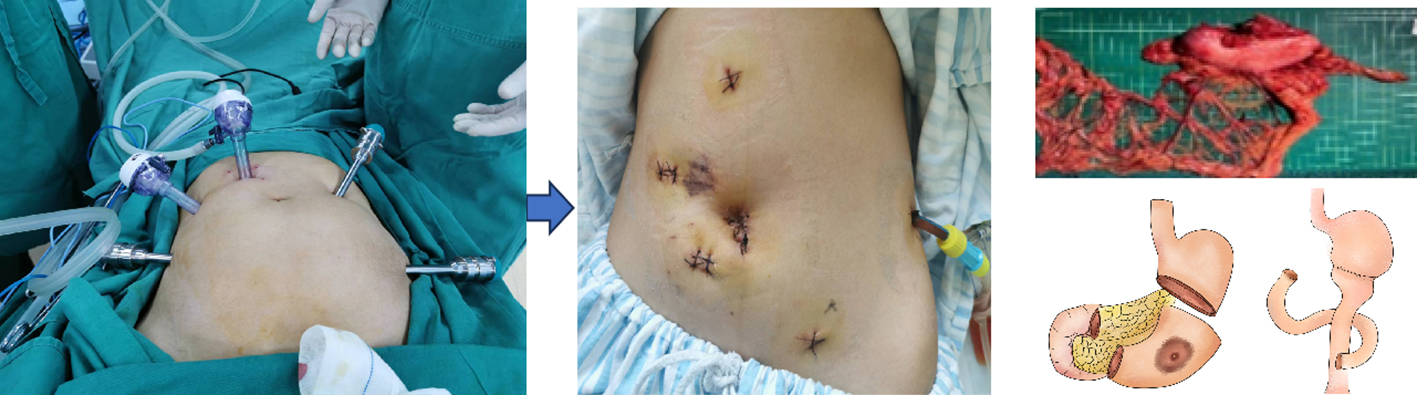

FIG. 1.2 — Endoscopic ‘organ resection + anatomical reconstruction’ surgical technique. Note: Endoscopic surgery can be performed on the skin, the nose, the gallbladder or the vaginal route; it is more minimally invasive, with faster recovery, but it does not change the ‘excision lesion + organ’ mode.
Minimally invasive surgery (MIS) is a medical science branch allowing us to conduct surgical operations inside the human body by using a micro-incision or route (figure 1.3). This surgical mode completes the treatments of the body’s internal lesions, deformities and trauma, etc., by special instruments, physical energy or chemical agents, including inactivation, excision, repair or reconstruction, aiming at achieving the treatment objective. The development history of minimally invasive surgery can be traced back to 1985, when the British urologist Payne and Wickham used for the first time the word of “minimally invasive procedure” in the treatment of urinary stones. After that, the French genecologist Mouret proposed the concept of “minimally invasive surgery” in 1987, initiating a new chapter in the history of minimally invasive treatment. After 1990, with the continuous refinement of the endoscopic surgery, minimally invasive surgery has been widely used in fields like general surgery, thoracic surgery, gynecology and obstetrics, urinary surgery, and pediatric surgery, etc.

FIG. 1.3 — Endoscopic ‘organ resection + anatomical reconstruction’ surgical technique. The concept of minimally invasive surgery is general, including therapeutic endoscope, robot and soft endoscopic surgery, but still with ‘excision of lesion + organ’ as the surgical technique.
Traditional surgery considers that the larger the incision, the clearer the exposure, the more extensive the surgery, the more thorough the treatment. But minimally invasive surgery is turning this concept on its head. Although minimally invasive surgery still uses the sequence “excision lesion + organ” as its basic mode, it induces smaller wounds compared to traditional thoracotomy or laparotomy. In clinical practice, we generally uniformly call the surgeries with smaller wounds than those induced by traditional surgery as minimally invasive surgery: these include laparoscopic surgery, robot-assisted surgery, and soft endoscopic surgery. However, because of the confusion that persists as to the exact definition of minimally invasive surgery, further discussion is needed to clarify it.
Although gastrointestinal surgery has experienced multiple development phases, the essence of the treatment is still “excision of lesion + partial or total organ resection + anatomical reconstruction.” With the continuous development of modern medicine and the humans’ quest to improve their quality of life, they cannot be satisfied with traditional open operation, laparoscopic surgery, and minimally invasive surgery: though these can meet curing demand, they cannot restore the human body to the state it was in before the onset of the disease. Therefore, it is necessary to seek a new surgical technique to guide the development of future surgical therapeutics. Such a surgical technique should eliminate diseases under the condition of maintaining unchanged the original anatomical structure of the human body, and make the patient return after the operation to his/her normal state before he/she fell ill. In 2016, Prof. Linghu Enqiang of PLA General Hospital, noting the drawbacks of the traditional surgical technique based on “partial or total organ resection + anatomical reconstruction,” first proposed a super minimally invasive surgical technique aiming at “curing diseases while preserving organs, not changing the human body’s overall anatomical structure, and maintaining normal physiological functions” (figure 1.4). Since then, the transformation of traditional surgical techniques into such an ideal surgical mode, which has been widely implemented, and the objective has been gradually extended to other related areas of surgical treatment.

FIG. 1.4 — Super minimally invasive ‘preserving organ + eliminating disease’ surgical technique. Note: Based on of keeping the organ’s structure unchanged, super minimally invasive surgery eliminates lesions, not resecting organs, not affecting anatomical structure, and not affecting the postoperative quality of life.
1.2 Super Minimally Invasive Theory Has Profound Guiding Significance for Future Surgical Therapeutics
Although the continuous progress of minimally invasive surgery, its core treatment strategy is always to achieve therapeutic objectives by removing parts of the lesion or the entire organ. While the organ is resected, the normal anatomical structure of the human body may need to be reconstructed. Such reconstruction may involve different degrees of complexity. Human evolution till today has given every organ its own unique function and value, although certain organs are more easily affected by diseases. At present, our research on the connections between human organs is still not comprehensive.
Although molecular medicine has penetrated deep into the microworld, our understanding of the human body remains relatively limited.
Based on anatomy, modern medicine decomposes the human body according to organs and systems, while conducting research and treatment. However, the human body is a complex whole, and its metabolism is complex and coordinated. Such a segmented research method may cannot totally capture the wholeness of the human body. In addition, minimally invasive surgery of human organs usually needs to excise lesions and organs, and it will inevitably bring a significant negative impact on the human body (figure 1.5). Some negative impacts can be obviously observed after the operation, for example, refractory reflux esophagitis, malnutrition, and lifestyle change, etc. after proximal gastrectomy. Research has evidenced that gastrectomy may have a significant impact on the activity of the posterior cingulate cortex. Other effects are not fully understood, such as whether the operation may affect the physiological functions of other organs, and its impact on human metabolism. Therefore, the chain effect of losing one particular organ may be beyond our current cognitive scope, so the maximum goal of illness treatment is to try to keep the original anatomical structure unchanged.
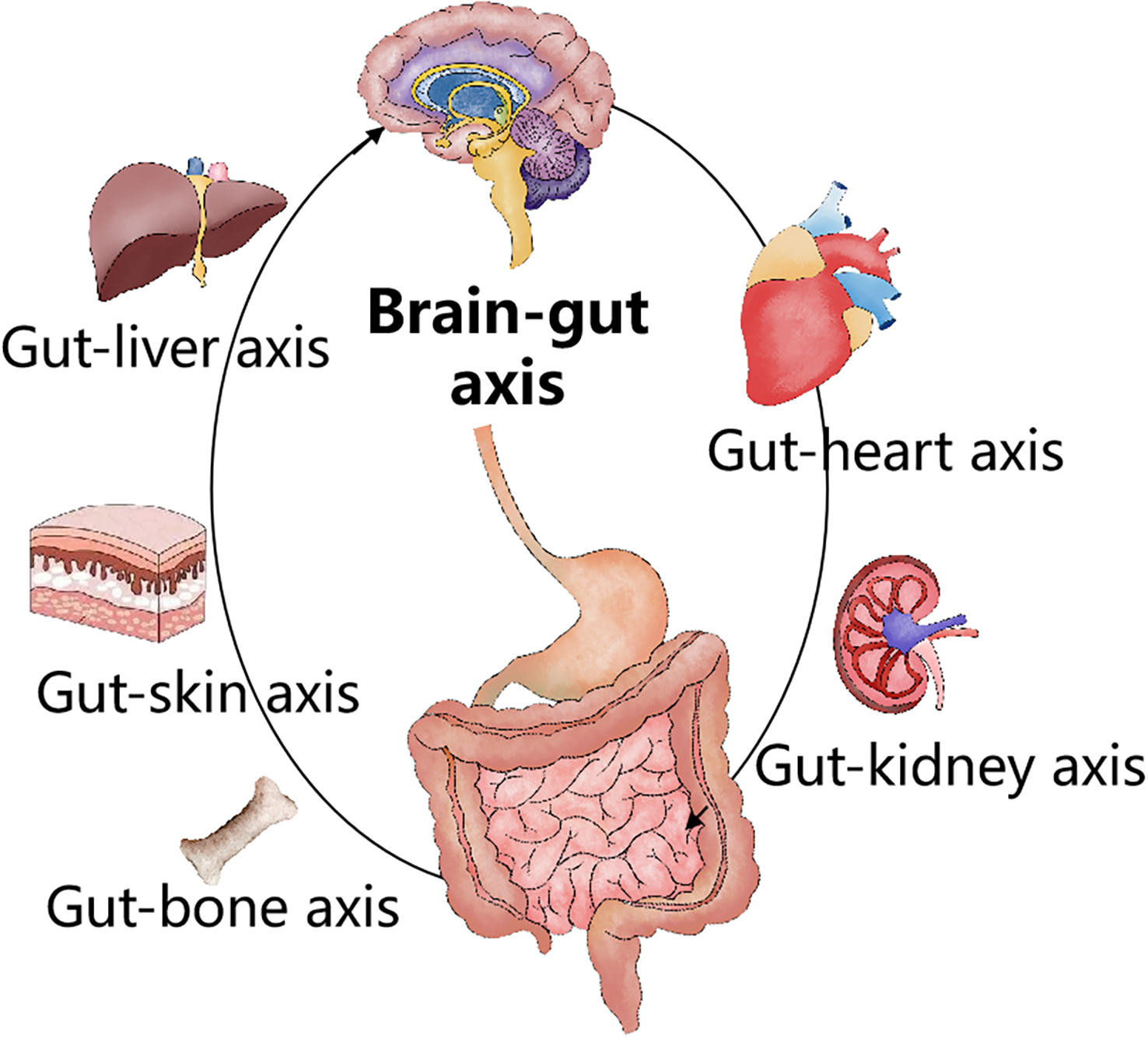

FIG. 1.5 — Connection axis among human organs.
Super minimally invasive surgery is a complex concept, which perfectly points out the best means of treating diseases. SMIS does not refer to the specific type of surgery or treatment, but rather to a comprehensive concept based on maintaining the integrity of the anatomical structure while achieving treatment objectives at the same time. Compared to traditional surgery, it guides the transformation of the concept of treatment, with the incomparable advantage over other surgical modals or techniques to preserve at a maximum the normal operation of the human physiological functions.
Super minimally invasive theory is a medical mode, aiming at planning and guiding the development of the future operation. With the rapid development of modern medicine, no matter the exploration and innovation of the basic medical research, or the huge progress of the clinical medical technologies, all are constantly marching toward the realization of the super minimally invasive goal. Super minimally invasive theory can be understood as a biological medical mode; it builds the development frame for the future therapeutic operation, and provides guidance for future medical breakthrough.
Super minimally invasive theory is an innovative thinking, it continuously stimulates and encourages research and the development of new technologies and new methods. The establishment of the super minimally invasive theory “growing out of from nothing” represents an innovative way of thinking, which breaks with the limits of the minimally invasive surgical concept of the past and introduces a new medical mode. As an innovative thinking, the super minimally invasive theory stimulates constant improvement and research into medical technology equipment and surgical techniques, and provides a major boost to the progress of modern medicine.
Super minimally invasive theory is a link connecting Internal Medicine and Surgery, closing the boundary between the two, making both become fuzzy. With improved knowledge of diseases, many symptoms of traditional internal medicine diseases can be treated, or now even cured, by super minimally invasive surgical treatment. Meanwhile, the treatment methods for a large number of traditional surgical diseases have also changed. This is not limited to a large scope of traumatic open surgery or to minimally invasive surgery alone, but it is extending towards super minimally invasive surgery, which reduces the occurrence of complications while gaining a better therapeutic effect.
Super minimally invasive theory can be viewed as the Noah’s Ark of modern medicine. It accompanies man’s quest and aspiration to remain mentally and physically healthy. Not limited to the gastrointestinal endoscopy field, super minimally invasive surgery also applies to other surgical fields. Modern medicine not only focuses on the curing rate of diseases, but attaches more importance to the patient’s quality of life, sense of comfort and experience, etc.
Super minimally invasive surgery is in line with the development trend in modern medicine, and its purpose lies in achieving maximum therapeutic effect with the least possible destruction to the physiological structure and with the lowest economic cost. This Noah’s Ark that carries mankind’s long-held desire for health, will surely unfurl its sails to guide the medical field towards a brighter future.
1.3 Guided by the Super Minimally Invasive Theory, Super Minimally Invasive Surgery is Booming
Super minimally invasive surgery has developed by leaps and bounds in clinical work, particularly by its wide application in the field of gastrointestinal endoscopy (figure 1.6). Since the “super minimally invasive” concept was proposed in 2016, it has been widely accepted by the medical colleagues and the patients. In 2019, the Association of Gastrointestinal Endoscopy, Chinese Society of Medicine, has established the “super minimally invasive collaboration group,” laying a solid foundation for the international development of super minimally invasive surgery. In the same year, the World Endoscopy Organization (WEO) established a “super minimally invasive” committee, and the China National Committee for Terminology in Science and Technology also listed the “super minimally invasive” professional terminology in the book Chinese Terms in Digestive Endoscopology. In 2022, super minimally invasive surgery obtained financial assistance from the 14th Five-Year National Key Research and Development Program of China and used the funds for the “establishment of the curative effect evaluating system of the super minimally invasive surgery for gastrointestinal tumors and research of the application model.” On that basis, research for new surgical technique has been continuously carried out to standardize and optimize the current operation plans and has been actively promoted and applied nationwide.
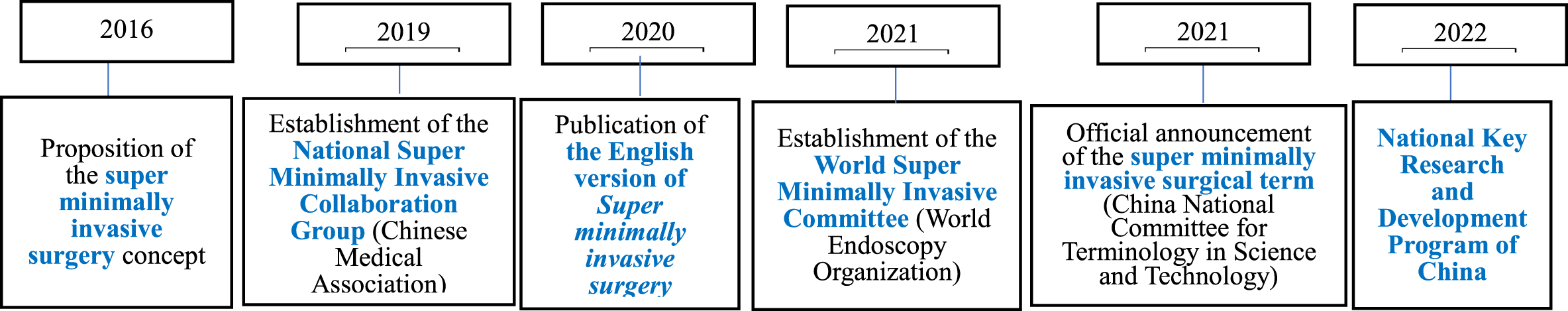

FIG. 1.6 — History of the development of super minimally invasive surgery.
Super minimally invasive surgery is continuously innovating and developing along four channels: natural, tunnel, puncture, and multiple cavity. The specific features are as follows.
1. Development of the natural channel
(1) Development of the non-full-thickness resection of the digestive tube: over the past 20 years, local tissue excision techniques like endoscopic submucosal dissection (ESD), endoscopic mucosal resection (EMR), etc., have become the first-choice therapies for the treatment of early gastrointestinal cancer; in the 20 years that followed, the exploration refinement of the expanded-indication SMIS excision technique and the sealing technique for large wounds will be the development directions.
(2) Development of pancreaticobiliary endoscopy: endoscopic retrograde cholangiao-pancreatography (ERCP) has developed steadily for more than half a century, and finally blazed the visible field of the pancreaticobiliary diseases after the emergence of biliary and pancreatic vision equipment, which explored the diagnosis and treatment scope of the endoscope, making precision diagnosis and treatment become a reality. On that basis, a series of SMIS surgeries for biliopancreatic diseases have obtained decisive progress, including laser lithotripsy under direct choledochoscope with preserved sphincter, exploratory pancreatic duct with cholangiopancreatic vision system, super minimally invasive photodynamic therapy under the pancreatic duct, radiofrequency ablation of the bile duct under direct choledochoscope, direct choledochoscopy with a papillary support for cholecystolithiasis and extraction of biliary calculus, etc.
(3) Development of robot therapy technology: this is the direction of technical development in the medical field in the future, including the implementation of the digestive endoscope robot undergoing SMIS channel resection via the natural route, of the soft medical robot, of the magnetic-driven medical robot, of operational navigation, of intraoperative imaging and visualization systems, of the gastrointestinal endoscopy robot for the treatment of colon diseases, etc.
2. Development of the tunnel channel
Tunnelling technique is a diagnosis and treatment technique of the mucosal side of the lateral muscularis propria of the digestive tube pass through the muscularis propria to the outside of the digestive lumen, so its development will be based on the following aspects:
(1) Non-full-thickness resection of the mucosal side of the digestive tube: the endoscopic submucosal tunnel dissection (ESTD) technique, whose purpose is non-full-thickness resection via the tunnel channel, has become the standard method for treating the large gastrointestinal area of early cancer, and it is certified as effective, safe, with high cure and resection rates. Therefore, the structured operation, standard quality control and base hospital promotion will be the direction for future development.
(2) Non-full-thickness resection of the muscularis propria side of the digestive tube: the STER of the resection of muscularis propria and submucosal tumors of the esophagus via the tunnel channel has become the standard SMIS; in the future, the excision site can be extended, for instance to the transtunnel resection of submucosal neoplasms in the prepyloric area, etc.
(3) The full-thickness dissection of the muscularis propria side of digestive tube: peroral endoscopic myotomy (POEM) is the first-choice treatment for achalasia, and endoscopic Ling typing effectively guides the selection of specific surgical methods for POEM. Gastric peroral endoscopic pyloromyotomy (G-POEM) evolved based on the fact it is presently regarded as one effective and safe operation for treating refractory gastroparesis. Therefore, improving and standardizing G-POEM and expanding indications will be the future development direction.
(4) The technique of passing through the muscularis propria for extraluminal treatment of the digestive ducts: this opens a new activity area for gastrointestinal endoscopy, and will enable decisive progress to be made in the SMIS operation.
3. Development of the puncture channel. Over the past 10 years, endoscopic ultrasonography (EUS) has gradually become the third eye for endoscopists. Its combination with SMIS brings a new type model for the diagnosis and treatment of organic lesions outside the digestive tubes. For example, EUS-guided bile drainage (EUS–BD) and EUS-guided pancreatic duct drainage (EUS–PD) have been used as effective treatments after the failure of trans-nipple drainage; meanwhile the treatment strategy for acute necrotizing pancreatitis has also changed greatly. EUS-guided super minimally invasive drainage by gastric parietal puncture for pancreatic encapsulated necrosis and EUS-guided pancreatic cystic lesion puncture laurosinol ablation have become the mainstream treatment methods. In the future, SIMS operations like exploring and refining super minimally invasive drainage via a gastric puncture channel for suppurative cholecystitis, EUS-guided super minimally invasive gallstone extraction via a gastric puncture channel, EUS-guided portosystemic shunts and EUS-guided drainage of the chest, the abdomen and pelvic abscess, etc. will be the development directions.
4. Development of the multi-cavity channel. As part of multidisciplinary treatment in the age of precision medicine, combined endoscopic surgery will create chances at sites where the single endoscope does not know what to do, to provide convenience to positions where treatment is difficult. Over the recent 10 years, development of this direction has focused on the endoscopic combined with laparoscopic treatment of upper gastrointestinal stromal tumors and refractory colon polyps. The surgical modes include laparoscopy-assisted endoscopic full-thickness resection (LAFEFR), laparoscopic and endoscopic cooperative surgery (LECS), etc. In the future, developments in this direction will challenge traditional surgical techniques. Endoscopic digestion combined with laparoscopic bi-endoscopic lesion resection + lymph node dissection will further expand the SMIS therapeutic indications in the field of gastrointestinal cancer.
Super minimally invasive surgery technique is making steady progress, heralding the new trend in future gastrointestinal surgery. Such a surgical method uses a cutting-edge endoscopic equipment, robot technology and an image navigation system, significantly improves the accuracy and visibility of operations, reduces injury to the patient’s body, protects the surrounding healthy tissues, thus improving the success rate of operations. With continuous advances in technologies and the improvement of surgical techniques, the super minimally invasive surgery has a lot of space for development in the future. For instance, by introducing technologies like artificial intelligence and machine learning, etc., automation and intelligence of the operative process can be realized, further improving the precision and effect of operations. In addition, with the continued application of robot technology, super minimally invasive surgery is expected to realize remote control and operation, expanding the quality of medical resources to wider areas. We believe that, with the continuous advances in technologies and medical innovation, super minimally invasive surgery will become an important means of treating gastrocolorectal diseases and will be more beneficial to patients’ health.
References
[1] Linghu E. (2016) A new stage in the history of surgery: super minimally invasive technology, Chin. J. Gastrointest. Endosc. (Electron. Ed.) 3, 97–98.
[2] Linghu E. (2024) Three stages of the development of gastrointestinal endoscopic therapeutics, Chin. J. Gastrointest. Endosc. (Electron. Ed.) 11(2), 73–74.
[3] Linghu E. (2022) A new stage of surgical treatment: super minimally invasive surgery, Chin. Med. J. (Engl.) 135, 1–3.
[4] Linghu E. (2024) New direction for surgery: super minimally invasive surgery, World J. Gastroenterol. 30(12), 1676–1679.
[5] Ronellenfitsch U., Najmeh S., Andalib A., et al. (2015) Functional outcomes and quality of life after proximal gastrectomy with esophagogastrostomy using a narrow gastric conduit, Ann. Surg. Oncol. 22, 772–779.
[6] Gu Y., Li G., Wang J., von Deneen K. M., et al. (2020) Comparing the impact of laparoscopic SELeve gastrectomy and gastric carcinoma surgery on resting-state brain activity and functional connectivity, Front. Neurosci. 14, 614092.
[7] Linghu E. (2019) The development direction and future of digestive endoscopology-the task of CCDE, Chin. J. Gastrointest. Endosc. (Electron. Ed.) 6, 161–162.
[8] Linghu E. (2012) Revisiting the new endoscopic diagnosis and treatment model for early gastrointestinal cancer, Chin. J. Laparosc. Surg. (Electron. Ed.) 5, 3.
[9] Linghu E. (2021) Terminology of digestive endoscopy, Chin. J. Gastrointest. Endosc. (Electron. Ed.) 08, 192.
[10] Linghu E., Feng J. (2008) Development and progress in the technique of endoscopic resection for precancerous lesions and early carcinomas in gastrointestinal tract, Dig. Dis. Endosc. (Z1), 27–29.
[11] Linghu E., Lu Z., Feng J., et al. (2008) Experience of endoscopic submucosal dissection (ESD) for 24 cases of gastrointdestinal early stage cancer or previous cancer lesion, Chin. J. Pract. Intern. Med. 28, 964–966.
[12] Lu Z., Linghu E., Huang Q., et al. (2008) Endoscopic submucosal dissection for early gastrointestinal cancer and precancerous lesions, Chin. J. Dig. Endosc. 25, 578–583.
[13] Meng J., Peng L., Sun G., et al. (2010) Cooperation of technicians in endoscopic submucosal dissection, J. Chin. PLA Postgrad. Med. Sch. 31, 253–254.
[14] Lu Z., Linghu E., Wang S., et al. (2011) Endoscopic submucosal dissection of superficial upper gastrointestinal lesions, J. Chin. PLA Postgrad. Med. Sch. 32, 111–113.
[15] Bu B., Linghu E. (2012) Research advances in endoscopic resection for early esophageal cancer, Chin. J. Laparosc. Surg. (Electron. Ed.) 5, 50–52.
[16] Cui Y., Lu Z., Linghu E., et al. (2013) The clinical value of endoscopic submucosal dissection for early gastric cancer, Chin. J. Gastroenterol. Hepatol. 22, 341–343.
[17] Cui Y., Lu Z., Linghu E., et al. (2013) Endoscopic submucosal dissection of esophageal squamous cell neoplasms, Chin. J. Gastroenterol. Hepatol. 22, 324–326.
[18] Sun X., Wang X., Lu Z., et al. (2013) How nova endoscopic procedures altered the diagnostic and therapeutic modality of early gastrointestinal cancer, Chin. J. Pract. Intern. Med. 33, 207–209.
[19] Wu B., Linghu E., Li H. (2014) Long-term effects of endoscopic submucosal dissection upon gastric mucosal lesions, Chin. J. Gastrointest. Endosc. (Electron. Ed.) 9–14.
[20] Zhang B., Linghu E., Chai N., et al. (2017) Long-term effects of endoscopic submucosal dissection upon gastric mucosal lesions, Chin. J. Gastrointest. Endosc. (Electron. Ed.) 4, 97–102.
[21] Zhang G., Zhang D., Cai F., et al. (2020) New development of endoscopic retrograde cholangiopancreatography in the diagnosis and treatment, Chin. J. Gastrointest. Endosc. (Electron. Ed.) 7, 205–210.
[22] Zhao D., Chai N., Linghu E., et al. (2021) Comparison of endoscopic ultrasound- guided drainage with metal stent and percutaneous nephroscopic necrosectomy for walled off necrosis, Chin. J. Laparosc. Surg. (Electron. Ed.) 14, 298–303.
[23] Linghu E. (2022) Innovative exploration of super minimally invasive concept in pancreaticobiliary direct vision field, Chin. J. Gastrointest. Endosc. (Electron. Ed.) 09, 240.
[24] Zhang B., Yao Y., Zhang W., et al. (2023) Peroral cholangioscopy treatment of biliary and pancreatic disease: how super minimally invasive surgery have changed ERCP, Chin. J. Gastrointest. Endosc. (Electron. Ed.) 10, 264–266.
[25] Zhai Y., Linghu E., Li H., et al. (2014) Comparison of endoscopic submucosal tunnel dissection with endoscopic submucosal dissection for large esophageal superficial neoplasms, J. South. Med. Univ. 36–40.
[26] Jin Z., Linghu E., Jin D. (2014) Animal experimental study on circular esophageal mucosal dissection using double-tunnel technique, Chin. J. Dig. Endosc. 31, 219–220.
[27] Linghu E., Wang N., Wang H. (2015) Application of inverted “T”-shaped incision in tunnel-based resection of cardia intramuscular tumors: a report of two cases, Chin. J. Dig. Endosc. 32, 483–484.
[28] Xiang J., Linghu E., Li L., et al. (2019) Safety and efficacy of endoscopic submucosal tunnel dissection for large early esophageal cancer, Chin. J. Laparosc. Surg. (Electron. Ed.) 12, 272–276.
[29] Ma L., Chai N., Chen Q., et al. (2018) Clinical efficacy analysis of submucosal tunneling endoscopic resection for upper gastrointestinal submucosal tumors, Chin. J. Dig. Endosc. 35, 18–22.
[30] Xiong Y., Hu H., Gao Y., et al. (2014) Endoscopic esophageal submucosal tunnel resection of cardiac benign tumors originating from muscularis propria, Natl. Med. J. China 94, 3655–3657.
[31] Linghu E. (2011) The foundation and principles of endoscopic technological innovation: insights from the development of endoscopic submucosal dissection and peroral endoscopic myotomy, Chin. J. Dig. Endosc. 28, 603–604.
[32] Linghu E. Q., Li H. (2011) Endoscopic classification for achalasia in tunnel technique, China Contin. Med. Edu. 03, 78–80.
[33] Ma X., Linghu E., Wang N., et al. (2014) Application of peroral endoscopic myotomy with short tunnel for Ling IIc type achalasia, Chin. J. Laparosc. Surg. (Electron. Ed.) 271–274.
[34] Linghu E., Ding H. (2014) Operational approach of peroral endoscopic myotomy, Chin. J. Gastrointest. Endosc. (Electron. Ed.) 55–59.
[35] Li H., Linghu E. (2012) Progress of peroral endoscopic myotomy in the treatment of achalasia, Chin. J. Laparosc. Surg. (Electron. Ed.) 5, 47–49.
[36] Linghu E. (2012) A new concept for lower esophageal sphincter incision in peroral endoscopic myotomy (POEM), Chin. J. Laparosc. Surg. (Electron. Ed.) 5, 1–2.
[37] Linghu E., Li H., Feng X. (2012) Efficacy and safety of transverse entry incision during peroral endoscopic myotomy for achalasia, Chin. J. Dig. Endosc. 29, 483–486.
[38] Linghu E., Li H., Wang X., et al. (2012) Primary results of a randomized study comparing peroral endoscopic myotomy, botulinum toxin injection and balloon dilation for achalasia, Chin. J. Laparosc. Surg. (Electron. Ed.) 5, 7–11.
[39] Linghu E., Li H., Wang X., et al. (2012) Feasibility report of full-thickness myotomy in peroral endoscopic myotomy (POEM): a report of two cases, Chin. J. Laparosc. Surg. (Electron. Ed.) 5, 63.
[40] Linghu E., Li H., Wang X., et al. (2012) Feasibility report of glasses-assisted peroral endoscopic myotomy (POEM): a report of two cases, Chin. J. Laparosc. Surg. (Electron. Ed.) 5, 33.
[41] Basile P., Gonzalez J. M., Le Mouel J. P., et al. (2020) Per-oral endoscopic myotomy with septotomy for the treatment of distal esophageal diverticula (D-POEM), Surg. Endosc. 34, 2321–2325.
[42] Mohan B. P., Chandan S., Jha L. K., et al. (2020) Clinical efficacy of gastric per-oral endoscopic myotomy (G-POEM) in the treatment of refractory gastroparesis and predictors of outcomes: a systematic review and meta-analysis using surgical pyloroplasty as a comparator group, Surg. Endosc. 34, 3352–3367.
[43] Linghu E. (2016) Development of gastrointestinal endoscopy tunneling technology, China Med. News 31, 14.
[44] Linghu E. (2017) Interpretation of the expert consensus on digestive endoscopic tunnel technique (2017, Beijing), Chin. J. Gastrointest. Endosc. (Electron. Ed.) 4, 159–161.
[45] Zhang B., Chai N., Li L., et al. (2022) Advances in minimally invasive therapy of pancreatic fluid collection, Chin. J. Gastrointest. Endosc. (Electron. Ed.) 09, 41–44.
[46] Ning B., Linghu E. (2021) New advances in different treatment channels of super minimally invasive surgery by digestive endoscopy, Chin. J. Dig. Endosc. 38, 969–973.
[47] Wen J., Liang J., Cai F., et al. (2014) Effectiveness and safety of endoscopic ultrasound-guided transgastric or transpapillary drainage in treating pancreatic pseudocyst, Acta Acad. Med. Sin. 36, 194–197.
[48] Liu Y., Wang Z., Wang X., et al. (2007) Application of butt joint of percutaneous transhepatic biliary drainage and endoscopic retrograde cholangiopancreatography in treatment of benign obstructive jaundice, Chin. J. Postgrad. Med. 30, 29–30, 40.
[49] Liu Y., Wang Z., Wang X., et al. (2006) Stent implantation through butt joint of PTBD and ERCP for the treatment of obstructive jaundice, Med. J. Chin. People’s Liberation Army 31, 1101–1102.
[50] Linghu E. (2023) Endoscopic ultrasound-guided lauric macrogol ablation, Chin. J. Gastrointest. Endosc. (Electron. Ed.) 10, 144.
[51] Li X., Chai N., Linghu E. (2022) Endoscopic ultrasonography in the diagnosis and treatment of groove pancreatitis combined with pancreatic pseudocyst: a case report, Chin. J. Dig. Endosc. 39, 658–659.
[52] Linghu E., Li H. (2021) Endoscopic ultrasonography in diagnosis and treatment of pancreatic cystic neoplasms, Chin. J. Pract. Intern. Med. 41, 378–381.
[53] Du C., Chai N., Linghu E., et al. (2020) Long-term effects of endoscopic ultrasonography-guided lauromacrogol ablation for the treatment of pancreatic cystic neoplasms: a prospective study (with video), Chin. J. Dig. Endosc. 37, 696–701.
[54] Linghu E., Feng X., Li H., et al. (2017) A case of endoscopic ultrasound-guided radiofrequency ablation combined with lauric macrogol ablation for pancreatic cystic tumors, Chin. J. Gastrointest. Endosc. (Electron. Ed.) 4, 44–46.
[55] Linghu E. (2022) Digestive endoscopy enters the new era of super-minimally invasive surgery, China Med. News 37, 15.
[56] Linghu E. (2022) To the new era of super minimally invasive surgery, Chin. J. Gastrointest. Endosc. (Electron. Ed.) 09, 61–62.
[57] Ning B., Chai N., Li H., et al. (2021) The efficacy of EUS-LA in the treatment of PCNs under the new evaluation system, Chin. J. Laparosc. Surg. (Electron. Ed.) 14, 293–297.
[58] Cai F., Cheng L., Linghu E., et al. (2003) Endoscopic interventional therapy for patients with acute biliary pancreatitis, J. Intern. Intensive Med. 9, 68–70.
[59] Xiong Y., Hu H., Wang A., Linghu E., et al. (2015) Preliminary experience with endoscopic gastric submucosal tumor resection through the submucosal tunnel using double tunnel and double flex endoscope, J. South. Med. Univ. 455–458.
[60] Zhang Y., Linghu E., Jin D., et al. (2014) An animal experimental study on access to mediastinum with flexible endoscope via suprasternal fossa, Chin. J. Dig. Endosc. 31, 269–271.
[61] Linghu E., Qin C., Tang P., et al. (2013) Endoscopic resection of gastric submucosal tumors originated from muscularis propria layer with double peroral endoscopes: a pilot study, Chin. J. Dig. Endosc. 30, 304–306.
[62] Oyama K., Ohuchida K., Shindo K., et al. (2020) Thoracoscopic surgery combined with endoscopic creation of a submucosal tunnel for a large complicated esophageal leiomyoma, Surg. Case Rep. 6, 92.
[63] Tsujimoto H., Yaguchi Y., Kumano I., et al. (2012) Successful gastric submucosal tumor resection using laparoscopic and endoscopic cooperative surgery, World J. Surg. 36, 327–330.
Chapter 2 The Super Minimally Invasive Theoretical System
2.1 The Definition and Implementation Principles of Super Minimally Invasive Surgery
In 2021, the book Chinese Terms in Digestive Endoscopology released by the China National Committee for Terminology in Science and Technology defined the technical term of “super minimally invasive surgery.” Super minimally invasive surgery refers to a surgical treatment plan that preserves organs and functions and removes lesions for diseases that require surgical resection or diseases that have poor long-term drug treatment effects. The disease range includes gastrointestinal tumors (gastric cancer, intestinal cancer, esophageal cancer, stromal tumor, etc.), functional diseases (achalasia, gastroparesis, etc.), retrogression (e.g., gastroesophageal reflux disease, etc.). In excising lesions or removing foci, super minimally invasive surgery does not resect organs, does not affect normal anatomical structure, as well as it does not affect both postoperative survival time and quality of life, thus realizing the goal of “curing the disease, recovering as before.”
Not all operations performed endoscopically can be classified as super minimally invasive surgery. Similarly, the endoscopic surgery like laparoscopic surgery, thoracoscopic surgery and so on are also not eliminated from the scope of super minimally invasive surgery. Only surgeries conforming to the following conditions can be regarded as super minimally invasive surgery: preserving the organ’s anatomical structure, ensuring that the body’s important functions are kept unchanged. Super minimally invasive surgery has no direct connection with the instruments and equipment used for the operations. For example, operations like laparoscope-assisted endoscopic full-thickness resection of gastrointestinal cancer, stromal tumor and polyp, etc. can preserve the function of the stomach and intestine, and meet the above conditions, so they can be classified as super minimally invasive surgery.
The implementation of gastrointestinal endoscopic SMIS must respect specific implementing principles, including the following 10 items, aiming at perform safe, effective surgical treatment.
1. Preserving organs, anatomical structure, and organ functions unchanged. One should try as much as possible to excise the lesion itself, without destroying the surrounding organs and tissues. Due to the limitation of factors like lesion depth and scope, etc., one cannot merely excise lesions; one should try as much as possible to preserve the normal organs and tissues during the complete excision of lesions, and one must keep the anatomical structure and functions unchanged.
2. Lacunar integrity principle. One should try as much as possible to put the operation’s accesses, routes and target positions into the same lacunar, and try to avoid multilacunar channel incision, to reduce the postoperative trauma and the recovery period. In eSMIS treatment, this principle should be considered at the first of the four channels.
3. Surgery sterile principle. In non-sterile and sterile conditions, the sterile condition should be the first-choice, this principle should be strictly observed especially for operations via multilacunar channels involving the surgery field or the percutaneous route.
4. In conditions with chemical stimulation and without chemical stimulation, the without chemical stimulation is the first-choice principle. In eSMIS puncture channel and multiple-cavity treatments, touching the pipeline system of the puncture access, such as vessels, lymph vessels, bile duct, gallbladder and pancreatic duct, etc., should be avoided, so as to prevent chemicals from flowing into abdomen and causing abdominal infection.
5. Natural channel preference principle. In both natural and without natural channel conditions, the natural channel should be the first choice; the selection of the natural channel should at first comply with the 1st to 4th principles.
6. Preference principle when the natural channel has taboos. When there are taboos in accessing the natural channel, or in situations like narrowing of the access to the natural channel, occlusion, etc. resulting from congenital or acquired factors, preventing the endoscope from entering, it should be the first choice for the puncture channel, and at first comply with the 1st to 4th principles.
7. Surgical route proximity principle. As regards the distance between access and operative sites, in addition to compliance with the above principle of the application of the four channels in eSMIS treatment, the proximity principle should be followed, in order to avoid damage to peripheral organs or tissues resulting from the long route. The shorter the surgical route, the smaller the time of operation and the difficulty.
8. Prevention of bleeding and timely hemostasis principle. The probability of bleeding from the lesion should be fully predicted before the operation, in order to confirm the good preventive and hemostatic techniques for such a lesion, as well as the substitution measures to ensure an almost 100% hemostasis rate. This is the principle that must be considered in the eSMIS treatment 4-route application.
9. Prevention of perforation and lacunar sealing principle. The lesion perforation probability should be fully predicted before the operation, to confirm the skillful prevention and interpenetration technique among the closed lacunas, to ensure that the human original lacunar integrity and closing state are restored. This is the principle that must be considered in the eSMIS treatment 4-route application.
10. Tumor treatment principle. For the eSMIS treatment of benign, malignant tumors, the principles of non-resection of the tumor, ‘en bloc’ resection, tumor free technique and prevention of metastasis should be followed.
2.2 The Four Channels of Super Minimally Invasive Surgery
SMIS is mainly realized by four channels: human natural channel, tunnel channel, puncture channel, multi cavity, and several dozens of methods.
(I) Approach via human natural channel
This refers to surgeries that enter the human’s naturally opening orifice and are performed in a natural cavity conforming to super minimally invasive requirements. For instance, through human natural channels such as the nasal cavity, the auditory meatus, the respiratory tract, the mouth, the anus, the vagina, the urinary tract, etc. Let’s take the example of single-channel SMIS surgery of the digestive tract (figure 2.1).
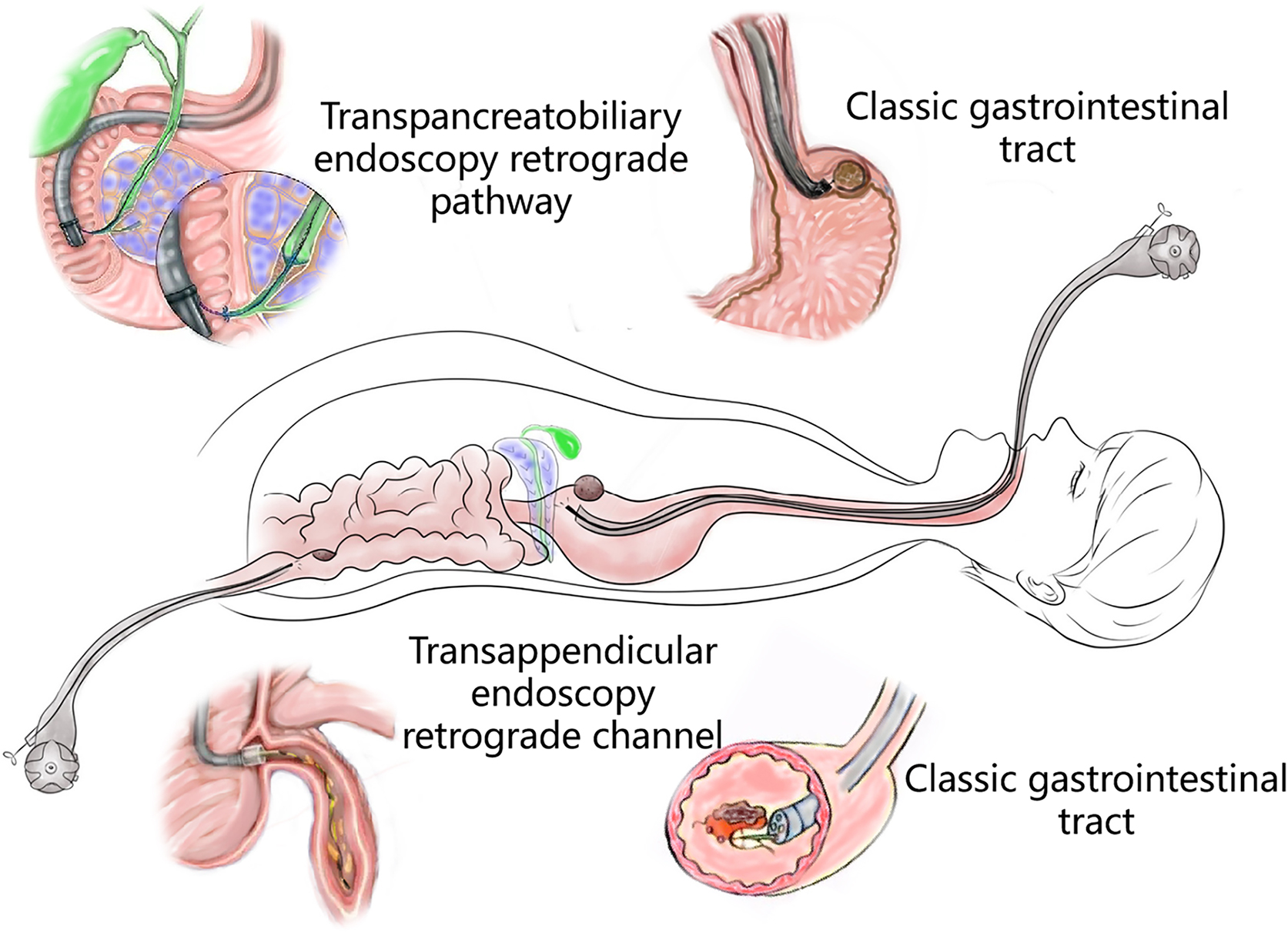

FIG. 2.1 — SMIS through a natural channel.
1. Classic digestive tube channel. The classic digestive tube approach refers to the path through the continuous tube from the mouth to the anus. The wall of the tube is generally divided into the mucosal layer, submucosal layer, muscularis propria layer and serous layer, from the intracavitary to the extracavitary layer. So far, most gastrointestinal endoscopy operations have carried out in this tube.
After endoscopic resection of circumferential esophageal lesions, almost 100% of patients experienced symptoms like esophageal stenosis and an inability to eat. Our team is the first in the world to carry out endoscopic autologous skin flap transplantation to prevent stenosis after endoscopic resection of esophageal circumferential lesions (figure 2.1), and 19 cases of autologous transplantation of skin flaps and simple placement of an esophageal stent for the prevention of esophageal stenosis were compared. The results showed that rate of stent removal for the group who had received an autologous skin flap transplantation was significantly reduced compared to the group with a simple esophageal stent (36.8%–78.9%). Currently, this research is also aimed at optimizing surgical and transplant methods, so as to improve the survival rate of skin flaps and to reduce the rate of occurrence of esophageal stenosis.
Endoscopic autologous skin flap grafting, with the elimination of the postoperative scar as the starting point, points the way to solving the postoperative benign stenosis that is widespread in various surgeries, and also provides a certain theoretical basis for the repair of injuries to the digestive duct. Endoscopic autologous skin flap transplant skillfully avoids “pain points” such as large trauma, slow recovery, and many complications during surgical resection, and goes deeper into the “blind area” of disease that cannot be reached by a surgical operation.
2. Transappendicular retrograde endoscopy. The appendiceal duct is a narrow, long and winding blind gut. Endoscopic retrograde appendicitis therapy (ERAT) complements endoscopic irrigation, drainage, etc., operations, thus enabling acute appendicitis to be cured. Although the appendix is generally regarded as a vestigial organ, it can be immune. It can therefore be of very great clinical significance to preserve the appendix as part of the treatment of acute appendicitis.
In 2015, a study was carried out on a group of 34 patients with acute pure appendicitis, among which 33 patients had successfully completed ERAT: abdominal pain was immediately alleviated in 32 patients, whereas during the follow-up period, two patients had recurring abdominal pain and underwent an appendicectomy, which proves that ERAT is an effective method for the treatment of acute pure appendicitis. In 2021, a study integrated 210 patients who were diagnosed with acute appendicitis. The researchers divided the patients into three groups: ERAT group, laparoscopic appendectomy group, and open appendectomy group. The results showed that ERAT is a safe and effective method for treating acute appendicitis and appendiceal abscess. Comparing to open appendectomy and laparoscopic appendectomy, ERAT was less traumatic, less expensive, with shorter time of operation and quicker recovery time. The effect of ERAT is more significant, especially in patients with acute appendicitis, without complications like fecal calculi, narrowing or abscess of the appendiceal cavity.
3. Retrograde transpancreatobiliary endoscopy. Super minimally invasive surgery via this channel includes endoscopic surgeries aimed at performing endoscopic lithotomy of the pancreatic biliary system, stent implantation, and biopsy, etc., via the duodenal papilla. The pancreatic duct runs through the entire length of the pancreas; its diameter gradually increases from left to right; the bile duct mainly includes the gallbladder duct, the hepatic duct and the common bile duct, etc. The pancreatic duct joins the bile duct and opens at the descending part of the duodenum, with the sphincter of Oddi surrounding the opening.
The normal common bile duct pressure is 10–15 mmHg (1 mmHg = 0.133 kPa) higher than that of the duodenum, but the basal pressure of the sphincter of Oddi is about 4 mmHg higher than that of the pancreatic biliary system, so that the sphincter of Oddi can effectively prevent the regurgitation of duodenal contents and avoid gastrointestinal bacterial retrograde infection. EST is usually performed before duodenal papillary intubation, which damages the integrity of the sphincter of Oddi, leading to sphincter relaxation and a loss of the anti-reflux physiological function. In 2020, a study was conducted to investigate the feasibility and effectiveness of duodenoplasty to restore the function of the sphincter of Oddi. The study prospectively included 30 patients who had undergone EST lithotomy and duodenal papilloplasty; 80% of patients had a basal pressure of the sphincter of Oddi after duodenal papilloplasty >10 mmHg, and the pressure parameter was resumed after the operation, and was unrelated to the healing process. Postoperative mild pancreatitis occurred in six cases, and a recurrence of the stone in one case. Studies have shown that duodenal papilloplasty can restore the basal pressure of the sphincter of Oddi, thus restoring its anti-reflux function and possibly preventing the reappearence of gallstones and cholangitis.
The intrahepatic bile duct is the relative blind area of SpyGlass. The treatment of intrahepatic bile ducts by retrograde transpancreatobiliary endoscopy can avoid the trauma of surgical choledochoscopy and sinus infection caused by percutaneous choledochoscopy. However, there are still few reports on the diagnosis and treatment of intrahepatic bile ducts by choledochoscopy in China and abroad, and they only concern grade-I and II intrahepatic bile ducts, even if currently the surgical choledochoscopic fiber can enter directly intrahepatic bile ducts above grade IV. In 2021, a study evaluated the effect of SpyGlass on grade-I and II intrahepatic bile duct lithotomy, whereas laser lithotripsis is performed for large or impacted gallstones at the same time. The results showed that the diagnostic rate of SpyGlass was 97.14%, the success rate of one-time lithotomy was 85.71%, the occurrence rate of complication was 8.57%, the postoperative residual stone rate was 14.28%, showing that SpyGlass can effectively diagnose and treat intrahepatic bile duct stones. But so far a multi-center study is still lacking to support its clinical promotion and application. In addition, SpyGlass can also be used to remove gallstones through the gallbladder duct while preserving it, with a high rate of stone removal, while being less traumatic, with rapid recovery and effective resumption of gallbladder function.
4. Videos of clinical cases
(1) A modified endoscopic resection of the entire thickness of a gastrointestinal stroma tumor: A new sealing technique based on the principles of super minimally invasive surgery (video 2.1 (See online resources)).
(2) Novel endoscopic papillectomy technique for reducing postoperative adverse events (video 2.2 (See online resources)).
(3) Successful endoscopic transgastric retrieval of a plastic stent that migrated into the abdominal cavity during pancreatic fistula drainage (video 2.3 (See online resources)).
(4) Endoscopic retrieval of a migrated lumen-apposing metal stent into the abdominal cavity during transluminal drainage (video 2.4 (See online resources)).
(5) Incidental discovery of a pancreatic ductal adenocarcinoma during percutaneous cholangioscopy through a T-tube tract (video 2.5 (See online resources)).
(6) Three pancreatic duct lesions discovered at an early stage in a patient by cholangioscopy (video 2.6 (See online resources)).
(II) Approach via tunnel channel
This refers to the operation that meets the super minimally invasive requirements in the artificially created space by reaching the surgical site through an artificially established channel in the tissues. For instance, thyroid surgery and breast surgery through subcutaneous channels, mediastinal surgery via a supraclavicular channel, surgery for lumbar disc herniation through the back channel, and a series of operations using the gastrointestinal endoscopic tunnel technique. The following text will take the SMIS through the digestive tunnel technique as an example to present this channel (figure 2.2).
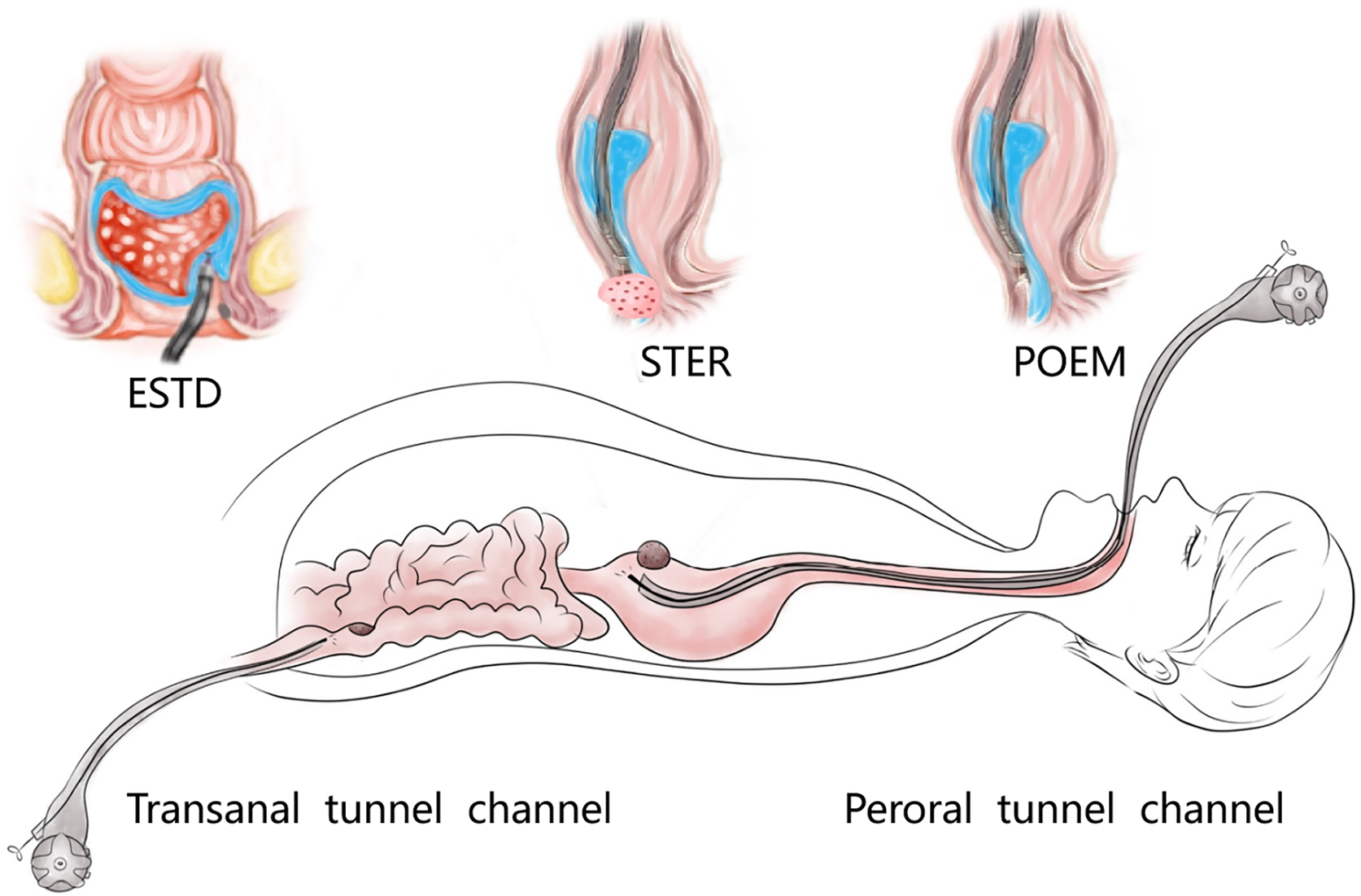

FIG. 2.2 — SMIS through a tunnel channel.
The digestive endoscopic tunnel technique (DETT) is a treatment technology using an endoscope to establish a channel between the mucosal muscle layer and the fixed muscle layer under the digestive mucosa, and leads to the mucosa side, to the muscularis propria side and, through muscularis propria, to the outside of the digestive lumen. DETT isolates the digestive lumen from other lacunae of the human body by preserving the integrity of the mucosal muscle layer or the proper muscle layer, avoiding the entry of gas and of digestive fluids, and maintaining the integrity of the structure of the human body during the treatment. Such therapeutic methods, including POEM, STER, and ESTD, are currently widely used in clinics.
1. Peroral endoscopic myotomy (POEM). POEM has been widely used in the clinical treatment of achalasia; recently, applications in other parts of the human body have also been gradually emerging. In 2020, a study was conducted on a group of eight patients esophageal diverticulum and esophageal diverticulum treated by using POEM, also known as peroral diverticulum myotomy (D-POEM). D-POEM establishes a tunnel under the esophageal mucosa and incises the ridge between the diverticulum in the tunnel to make the diverticulum disappear. Postoperative follow-up of all patients, their preoperative symptoms were improved, and no adverse events were reported. Studies have shown that D-POEM has a certain effect on esophageal diverticulum, and that it is not affected by the position of the diverticulum and the length of the ridge between them, making it a safe and effective operation.
Gastric peroral endoscopic pyloromyotomy (G-POEM) is an operation creating a tunnel through the gastric antrum to dissect the pyloric muscle. Gastroparesis is a delay in gastric emptying due to insufficient gastric dynamics and reduced gastrointestinal peristalsis. Gastroparesis occurs after proximal gastric surgery such as cardiac cancer and is also a complication of diabetes. Hyperglycemia causes paralysis of the nervous system, thus triggering pylorospasm. By cutting down the spasmodic and constrictive pyloric muscle, G-POEM makes the pylorus loose its sealing function, and food can naturally flow into the intestine. So far, it has become a new treatment method for improving the symptoms of patients with refractory gastroparesis. A meta-analysis evaluated the clinical efficacy of G-POEM and pyloroplasty in the treatment of gastroparesis. The results showed that G-POEM is comparable to phaneroplasty in the treatment of refractory gastroparesis, but that it is significantly less invasive.
2. Submucosal tunnel endoscopic resection (STER). STER is a new endoscopic technique for the treatment of gastrointestinal muscularis propria tumors; it is an endoscopic surgical modality evolved from POEM, which mainly applies to the excision of gastrointestinal muscularis propria tumors. In sites like the esophagus, gastroesophageal junction, gastric antrum, firstly, one starts by making a small incision in the mucosal surface, then the endoscope goes deeper into it, to create a tunnel in the submucosa, to completely remove the tumor under direct vision, while preserving the integrity of the mucous surface of the digestive tract. After complete resection of the tumor, the opening of the mucosal tunnel was closed with metal clips to avoid perforation of the digestive tract and to reduce postoperative complications such as digestive tract leakage, etc. This method has the advantages of short operation time, less trauma, less pain, low cost, and quick recovery of patients. It can completely dissect the tumor in a single operation, while maintaining the integrity of the digestive tract, and has the same therapeutic effect as classical surgery, and there is no postoperative scar on the surface of the body.
3. Endoscopic submucosal tunnel dissection (ESTD). For extensive colorectal lesions, research is currently being conducted into the use of ESTD to excise lesions by establishing a submucosal tunnel between the colon and the rectum. Using endoscopy, the mucosa was first incised on the anal side, then by the oral side, and then a submucosal tunnel was created from the oral side to the anal side. Finally, both sides of the mucosa were incised synchronously along the border until complete resection.
However, the space of the colorectal cavity is not a straight tube, so it is relatively difficult to construct a complete submucosal tunnel. In 2018, a study has shown that colorectal lesions can be treated using the tunnel technique. Compared to traditional ESD, it shortens operation time, reduces bleeding and perforation risks. However, due to the small sample size, this work is not sufficient to fully assess the applicability of the tunnel technique in the treatment of colorectal lesions.
4. Video of a classic case
(1) Autologous skin-grafting surgery for the prevention of esophageal stenosis after complete circular endoscopic submucosal tunnel dissection (video 2.7 (See online resources)).
(2) Challenging use of the digestive endoscopic tunnel technique to treat schwannoma originating in the trachea (video 2.8 (See online resources)).
(3) Endoscopic submucosal tunnel dissection with an elastic traction device for a circumferential superficial esophageal neoplasm (video 2.9 (See online resources)).
(4) Magnetic multi-directional anchoring-guided endoscopic submucosal tunnel dissection for large gastric lesions (video 2.10 (See online resources)).
(5) Simultaneous performance of one-tunnel per-oral endoscopic myotomy, submucosal tunnel endoscopic resection, and peroral diverticulotomy (video 2.11 (See online resources)).
(6) Peroral endoscopic myotomy with simultaneous submucosal and muscle dissection for achalasia with severe interlayer adhesions (video 2.12 (See online resources)).
(7) Moving the knife’s tip on the thoracic aorta: high-risk submucosal tunneling endoscopic resection procedure for a puzzling submucosal tumor in the esophagus ((video 2.13 (See online resources)).
(8) Prepyloric submucosal tunneling endoscopic resection for a case of inflammatory mass (video 2.14 (See online resources)).
(III) Approach via the puncture channel
This refers to the operation that meets the super minimally invasive requirements by entering the lacunar or lumen through the puncture route. For instance, laparoscopic, thoracoscopic, arthroscopic-aided operations, related operations after body surface guided puncture bile duct, gallbladder, related operations conducted after ultrasonic endoscopic guided puncture chambers, etc. (figure 2.3).
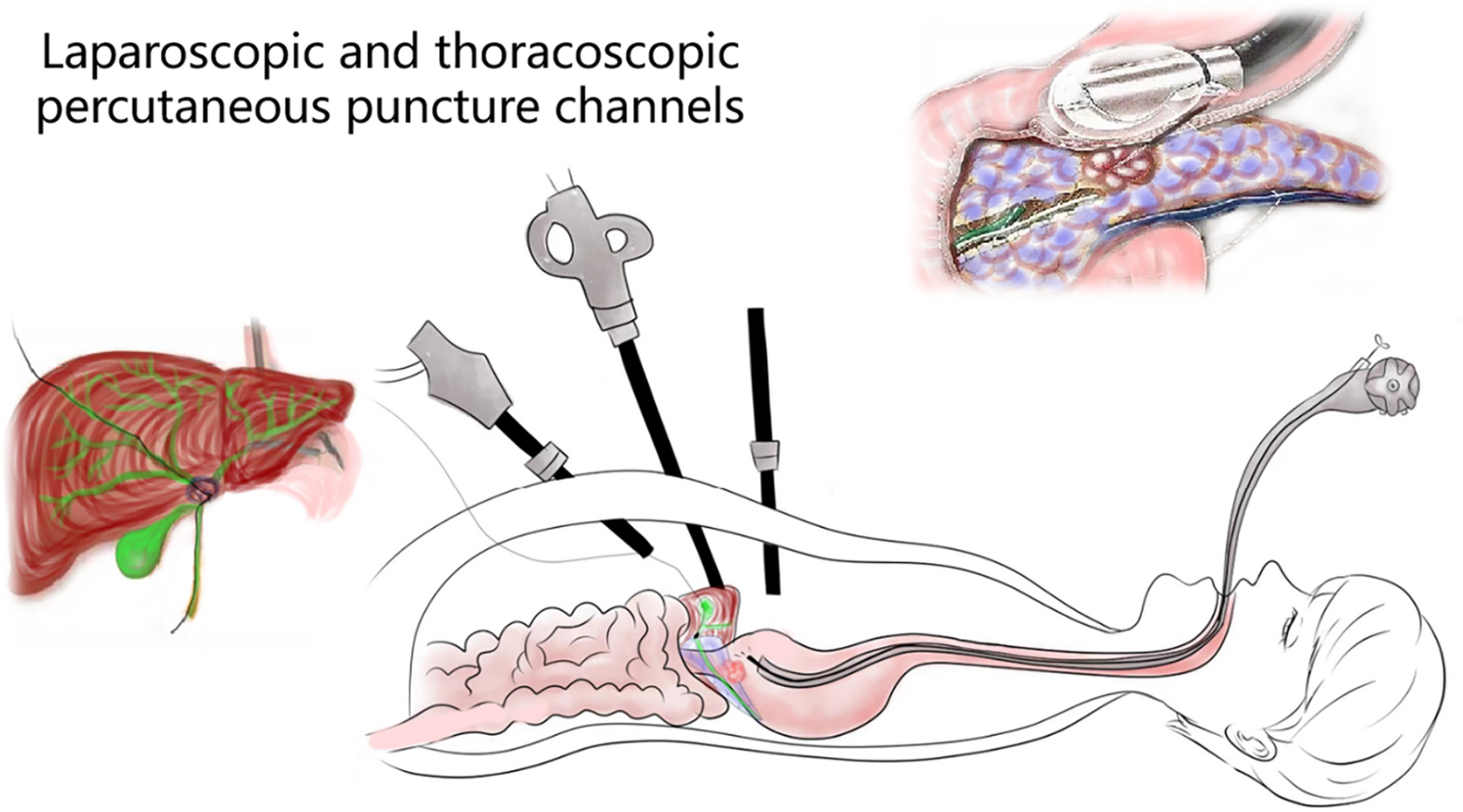

FIG. 2.3 — SMIS through a puncture channel.
1. Digestive organ puncture stent channel: It establishes convenient pathways for various organs of the digestive system through stent. The organs of the digestive system communicate through the main passage of the digestive tract, the bile duct, the pancreatic duct, the appendix tube and other branch channels to ensure the normal operation of water, food, digestive fluids, and so on. When situations like tumors, inflammations, stones occur, they cause a certain degree of obstruction. At this stage, it is necessary to establish appropriate channels for the drainage of various organs of the digestive system through the stent, and even to intervene surgically. The anatomical proximity of the digestive organs means that the establishment of a convenient channel through the stent is a prerequisite.
The development of bilateral lumen attached metal stents (LAMS) is the most remarkable, with low drainage leakage rate and easy removal. By using endoscopic ultrasound to release the puncture, LAMS makes the lumen close to the wall, so as to drain, relieve the obstruction, or even allow intervention. In 2014, Teoh et al. first reported to have used a new device, the electrically cauterized light-assisted metal stent (CE-LAMS), to perform EUSGBD. This new stent can perform the puncture, expansion and release of the stent in a single operation without changing the guide wire, which reduces the number of exchanges and the operation time, whereas no X-ray assistance is required, which presents obvious advantages.
(1) Pancreatic channel. The gastro-pancreatic channel is a duct formed by endoscopic ultrasound to puncture pancreatic lesions in the gastrointestinal cavity, which is mainly used for the treatment of pancreatic pseudocysts and encapsulated pancreatic necrosis, etc. (figure 2.3). The establishment of continuous drainage of the gastro-pancreatic channel with LAMS has been recommended by international experts as the standard method for endoscopic debridement of the drainage of encapsulated necrosis. A study evaluated the safety and effectiveness of 20-mm LAMS drainage of peripancreatic effusion. The technical success rate was 100%, the clinical success rate was 93.9%, and serious adverse events occurred in 8.16% of cases, indicating that the technique for treating peripancreatic effusion with 20-mm LAMS, which has a high clinical success rate, has good prospects for application.
(2) Gastro-gallbladder, duodenal-gallbladder, jejunum-gallbladder channels. These three channels are used for gallbladder puncture by endoscopic ultrasound in the gastrointestinal cavity, duodenal lumen, and jejunal lumen, which are mainly utilized to treat diseases like acute cholecystitis, gallstones, gallbladder polyps, etc. Kwan et al. were the first to report EUS-guided gallbladder puncture, bile extraction, and the deployment of a double pigtail plastic stent in 2007. The plastic stents or self-expanding metallic stents were mainly mentioned in subsequent reports, but these two stents were at high risk of displacement and bile leakage.
Currently there is little data on the advantages and disadvantages of the three channels. Jain et al. reported patients who had undergone gallbladder puncture drainage by endoscopic ultrasonography (EUS–GBD), of whom 59.8% had duodenal drainage, 39.7% had gastric drainage, and 0.5% had jejunal drainage. The duodenum is retroperitoneally close to the gallbladder and is less peristaltic than the stomach, so the duodenal-gallbladder channel is more stable and is the routine choice for EUS–GBD. However, if subsequent interventional therapy is required, the gastro-gallbladder channel is chosen because it is easier to seal the gastric laceration under endoscopy.
A study of 373 acute cholecystitis patients who had gallbladder drainage, among whom 146 had percutaneous transhepatic gallbladder drainage, 124 had T-tube drainage, 102 had EUS–GBD, showed success rates of gallbladder drainage of 98%, 88%, and 94% respectively, whereas the clinical symptom alleviation rates were 97%, 90%, and 80%, respectively, the complication occurrence rates were 20%, 2%, and 5%, respectively. Therefore, EUS–GBD can become the effective and safe alternative plan for percutaneous drainage and T-tube drainage. Another study evaluated endoscopic cholecystectomy and lithotomy. But considering the complications and recurrence of calculus, this method still needs further research to be clinically recommended.
(3) Gastro-jejunal channel. The gastro-jejunal channel is a stent channel formed through the gastral cavity to puncture the jejunal lumen under endoscopic ultrasound. Generally referred to as EUS-guided gastroenterostomy (EUS-GE) and used to treat gastric outflow tract obstruction (GOO) with obvious symptoms, afferent loop syndrome, etc., EUS-GE can be divided into the direct method and the balloon-assisted method according to the method of filling the enteric cavity.
A study of 35 GOO patients for whom a gastro-jejunal channel was used for drainage showed a technical success rate of 80.0%, an adverse event rate of 14.3%, and a clinical alleviation rate of 74.3%. The success of the technique depends on the distance of the two lumens connected by the LAMS, as well as on the experience of the endoscopist. Another study evidenced that EUS-GE could treat 83.3% patients with GOO, and that the LAMS needs to remain for 8.5 months at its original location. The GOO recurrence rate was low after the LAMS was removed.
(4) Duodenal-common bile duct channel. The duodenal-common bile duct channel is the channel formed through the duodenal lumen to puncture the common bile duct, and place it in the stent under endoscopic ultrasound, in order to allow the drainage of bile into the duodenum, which can be used to solve the bile drainage in patients in whom ERCP has failed. This is known as EUS-guided common bile duct duodenostomy. A meta-analysis included seven researches on a total of 284 patients. Neostomy was performed using the duodenal-common bile duct channel, with a combined technical success rate of 95.7%, a clinical success rate of 95.9%, an adverse event occurrence rate of 5.2%, and a jaundice recurrence rate of 8.7%; in addition, there has also been five similar studies of LAMS with cauterization, with a combined technical success rate of 93.8%, a clinical success rate of 95.9%, an adverse event occurrence rate of 5.6%, and a jaundice recurrence rate of 11.3%, showing that LAMS can be used to resolve biliary decompression after ERCP failure. But further research is needed for establish it as the main method of resolving biliary obstruction.
2. Percutaneous puncture choledochoscopic channel. The percutaneous puncture choledochoscopic channel allows for a complete diagnosis and treatment by percutaneous puncture choledochoscope to penetrate the biliary system. The PTCS is the channel established by percutaneous transhepatic cholangial drainage (PTCD), to dilate the channel, and finally enter the biliary tract. PTCS can be chosen for patients with biliary stone or obstruction of the biliary tract who cannot tolerate surgery or ERCP.
The PTCS technique includes two methods (staged sinus dilation and stage-I sinus dilation). Staged sinus dilation is the establishment of a channel through multiple expansions using drainage tubes in PTCD. It needs several weeks, is safe and easy to be promoted. Stage-I sinus dilation consists in directly dilating the sinus to the target diameter after the PTCD operation, which saves time, but promotes bile leakage and hemorrhage. So far, PTCS can be combined with laser lithotripsy for the treatment of intrahepatic bile duct stones, such as diffuse intrahepatic bile duct stones, grade-III and grade-IV intrahepatic bile duct stones. In 2016, a study of 118 patients with intrahepatic bile duct stone, 67 of whom had undergone PTCSL, 51 of whom had undergone routine PTCS, evaluated percutaneous transhepatic choledochoscopy lithotripsy (PTCSL) and traditional PTCS. The results showed that the stone clearance rate of the PTCSL group was significantly improved, with a low recurrence rate, and that the postoperative hospitalization time was significantly shortened.
3. Video of a classic case
(1) Rendezvous-assisted endoscopic retrograde pancreatography in a patient with annular pancreas and coexisting pancreas divisum (video 2.15 (See online resources)).
(2) Novel treatment of pancreatic cystic neoplasms: EUS-guided radiofrequency ablation combined with lauromacrogol ablation (video 2.16 (See online resources)).
(3) Spyglass discovery of a mucinous cystic neoplasm by introducing an optical fiber into the cyst through a 19G needle during EUS examination (video 2.17 (See online resources)).
(IV) Approach via multiple-cavity channel
This refers to the operation that is performed in the lacuna and is carried out in accordance with super-minimally-invasive requirements through more than two channels, and with more than two endoscopes. For example, duodenoscope combined with laparoscopic surgery, gastroscopy combined with thoracoscopic surgery, etc. Let us take the example of the SMIS of the multilacunar dual channel of the digestive system (figure 2.4).
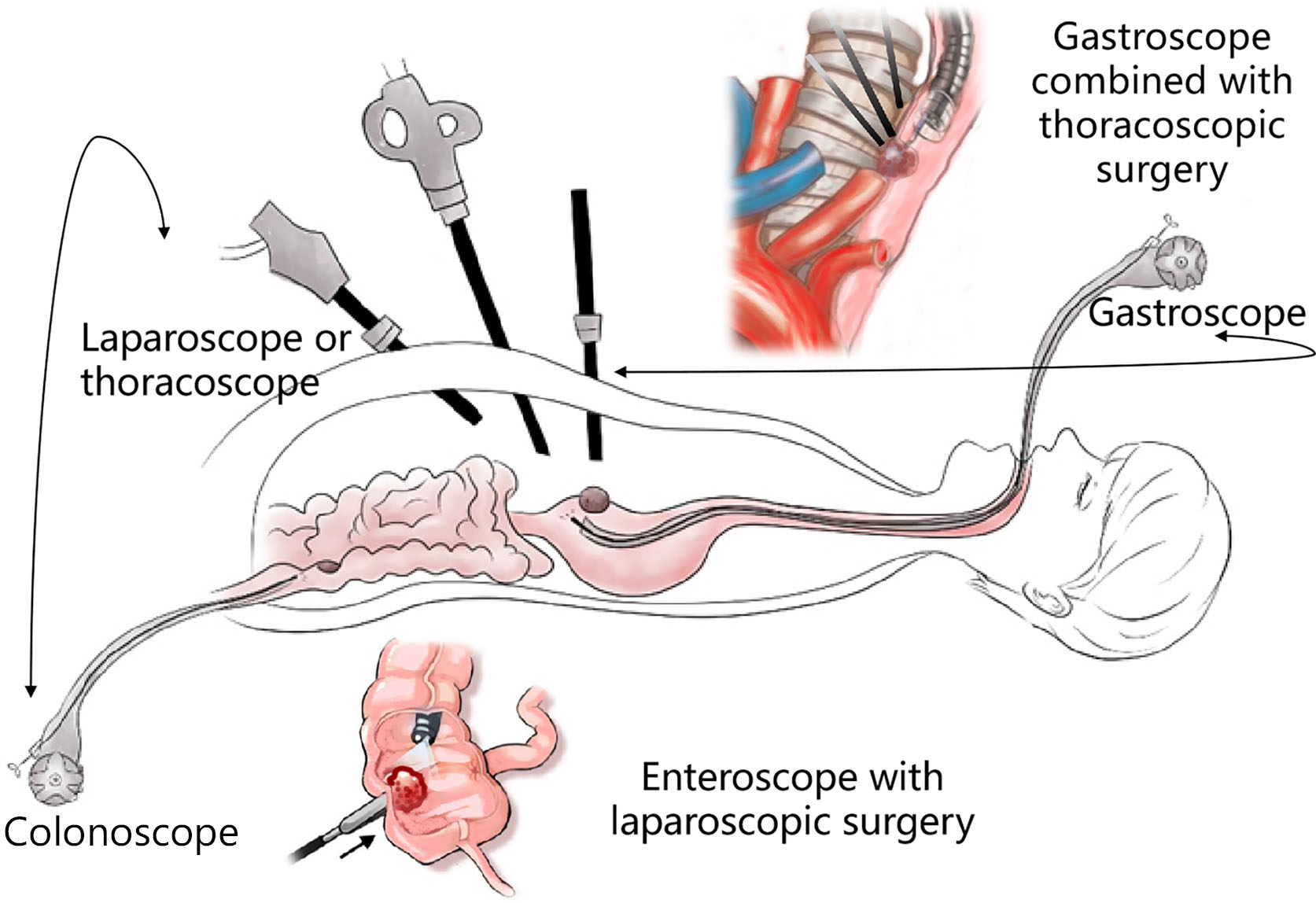

FIG. 2.4 — SMIS through a multiple-cavity channel.
1. Gastrointesinal endoscopy combined with thoracoscopy channel. Gastrointestinal endoscopy combined with the use of a thoracoscopic channel is a complete surgery through the digestive lumen and the chest cavity using an endoscope and a thoracoscope. Benign esophageal tumors can generally be resected directly by endoscopy. But for giant benign esophageal tumors, as when the tumors have begun to compress the esophagus and the narrow esophageal lumen is narrow, with small working space, and also for many tumors that are located at the mediastinum site with abundant vessels and nerves, or when the endoscope is in the esophagus, there is a high risk of damaging the great vessels outside of the esophagus when tumors are resected from the inside to the outside. Gastrointestinal endoscopy combined with thoracoscopy integrates the characteristics of the endoscope and the thoracoscope, to utilize the respective benefits of the two endoscopes to complete the operation through joint cooperation. Firstly, the gastroscope establishes a submucosal tunnel inside the esophagus to remove the tumor from the esophageal mucosa, and then the tumor is taken out of the body after it is resected by the thoracoscope from the lateral esophagus. This gastrointestinal endoscopic and thoracoscopic combined channel surgery greatly reduces surgical risks and shortens operation time. The most important is that the integrity of the organs is preserved during the process of lesion excision. There have been cases in which the above gastrointestinal endoscopic/thoracoscopic surgery technique was used to safely remove 6-cm benign tumors in the lower esophagus. The submucosal tunnel established by endoscopy increases tumor activity, minimizes the absence of muscle layers and the risk of esophageal mucosal injury, lowers esophageal stenosis and the rate of local infection.
2. Gastrointestinal endoscopy combined with the laparoscopic channel. Gastrointestinal endoscopic combined with the laparoscopic channel is a complete surgery through the digestive lumen and the abdominal cavity by using an endoscope and a laparoscope. The gastrointestinal endoscope is a soft endoscope that is operated in the gastrointestinal cavity, the laparoscope is a rigid endoscope that is operated outside the gastrointestinal cavity. The two in cooperation can clearly find the lesion’s location, determine the lesion’s size, shape, growing model and its distance to the important anatomical structures (e.g., cardia, pylorus, etc.). On that basis, the surgeon can prepare personal, accurate excision plans aiming at removing the lesion. Such dual endoscopic combined mode is generally used for the precise removal of giant benign tumors in the stomach and the intestines, while preserving the organs’ integrity. Traditional esophageal–gastric anastomosis not only presents a risk of significant injury, but the surgeon must also judge the incisive edge for a “blind excision” based on the feel of his hand, which means that the cardia that was originally preserved can be resected due to inaccurate positioning. In this way, it expands the scope of surgery, and the postoperative gastroesophageal reflux seriously reduces the patient’s quality of life. A study of 20 patients having undergone resection of the gastric submucous mass by combined gastroscopy and laparoscopy has shown that the operations were all successful, and without complications, confirming that the combined operation of gastroscopy and laparoscopy is safe, simple and beneficial for the resection of gastric submucosal tumors; nevertheless, contamination of the gastric fluid should be avoided.
3. Video of a classic case. Flexible choledochoscopy via the cystic duct combined with laparoscopic cholecystectomy for the treatment of cholelithiasis (video 2.18 (See online resources)).
2.3 The Techniques of Super Minimally Invasive Surgery
(I) Operation method per natural channel
1. Non-full-thickness excision method. It refers to a super minimally invasive treatment method of lesion resection through natural channel surgery, where any layer or multiple layers of the original digestive tract wall are preserved at the beginning of the operation of the digestive tract mucosal layer. The wall of the digestive tract in the non-full-thickness excision method has a layered structure, comprising the mucosal layer (mucosal epithelium, mucosal lamina propria and mucosal musculature), the submucosa, the muscularis propria and the outer membrane. Such a method of non-full-thickness excision is valid in the diagnosis and treatment of early tumors of the digestive tract, polyps, and other superficial gastrointestinal tumors.
The non-full-thickness excision are as follows.
(1) Endoscopic mucosal resection (EMR) (figure 2.5). First, the precise boundaries of the lesion are accurately determined under endoscopy and marked. Next, submucosal injection or other methods are used to separate the submucosa from the muscularis propria. Subsequently, the total or partial mucosal lesions are removed with a snare, and the diseased tissue is collected. Finally, the necessary treatment techniques are applied to the surface of the wound.
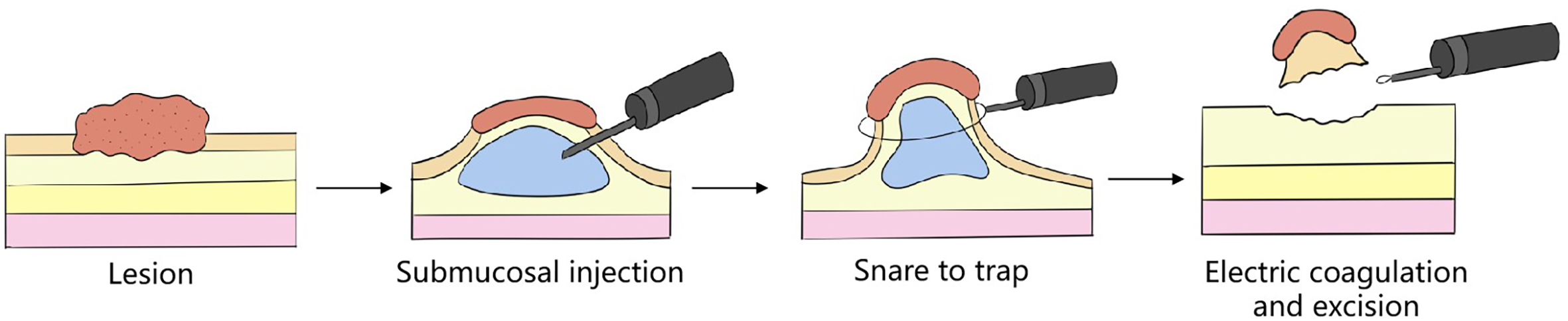

FIG. 2.5 — Operating steps of SMIS–EMR.
The invention of EMR is a multiphase process, with no single inventor. In 1984, A. Nakajima in Japan and his team first proposed and tried endoscopic mucosal resection to cure early gastric cancer. This is the prototype of the EMR concept. With the continuous improvement of endoscopic equipment and techniques, EMR became widespread in Japan in 1995, particularly for the treatment of early gastric cancer. From the end of the 1990s and into the early 2000s, it spread rapidly throughout the world.
(2) Precutting endoscopic mucosal resection (precutting-EMR) (figure 2.6). First, the boundaries of the lesion are accurately determined under the endoscope and marked. Submucosal injections of substances such as normal saline solution or methylene blue, etc., are then administered in the area of the lesion to achieve full lift of the lesion. After that, the mucosa is incised circumferentially around the lesion using an electric knife or the tip of the snare. In this step, the submucosal layer does not need to be separated. Subsequently, the lesion was loop-ligatured directly with a snare, resected and collected. Finally, after excision, the wound is treated accordingly. Precutting-EMR has been validated as a technique by The Japan Gastroenterological Endoscopy Society.
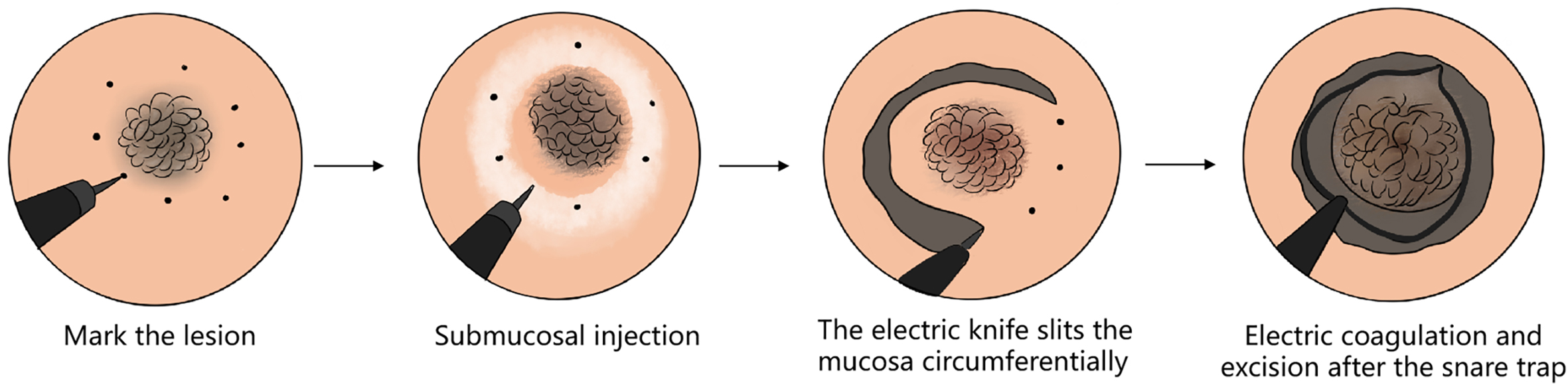

FIG. 2.6 — Operating steps of SMIS-precutting-EMR.
(3) Piecemeal endoscopic mucosal resection (piecemeal-EMR) (figure 2.7). First, the lesion is carefully observed using an endoscope to determine the part that cannot be completely removed at one time only. Next, the boundaries of the lesion are determined and marked around the lesion for accuracy and to enable visualization in subsequent operations. After that, a submucosal injection is performed around the marked lesion to lift up the tissue in the lesion so that it can be easily operated and removed. Then, starting from the lesion’s side, piecemeal snare resection is performed subsequently. The lesion is gradually dissected by removal of the block of tissue surrounding it, while ensuring that the dissected tissues are recycled for subsequent pathological examinations and analyses. Finally, after completing lesion resection, appropriate treatment is carried out on the surface of wound, which can include steps like hemostasis, cleaning and sealing, etc., to ensure the wound’s healing and recovery.
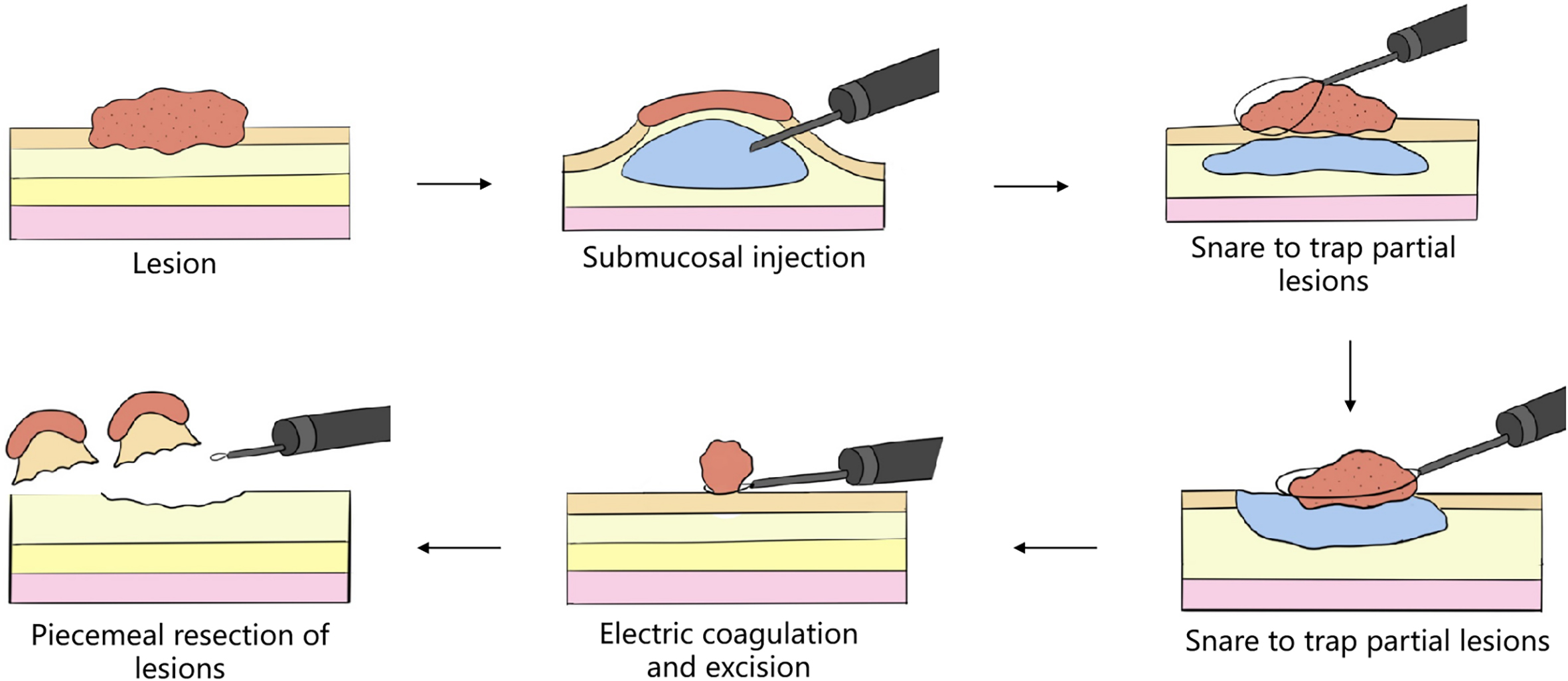

FIG. 2.7 — Operating steps of SMIS piecemeal EMR.
At first, piecemeal EMR was proposed by Japanese endoscope specialist Hisao Oyama in the 1990s. He and his team developed the technique of lesion piecemeal dissection by training on large or complicated lesions. This technique was initially proposed to address the limitation of traditional EMR, which cannot totally dissect the major lesions, and has been gradually promoted and applied to the treatment a wider range of gastrointestinal early tumors.
(4) Cap-assisted endoscopic mucosal resection (cap-assisted EMR, EMRC for short) (figure 2.8). First, the lesion is carefully observed using endoscopy to determine and mark the boundaries of the lesion. After that, submucosal injection is administered in the area of the lesion to fully lift it. Then, the snare is prepositioned inside the cap, to attract the lesion into the cap. After that, the lesion is removed with a snare and collected. Finally, the wound is treated with hemostasis and sealed, etc. At first, EMRC was proposed by Hiroshi Inoue, a Japanese endoscopist, to improve EMR for the treatment of esophageal cancer.
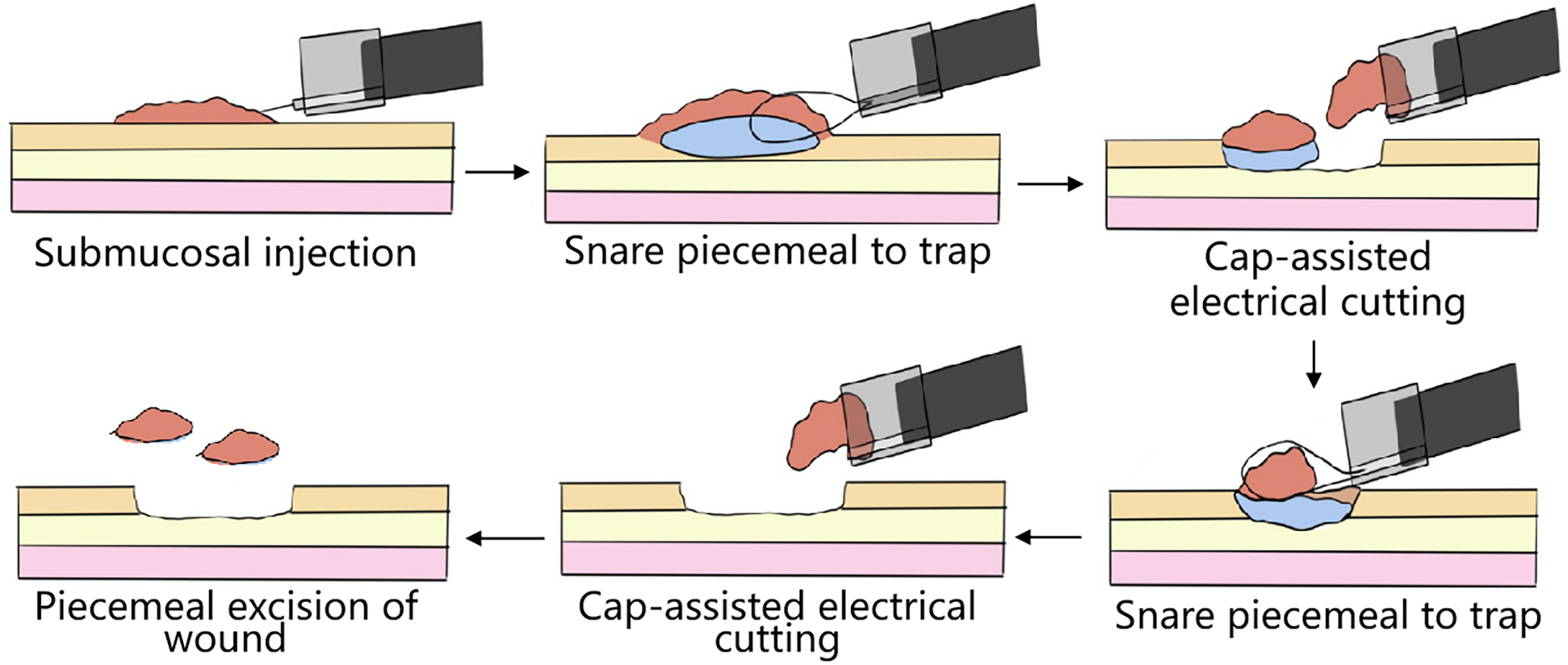

FIG. 2.8 — Operating steps of SMIS-cap-assisted EMR.
(5) Ligation-assisted endoscopic mucosal resection (ligation-assisted EMR) (figure 2.9). The operating steps are as follows. First, the shape and extent of the lesion are determined under endoscopy, and its boundaries are marked using an electric knife or argon gas. After that, a mixture of normal saline solution and methylene blue is injected into the submucosa of the lesion area in order to fully lift it. Subsequently, the lesion to be ligated is directly attracted with a ligation device containing a ligation ring, and the red color of the endoscopic visual field indicates that the lesion is fully attracted in the cap. Then the lesion is removed with a snare at the bottom of the ligation ring and tissue samples are collected. Finally, the surgeon observes the wound to detect any bleeding and perforation, in order to determine the appropriate technology to treat it. At first, ligation-assisted EMR was proposed by M.A. Soehendra and S.L.S.J.D. Haese. This technique, which was firstly introduced in 1993, has been used since then to improve the effect of endoscopic mucosal resection.
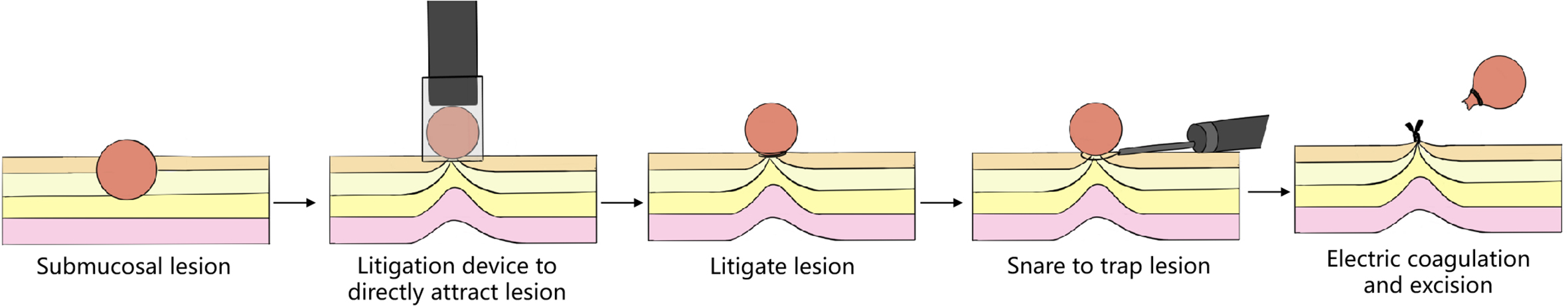

FIG. 2.9 — Operating steps of SMIS ligation-assisted EMR.
(6) Multi band mucosa resection (MBM) (figure 2.10). First, the lesion boundary is determined and marked endoscopically. After that, mucosa ligation is carried out using a specially designed multi-ring ligation device. After that, the mucosa is removed with a snare. Note that each lesion attracted is removed by the snare, and the operation of attraction-ligation-excision is repeated several times to achieve piecemeal resection and recover of the whole lesion. Finally, the wounds are treated.
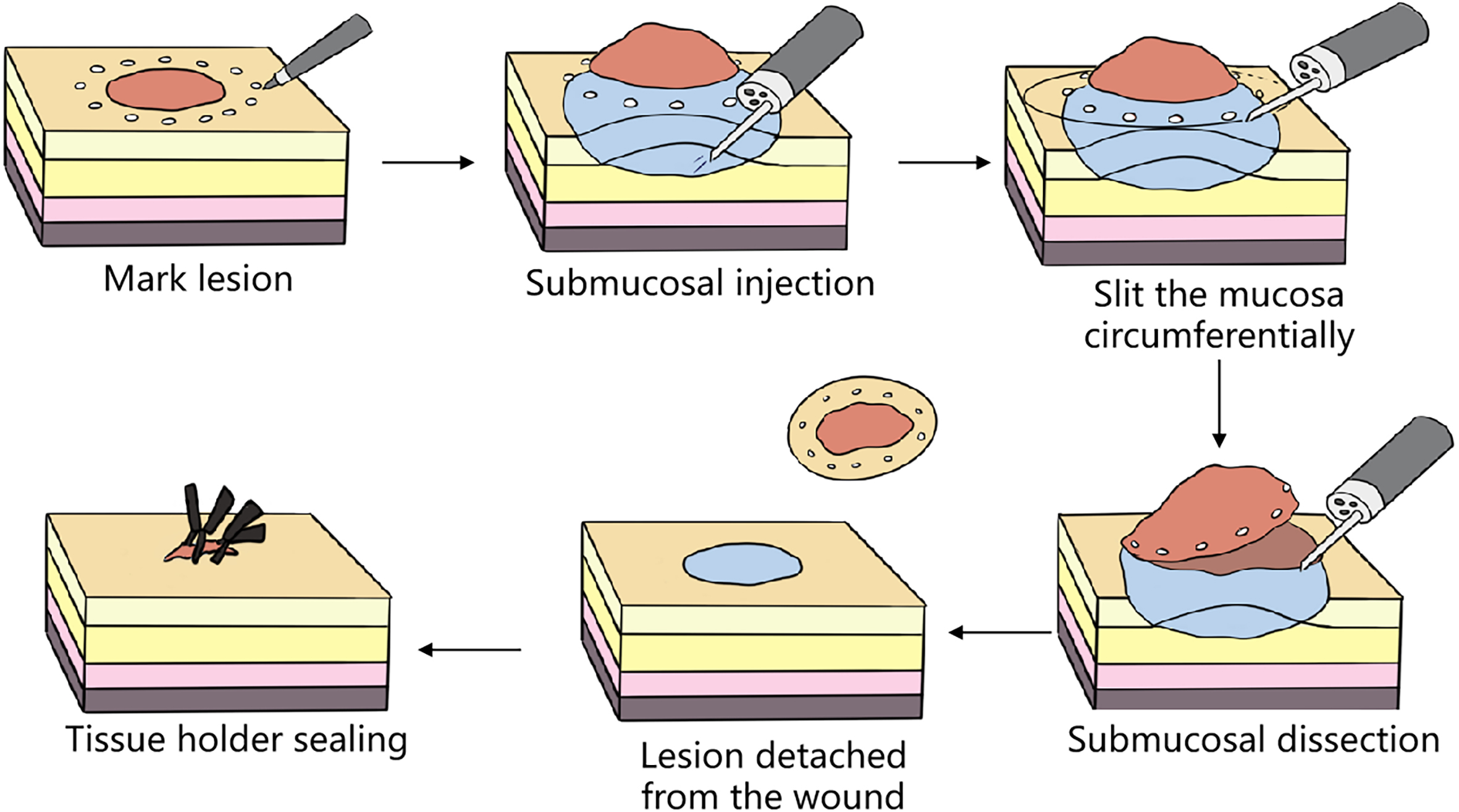

FIG. 2.10 — Operating steps of SMIS–ESD.
(7) Endoscopic submucosal dissection (ESD) (figure 2.10). First, after the shape and scope of the lesion have been determined under endoscopy, the boundary of the lesion is circumferentially marked. After that, submucosal injection is carried out to fully lift the lesion. Subsequently, the tissues between the muscularis mucosa and muscularis propria of the lesion are gradually dissected using an electric knife to completely dissect the lesion’s mucosa and submucosa. Finally, the wound is treated after collection of specimens of pathological tissues.
In 1993, the Japanese physician Hiroshi Watanabe and Noboru Oda first proposed to apply submucosal dissection to the treatment of gastric cancer. This technique was at first used for addressing the limitation of EMR in the treatment of relatively major lesions. In 1999, Hiroshi Watanabe and Yoshinobu Watanabe et al. further developed this technique by improving the operating procedures and technical details, making ESD a reliable endoscopic treatment method, especially for the treatment of early gastric cancer. In the early 2000s, ESD technology was gradually promoted and applied in Japan and other countries. Research has shown that ESD can effectively improve the resection rate of early gastric cancer and reduce the recurrence risk.
(8) Endoscopic submucosal dissection with traction (ESD with traction) (figure 2.11). Marking and submucosal injection are performed in the same way as in the case of submucosal dissection. After that, partial lesions are dissected by the pull of an external force, which makes it possible to completely separate the mucosal layer of the lesion from the muscularis propria to obtain a better surgical field of view. Subsequently, the tissue between the muscularis mucosa of the lesion and the muscularis propria is gradually separated using an electric knife until complete dissection. Finally, the lesion is collected and the wound is treated.
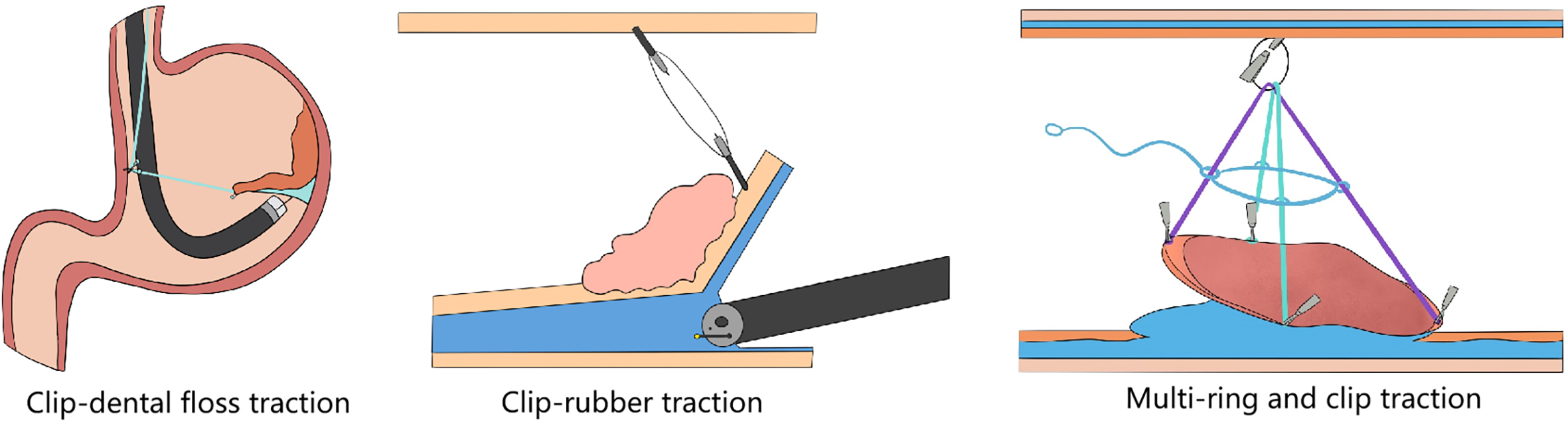

FIG. 2.11 — Operating steps of the SMIS–ESD technique with traction.
(9) Endoscopic submucosal excavation (ESE). This is a new type of super minimally invasive gastrointestinal surgery studied and explored on the basis of endoscopic submucosal dissection (ESD). Chinese researcher Zhou Pinghong was the first to name this technique ESE. It means that, under endoscopy, an electric knife is used to incise the mucosa of the lesion’s surface to fully expose it, and finely and completely dissect the junction between it and the muscularis propria; finally, the wound is suture with titanium clips, and so on. This is a new method for the treatment of gastrointestinal mucosal tumors in China and abroad.
2. Full-thickness excision method. A technique in which the boundaries of the lesion are determined endoscopically, marking, and submucosal injection to completely lift the lesion. The entire local layer (including the mucosa, muscularis mucosa, submucosa, muscularis propria, and/or serosa) is excised using an electric knife, causing iatrogenic active perforation and damage to the wall of the tube, then the lesion is collected, and finally the perforated wound is sealed. The implementation of full-thickness resection creates the conditions for a wider indication of super minimally invasive surgery.
The full-thickness resection methods are usually as follows.
(1) Endoscopic full-thickness resection (EFTR) (figure 2.12). This method allows for the complete resection of lesions originating from the wall of the digestive tract, especially those located deep in the muscularis propria, the tumor must be resected along with the entire wall of the digestive tract. Initially, Thomas Rösch and Klaus W. D. R. W. described the method of using endoscopic techniques for full-thickness resection. This technique is mainly used for the endoscopic treatment of gastrointestinal tumors, especially lesions that are difficult to dissect using traditional methods.
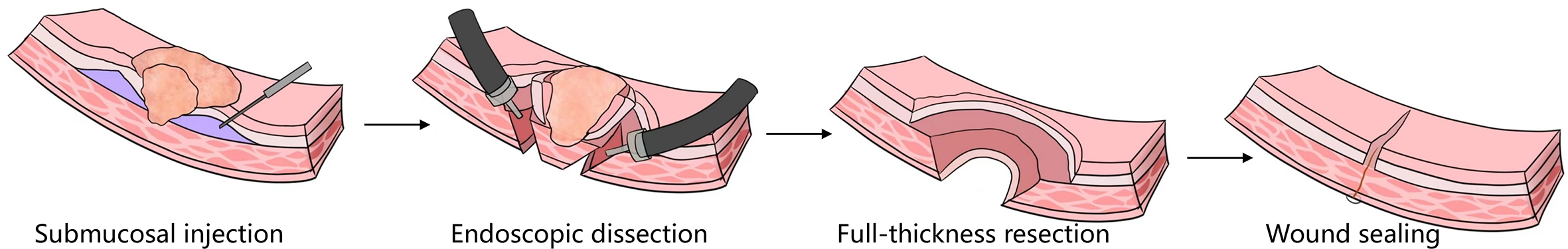

FIG. 2.12 — Operating steps of the SMIS–EFTR technique.
(2) Hybrid ESD and FTR methods (figure 2.13). The operating process is as follows. First, the boundaries of the lesion are clearly observed under the endoscope, indigo carmine is sprayed, and the first circle is marked with argon gas 0.5 cm away from the lesion; Mark the second circle is marked with argon gas at a distance of 1 cm from the lesion, and another point is marked on the oral side. Next, after submucosal injection, the edge of the lesion is lifted, the marked outer mucosa is circumferentially incised, and part of the submucosal is removed. Then, two sets of tissue clips and dental floss traction are applied to make the boundary between mucosa and muscularis propria clear, and a submucosal injection is carried out while the submucosal dissection is carried out up to the inner ring mark. Subsequently, the traction line is pulled up together with the muscularis propria, and tissue clips are applied to locate and seal the marked inner-ring muscularis propria, then full-thickness resection is performed; after active perforation, tissue clips are applied to seal the perforation, while full-thickness resection is carried out until the lesion of the inner circle is completely detached from the wound. Finally, the wound is carefully healed, tissue clips are added to seal the wound strictly, and biological protein glue is sprayed to cover the wound again.
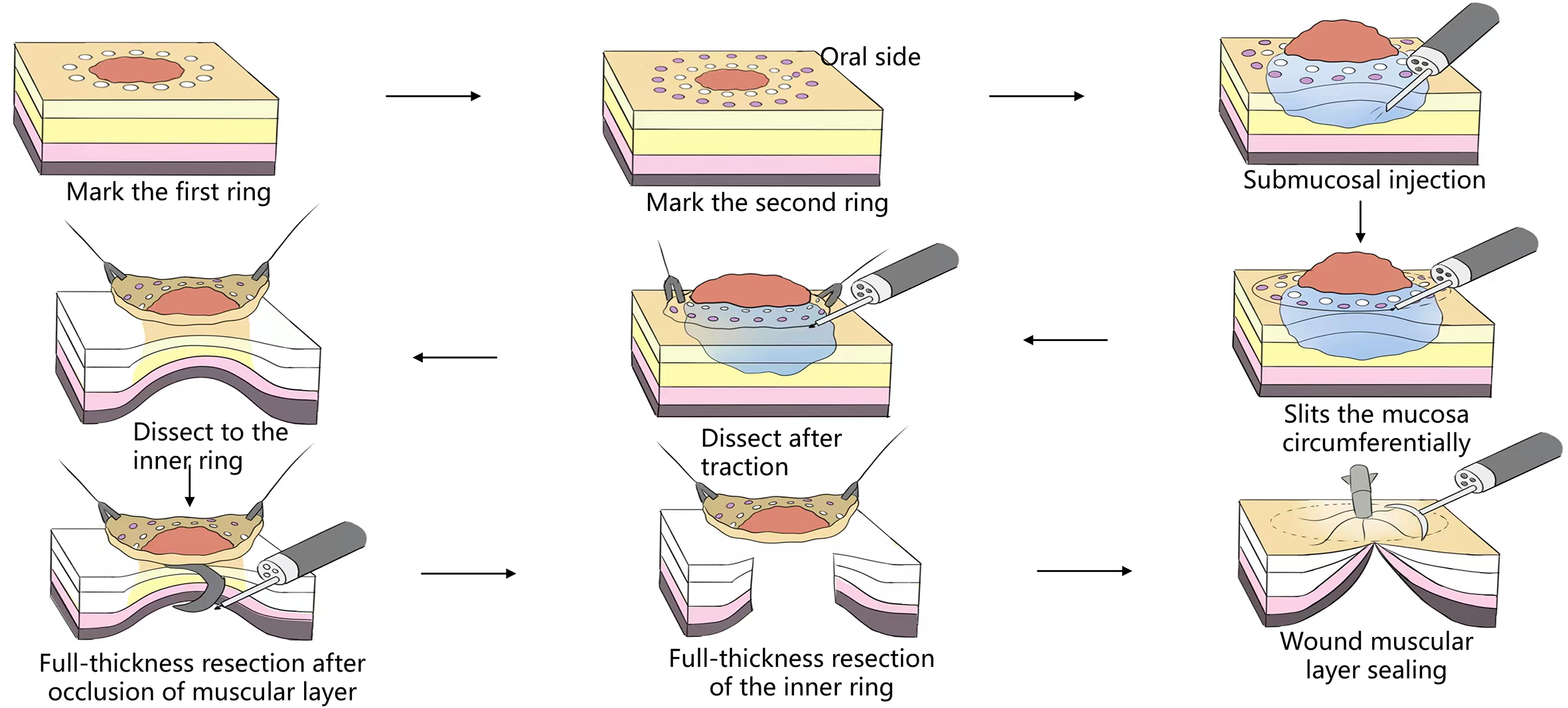

FIG. 2.13 — Operating steps of the hybrid ESD and FTR technique.
(3) Drainage method. For diseases requiring surgery or diseases for which the effects of long-term drug therapy are unsatisfactory, endoscopic gastrointestinal drainage is performed in order to preserve the structural integrity of human organs. This consists in draining food residues, digestive juice or pus out of the body through the natural channel of the digestive tract, so as to treat diseases or relieve symptoms.
The common drainage methods include the following steps.
(a) Super minimally invasive drainage for infection of the digestive tract with one side blind. This is a surgical procedure that aims to cure a blind end of the digestive tract infection by draining it while preserving the integrity of the original tissue and organ anatomical structure. It includes:
Esophageal mediastinal fistula gastric tube insertion method: Under the guidance of a guide wire, a gastric tube is temporarily inserted into the mouth of the esophageal mediastinal fistula through oral endoscopy to drain pus from the mediastinum. Transanal super minimally invasive surgery for appendicitis (transanal SMIS for appendicitis), a surgery involving intubation, contrast, irrigation, and drainage of the appendix by transanal endoscopy to relieve appendiceal obstruction and treat appendicitis (figure 2.14). Currently, the effect of this treatment is basically equivalent to that of surgery.
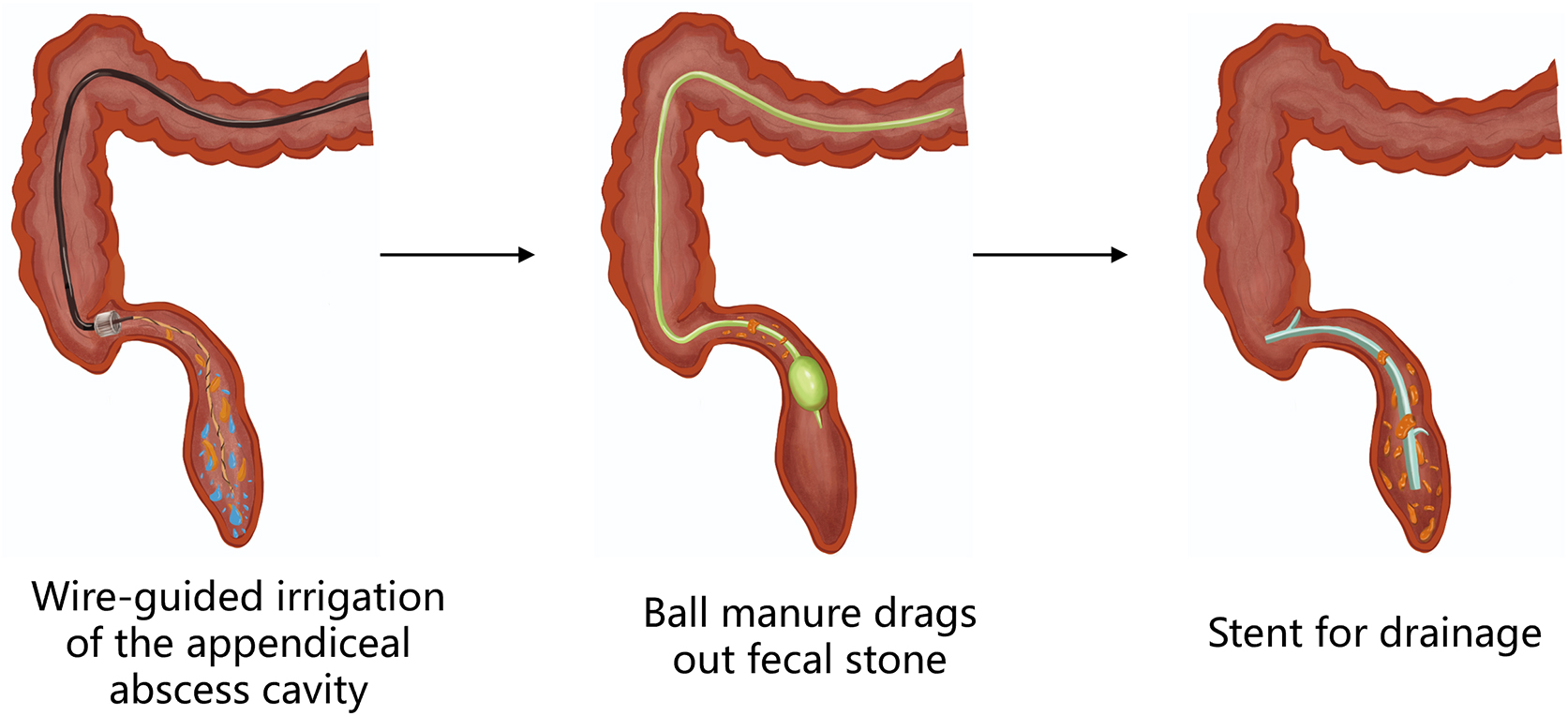

FIG. 2.14 — Operating steps of transanal super minimally invasive surgery for appendicitis.
One of the most representative examples is endoscopic retrograde appendicitis therapy (ERAT). ERAT was first reported by the Austrian team of Said et al. in 1995. Acute appendicitis (AA) was treated by endoscopy of the intubated appendix, aspiration of the pus and irrigation of the appendix lumen, etc. In 2012, Professor Liu Bingrong et al. formally proposed the ERAT concept and used it for treating uncomplicated AA. This technique allows both diagnosis and therapy, with rapid, effective, and minimally invasive treatment of appendicitis while preserving the normal function of the appendix. In addition, there is no abdominal incision cutting, which also improves the cosmetic effect after appendicitis. Researches showed that the overall treatment success rate of ERAT is higher than 95%, with a complication occurrence rate of about 3%; the longest follow-up was 33 months, the recurrence rate was 6.2%–9%, and the recurrent patients underwent appendicitis.
(b) Biliary endoscopy. The duodenoscope is inserted perorally through the natural channel at the level of the papilla, the patient is intubated into the bile duct for super minimally invasive drainage, and can be operated under choledochoscopy mostly at present. The technique mainly applies to the treatment of obstructive jaundice. It includes the following: placement of a nasobiliary drainage tube by a method consisting of placing endoscopically perorally one end of the nasobiliary drainage tube in the bile duct through the working channel endoscopic, and after the duodenoscope is removed, the other end of the drainage tube is extracted through the nasal cavity; endoscopic placement of a plastic biliary stent by a peroral endoscopic method consisting of pushing the plastic stent into the bile duct through the working channel along the guide wire, and leaving the rear end in the duodenum for drainage. The endoscopic placement of a metallic biliary stent is performed by a method consisting of endoscopically inserting a guide wire into the bile duct through the working channel and pushing the metallic stent along the guide wire to pass the metallic stent through the narrow segment for drainage.
(c) Endoscopic pancreatic drainage. This is an operating technique involving bidirectional peroral endoscopic drainage to the pancreatic duct.
According to the drainage method, it can be divided into internal stent drainage and external nasopancreatic drainage; according to the stent type, it can be divided into plastic stent placement and metal stent placement. It includes: endoscopic placement of a nasopancreatic drainage duct by a peroral endoscopic method consisting of placing one end of the nasopancreatic drainage tube in the pancreatic duct through the duodenal papilla through the working channel, whereas the other end of the drainage tube is drawn out through the nasal cavity; endoscopic placement of a plastic stent in the pancreatic duct by a peroral endoscopy method allowing placement of a plastic stent in the pancreatic duct through the duodenal papilla using the endoscopic working channel for internal drainage of pancreatic fluid.
(II) Operation via tunnel channel
Gastrointestinal endoscopic tunnel technique. This is a treatment technology in which, after endoscopic gastrointestinal submucosal injection, the mucosal layer is incised to establish the tunnel opening; then the endoscope enters the submucosal layer to dissect through the tunnel opening and establishes a tunnel located between muscularis propria and mucosa, and through the tunnel conducts to the mucosal layer and the muscularis propria, and through the muscularis propria to the outside of the digestive lumen.
A successful case obtained by the tunnel technique has been published by Professor Linghu Enqiang in 2009.
The resection of a large area of esophageal early cancer on a human body using the tunnel technique for the first time in the world was a success. After that, animal and clinical research into this technique continued, and it was used to treat achalasia, tumors of the submucosal muscularis propria, continually perfecting the tunnel technique and implementing a comprehensive system. Professor Linghu Enqiang also perfected the system and compiled his work in a book, which was published by the world-famous publisher Springer. It is the first monograph on the gastrointestinal endoscopic tunnel technique in the world, and also first monograph in English comprised of Chinese original elements in the world and edited by the scholar in China.
1. Excision method
(1) Endoscopic submucosal tunnel dissection (ESTD) (figure 2.15). First, the boundaries of the lesion are accurately identified by endoscopy and marked on its oral and anal sides, respectively. Next, a routine submucosal injection is performed to lift the mucosal layer, and an incision is made in the mucosa on the oral and anal sides of the lesion. Then the endoscope enters the submucosal to dissect through the oral side tunnel, the tunnel between the mucosal layer and the muscularis propria is established while dissecting. Subsequently, after having been dissected up to the lateral anal opening, the entire lesion is resected in the tunnel on both sides. Finally, the specimen is collected and the wound is treated. In 2009, Professor Linghu Enqiang accidentally established a submucosal tunnel from the lesion mouth to the anus during an endoscopic resection of high-grade epithelial neoplasia around the esophagus in 2009. This tunnel helped him safely and quickly remove the lesion. At the same year’s Beijing Gastrointestinal Endoscopy Annual Conference, Professor Linghu Enqiang gave a special report on the topic and named it ESTD surgery. And then he modified it in 2013, proposing single-tunnel, double-tunnel, and multi-tunnel approaches for the treatment of esophageal large area early cancer. Numerous studies have shown that ESTD has a better effect and a lower complication rate in the treatment of early gastric cancer, esophageal cancer, and other digestive diseases.
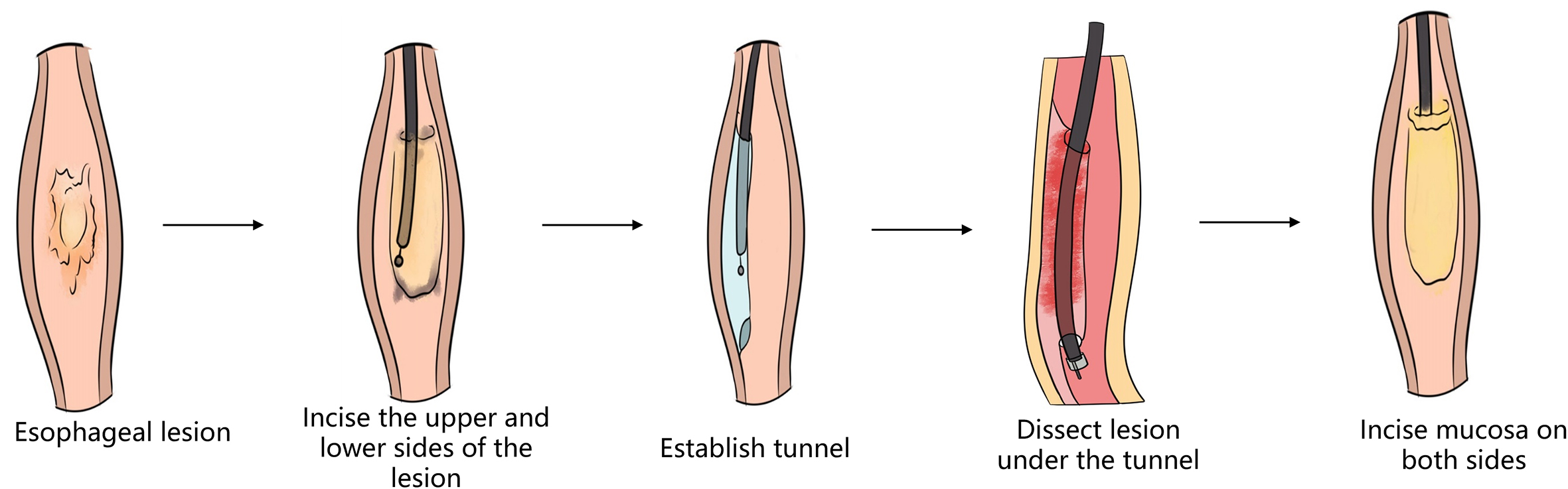

FIG. 2.15 — Operating steps of SMIS–ESTD.
1) Single tunnel endoscopic submucosal tunnel dissection: the operation steps are the same as in ESTD. Note that this method completes the excision of the lesion by creating a single tunnel.
2) Double-tunnel endoscopic submucosal tunnel dissection: the operation steps are the same as in ESTD. Note that this method completes the excision of the lesion by creating two tunnels.
3) Multi tunnel endoscopic submucosal tunnel dissection: the operation steps are the same as in ESTD. Note that this method completes the excision of the lesion by creating multiple tunnels.
(2) Tunnel endoscopic resection of the muscularis propria (ESTD of the muscularis propria). First, the lesions are detected using conventional endoscopy or endoscopic ultrasound. Next, after continuing the submucosal injection 5 cm away from one side of the lesion, an incision is made in the mucosal layer to create a tunnel-shaped opening. After that, the endoscope enters the submucosal to dissect and create a tunnel between the mucosal layer and the muscularis propria, until the tumor of the muscularis propria is found. Subsequently, the tunnel around the tumor is widened and the tumor is gradually resected and recycled. Finally, the liquid is clearly absorbed in the tunnel; then, after hemostasis of the vessels in the tunnel, the tunnel opening is sealed.
(3) Submucosal tunnel endoscopic resection (STER). The operating steps are the same as in the tunnel endoscopic resection if the muscularis propria. A tunnel opening is created, the submucosal tunnel is established, the tumor is resected, the wounds are treated and the tunnel opening is sealed (figure 2.16). In 2011, Professor Linghu Enqiang officially published a feasibility study on esophageal resection through a tunnel in the muscularis propria. Professor Xu Meidong published the submucosal tunnel endoscopic resection (STER) technique in 2012. Professor Inoue named this technique peroral endoscopic tumor resection (POET).
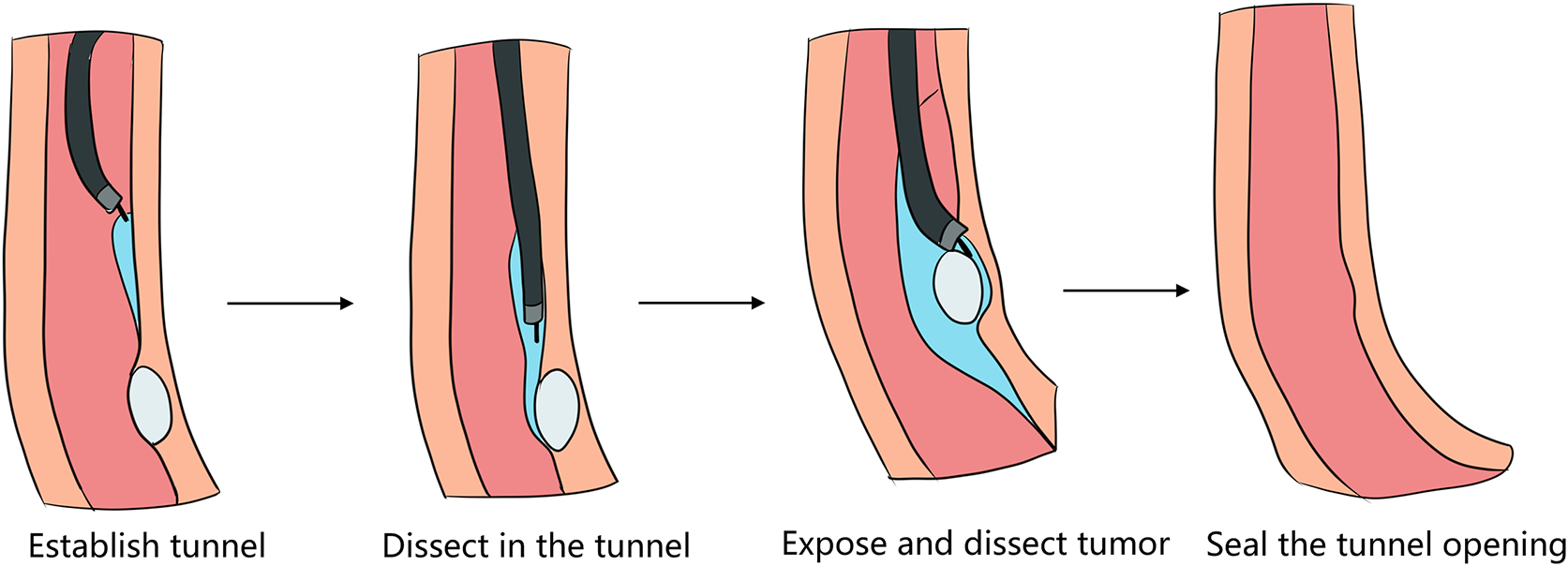

FIG. 2.16 — Operating steps of SMIS–STER.
2. Myotomy method
(1) Two options are available depending on the incision made in the muscularis site.
(a) Peroral endoscopic myotomy (POEM) (figure 2.17). After submucosal injection 7–12 cm from the cardia, the mucosal layer is incised to create the tunnel opening. The endoscope enters the submucosal layer through the tunnel opening to dissect and establishes a tunnel between the muscularis mucosa and the muscularis propria; in the tunnel, the muscularis propria of the lower esophagus and the cardia are cut with an electric knife, and the tunnel opening is sealed, then the wound is treated. The procedure combines surgical myotomy and endoscopy techniques to avoid the trauma of traditional surgery.
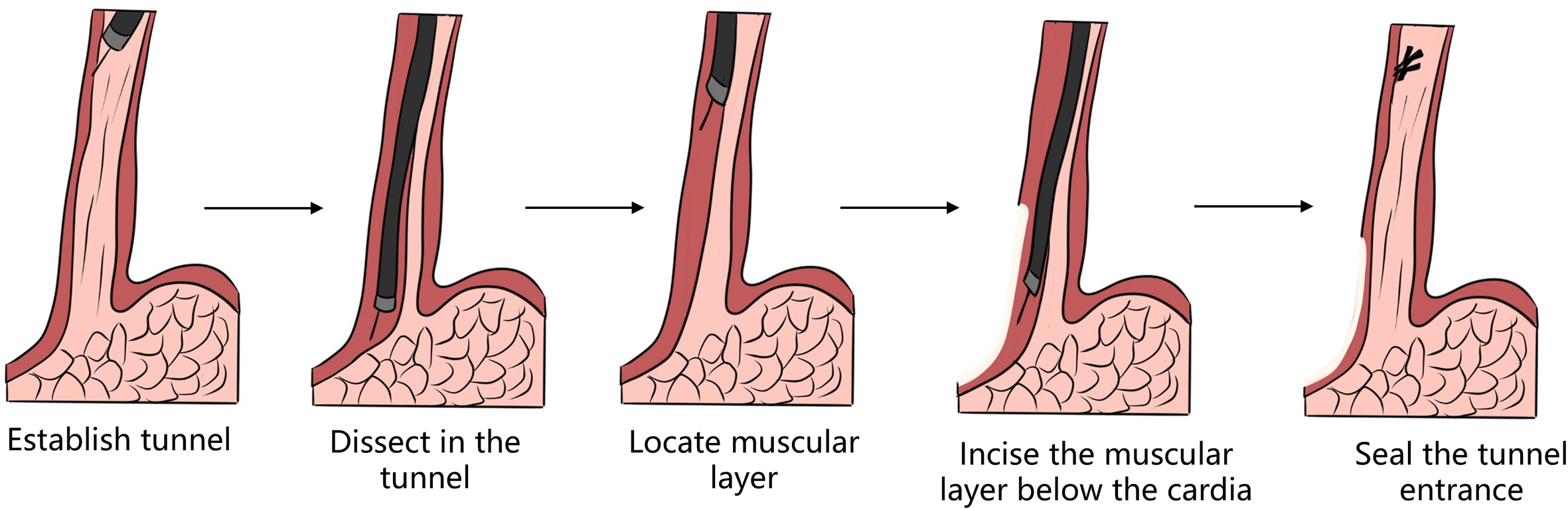

FIG. 2.17 — Operating steps of SMIS–POEM.
(b) Gastric peroral endoscopic myotomy (G-POEM) (figure 2.18). After submucosal injection at about 5 cm from the pyloric opening, the mucosal layer is incised to create the tunnel opening, then the endoscope enters the submucosal layer to dissect through the tunnel opening and establishes a tunnel until it crosses 1 cm of the pyloric canal; in the tunnel, the pyloric sphincter is incised with an electric knife, and finally the wound is treated and sealed. This technique, in which a tunnel is created at the gastric prepyloric area to perform submucosal tumor resection, was first reported in 2012 by Professor Linghu Enqiang in his book Digestive Endoscopic Tunnel Technique Therapeutics.
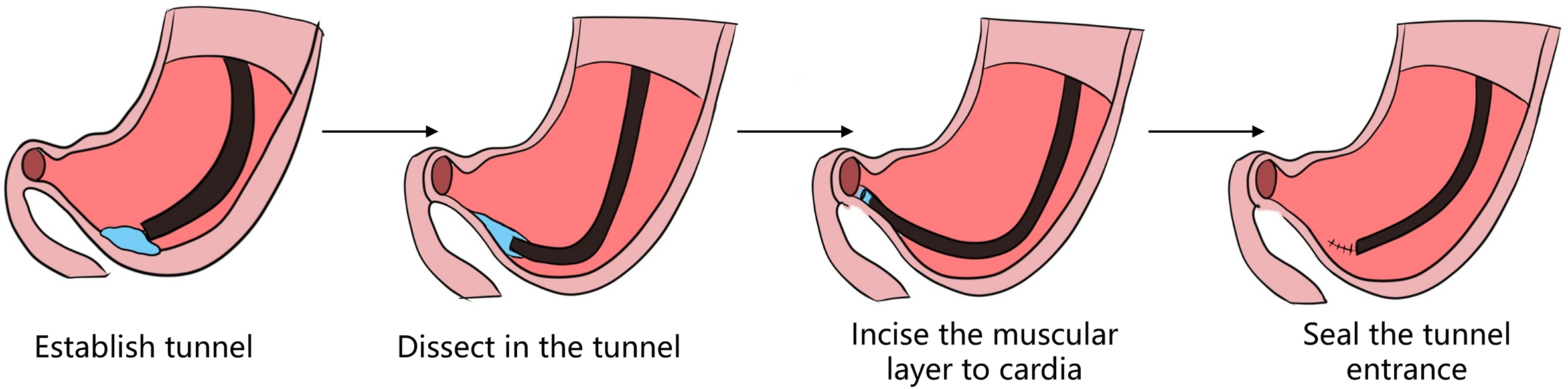

FIG. 2.18 — Operating steps of SMIS–G-POEM.
(c) Submucosal tunneling endoscopic septum division (STESD). A tunnel entrance is created 3 cm from the diverticular septum by the tunnel technique, then a short tunnel is established at the submucosal layer, the diverticulum in the tunnel is fond and completely exposed; finally, the division incision of the esophageal septum is completed. The technique was reported by Professor Zhou Pinghong in 2013. Its advantages lie in the fact that it allows the diverticular septum to be sectioned while preserving the integrity of the mucosal layer, reducing the risk of postoperative perforation, infection, and lowering the risk of complications; it allows the muscular layers between the esophageal wall and the diverticulum to be fully exposed and the muscle in the septum division to be completely and thoroughly incised (figure 2.19).
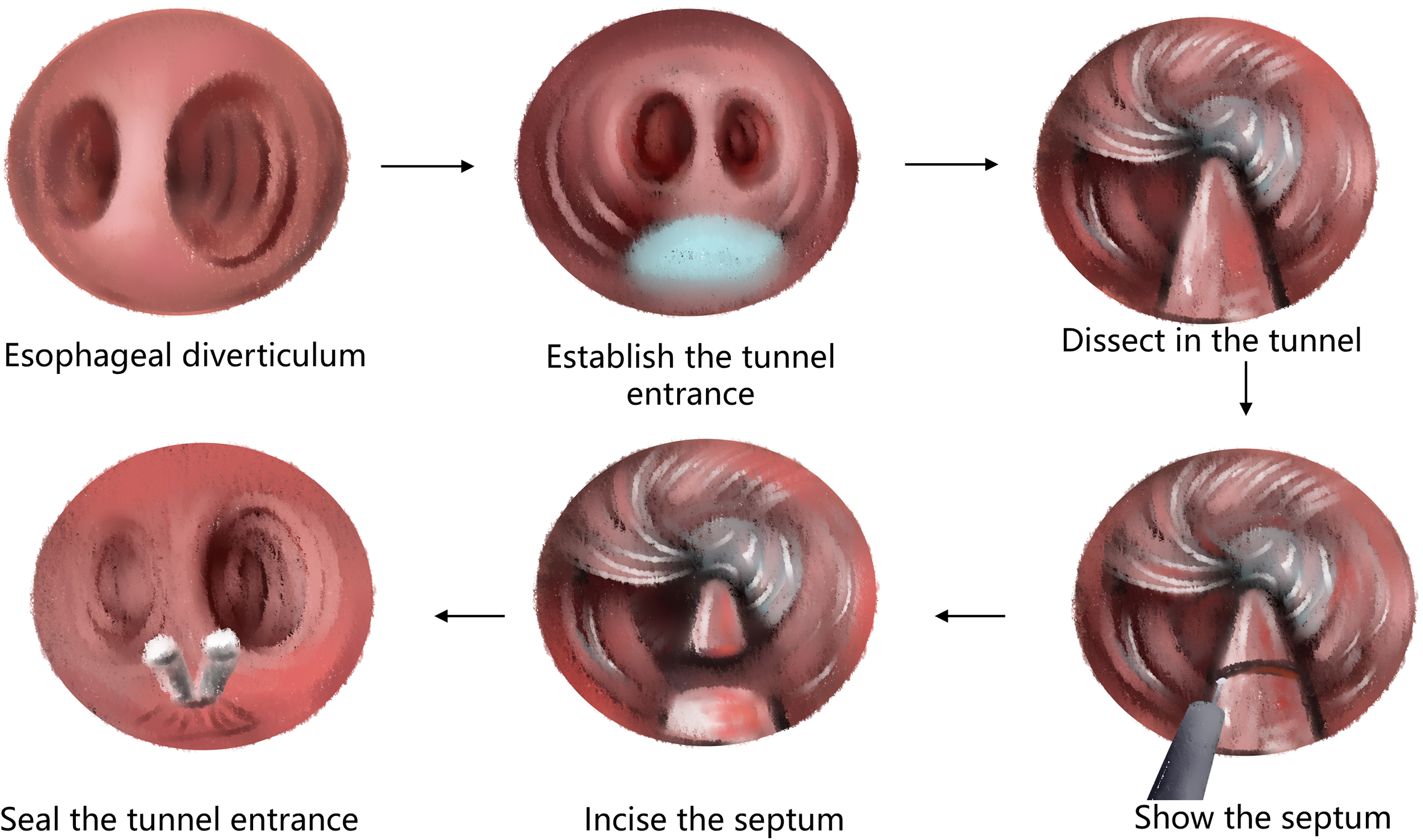

FIG. 2.19 — Operating steps of SMIS–STESD.
(d) Peroral endoscopic congenital esophageal cystotomy. It is a technique using a peroral endoscopic tunnel, in which, after finding the cyst and partially dissecting it, the wall of the cyst is directly incised to achieve the objective of draining cystic fluid, and in which the wall of the outer capsule can be broken by local injection of lauric alcohol or anhydrous alcohol.
(2) The common myotomy method is peroral endoscopic circular muscle myotomy: it is a method in which, in the tunnel, the circular muscle layer of the lower esophagus and the cardia are selectively incised with an electric knife, the longitudinal muscle layer and the outer membrane being preserved.
(a) Peroral endoscopic full-thickness myotomy. In the tunnel, the whole muscular layers of the lower esophagus and cardiac (circular muscle + longitudinal muscle layer) are incised with an electric knife.
(b) Peroral endoscopic progressive full-thickness myotomy. In the tunnel, starting from the lower esophagus, the circular muscle is incised up to the longitudinal muscle layer, from the shallow to the deep layer, with an electric knife, until the whole muscular layer of the esophagogastric junction reaches full-thickness resection.
(c) Peroral endoscopic glasses-style myotomy. In the tunnel, about 1 cm of the muscularis propria of the esophagogastric junction is preserved; the full thickness of the muscularis propria (circular muscle + longitudinal muscle layer) of the muscles on the oral and anal sides is incised using an electric knife.
(d) Peroral endoscopic circular muscle myotomy plus balloon plasty. In the tunnel, the circular muscle of the lower esophageal muscularis propria is selectively incised with an electric knife, and a dilator balloon is used in the esophageal cavity to further increase the separation distance and the depth of the muscle layer.
(e) Peroral endoscopic myotomy with simultaneous submucosal and muscle dissection (POEM–SSMD). The operating steps are as follows: first, after peroral endoscopic submucosal injection, the mucosal layer is incised to establish the tunnel opening; when the submucosa and the muscularis propria of the lower esophagus are completely adherent, then the latter as well as the cardia are incised simultaneously in the tunnel, and finally the wound is treated and the tunnel opening is sealed. This technique can be used for the treatment achalasia with grade-3 local adhesion.
(3) Tunnel opening method. The entrance to the submucosal tunnel was established after submucosal injection in the digestive duct, including transverse incision, longitudinal incision, inverted T incision and arc-shaped incision. In 2012, Professor Linghu Enqiang first introduced in his book Digestive Endoscopic Tunnel Technique Therapeutics the techniques of tunnel horizontal incision, longitudinal incision, and arc-shaped incision; he modified the tunnel incision technique again in 2014 by introducing for the first time the inverted the T-incision method to establish a tunnel opening.
A horizontal incision of about 1.2 cm is made in the mucosa to create an opening in the submucosal tunnel. With such an opening incision, it is convenient to insert and remove the endoscope in the tunnel, and the gas can also easily overflow from the tunnel opening, which is beneficial for reducing the gas pressure in the tunnel cavity. The disadvantage is that it is relatively difficult to close the incision after surgery.
A longitudinal incision of about 1.8–2.0 cm is made in the mucosa to create an opening in the submucosal tunnel. Postoperative sealing of the incision is simple in this case, but has many disadvantages, notably: it is more difficult for the endoscope to enter and exit the tunnel, more tissue clips are needed for sealing the opening, and gas overflow from the tunnel opening is not facilitated, resulting in higher pressure in the tunnel cavity.
This is a method first proposed and promoted by Professor Linghu Enqiang, which is now widely used. First, the mucosa is horizontally incised on about 0.5 cm, then it is incised longitudinally on about 1.0 cm along the midpoint of the transverse incision; finally the inverted T-shape submucosal tunnel opening is formed. This tunnel incision combines the advantages of horizontal and longitudinal incisions and avoids their disadvantages. Furthermore, with such an incision, it is easy for the endoscope to enter the tunnel, with a simple postoperative sealing incision; in addition, low pressure prevails in the tunnel cavity, which is beneficial for reducing the risks of gas-related adverse events.
First proposed and developed by Professor Linghu Enqiang, this method consists of creating a submucosal tunnel by making an arc-shaped incision in the mucosa according to the characteristics of the lesion when performing the endoscopic digestive tunnel technique. This is the common incision for resection of the submucosa or the muscularis propria.
(4) Auxiliary traction. The traction device is used to build a clear space so that the mucosal layer is fully separated from the muscularis propria, which is convenient for an endoscopic operation. It can be applied to early esophageal cancer, gastric cancer, colorectal cancer, lateral developmental polyps, and submucosal tumors. First, a complete circumferential mucosal incision is made around the lesion, and a single-ring, multiple-ring, or another traction device is then attached to the gastric wall on the side opposite to the lesion, and the other end of the traction device is attached to the flap located at the edge of the proximal end of the lesion. Afterwards, the submucosal layer was stretched and the field of view became clear before continuing to peel off the submucosal layer until the lesion detached from the wound.
3. Method of operation per puncture channel
(1) Drainage method. Its aim is to preserve the anatomical structural integrity of the original tissues and organs, to resolve problems of obstruction of the bile or pancreatic duct or encapsulated effusion, to allow necrotic drainage, and to achieve the objective of curing diseases. Drainage by puncture channel requires the application of endoscopic ultrasound as a guide in the digestive lumen using a puncture technique and a drainage device to perform the drainage and debridement therapy.
The common drainage methods include mainly the following techniques.
1) Endoscopic ultrasound-guided pancreatic pseudocyst drainage (EUS–PPD). This is a the method in which an endoscopic ultrasound-guided puncture needle is inserted into the pseudocyst, by placing a guide wire; the drainage stent or nasobiliary duct is placed in the cyst to drain the cystic fluid into the pseudocyst. This can be divided into internal drainage and external drainage. The placement of a stent between the cyst and the stomach or duodenum constitutes a fistula channel, which is an internal drainage system; one end of the nasobiliary duct is placed outside the body, so it belongs to the category of external drainage (figure 2.20). In 1992, H. Grimm et al. in Germany first proposed the application of EUS-guided drainage for the treatment pancreatic pseudocyst, which overcame the problems of high pancreatitis recurrence rate, high postoperative infection rate and death rate, etc., associated with percutaneous puncture drainage.
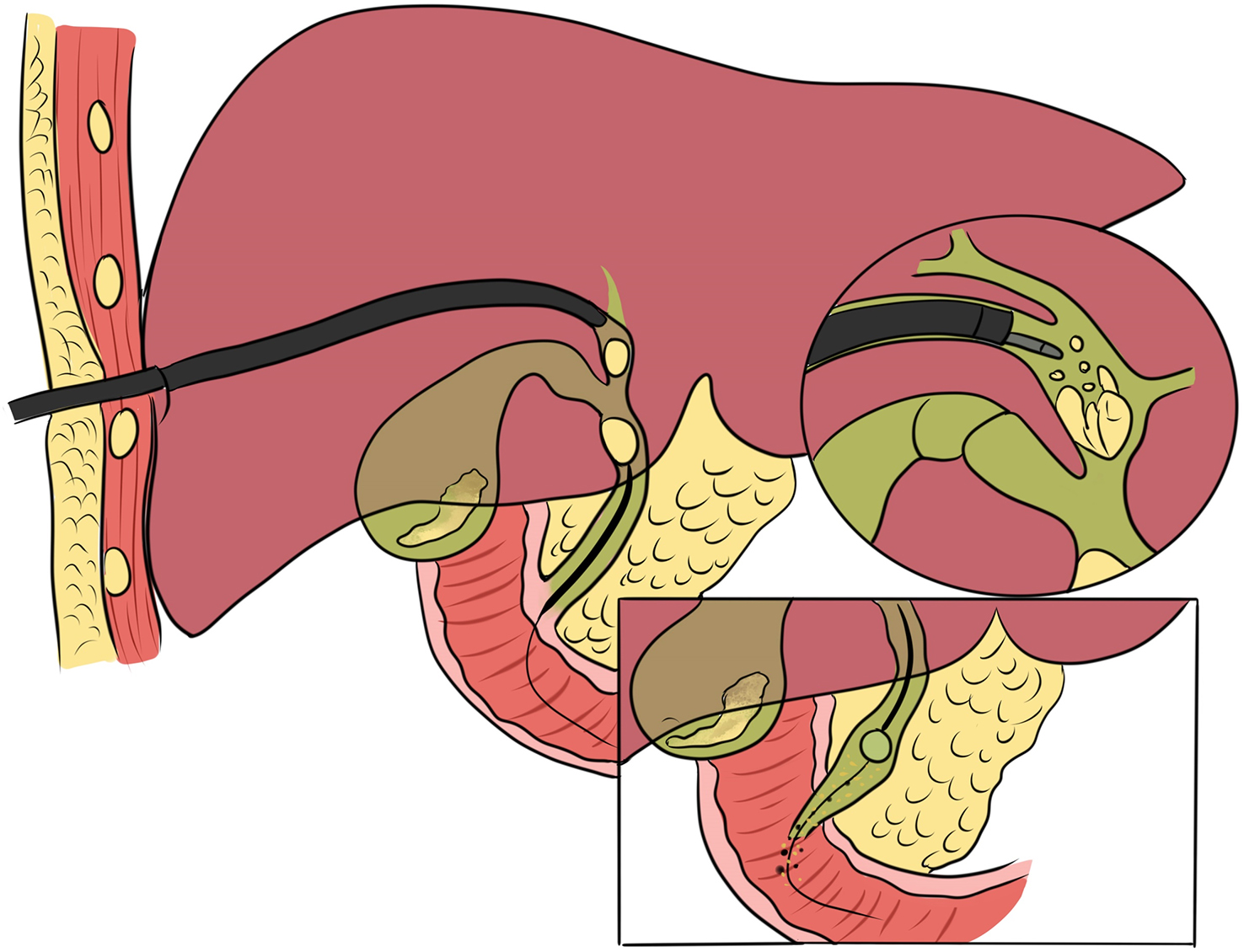

FIG. 2.20 — Operating steps of SMIS–EUS–PPD.
2) Endoscopic ultrasound-guided biliary drainage (EUS–BD). This is an endoscopic ultrasound-guided method that involves dilating the intra- or extra-hepatic biliary tract by puncture, using a stent to establish a channel between the bile duct and the digestive tract, thus resolving the obstruction of the biliary tract. If a nasobiliary duct is inserted, it is a form of external drainage. In 2001, Giovannini et al. reported EUS-guided common bile duct puncture and drainage; this pioneering work marks the birth of EUS–BD.
3) Endoscopic ultrasound-guided pancreatic drainage. This is an endoscopic ultrasound-guided method. The pancreatic duct is dilated by puncture, and a pancreatic duct stent is placed to drain the pancreatic fluid and relieve the obstruction. Erik François et al. in Belgium reported this technique in 2002. At that time, they performed an EUS–PD in four patients with chronic pancreatitis associated with hypertensive abdominal pain of the pancreatic duct, who all successfully received stents, three of which with satisfactory postoperative pain alleviation.
4) Endoscopic ultrasound-guided hepaticogastrostomy (EUS–HGS). An endoscopic ultrasound-guided puncture needle enters the intrahepatic bile ducts through the stomach, the guide wire is placed, a stent is placed in the stomach and in the intrahepatic bile ducts along the guide wire to establish a channel for drainage. In 2003, the EUS–HGS technique was first reported by Japanese endoscopist Kazutaka Kitano and his team. The innovation of this technique lies in the fact that, thanks to endoscopic ultrasound guidance, biliary drainage can be successfully performed without surgery. This technique is particularly suitable for patients who cannot undergo traditional ERCP or percutaneous biliary drainage.
5) Endoscopic ultrasound-guided choledochoduodenostomy (EUS–CDS). This is an endoscopic ultrasound-guided puncture method in the common bile duct at the level of the duodenum, in which a guide wire is placed in the common bile duct, and a stent is placed along the guide wire in the duodenum and in the common bile duct to establish a channel for drainage. At early as in 2001, Professor Marc Giovannini first reported and proposed this technique. The EUS–CDS technique involves the endoscopic ultrasound-guided creation of a new channel through the common bile duct and the duodenum, and bile drainage is realized by placing a stent. This technique mainly applies to patients who cannot undergo traditional endoscopic retrograde cholangio-pancreatography (ERCP) due to reasons like bile papillitis disease, peripapillary diverticulum, anatomic abnormality or malignant obstruction of the biliary tract, etc.
6) Endoscopic ultrasound-guided gallbladder drainage. An endoscopic ultrasound-guided gallbladder puncture is made through the stomach and duodenum at close distance and the drainage instrument is placed, thus realizing gallbladder drainage.
7) Endoscopic ultrasound-guided pelvic effusion drainage. An endoscopic ultrasound-guided channel puncture is made through the pelvic effusion area to place a drainage tube, enabling effective drainage.
(2) Puncture method: the puncture channels for super minimally invasive surgery include the digestive lumen channels, and are created using endoscopic ultrasound guidance. In addition, there are also super minimally invasive procedures for percutaneous puncture in the chest, abdomen, and pelvic cavity up to the diseased area, and they are mostly applied to obstructive biliary diseases and cystic pancreatic tumors. ERCP may damage the duodenal papilla in patients who require clinical relief of obstructive jaundice, so this defect limits the applicability of this treatment to certain remodeled organs, especially when the papillae develop lesions. In this case, ultrasound-guided puncture drainage is more popular. As the biliary tract is anatomically adjacent to the digestive tract in many places, it can be clearly explored by endoscopic ultrasound in case of obstruction, which makes endoscopic ultrasound puncture possible and provides a basis for further treatment.
Puncture drainage combines the advantages of endoscopy and of percutaneous puncture without the need to place drainage devices outside the body. On the other hand, the method of puncture depends on the patient’s own anatomy, no matter it is pathological or normal, as well as the site of obstruction (occupying) or the patient’s own underlying disease. The two approaches of EUS–BD are the transgastrohepatic approach and the transduodenal extrahepatic approach. Super-minimally-invasive-guided puncture can resolve obstructions that cannot be resolved by ERCP, with high applicability and large space for innovation.
The main puncture methods are as follows.
1) Gastrointestinal puncture (figure 2.21). Super minimally invasive gastropuncture surgery mainly includes gastropancreatic, gastrogallbladder, gastrojejunal surgeries. The puncture path can be either intrahepatic or extrahepatic. The intrahepatic approach utilizes mainly the gastropuncture channel, which involves puncture and intubation of the intrahepatic biliary tree. This channel can be used for EUS-guided rendezvous (EUS–RV), anterograde stent placement (EUS–AS) or hepatogastrostomy (EUS–HGS). The extrahepatic channel utilizes mainly the duodenal puncture channel. EUS-guided puncture enters directly the common bile duct (CBD) through an extrahepatic channel, and can be used in EUS–RV. It is most commonly used for choledochoduodenostomy (EUS–CDS). In these two methods, the stent can be placed across the narrowing and/or the papillae, while respecting anatomy (EUS–RV, EUS–AS), or connecting two organs and creating a new anastomosis (EUS–CDS, EUS–HGS). In case ERCP fails, EUS–BD will provide good results. Therefore, EUS–BD is also regarded as the preferred drainage strategy for distal malignant obstruction. In addition, SMIS of the duodenal puncture channel mainly includes EUS-guided choledochoduodenostomy and ablative therapy of pancreatic cystic tumors (the same as with the operating methods of the gastro puncture channel).
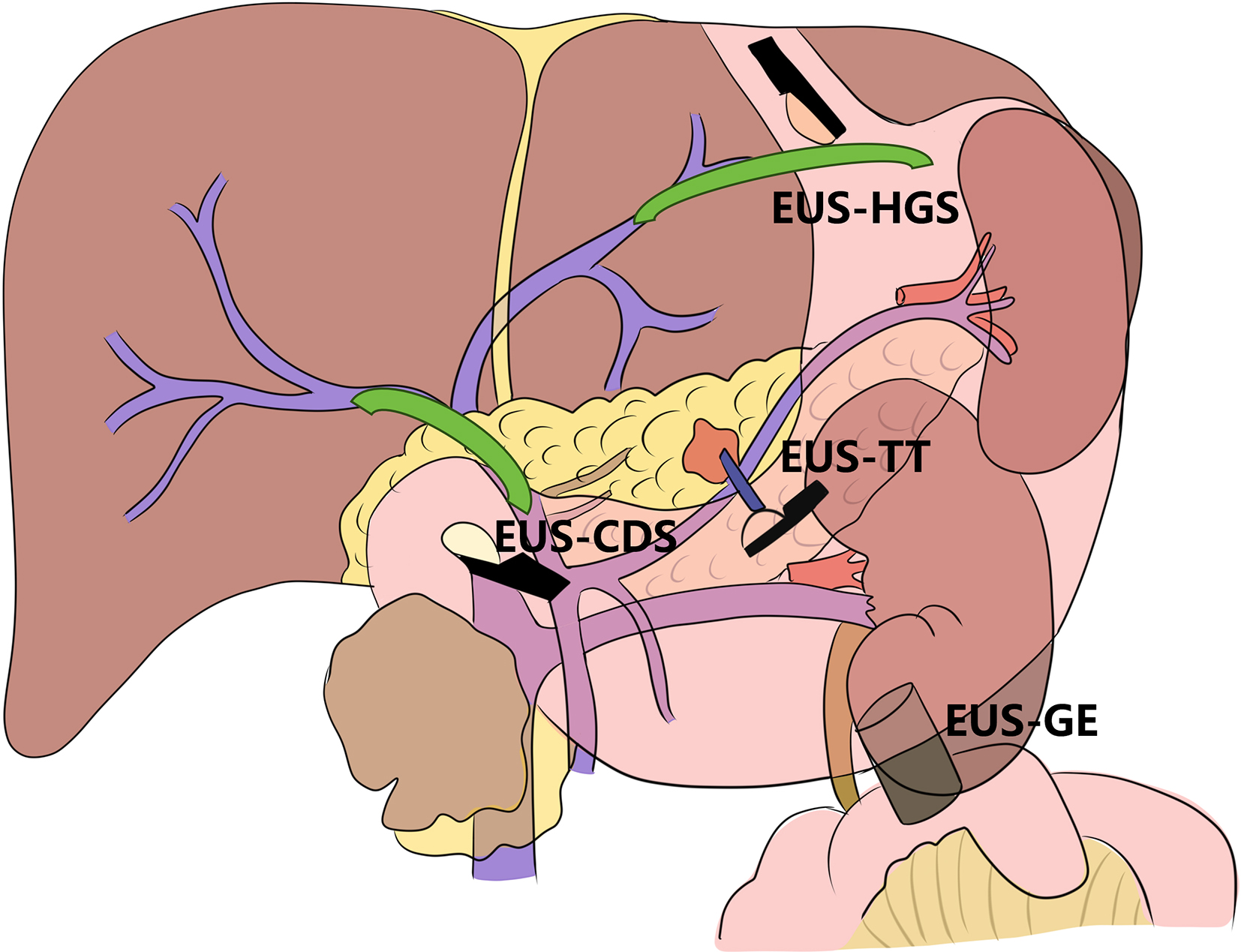

FIG. 2.21 — Surgical operation through the SMIS-gastrointestinal puncture channel.
A few methods through puncture channels are available.
A. Endoscopic ultrasound-guided necrosetomy. The gastric or duodenal wall is penetrated in the necrotic area under the guidance of endoscopic ultrasound, to establish a channel between the necrotic area and the digestive wall, and using the balloon dilator to expand and allow the endoscope to enter, and to remove the necrotic tissues by using multiple tools like a mesh basket, foreign body forceps, a snare, etc.
B. Pancreatic cystic tumor ablation. It is mainly is divided into three types. Endoscopic ultrasound-guided chemoablation is a method in which an endoscopic ultrasound-guided puncture needle is inserted into the tumor to inject a drug, thus killing the tumor cells. Endoscopic ultrasound-guided ethanol ablation is a method in which an endoscopic ultrasound-guided puncture needle is inserted into the tumor to inject absolute ethyl alcohol injection, thus killing the tumor cells. Endoscopic ultrasound-guided lauromacrogol ablation is a method in which an endoscopic ultrasound-guided puncture needle is inserted into the cystic tumor to inject lauromacrogol to destroy the cells of the cystic wall, thus killing tumor cells. Endoscopic ultrasound-guided ablation therapy of pancreatic cystic tumors was proposed and used for the first time by Professor Anthony Yuen-Bun. He and the international expert group jointly published a position statement on the endoscopic ultrasound-guided ablation of pancreatic cystic tumor.
C. Endoscopic ultrasound-guided through-the-needle cystoscopy. It is a method in which an ultrasound-guided fiberoptic endoscope is inserted into the cystic cavity for examination.
2) Percutaneous puncture. It consists mainly of percutaneous transhepatic cholangioscopy (PTCS) and can be used to assess biliary stricture (benign or malignant), for biliary biopsy, lithotripsy and lithotomy, etc. The first step of percutaneous transhepatic cholangioscopy is the creation of an entry path. The catheter is placed percutaneously using imaging examinations like ultrasound, and one waits for the fistula to form naturally. Then, under direct visualization, the choledochoscope is inserted into the bile duct tree guided by a guide wire through the fistula. When biliary stenosis is present, or when there is insufficient time for the formation of a mature biliary-cutaneous fistula tract, the cholangioscope is advanced over the guidewire after withdrawing the guidewire through the endoscopic biopsy channel. Therapeutic PTCS is often used to treat complicated biliary stones that have failed or are not ERCP candidates (e.g., recurrent suppurative cholangitis) or cholangiolithiasis, and is an effective and safe approach. In addition, complicated intrahepatic stones can be treated by PTCS combined with lithotripsy. Other therapeutic applications of PTCS include the dilation of benign biliary stricture, endoscopic sphincterotomy, laser therapy, photodynamic therapy, and microwave coagulation for biliary malignancies.
4. Method of operation via multiple-cavity channel
In the age of SMIS operation, when facing with lesions that need to be resected, we usually need to consider at least two points: the first one is that lesions must be excised provided that the normal anatomical structure and functions are preserved, so as to maintain the natural physiological state of the human being as much as possible; the second one is that the excised lesions need to have a sufficient excision edge to ensure a negative incisal margin and avoid retention, implantation and metastasis of tumorous tissues. It seems that these two goals are in opposition to each other, but the breakthrough of the multilacunar channels surgery has opened up new possibilities for us in our search for a balance between opposites. For example, laparoscopy-assisted endoscopic full-thickness resection for the treatment of large gastrointestinal submucosal masses, endoscopic and laparoscopic focal resection + lymph node dissection in the treatment of partial stages T1 and T2 of gastric cancer, etc.
Submucosal tumors include gastrointestinal stroma tumor (GIST) lipoma, leiomyoma and neuroma, etc., with GIST being the most common ones. Most GIST have a certain degree of malignant potential, and 20%–25% of gastric GIST show malignant activity, whereas regional lymph node metastasis only covers 1.1%–3.4% of the cases. Owing to GIST’s unpredictable malignant activity and rare lymph node metastasis, it is recommended to carry out a local full-thickness resection rather than a primary lymph node dissection. The goal of the surgery is to totally resect the lesion without injuring the capsule of the masses, because capsular rupture may lead to intraperitoneal metastasis and is associated with poor postoperative adhesion. Therefore, laparoscopy-assisted endoscopic full-thickness resection is an effective means for treating submucosal tumors of the digestive tract.
In recent years, the development of the lymph node tracer technique has created the necessary conditions for imaging metastatic lymph nodes and for the dissection of local lymph nodes. The combined endoscopic and laparoscopic surgery for lesion excision + lymph node dissection should bring good prospects to some patients with digestive tract cancer, and reduce the damage caused to the body while effectively excising lesions.
(1) Laparoscopy-assisted endoscopic full-thickness resection. The first step is to do an endoscopic deep submucosal incision around the lesion; the incision length is about 3/4 or 2/3 of the circumference. Then, with the assistance of a laparoscope, an endoscopic reverse traction is performed on the full thickness of the incision, then the endoscope completes the full incision of the remaining annulus.
(2) Endoscopic and laparoscopic combined surgery. The first step consists of an endoscopic and laparoscopic examination, while determining the site and extent of the lesion, etc. Then the endoscopic vascular preparation of the surgically resected area is carried out using an ultrasonic laser device. Subsequently, a mark is made endoscopically with APC around the lesion, the marking points having to be as close as possible to the edge of the tumor. After submucosal injection of substances such as normal saline solution and methylene blue, the tip end of an IT knife is inserted into the submucosa to incise it along the marks to the 3/4 of the delimited area. The tip of the laparoscopic ultrasound activation device is then inserted into the incised area, making it penetrate the sarcoplasmic muscle layer to also incise 3/4 of the tumor perimeter, creating an artificial perforation. At this point, the tumor is suspended in the abdominal cavity, attached only by the tissue pedicle that retains 1/4 of its full circumference. Finally, the assistant uses tweezers to lift the tumor and the edge of the incision line, and the surgeon uses a laparoscopic stapler to simultaneously close the incision and complete tumor removal.
(3) Inverted endoscopic and laparoscopic combined surgery. First, the laparoscope and the endoscope jointly confirm the location of the tumor. Then the vascular preparation of the area to be surgically resected is carried out using the ultrasound activated device. The marking point should be about 1 cm from the periphery of the tumor. Subsequently, the IT knife and the ultrasound activated instrument are used successively to incise 3/4 of the circumference of the tube wall From the luminal and abdominal sides of the digestive tract wall, respectively. To prevent the tumor from coming into contact with an internal organ while exiting the edge of the lesion it must be inverted using suture traction, directing it towards the internal digestive cavity. With this method, the removed tumor tissue can be passed through the oral/anal route.
(4) Combined endoscopic and laparoscopic technique to treat tumors without exposure. After endoscopic marking of the site of the lesion with indocyanine green, the mucosa layer is fixed on the sarcomuscular layer with four full-layer sutures. After submucosal injection of a solution, the serosa and muscularis propria are incised with a laparoscopic electric knife along the lateral side of four indentable sutures. Then, four indentured sutures are used to lift the full-thickness specimen, and the mucosa surrounding the full-thickness specimen is also pulled up. Finally, a laparoscopic stapler is used to close the incision while removing the lesion. This method can effectively eliminate lesions without the risk of exposing the tumoral tissue to the peritoneal cavity.
(5) Non-exposure endoscopic intramural flip surgery. First, an endoscopic incision of the serous layer and the muscularis propria is carried out around the markers of the lesion. Subsequently, the serous and muscular layers are sutured from the abdominal surface by placing muscle sutures to achieve flipping of the lesion in the gastrointestinal cavity. Finally, the ESD technique is imitated to achieve the excision without exposing the lesion.
(6) Laparoscopic transgastric surgery. The laparoscope and the gastroscope are inserted into the stomach, and the laparoscopic grip holds the wall of the stomach. Then, guided by the gastroscope, the laparoscopic stapler is inserted into the stomach, and the lesion is excised from the side of the lumen using a laparoscopic grip.
(7) Laparoscopic full-thickness resection. First, a mark is made at 1 cm from the external side of the lesion with APC. After that, under endoscopic reverse pressure-guiding, hook diathermy marking is carried out on the serous membrane side at 1 cm outside the marking. After that, brace rods are placed in the abdominal cavity; then all brace rods are tightened, the tissues wrapping the form of the lesion folding over to flip into the digestive lumen. The brace is sutured laparoscopically using two layers of continuous sutures to achieve a ‘pre-closure’ of the expected full-thickness defect. Finally, the lesion is completely resected using the digestive endoscopic clamp.
(8) Eversion full-thickness laparoscopic resection. After an endoscopic mark is made using APC and four points have been marked using hook diathermy on the abdominal wall of the tube, a support rod is placed through the endoscope from the side of the digestive lumen, the support rod is tightened so that the lesion and the surrounding tissues exert pressure towards the peritoneum. Finally, the tissues including the lesion are excised using the laparoscope to achieve full-thickness resection.
References
[1] Digestive Endoscopy Terminology Examination Subcommittee. (2021) Digestive endoscopy terminology. Science Press, Beijing.
[2] Linghu E. (2023) Interpretation of the terminology of super minimally invasive surgery in the National Terminology Committee’s Digestive Endoscopy Terminology, Chin. J. Gastrointest. Endosc. (Electron. Ed.) 10, F3.
[3] Linghu E. (2016) A new stage in the history of surgery: super minimally invasive surgery, Chin. J. Gastrointest. Endosc. (Electron. Ed.) 3, 97–98.
[4] Ning B., Linghu E. (2021) New advances in different treatment channels of super minimally invasive surgery by digestive endoscopy, Chin. J. Dig. Endosc. 38, 969–973.
[5] Zou J., Chai N., Linghu E., et al. (2021) Autologous skin-grafting surgery to prevent esophageal stenosis after complete circular endoscopic submucosal tunnel dissection: a case-matched controlled study, Surg. Endosc. 35, 5962–5970.
[6] Feng Y. (2008) Study on the impact of new understanding of appendix function on the prevention and treatment of appendicitis, Contemp. Med., 19–21.
[7] Liu B. R., Ma X., Feng J., Yang Z., et al. (2015) Endoscopic retrograde appendicitis therapy (ERAT): a multicenter retrospective study in China, Surg. Endosc. 29, 905–909.
[8] Ding W., Du Z., Zhou X. (2022) Endoscopic retrograde appendicitis therapy for management of acute appendicitis, Surg. Endosc. 36, 2480–2487.
[9] Afghani E., Lo S. K., Covington P. S., et al. (2017) Sphincter of Oddi function and risk factors for dysfunction, Front. Nutr. 4, 1.
[10] Kong J., Wu S. D., Xian G. Z., Yang S. (2010) Complications analysis with postoperative choledochoscopy for residual bile duct stones, World J. Surg. 34, 574–580.
[11] Sun M., Wang H., Wang M., et al. (2021) Application of SpyGlass DS choledochoscope in intrahepatic bile duct stones, China J. Endosc. 27, 78–83.
[12] Zhang H., Liu D., Xiao L., et al. (2018) ERCP combined with SpyGlass system for the treatment of concomitant gallbladder stones and secondary common bile duct stones in high-risky surgical patients, Chin. J. Pract. Surg. 38, 1310–1313.
[13] Li X., Zhang W., Gao F., Dong H., Wang J., Chai N., Linghu E. (2023) A modified endoscopic full-thickness resection for gastrointestinal stromal tumors: a new closure technique based on the instruction of super minimally invasive surgery, Endoscopy 55(S 01), E561–E562. DOI: 10.1055/a-2040-3927.
[14] Jiang L., Ling-Hu E. Q., Chai N. L., Li W., Cai F. C., Li M. Y., Guo X., Meng J. Y., Wang X. D., Tang P., et al. (2020) Novel endoscopic papillectomy for reducing postoperative adverse events (with videos), World J. Gastroenterol. 26(40), 6250–6259. DOI: 10.3748/wjg.v26.i40.6250.
[15] Li L., Linghu E., Chai N. (2020) Successful endoscopic transgastric retrieval of a plastic stent that migrated into the abdominal cavity during pancreatic fistula drainage, Endoscopy 52(8), E289–E290. DOI: 10.1055/a-1103-1774.
[16] Li L., Linghu E., Wang Z., Chai N. (2020 Apr) Endoscopic retrieval of a lumen-apposing metal stent that migrated into the abdominal cavity during transluminal drainage, Am. J. Gastroenterol. 115(4), 504.
[17] Zhang W., Chai N., Zhai Y., et al. (2023) The accidental discovery of pancreatic ductal adenocarcinoma on percutaneous cholangioscopy through a T-tube tract, Endoscopy 55, E404–E405.
[18] Zhang W., Chai N., Zhai Y., et al. (2023) Three pancreatic duct lesions in one patient found at an early stage by cholangioscopy, Endoscopy 55, E332–E333.
[19] Chai N. L., Li H. K., Linghu E. Q., et al. (2019) Consensus on the digestive endoscopic tunnel technique, World J. Gastroenterol. 25, 744–776.
[20] Linghu E. (2011) The creation and prospects of tunnel technology, Chin. J. Laparosc. Surg. (Electron. Ed.) 04, 326–327.
[21] Basile P., Gonzalez J. M., Le Mouel J. P., et al. (2020) Per-oral endoscopic myotomy with septotomy for the treatment of distal esophageal diverticula (D-POEM), Surg. Endosc. 34, 2321–2325.
[22] Mohan B. P., Chandan S., Jha L. K., et al. (2020) Clinical efficacy of gastric per-oral endoscopic myotomy (G-POEM) in the treatment of refractory gastroparesis and predictors of outcomes: a systematic review and meta-analysis using surgical pyloroplasty as a comparator group, Surg. Endosc. 34, 3352–3367.
[23] Linghu E. (2013) Endoscopic submucosal tunnel dissection for large esophageal neoplastic lesions, Endosc.: J. Clin. Use Biopsy Tech. 45, 60–62.
[24] Xu M. D., Cai M. Y., Zhou P. H., et al. (2012) Submucosal tunneling endoscopic resection: a new technique for treating upper GI submucosal tumors originating from the muscularis propria layer (with videos), Gastrointest. Endosc. 75, 195–199.
[25] Ma L., Chai N., Chen Q., et al. (2018) Clinical efficacy analysis of submucosal tunneling endoscopic resection for upper gastrointestinal submucosal tumors, Chin. J. Dig. Endosc. 35, 18–22.
[26] Zhai Y. Q., Li H. K., Linghu E. Q. (2016) Endoscopic submucosal tunnel dissection for large superficial esophageal squamous cell neoplasms, World J. Gastroenterol. 22, 435–445.
[27] Zhou W., Yu H. (2018) Submucosal tunneling endoscopic resection for colorectal lesions, China J. Endosc. 24, 28–33.
[28] Liu S., Chai N., Lin Y., Wang N., Li L., Zhang N., Linghu E. (2024) Autologous skin-grafting surgery with novel continuous liquid infusion stent for prevention of esophageal stenosis after complete circular endoscopic submucosal tunnel dissection, Endoscopy 56(S 01), E290–E291. DOI: 10.1055/a-2268-5590.
[29] Chai N., Wang S., Xiang J., Zhai Y., Li L., Xu S., Linghu E. (2020) Challenging use of digestive endoscopic tunnel technique to treat schwannoma originating in the trachea, Am. J. Gastroenterol. 115(5), 650. DOI: 10.14309/ajg.0000000000000629.
[30] Ru N., Linghu E., Chai N. (2023) Endoscopic submucosal tunnel dissection with an elastic traction device for a circumferential superficial esophageal neoplasm, Endoscopy 55(S 01), E388–E389. DOI: 10.1055/a-2008-0272.
[31] Liu S., Chai Y., Linghu E., Zhang B., Chai N., Lv Y. (2021) Magnetic multidirectional anchor-guided endoscopic submucosal tunnel dissection for large gastric lesions, Endoscopy 53(10), E382–E383. DOI: 10.1055/a-1298-3984.
[32] Chai N., Linghu E., Zhang X. (2016) Simultaneous performance of one-tunnel per-oral endoscopic myotomy, submucosal tunneling endoscopic resection, and diverticulotomy, Gastrointest. Endosc. 84(5), 846–847. DOI: 10.1016/j.gie.2016.05.025.
[33] Li Y., LingHu E., Ding H., Zhang X., Li M., Xiong Y., Wang X. (2016) Peroral endoscopic myotomy with simultaneous submucosal and muscle dissection for achalasia with severe interlayer adhesions, Gastrointest. Endosc. 83(3), 651–652. DOI: 10.1016/j.gie.2015.09.030.
[34] Zhang W., Chai N., Linghu E. (2018) Moving knife tip on the thoracic aorta: high-risk submucosal tunneling endoscopic resection procedure for a puzzling submucosal mass in the esophagus, Dig. Endosc. 30(3), 397–398. DOI: 10.1111/den.13014.
[35] Chai N., Zhang W., Linghu E. Q. (2018) Prepyloric submucosal tunneling endoscopic resection for a case of inflammatory mass (with video), Dig. Endosc. 30(4), 546–547. DOI: 10.1111/den.13175.
[36] Sharma P., McCarty T. R., Chhoda A., et al. (2020) Alternative uses of lumen apposing metal stents, World J. Gastroenterol. 26, 2715–2728.
[37] Zhai Y., Chai N., Li H., Linghu E. (2020) Lumen-apposing metal stent bridges space between medicine and surgery, Chin. J. Dig. Endosc. 37, 233–238.
[38] Guo J., Saftoiu A., Vilmann P., et al. (2017) A multi-institutional consensus on how to perform endoscopic ultrasound-guided peri-pancreatic fluid collection drainage and endoscopic necrosectomy, Endosc. Ultrasound 6, 285–291.
[39] Anderloni A., Fabbri C., Nieto J., et al. (2021) The safety and efficacy of a new 20-mm lumen apposing metal stent (lams) for the endoscopic treatment of pancreatic and peripancreatic fluid collections: a large international, multicenter study, Surg. Endosc. 35, 1741–1748.
[40] Jain D., Bhandari B. S., Agrawal N., Singhal S. (2018) Endoscopic ultrasound-guided gallbladder drainage using a lumen-apposing metal stent for acute cholecystitis: a systematic review, Clin. Endosc. 51, 450–462.
[41] Sobani Z. A., Ling C., Rustagi T. (2021) Endoscopic ultrasound-guided gallbladder drainage, Dig. Dis. Sci. 66, 2154–2161.
[42] Siddiqui A., Kunda R., Tyberg A., et al. (2019) Three-way comparative study of endoscopic ultrasound-guided transmural gallbladder drainage using lumen-apposing metal stents versus endoscopic transpapillary drainage versus percutaneous cholecystostomy for gallbladder drainage in high-risk surgical patients with acute cholecystitis: clinical outcomes and success in an International, Multicenter Study, Surg. Endosc. 33, 1260–1270.
[43] Shen Y., Cao J., Zhou X., Zhang S., et al. (2020) Endoscopic ultrasound-guided cholecystostomy for resection of gallbladder polyps with lumen-apposing metal stent, Medicine (Baltimore) 99, e22903.
[44] Zhai Y., Li M., Li H., Cai F., Linghu E. (2023) Rendezvous-assisted endoscopic retrograde pancreatography in a patient with annular pancreas and coexisting pancreas divisum, Endoscopy 55(S 01), E559–E560. DOI: 10.1055/a-2032-3462.
[45] Feng X., Linghu E., Chai N., Li H. (2018) New treatment of the pancreatic cystic neoplasm: endoscopic ultrasonography-guided radiofrequency ablation combined with lauromacrogol ablation, Turk. J. Gastroenterol. 29(1), 101–104. DOI: 10.5152/tjg.2017.17340.
[46] Zhang W., Linghu E. (2019) SpyGlass findings of mucinous cystic neoplasm by introducing the fiber-optic into the cyst through a 19-gauge needle during endoscopic ultrasound, Endosc. Ultrasound, 8(1), 60–62. DOI: 10.4103/eus.eus_24_17.
[47] Rimbas M., Larghi A., Costamagna G. (2017) Endoscopic ultrasound-guided gastroenterostomy: are we ready for prime time? Endosc. Ultrasound 6, 235–240.
[48] Tsujimoto H., Yaguchi Y., Kumano I., et al. (2012) Successful gastric submucosal tumor resection using laparoscopic and endoscopic cooperative surgery, World J. Surg. 36, 327–330.
[49] Zhang W., Ding H., Li Z., Linghu E. (2023) Laparoscopic common bile duct exploration through the cystic duct using flexible cholangioscopy combined with cholecystectomy for managing cholecysto-choledocholithiasis, Endoscopy 55(S 01), E659–E661. DOI: 10.1055/a-2067-4587.
[50] Hamada T., Kondo K., Saito S., Kitamura S. (2001) Nihon Geka Gakkai Zasshi. 102(10), 745–748.
[51] Tanaka S., Kashida H., Saito Y., et al. (2015) JGES guidelines for colorectal endoscopic submucosal dissection/endoscopic mucosal resection, Dig. Endosc. 27(4), 417–434.
[52] Oyama T., Tomori A., Hotta K., et al. (2005) Endoscopic submucosal dissection of early esophageal cancer, Clin. Gastroenterol. Hepatol. 3(7 Suppl 1), S67–S70.
[53] Inoue H., Endo M., Takeshita K., Yoshino K., Muraoka Y., Yoneshima H. (1992) A new simplified technique of endoscopic esophageal mucosal resection using a cap-fitted panendoscope (EMRC), Surg. Endosc. 6(5), 264–265.
[54] Kim H. S., Lee D. K., Baik S. K., et al. (2000) Endoscopic mucosal resection with a ligation device for early gastric cancer and precancerous lesions: comparison of its therapeutic efficacy with surgical resection, Yonsei Med. J. 41(5), 577–583. DOI: 10.3349/ymj.2000.41.5.577.
[55] Zhang B., Linghu E., Chen Y., Yuan F. (2018) Endoscopic diagnosis and treatment of early gastric cancer, Chin. J. Clin. Healthcare 21, 279–283.
[56] Shao Q., Wang X., Han K., Li J., Cai Z., Linghu E., Chen Q. (2024) Application of` the variable angle traction method by using clip with dental in super minimally invasive non full thickness resection of early gastric cancer, Chin. J. Postgrad. Med. 47, 221–225.
[57] Zhou P. H., Shi Q., Zhong Y. S., Yao L. Q. (2013 Nov 7) New progress in endoscopic treatment of esophageal diseases, World J. Gastroenterol. 19(41), 6962–6968.
[58] Rösch T., Sarbia M., Schumacher B., et al. (2004) Attempted endoscopic en bloc resection of mucosal and submucosal tumors using insulated-tip knives: a pilot series, Endoscopy 36(9), 788–801. DOI: 10.1055/s-2004-825838.
[59] Wang Z., Yang Y., Li W., Sun G., Peng L., Wang X. (2021) Current status and prospect of endoscopic therapeutic strategy for acute appendicitis, Chin. J. Dig. Endosc. 38(12), 976–979.
[60] Linghu E. (2013) Therapeutics of digestive endoscopic tunnel technique, 1st edn. Springer, Germany.
[61] Zhang W., Zhai Y., Chai N., et al. (2018) Single- and double-tunnel endoscopic submucosal tunnel dissection for large superficial esophageal squamous cell neoplasms, Endoscopy 50, 505–510.
[62] Linghu E., Ding H. (2014) Operational approach of peroral endoscopic myotomy, Chin. J. Gastrointest. Endosc. (Electron. Ed.), 55–59.
[63] Linghu E. (2012) New concepts of esophageal lower sphincterotomy in Peroral Endoscopic Myotomy (POEM), Chin. J. Laparosc. Surg. (Electron. Ed.) 5, 1–2.
[64] Fan K. Y., He M. J., Wang L., Xu J. Q., Li Q. L., Zhou P. H. (2024 Dec) Management of recurrence after peroral endoscopic myotomy and submucosal tunneling endoscopic septum division, Endoscopy 56(S 01), E548–E549.
[65] Linghu E., Li H., Wang X., et al. (2012) Feasibility of full-thickness myotomy in peroral endoscopic myotomy: two case reports, Chin. J. Laparosc. Surg. (Electron. Ed.) 5, 63.
[66] Linghu E., Zhang X., Du H., et al. (2014) Peroral endoscopic myotomy with inverted T-incision, short tunnel, and progressive full-thickness myotomy: a case report, Chin. J. Gastrointest. Endosc. (Electron. Ed.), 32–33.
[67] Linghu E., Li H., Wang X., et al. (2012) Feasibility of glasses-assisted peroral endoscopic myotomy: two case reports, Chin. J. Laparosc. Surg. (Electron. Ed.) 5, 33.
[68] Linghu E., Li H., Wang X., et al. (2012) Peroral endoscopic myotomy combined with balloon dilation for the treatment of achalasia: two case reports, Chin. J. Laparosc. Surg. (Electron. Ed.) 5, 59.
[69] Zhai Y., Linghu E., Li H., et al. (2013) Comparison of peroral endoscopic myotomy with transverse entry incision versus longitudinal entry incision for achalasia, J. South. Med. Univ., 1399–1402.
[70] Linghu E., Wang N., Wang H. (2015) Application of inverted ‘T’-shaped incision in tunneling resection of cardia submucosal tumors: two case reports, Chin. J. Dig. Endosc. 32, 483–484.
[71] Xiong Y., Hu H., Wang A., et al. (2015) Preliminary experience with endoscopic gastric submucosal tumor resection through the submucosal tunnel using double tunnel and double flex endoscope, J. South. Med. Univ., 455–458.
[72] Zhao D., Chai N., Linghu E., et al. (2021) Comparison of endoscopic ultrasound-guided drainage with metal stent and percutaneous nephroscopic necrosectomy for walled off necrosis, Chin. J. Laparosc. Surg. (Electron. Ed.) 14, 298–303.
[73] Wen J., Liang H., Cai F., et al. (2014) Effectiveness and safety of endoscopic ultrasound-guided transgastric or transpapillary drainage in treating pancreatic pseudocyst, Acta Acad. Med. Sin. 36, 194–197.
[74] Liu Y., Wang Z., Wang X., et al. (2006) Stent implantation through butt joint of PTBD and ERCP for the treatment of obstructive jaundice, Med. J. Chin. People’s Liberation Army 31, 1101–1102.
[75] Wang X., Cai F., Linghu E., et al. (2012) Emergency endoscopic intervention versus surgery in the management of acute obstructive suppurative cholangitis, World Chin. J. Digestol. 20, 866–869.
[76] Linghu E., Huang Q., Wang Z., et al. (2007) Review analysis of minimally invasive intervention for acute suppurative cholangitis, Chin. J. Pract. Intern. Med. 27, 775–776.
[77] Zhai Y., Chai N., Linghu E., Li H. (2019) Postoperative pancreatic fistula: a new indication for endoscopic intervention, Chin. J. Gastrointest. Endosc. (Electron. Ed.) 6, 77–81.
[78] Cheng L., Cai F., Linghu E. (2004) Endoscopic pancreatic stent insertion and drainage, Chin. J. Pract. Intern. Med. 24, 612–613.
[79] Krishnamoorthi R., Dasari C. S., Thoguluva Chandrasekar V., et al. (2020) Effectiveness and safety of EUS-guided choledochoduodenostomy using lumen-apposing metal stents (LAMS): a systematic review and meta-analysis, Surg. Endosc. 34, 2866–2877.
[80] Dhar J., Samanta J. (2022) Role of therapeutic endoscopic ultrasound in gastrointestinal malignancy-current evidence and future directions, Clin. J. Gastroenterol. 15, 11–29.
[81] Linghu E. (2023) Endoscopic ultrasonography-guided lauromacrogol ablation, Chin. J. Gastrointest. Endosc. (Electron. Ed.) 10, 144.
[82] Du C., Chai N., Linghu E., et al. (2020) Long-term effects of endoscopic ultrasonography-guided lauromacrogol ablation for the treatment of pancreatic cystic neoplasms: a prospective study (with video), Chin. J. Dig. Endosc. 37, 696–701.
[83] Gao F., Chai N., Li H., et al. (2022) Research progress in lauromacrogol ablation for digestive cystic lesions, Chin. J. Dig. Endosc. 39, 1029–1031.
[84] Du C., Linghu E., Chai N., et al. (2016) A review of endoscopic ultrasound-guided pancreatic cyst ablation, Chin. J. Gastrointest. Endosc. (Electron. Ed.) 3, 74–78.
[85] Liu Y., Wang Z., Wang X., et al. (2007) Application of butt joint of percutaneous transhepatic biliary drainage and endoscopic retrograde cholangiopancreatography in treatment of benign obstructive jaundice, Chin. J. Postgrad. Med. 30, 29–30, 40.
[86] Li J., Chai N., Linghu E. (2022) Application value and opportunity of endoscopic ultrasonography in super minimally invasive resection, China J. Mod. Med. 32, 1–6.
[87] Wang J., Wang X., Guan D., et al. (2024) Clinical analysis of patients with early gastric cancer undergoing non-curative endoscopic resection followed by additional surgical procedures, Chin. J. Dig. Endosc. 41, 30–34.
[88] Hiki N., Yamamoto Y., Fukunaga T., et al. (2008) Laparoscopic and endoscopic cooperative surgery for gastrointestinal stromal tumor dissection, Surg. Endosc. 22, 1729–1735.
[89] Kim H. H., Uedo N. (2016 Apr) Hybrid NOTES: combined laparo-endoscopic full-thickness resection techniques, Gastrointest. Endosc. Clin. N. Am. 26(2), 335–373.
[90] Grimm H., Binmoeller K. F., Soehendra N. (1992) Endosonography-guided drainage of a pancreatic pseudocyst, Gastrointest. Endosc. 38(2), 170–171.
[91] Giovannini M., Moutardier V., Pesenti C., Bories E., Lelong B., Delpero J. R. (2001) Endoscopic ultrasound-guided bilioduodenal anastomosis: a new technique for biliary drainage, Endoscopy 33(10), 898–900.
[92] François E., Kahaleh M., Giovannini M., et al. (2002) EUS-guided pancreaticogastrostomy, Gastrointest. Endosc. 56(1), 128–133.
[93] Teoh A. Y.-B., Seo D. W., Brugge W., et al. (2019) Position statement on EUS-guided ablation of pancreatic cystic neoplasms from an international expert panel, Chin. J. Gastrointest. Endosc. (Electron. Ed.) 06(04), 145–158.
[94] Chen Q., Linghu E. (2024) Super minimally invasive surgery to reduce over-treatment of early-stage gastric cancer, Chin. J. Postgrad. Med. 47(4), 289–291.
[95] Wang L., Lv K., Li Y., Chen Q. (2024) Feasibility analysis of digestive endoscopic super minimally invasive surgery of non-ampullary duodenal lesions, Chin. J. Postgrad. Med. 47(4), 296–300.
[96] Wang J., Sun Y., Li W., Xiao P., Chen Q. (2024) Clinical analytical results of enhanced abdominal CT detection of lymph node metastasis in patients with non-curable resection of early gastric cancer to guide the rational treatment of super minimally invasive surgery, Chin. J. Postgrad. Med. 47(4), 306–309.
[97] Sun G., Wang L., Lv K., Dong X., Chen Q., Shi H. (2024) Therapeutic efficacy assessment of super minimally invasive surgery in the treatment of early colorectal cancer and precancerous lesions, Chin. J. Postgrad. Med. 47(4), 439–443.
[98] Chen Q., Linghu E. (2024) The development, implementation principles and application of the endoscopic super minimally invasive surgery, Natl. Med. J. China 104(35), 3279–3281.
[99] Linghu E. (2012) Therapeutics of digestive endoscopic tunnel technique. Beijing Publishing House Group.
Chapter 3 Nomenclature in Super Minimally Invasive Surgery and Surgical Record Rules
Given the broad range of super minimally invasive surgeries (SMIS), and their huge potential for development and application prospects, the correct nomenclature and specific surgical record of super minimally invasive surgery will provide important reference for clinician learning and the implementation of clinical work, and will be helpful for discovering and treating complications in a timely manner, to ensure the safety and effectiveness of operations. Due to the diversification of SMIS techniques to date, and to the constant input of new techniques, there is an urgent need for a unified nomenclature of the current techniques that have been applied to clinical practice and of newly developed related techniques in the future, as well as for detailed surgical record in a uniform and modular manner.
3.1 Rules of Surgical Nomenclature
In order to catalogue diseases and their methods of treatment concisely and comprehensively, to make this record convenient for clinical recall and application, the SMIS nomenclature rules determined currently in force are as follows. From a general perspective, the nomenclature must contain the site of the lesion, its nature, the surgical channel and the method of treatment. It is recommended to adopt the nomenclature ‘site of the lesion + nature of the lesion + channel + super minimally invasive resection/incision/extraction/drainage/ablation.’ This nomenclature rule is the final diagnosis, combined with the pathological results after the operation is over. The endoscopic techniques applied in clinical practice in the past – for instance, the SMIS methods of endoscopic mucosal resection, endoscopic submucosal dissection, etc. – will not appear in surgical nomenclature, but the treatment process and the specific techniques applied need to be specifically described in the surgical record.
1. Rules of nomenclature in gastrointestinal super minimally invasive surgery
Referring to the book Chinese Terms in Digestive Endoscopology, for diseases that need surgical intervention or diseases with unsatisfactory long-term medication effect, the surgery aimed at preserving the integrity of the structure of the human organ while achieving the objective of curing a disease by endoscopic excision of lesions is called super minimally invasive resection (SMIR). Meanwhile, depending on the depth of the excised lesion in the digestive tract, SMIR is subdivided into SMIS by full-thickness resection (SMIS–FTR) and SMIS by non-full-thickness resection (SMIS-non FTR).
It is recommended to use the nomenclature ‘lesion site + lesion nature + channel + super minimally rection’ at the present stage.
(1) Site. It is the surgical site that needs to be accurately positioned on the digestive tract, such as esophageal superior/middle/inferior segment, greater/lesser curvature of gastric fundus, anterior/posterior wall of the gastric antrum. Duodenal bulb/duodenal descending part, jejunum, ileum, appendix, cecum, colon, rectum and anal tube, etc.
(2) Nature of the lesion. Adenocarcinoma, squamous cell carcinoma, high-grade intraepithelial neoplasia, low-grade intraepithelial neoplasia, stromal tumor, leiomyoma, giant adenoma, lipoma, lateral developmental tumor, etc.
(3) Channel. Peroral, transanal, per tunnel, per multiple cavity, etc.
See tables 3.1–3.4 for reference examples of specific nomenclature.
TAB. 3.1 — Examples of nomenclature in esophageal SMIS surgery.
| No. | Name | Abbreviation |
| 1 | Peroral super minimally invasive surgery by non-full-thickness resection of a superior esophageal papilloma | Peroral SMIS-non FTR for SEP |
| 2 | Peroral super minimally invasive surgery by non-full-thickness resection of a middle esophageal high-grade intraepithelial neoplasm | Peroral SMIS-non FTR for MEHIN |
| 3 | Peroral super minimally invasive surgery by non-full-thickness resection of an inferior esophageal squamous cell carcinoma | Peroral SMIS-non FTR for IESCC |
| 4 | Peroral super minimally invasive surgery by non-full-thickness resection of a middle esophageal granular cell tumor | Peroral SMIS-non FTR for MEGCT |
| 5 | Peroral super minimally invasive surgery by full-thickness resection of an early adenocarcinoma on the gastroesophageal junction | Peroral SMIS–FTR for EAC on GEJ |
| 6 | Peroral super minimally invasive surgery by full-thickness resection of an inferior esophageal leiomyoma | Peroral SMIS–FTR for IEL |
| 7 | Per-tunnel super minimally invasive surgery by non-full-thickness resection of a middle esophageal carcinoma | Per-tunnel SMIS-non FTR for MEC |
| 8 | Per-tunnel super minimally invasive resection of an inferior esophageal leiomyoma | Per-tunnel SMIR for IEL |
| 9 | Per-tunnel super minimally invasive resection of a gastric stromal tumor on the gastroesophageal junction | Per-tunnel SMIR for GST on GEJ |
| 10 | Per-multi-cavity super minimally invasive resection of a middle esophageal leiomyoma | Per-multi-cavity SMIR for MEL |
TAB. 3.2 — Examples of nomenclature in gastric SMIS surgery.
| No. | Name | Abbreviation |
| 1 | Peroral super minimally invasive surgery by non-full-thickness resection for gastric high-grade intraepithelial neoplasia on the greater curvature of the gastric body | Peroral SMIS-non FTR for GHIN on the greater curvature of the gastric body |
| 2 | Peroral super minimally invasive surgery by non-full-thickness resection of a gastric carcinoma on the anterior wall of the gastric antrum | Peroral SMIS-non FTR for GC on the anterior wall of the gastric antrum |
| 3 | Peroral super minimally invasive surgery by non-full-thickness resection of a gastric leiomyoma on the lesser curvature of the gastric body | Peroral SMIS-non FTR for GL on the lesser curvature of the gastric body |
| 4 | Peroral super minimally invasive surgery by non-full-thickness resection of a gastric neuroendocrine tumor on the posterior wall of the gastric antrum | Peroral SMIS-non FTR for GNET on the posterior wall of the gastric antrum |
| 5 | Peroral super minimally invasive surgery by non-full-thickness resection of a gastric lipoma on the posterior wall of the gastric body | Peroral SMIS-non FTR for GL on the posterior wall of the gastric body |
| 6 | Peroral super minimally invasive surgery by full-thickness resection of a gastric glomus tumor on the gastric angle | Peroral SMIS–FTR for GGT on the gastric angle |
| 7 | Peroral super minimally invasive surgery by full-thickness resection of a gastric carcinoma on the greater curvature of the gastric body | Peroral SMIS–FTR for GC on the greater curvature of the gastric body |
| 8 | Peroral super minimally invasive surgery by full-thickness resection of a gastric stromal tumor on the greater curvature of gastric fundus | Peroral SMIS–FTR for GST on the greater curvature of the gastric fundus |
| 9 | Per-tunnel super minimally invasive surgery by non-full-thickness resection of a gastric carcinoma on the greater curvature of the gastric antrum | Per-tunnel SMIS-non FTR for GC on the greater curvature of the gastric antrum |
| 10 | Per-multi-cavity super minimally invasive resection of a gastric carcinoma on the anterior wall of the gastric body | Per-multi-cavity SMIR for GC on the anterior wall of the gastric body |
| 11 | Per-multi-cavity super minimally invasive resection of a gastric stromal tumor on the gastric angle | Per-multi-cavity SMIR for GST on the gastric angle |
TAB. 3.3 — Examples of nomenclature in intestinal SMIS surgery.
| No. | Name | Abbreviation |
| 1 | Peroral super minimally invasive surgery by non-full-thickness resection of a duodenal bulb neuroendocrine tumor | Peroral SMIS-non FTR for DBNET |
| 2 | Peroral super minimally invasive surgery by non-full-thickness resection of a duodenal adenoma on the duodenal descending part | Peroral SMIS-non FTR for DA on the duodenal descending part |
| 3 | Peroral super minimally invasive surgery by non-full-thickness resection of a duodenal carcinoma on the duodenal descending part | Peroral SMIS-non FTR for DC on the duodenal descending part |
| 4 | Peroral super minimally invasive surgery by full-thickness resection of a duodenal bulb leiomyoma | Peroral SMIS–FTR for DBL |
| 5 | Peroral super minimally invasive resection of a duodenal papillary adenoma | Peroral SMIR for DPA |
| 6 | Peroral super minimally invasive resection of an early duodenal papillary carcinoma | Peroral SMIR for EDPC |
| 7 | Per-tunnel super minimally invasive resection of a duodenal bulb stromal tumor | Per-tunnel SMIR for DBST |
| 8 | Per-multi-cavity super minimally invasive resection of a duodenal carcinoma on the duodenal descending part | Per-multi-cavity SMIR for DC on the duodenal descending part |
| 9 | Peroral super minimally invasive surgery by non-full-thickness resection of a jejunum adenoma | Peroral SMIS-non FTR for JA |
| 10 | Transanal super minimally invasive resection of an ileal stromal tumor | Transanal SMIR for IST |
TAB. 3.4 — Examples of nomenclature in colorectal SMIS surgery.
| No. | Name | Abbreviation |
| 1 | Transanal super minimally invasive surgery by non-full-thickness resection of a sigmoid colon adenoma | Transanal SMIS-non FTR for SCA |
| 2 | Transanal super minimally invasive surgery by non-full-thickness resection of a descending colon lipoma | Transanal SMIS-non FTR for DCL |
| 3 | Transanal super minimally invasive surgery by full-thickness resection for an ascending colon adenocarcinoma | Transanal SMIS-non FTR for ACAC |
| 4 | Per-tunnel super minimally invasive surgery by non-full-thickness resection of a rectal adenocarcinoma | Per-tunnel SMIS-non FTR for RAC |
| 5 | Per-multi-cavity super minimally invasive resection of a neuroendocrine tumor of the appendix | Per-multi-cavity SMIR for ANET |
| 6 | Per-multi-cavity super minimally invasive resection of a cecal stromal tumor | Per-multi-cavity SMIR for CST |
2. Rules of nomenclature in gastrointestinal super minimally invasive incision
Referring to the book Chinese Terms in Digestive Endoscopology, for diseases that need surgical intervention or diseases with unsatisfactory long-term medication effect, the surgery aimed at preserving the integrity of the structure of the human organ while achieving the objective of curing a disease by endoscopic excision of lesions is called super minimally invasive incision (SMII).
At the present stage, the nomenclature ‘lesion site + lesion nature + channel + super minimally invasive incision’ is recommended. The sites can be the esophagus, the intestine, the colorectum etc., and the channels are mostly natural. Depending on the incision method, the site, SMII can be divided into SMIS by full-thickness incision, SMIS by lower esophageal sphincter myotomy, and SMIS by pyloric sphincter myotomy, etc.
See table 3.5 for reference examples of specific nomenclature.
TAB. 3.5 — Example of nomenclature in gastrointestinal endoscopic super minimally invasive incision.
| No. | Name | Abbreviation |
| 1 | Peroral super minimally invasive incision for middle esophageal cyst | Peroral SMII for MEC |
| 2 | Peroral super minimally invasive incision for superior esophageal diverticulum | Peroral SMII for SED |
| 3 | Peroral super minimally invasive incision for inferior benign esophageal stenosis | Peroral SMII for IBES |
| 4 | Peroral super minimally invasive E myotomy for achalasia | Peroral SMIM for achalasia |
| 5 | Peroral super minimally invasive myotomy for diffuse esophageal spasm | Peroral SMIM for DES |
| 6 | Peroral super minimally invasive myotomy for hypertrophic pyloric | Peroral SMIM for HP |
| 7 | Transanal super minimally invasive incision for benign jejunum stenosis | Transanal SMII for BJS |
| 8 | Transanal super minimally invasive incision for benign rectal stenosis | Transanal SMII for BRS |
3. Rules of nomenclature in gastrointestinal endoscopic super minimally invasive drainage
Referring to the book Chinese Terms in Digestive Endoscopology, for diseases that need surgical intervention or diseases with unsatisfactory long-term medication effect, the surgery aimed at preserving the integrity of the structure of the human organ while achieving the objective of curing a disease by endoscopic drainage of lesions is called super minimally invasive drainage (SMID).
At the present stage, the nomenclature ‘lesion site + lesion nature + channel + super minimally invasive drainage’ is recommended, including for drainage carried out in cases such as those of pancreatic pseudocyst, encapsulated effusion, obstruction of the biliary tract, etc. The lesion sites can be the pancreas, the bile duct, the mediastinum, the abdomen, the pelvic cavity, etc. Channels are mostly per puncture channel or per natural channel drainages, such as peroral, transanal, per duodenal papilla, per gastric puncture channel, per duodenal puncture channel and percutaneous puncture channel, etc.
See table 3.6 for reference examples of specific nomenclature.
TAB. 3.6 — Reference examples of nomenclature in gastrointestinal endoscopic super minimally invasive drainage.
| No. | Name | Abbreviation |
| 1 | Per-cutaneous-puncture ultrasound-guided super minimally invasive biliary drainage for primary sclerosing cholangitis | Per-cutaneous-puncture US-guided SMIBD for PSC |
| 2 | Per-gastric-puncture endoscopic ultrasound-guided super minimally invasive biliary drainage for acute suppurative cholecystitis | Per-gastric-puncture EUS-guided SMIBD for ASC |
| 3 | Per-duodenal-papilla super minimally invasive biliary drainage for acute suppurative cholangitis | Per-duodenal-papilla SMIBD for ASC |
| 4 | Per-duodenal-papilla super minimally invasive biliary drainage for choledochoduodenal fistula | Per-duodenal-papilla SMIBD for CF |
| 5 | Peroral super minimally invasive biliary drainage for biliary perforation | Peroral SMIBD for BP |
| 6 | Peroral super minimally invasive drainage for choledochoduodenal fistula | Peroral SMID for CF |
| 7 | Per-duodenal-papilla super minimally invasive drainage for biliary stenosis | Per-duodenal-papilla SMID for BS |
| 8 | Per-duodenal-papilla super minimally invasive pancreatic duct drainage for incomplete pancreas divisum | Per-duodenal-papilla SMIPD for incomplete PD |
| 9 | Per-duodenal-papilla super minimally invasive pancreatic drainage for complete pancreas divisum | Per-duodenal-papilla SMIPD for complete PD |
| 10 | Per-gastric-puncture endoscopic ultrasound-guided super minimally invasive drainage for pancreatic pseudocyst | Per-gastric-puncture EUS-guided SMID for PPC |
| 11 | Per-gastric-puncture endoscopic ultrasound-guided super minimally invasive drainage for pancreatic walled-off necrosis | Per-gastric-puncture EUS-guided SMID for PWN |
| 12 | Peroral super minimally invasive pancreatic drainage for chronic pancreatitis | Peroral SMIPD for CP |
| 13 | Peroral super minimally invasive pancreatic drainage for pancreatic fistula | Peroral SMIPD for PF |
| 14 | Per-duodenal-papilla super minimally invasive pancreatic drainage for pancreatic stenosis | Per-duodenal-papilla SMIPD for PS |
| 15 | Per-duodenal-papilla super minimally invasive pancreatic drainage for disconnected pancreatic syndrome | Per-duodenal-papilla SMIPD for DPS |
4. Gastrointestinal endoscopic super minimally invasive extraction
Referring to the book Chinese Terms in Digestive Endoscopology, for diseases that need surgical intervention or diseases with unsatisfactory long-term medication effect, the surgery aimed at preserving the integrity of the structure of the human organ while achieving the objective of curing a disease by endoscopic extraction of lesions is called super minimally invasive extraction (SMIE). Nowadays, with the development of endoscopic techniques, it is possible to preserve the structural and functional integrity of the gallbladder and the duodenal papillary sphincter while curing the disease.
At the present stage, the nomenclature ‘lesion site + lesion nature + channel + super minimally invasive extract’ is recommended.
These surgeries are mainly the extraction of these stones from the pancreatic and bile duct. Lesion sites can be the bile duct, the pancreatic duct, etc. Channels can be the duodenal papilla, a percutaneous puncture channel, peroral, transanal channels, etc.
See table 3.7 for reference examples of specific nomenclature.
TAB. 3.7 — Reference examples of nomenclature in gastrointestinal endoscopic super minimally invasive extraction.
| No. | Name | Abbreviation |
| 1 | Per-duodenal-papilla super minimally invasive extract for cholecystolithiasis | Per-duodenal-papilla SMIE for cholecystolithiasis |
| 2 | Peroral endoscopic ultrasound-guided super minimally invasive extract for cholecystolithiasis | Peroral EUS-guided SMIE for cholecystolithiasis |
| 3 | Per-duodenal-papilla super minimally invasive extract under cholangioscopy for biliary stone | Per-duodenal-papilla SMIE under cholangioscopy for BS |
| 4 | Peroral super minimally invasive extract for biliary stone after liver transplantation | Peroral SMIE for BS after LT |
| 5 | Peroral super minimally invasive extract for extrahepatic biliary stone | Peroral SMIE for EBS |
| 6 | Per-duodenal-papilla super minimally invasive extract for biliary pancreatitis | Per-duodenal-papilla SMIE for BP |
| 7 | Peroral super minimally invasive extract for pancreatic stone | Peroral SMIE for PS |
5. Other super minimally invasive surgeries
Referring to the book Chinese Terms in Digestive Endoscopology, gastrointestinal endoscopic super minimally invasive anti-reflux surgery, gastrointestinal endoscopic super minimally invasive closure, gastrointestinal endoscopic super minimally invasive incision, etc., are examples of super minimally invasive surgeries. Currently, the nomenclature ‘lesion site + lesion nature + channel + gastrointestinal endoscopic super minimally invasive closure’ is recommended. Lesion sites are mainly digestive lumens such as esophagus, stomach, colorectum, etc.
Other SMIS surgeries are listed below.
This is a surgery using the anti-reflux method, aimed at preserving the integrity of the structure of the human organ while achieving the objective of curing a disease.
This is a surgery for diseases that need surgical intervention or diseases with unsatisfactory long-term medication effect, aimed at preserving the integrity of the structure of the human organ while achieving the objective of curing a disease by endoscopic closure of the lesions.
This endoscopic technique aims to treat injuries to the target tissue as well as cell necrosis using different types of physical energies or chemical substances.
See table 3.8 for the reference example of specific nomenclature.
TAB. 3.8 — Reference examples of nomenclature in other types of SMIS surgeries.
| No. | Name | Abbreviation |
| 1 | Peroral super minimally invasive anti-reflux surgery for gastroesophageal reflux disease | Peroral SMIARS for GERD |
| 2 | Peroral super minimally invasive radiofrequency ablation for gastroesophageal reflux disease | Peroral SMIRA for GERD |
| 3 | Peroral super minimally invasive ablation for middle esophageal cyst | Peroral SMIA for MEC |
| 4 | Peroral super minimally invasive radiofrequency ablation for superior esophageal low-grade intraepithelial neoplasia | Peroral SMIRA for SELIN |
| 5 | Peroral super minimally invasive radiofrequency ablation for low-grade intraepithelial neoplasia on the anterior wall of the gastric antrum | Peroral SMIRA for GLIN on the Anterior Wall of the gastric antrum |
| 6 | Peroral super minimally invasive ablation for gastric antral telangiectasia | Peroral SMIA for GAT |
| 7 | Per-gastric-puncture endoscopic ultrasound-guided super minimally invasive ablation for pancreatic cystic neoplasm | Per-gastric-puncture EUS-guided SMIA for PCN |
| 8 | Peroral super minimally invasive closure for esophagomediastinal fistula | Peroral SMIC for EMF |
| 9 | Peroral super minimally invasive closure for esophagotracheal fistula | Peroral SMIC for ETF |
3.2 Recording Rules for Super Minimally Invasive Surgery
The endoscopic surgical record is an essential component of medical documentation. It contains information such as the surgical procedure, the patient’s condition, the steps of the operation, the medical equipment used, and the drugs administered, all of which should be recorded accurately and specifically. These records are of significant importance for subsequent medical decisions, patient management and legal responsibilities, etc. Super minimally invasive surgical records should be divided into modules: the position of the patient (left lateral position, right shoulder elevation position, prone position, supine position, etc.), the mode of anesthesia (tracheal intubation, intravenous anesthesia, basic anesthesia, no anesthesia, etc.), the surgical process (including the detailed steps, the super minimally invasive surgery (SMIS) excision methods applied, the sealing method, the hemostasis method, etc.), the duration of the operation, the intraoperative complications and their treatment (intraoperative bleeding amount, intraoperative perforation condition, whether intraoperative-assisted abdominal puncture exhausts the patient, whether there is subcutaneous emphysema, etc.), the initial diagnosis of the operation, the final diagnosis of the operation. Safe and effective endoscopic techniques and method names used in the past can be specifically recorded according to the modules in the surgical record, so as to guide the understanding that clinicians have of surgery and improve the innovative awareness of surgeons.
3.2.1 SMIS Excision Methods and the Nomenclature of Endoscopic Techniques
This section refers to the book Chinese Terms in Digestive Endoscopology.
1. Endoscopic techniques used for endoscopic super minimally invasive resection: endoscopic treatment of digestive polyps, endoscopic high-frequency electrical polypectomy, endoscopic hot-forceps polypectomy, endoscopic hemoclip-assisted polypectomy, endoscopic nylon-loop ligation-assisted polypectomy, endoscopic rubber band ligation of polyps, endoscopic rubber band ligation-assisted polypectomy, endoscopic cold snare polypectomy, endoscopic mucosal resection, endoscopic mucosal resection, precutting endoscopic mucosal resection, piecemeal endoscopic mucosal resection, cap-assisted endoscopic mucosal resection, ligation-assisted endoscopic mucosal resection, multi-band mucosectomy, endoscopic submucosal dissection, endoscopic submucosal dissection with traction, endoscopic submucosal tunnel dissection, endoscopic papillectomy, endoscopic submucosal excavation, endoscopic full-thickness resection, etc.
2. Endoscopic techniques used for endoscopic super minimally invasive incision: endoscopic full-thickness incision, peroral endoscopic myotomy, peroral endoscopic circular muscle myotomy, peroral endoscopic full-thickness myotomy, peroral endoscopic progressive full-thickness myotomy, peroral endoscopic glasses-style myotomy, peroral endoscopic circular muscle myotomy plus balloon plasty, short-tunnel peroral endoscopic myotomy, peroral endoscopic myotomy with simultaneous submucosal and muscle dissection, gastric peroral endoscopic myotomy, etc.
3. Endoscopic techniques used for endoscopic super minimally invasive drainage: according to the difference of methods and sites, it can be divided into super minimally invasive drainage for digestive tract infection with one side blind, super minimally invasive drainage for encapsulated effusion, endoscopic ultrasound-guided drainage, endoscopic biliary drainage, endoscopic pancreatic drainage, endoscopic puncture drainage of pancreatic pseudocyst, endoscopic ultrasound-guided biliary drainage, endoscopic ultrasound-guided pancreatic drainage, endoscopic ultrasound-guided gallbladder drainage, endoscopic ultrasound-guided pelvic effusion drainage.
4. Endoscopic techniques used for endoscopic super minimally invasive extraction: depending on the nature of the extraction and the method, it can be divided into endoscopic gallbladder-preserving cholecystolithotomy, sphincter-preserving biliary stone removal, sphincter-preserving pancreatic stone removal, super minimally invasive removal of cholecystolithiasis, endoscopic ultrasound-guided peroral cholecystolithotomy, endoscopic removal of biliary stones after liver transplantation, peroral super minimally invasive removal of extrahepatic biliary stones, endoscopic removal of biliary stones for the treatment of biliary pancreatitis, peroral super minimally invasive removal of pancreatic stones, etc.
5. Endoscopic techniques used for endoscopic super minimally anti-reflux surgery: peroral incisionless fundoplication for gastroesophageal reflux disease, clip-band ligation anti-reflux therapy for gastroesophageal reflux disease, C-BLART for gastroesophageal reflux disease, anti-reflux mucosa resection for the treatment of gastro-esophageal reflux, etc.
6. Endoscopic techniques used for endoscopic super minimally closure: endoscopic metal clip closure, endoscopic suturing, endoscopic purse-string suture with a nylon rope, endoscopic closing with fibrin sealant, endoscopic wound omental tamponade, etc.
7. Endoscopic techniques used for endoscopic super minimally ablation: depending on the nature of the substance to be ablated, endoscopic physical ablation, endoscopic radiofrequency ablation, endoscopic laser therapy, endoscopic photodynamic therapy, endoscopic high-frequency electrocoagulation therapy, endoscopic thermoprobe therapy, endoscopic microwave therapy, endoscopic chemoablation, endoscopic ethanol ablation, endoscopic lauromacrogol ablation, endoscopic chemotherapeutic drug ablation, etc.
8. Endoscopic techniques used for super minimally invasive endoscopic surgery for the prevention and treatment of gastrointestinal bleeding: endoscopic drug-spraying hemostasis, endoscopic hemostasis by injection, endoscopic hemostasis by thermocoagulation, endoscopic electrocoagulation hemostasis, endoscopic argon plasma coagulation, endoscopic microwave hemostasis, endoscopic thermoprobe hemostasis, endoscopic ligation hemostasis, endoscopic chemical cautery hemostasis, etc.
3.2.2 Examples of SMIS Surgical Records
Surgical record of peroral super minimally invasive resection for middle-esophageal cancer
Left lateral position, tracheal intubation under general anesthesia.
Description of the lesion. A type-IIa + IIc lesion was found in the middle part of the esophagus, 26–28 cm from the incisor, with rough and red surface, occupying 1/2 circumference of the lumen; magnifying endoscopy + narrow-band light imaging observation: the lesion boundary was clear, IPCL was mainly of type B1, and type-B2 vessels were seen; the lesion area was resistant to Lugol’s iodine staining.
Surgical process. The normal mucosa surrounding the lesion was marked circumferentially with argon; a normal saline solution/adrenaline/methylene blue mixture was injected into the submucosa outside of the marking spots, and the mucosal layer was lifted. The mucosal layer was scratched circumferentially using a DualKnife; once the submucosa was exposed, the circumferential incision was continued using the DualKnife to cut through the submucosa; tissue clips and floss were used to pull on the lesion, enabling the submucosa to be fully separated from the muscularis propria; the mixture was injected submucosally to make the field of vision clearer; the triangular knife was replaced to continue dissecting the lesion, until it was totally removed from the wound; the hemostatic forceps was then applied to carefully heal the wound, so that no naked vessels and bleeding could be observed; finally, one proceeded to inhalation and withdrawal of the endoscope.
Intraoperative complications. The amount of bleeding was about 5 ml, no muscularis propria injury and perforation, no subcutaneous emphysema.
SMIS methods or past endoscopic technique names. Endoscopic submucosal dissection with traction (ESD with traction), endoscopic electric coagulation.
Initial diagnosis. Peroral super minimally invasive resection for middle-esophageal cancer
Surgical record of per-tunnel super minimally invasive myotomy for the treatment of achalasia
Right shoulder elevation position, tracheal intubation general anesthesia.
Description of the lesion. The endoscopic examination showed that the esophageal lumen was enlarged and distorted; the annular structure was visible and exceeded 1/3 of the lumen; an edema of the esophageal mucosa was present, manifested by granular changes. Tight cardiac ostium, several strips of ulcer erosion were observed in the mucosa above the cardiac ostium, about 39 cm from the incisor. There was some resistance when the endoscope passing through the cardia.
Surgical process. 6 ml of a mixture of methylene blue/adrenaline/normal saline solution were injected into the submucosa about 32 cm from the incisor; the mucosal layer was incised to the submucosal layer with the triangle knife, an inverted T-shaped incision was made at the tunnel entry; a short tunnel was established, whereas one continued to use the triangle knife for submucosal dissection until 3 cm under the cardia; the adhesion was obvious inside the tunnel. The progressive myotomy was conducted starting 36 cm from the incisor with the triangle knife; starting from 38 cm from incisor, the full-thickness muscle layer was incised until the bottom of the tunnel; the bleeding points in the tunnel cavity and the broken end of the muscle were treated with thermal hemostatic forceps; the endoscope exited the tunnel and passed through the cardiac again without resistance; several pieces of tissue clips were used to close the entry of the tunnel; once the operation was completed, one proceeded to inhalation and withdrawal of the endoscope.
Intraoperative complications. The amount of bleeding was about 5 ml, no subcutaneous emphysema was observed.
SMIS methods or past endoscopic technique names. Short tunnel peroral endoscopic myotomy (short-tunnel POEM), peroral endoscopic progressive full-thickness myotomy, endoscopic metal clip closure, endoscopic electric coagulation.
Initial diagnosis. Achalasia (Ling II type c, grading of mucosal inflammation, grade E, adhesiveness grading grade 2), per-tunnel super minimally invasive myotomy.
Per-duodenal papilla super minimally invasive extract under the cholangioscope for biliary stone removal
Prone position, intravenous anesthesia.
Description of the lesion. Preoperative MRCP imaging showed defects in the lower segment of the common bile duct, a slighted dilated upper segment of the common bile duct, considering a single-block CBD stone.
Surgical process. The duodenoscope was inserted at the site of the duodenal papilla; a bow-shaped knife carrying the guide wire was used for biliary intubation; after successful intubation of the biliary duct, a single mushroom-nipple support was placed between the distal common bile duct and the papillae (the diameter was 12 mm, the length was 25–30 mm); the peroral choledochoscope was inserted into the common bile duct, the lithotomy basket was inserted through the peroral choledochoscopic channel under direct vision; the peroral choledochoscope was again inserted into the CBD, to confirm whether there were stone residues; finally, the nipple support was removed.
SMIS methods or past endoscopic technique names. Sphincter-preserving removal of biliary stones.
Initial diagnosis. Per-duodenal papilla super minimally invasive extraction of common bile duct stones under the cholangioscope.
3.3 Surgical Items and Diseases
Super minimally invasive surgery has been widely used in the treatment of a variety of diseases of the digestive system, and was gradually expanded to more diseases of organs and systems, promoting the continuous improvement of this new medical concept and these new goals. These chapters (tables 3.9–3.12) will briefly introduce the most widely used surgical items by taking the examples of super minimally invasive surgeries via the human natural channel, the tunnel channel, the puncture channel, and the multi-cavity channel.
TAB. 3.9 — The surgical items and diseases via human natural cavity.
| Disease | SMIS Item | Definition |
| Biliary stone | Peroral super minimally invasive extraction | Peroral endoscopic removal of extrahepatic bile duct stone while preserving the function of the papillary sphincter, including basket extraction under direct vision, balloon extraction under direct vision, extraction by attraction under direct vision |
| Gallbladder stone | Peroral super minimally invasive extraction | Peroral endoscopic removal of gallbladder stones |
| Acute suppurative cholangitis | Peroral super minimally invasive drainage | Peroral endoscopic drainage of acute suppurative cholangitis |
| Gastric stromal tumor | Peroral super minimally invasive resection | Peroral endoscopic total resection of local gastric stromal tumors |
| Early gastric cancer | Peroral super minimally invasive resection | Peroral endoscopic total resection of local early gastric cancer |
| Early esophageal cancer | Peroral super minimally invasive resection | The technique of peroral endoscopic total resection locally of early esophageal cancer |
| Duodenal papillary adenoma | Peroral super minimally invasive resection | Peroral endoscopic total resection of a local duodenal papillary adenoma |
| Early colorectal cancer | Transanal super minimally invasive resection | Transanal endoscopic total resection of local early colorectal cancer |
| Gastroesophageal reflux disease | Peroral super minimally invasive anti-reflux | Peroral endoscopic treatment of gastroesophageal reflux |
| Barrett esophagus | Peroral super minimally invasive resection | Peroral endoscopic total resection of local esophageal lesions |
| Esophagomediastinal fistula | Peroral super minimally invasive closure for esophagomediastinal fistula | Peroral endoscopic closure of esophagome diastinal fistula |
| Appendicitis | Transanal super minimally invasive surgery | Transanal endoscopic removal of appendicular lithiasis |
| Benign esophageal stricture | Peroral super minimally invasive incision | Peroral endoscopic incision of the benign stricture segment to treat swallowing difficulties |
| Biliary stenosis, biliary fistula | Per-nasal super minimally invasive drainage | Used to place the nasobiliary drainage tube nasally; it thus plays the role of a drain |
| Gallbladder polyp | Peroral super minimally invasive resection | Peroral endoscopic resection of gallbladder polyps |
TAB. 3.10 — Surgery via tunnel channel.
| Disease | SMIS Item | Definition |
| Early esophageal cancer | Per-tunnel super minimally invasive resection | Per-tunnel endoscopic resection of early esophageal cancer |
| Early gastric cancer | Per-tunnel super minimally invasive resection | Per-tunnel endoscopic resection of early gastric cancer |
| Early colorectal cancer | Per-tunnel super minimally invasive resection | Per-tunnel endoscopic resection of early colorectal cancer |
| Esophageal stromal tumor | Per-tunnel super minimally invasive resection | Per-tunnel endoscopic resection of esophageal stromal tumors |
| Submucosal mass at the esophagogastric junction | Per-tunnel super minimally invasive resection | Per-tunnel endoscopic resection of the submucosal mass at the esophagogastric junction |
| Small intestinal submucosal mass | Per-tunnel super minimally invasive resection | Per-tunnel endoscopic resection of the submucosal mass of the small intestine |
| Prepyloric submucosal mass | Per-tunnel super minimally invasive resection | Per-tunnel endoscopic resection of the prepyloric submucosal mass |
| Achalasia | Per-tunnel super minimally invasive incision | Per-tunnel endoscopic myotomy of the lower esophageal sphincter to treat achalasia, so as to relieve swallowing difficulties |
| Gastroparesis | Per-tunnel super minimally invasive incision | Per-tunnel endoscopic myotomy of the pyloric sphincter to treat gastroparesis |
| Esophageal diverticulum | Per-tunnel super minimally invasive incision | Per-tunnel penetration of the submucosal layer of the lower edge of the esophageal diverticulum and incision of the muscle layer of the ridge, so as to cure the disease |
TAB. 3.11 — Surgery via puncture channel.
| Disease | SMIS Item | Definition |
| Encapsulated effusion | Per-puncture channel super minimally invasive drainage | Encapsulated effusion aimed at preserving the integrity of the original tissue and the anatomical structure of the organ while achieving the objective of curing a disease |
| Pancreatic pseudocyst | Per-puncture channel super minimally invasive drainage | Endoscopic ultrasound-guided insertion of a puncture needle into the pseudocyst, fitting of a drainage stent in the cyst using a guide wire, to perform a gastrostomy or a cystoduodenostomy, to drain the internal cystic fluid of the pseudocyst |
| Biliary obstruction | Per-puncture channel super minimally invasive drainage | Endoscopic ultrasound-guided puncture of the dilated intra-and extrahepatic bile duct, to establish a channel between the bile duct and the digestive tract with the aid of a stent, to resolve biliary obstruction |
| Pancreatic cystic tumor | Per-puncture channel super minimally invasive ablation | Endoscopic ultrasound-guided entry into the pancreatic cystic tumor by puncturing the wall of the digestive tract to carry out chemical or physical ablation therapies |
| Gallbladder stone | Per-puncture channel super minimally invasive extraction | Endoscopic ultrasound-guided entry into the gallbladder to perform stone extraction |
| Encapsulated necrosis | Per-puncture channel super minimally invasive extraction | Endoscopic ultrasound-guided penetration of the gastric wall or the duodenal wall to enter the necrotic area, to establish a channel between the necrotic area and the wall of the digestive tract, dilatation using a balloon dilator to allow the endoscope to enter, and removal of the necrotic tissues by applying several tools such as a basket, foreign body forceps, a snare, etc. |
| Mediastinal, abdominal, pelvic abscess | Per-puncture channel super minimally invasive drainage | Endoscopic ultrasound-guided entry into the abscess site by puncturing the wall of the digestive tract for drainage |
TAB. 3.12 — Surgery via multi-cavity channel.
| Disease | SMIS Item | Definition |
| Gastric stromal tumor | Per-multi-cavity-channel super minimally invasive resection | Peroral insertion of the gastroscope to perform the operation through a natural channel, the percutaneous puncture channel allowing the laparoscope to be inserted, in order to resect a gastric stromal tumor by the combination of the two endoscopes. |
| Early gastric cancer | Per-multi-cavity-channel super minimally invasive resection | Peroral insertion of the gastroscope to perform the operation through a natural channel, the percutaneous puncture channel allowing the laparoscope to be inserted, in order to resect the early gastric cancer foci and the D1 lymph nodes station by the combination of the two endoscopes. |
| Early colorectal cancer | Per-multi-cavity-channel super minimally invasive resection | Peroral insertion of the enteroscope to perform the operation through a natural channel, in order to resect the early colorectal cancer foci, the percutaneous puncture channel allowing the laparoscope to enter to carry out assisted resection and suture |
| Colorectal large polyp | Per-multi-cavity-channel super minimally invasive resection | Peroral insertion of the enteroscope to perform the operation through a natural channel, in order to resect the early colorectal cancer foci, the percutaneous puncture channel allowing the laparoscope to enter to carry out assisted resection and suture |
| Esophageal large muscularis propria mass | Per-multi-cavity-channel super minimally invasive resection | Peroral insertion of the gastroscope to perform the operation through a tunnel channel, in order to dissect the muscularis propria mass in the tunnel, the percutaneous puncture channel allowing the thoracoscope to enter to carry out assisted resection and suture |
| Biliary stone | Per-multi-cavity-channel super minimally invasive extract | Surgery using a percutaneous puncture channel to insert the laparoscope, and the natural channel of the bile duct to insert the choledochoscope, and combining the two endoscopes for the extraction of a biliary stone |
References
[1] Linghu E. (2023) Interpretation of the terminology of super minimally invasive surgery in the national terminology committee’s digestive endoscopy terminology, Chin. J. Gastrol. Endosc. (Electron. Ed.) 10, F3.
[2] Digestive Endoscopy Terminology Examination Subcommittee. (2021) Digestive endoscopy terminology. Science Press, Beijing.
[3] Linghu E. (2022) A new stage of surgical treatment: super minimally invasive surgery, Chin. Med. J. (Engl.) 135, 1–3.
[4] Linghu E. Q. (2024) New direction for surgery: super minimally invasive surgery, World J. Gastrointest. 30, 1676–1679.
Chapter 4 Common Super Minimally Invasive Surgery Operations
4.1 Super Minimally Invasive Closure
These are surgeries carried out when diseases require surgical interventions or diseases with long-term unsatisfactory medication curative effect, which aim to close lesions by endoscopic closing while preserving the integrity of the human anatomical structure and achieving the objective of curing a disease. So far, they mainly include endoscopic closure with metal clips, endoscopic suturing, endoscopic purse-string suturing with a nylon rope, and endoscopic sealing with fibrin glue, etc.
4.1.1 SMIS Wound Closure Method
(I) Endoscopic closure with metal clips (figure 4.1).


FIG. 4.1 — An endoscopic metal clip-closure system.
Endoscopic closure with metal clips is the most widely used for closing gastrointestinal perforations. So far, the most widely used in clinical practice is the per-endoscopic forceps with metal clips, the most common being Olympus metal clip, Boston metal clip, Nanjing minimally invasive SureClip, etc. In a specific operation, the metal clip must be applied gradually from one side of the perforation towards its center, with the two legs of the clip close to the mucosa on either side of the perforation site, and the area of the lesion must be at an angle of 60–90-degrees. The clip must be straight after being successfully positioned.
The characteristics of the metal clips are as follows:
they are simple and convenient to use;
their weak closing force generally only allows the mucosal layer to be closed rather than the full thickness of the digestive tract wall. For large perforations, clamping is often ineffective, as the clip may fall off;
in some cases where the lesions are difficult to visualize completely, such as perforations of the descending junction of the duodenum, manipulation is difficult or even impossible, and capsule protection is helpful to complete the operation if necessary. To improve suture efficiency, Ethicon Endo-Surgery has developed a multiple-clamp release device, which can release multiple metal clamps at once to act on the target tissues. Its clinical effect has yet to be verified.
Ovesco Endoscopy AG launched the Over-The-Scope-Clipsys (OTSC), and some endoscopic surgery centers in China have applied it. The operation procedure is as follows: the OTSC is installed at the front end of the endoscope, the paracentral tissue is grasped by the matching grasping forceps, combined with vacuum aspiration, the lesion and its surrounding tissues are placed in a cap at the front end of the endoscope, the OTSC is then released using a rotary trigger system, then the OTSC is released from the cap, the tissues are quickly occluded together to close the perforation. This device can effectively close perforations with a diameter of 3 cm, and its seaming force is obviously greater than that of normal metal clips. It can close the full thickness of the wall of the digestive tract to reduce the risk of localized leakage. But this device has also some drawbacks: the endoscope must be withdrawn to install the OTSC, the clip falling time is undetermined, and it is expensive.
(II) Endoscopic suturing
Endoscopic suturing uses a device for suturing the wall of the digestive tract.
The endoscopic sutures currently available in the market for closing perforations and fistulas include Over Stitch by Apollo Endosurgery (figure 4.2). It mimics surgical suture technology by first grasping the tissue with an anchor device, followed by penetrating it on one side with a curved needle fitted with suture thread, before finally capturing the suture thread on the opposite side. Through repeated operations, it can complete the intermittent or continuous suture of the tissue, and finally a special device is used to fix the suture. The perforation closure process must be completed without tying. Compared to metal clips, it can provide a more effective and reliable suturing effect, but its operational process is complicated and lacks flexibility while being costly. It is not widely applied clinically. Recently, Osamu Goto et al. reported a new suturing device in the magazine Endoscopy. They inserted a curved needle with sutures and a special needle-holding device into the body and sutured in vitro the wound in the digestive tract. The process was similar to surgical suturing and good results have been obtained in animal and human studies. In addition, a variety of special suture instruments for use on animals or humans are currently being studied.

FIG. 4.2 — Overstitch device.
(III) Endoscopic purse-string suturing with a nylon rope consists of the endoscopic application of multiple metal clips to fix the nylon rope to the surrounding of the perforations or defects in the digestive tract, and subsequently to tighten the nylon rope to carry out closure.
Titanium clip suturing is the most widely used endoscopic wound-suture technique. However, the opening angle of the titanium clip arm is limited, and it is sometimes difficult to use a titanium clip alone if the wound is large. In 2004, Matsuda et al. in Japan first introduced a method using a metal clip combined with purse-string suture with nylon rope to close a giant wound after endoscopic mucosal resection. This technique has been used to suture defects in the wall of the digestive tract after full-thickness resection and ESD perforation, and very good results have been obtained. As for purse-string suture after full-thickness resection, the related animal study showed that a perforated surface in the gastric wall with a diameter of 20 mm was sutured by using purse-string suture with nylon rope; both endoscopic and imaging examinations within two weeks after the operation indicated that the perforation healed satisfactorily with good apposition of the mucosal, muscular and serosal layers, which demonstrated the histological reliability of endoscopic purse-string wound closure.
The nylon rope suturing techniques reported here are as follows:
Nylon rope reinforcement after metal-clip suturing;
Metal clip combined with purse-string suture with nylon rope;
Metal clip combined with purse-string intermittent suture with nylon rope;
these techniques are again divided into double-forceps channel method and single-forceps channel method, depending on the manner the endoscope is used. They will be presented below.
1. Nylon rope reinforcement after metal clip suturing (figure 4.3). This method is generally used for wound clamping by means of a metal clip after full-thickness resection and ESD perforation. It can be used if one is unsatisfied with metal clip suture or worries that the metal clips will fall out. By using a double-forceps channel endoscope, the foreign-body forceps are inserted into one hole and the nylon rope into the other. The nylon rope is stretched, the foreign body forceps was passed through the nylon rope string to widen it and clamp the sutured wound, which is lifted up to make the mucosa around the wounds swell; the nylon rope string is tightened, and one tries to wrap all sutured wounds in the nylon rope. Although this method reinforces the suture, care and caution as well as moderate force are needed in the operating process, so as to avoid rupture of the wound after it has been sutured by the metal clip. Someone also used a single-forceps endoscope for nylon rope reinforcement. By suction, this makes the mucosa relax and swell around the wound, which then can be ligatured with a loop. Or one can make a loop using only the metal clip as a support point, tightening slowly, making the metal clips on the wound to come together, to obtain a reinforcement effect.

FIG. 4.3 — Realization of a suture reinforced by a nylon rope following a suture made using an SMIS metal clip.
2. Metal clip combined with purse-string suture with nylon rope (figure 4.4). This method is the most economic and effective method so far for suturing defects in the wall of the digestive tract after endoscopic full-thickness resection and ESD perforation. The method of implementing double-forceps endoscopic purse-string suture in a defective digestive tract is as follows: the nylon rope is sent through one forceps channel, the nylon rope loop string is stretched, its position is adjusted to fit the wound; the titanium clip is sent into the other forceps channel of the endoscope, and the nylon rope is anchored to the first titanium clip at the edge of the wound, and one tries to make the titanium clip solidly fixed. After that, titanium clips are inserted and the above steps are repeated until the titanium clips anchored at the edge of the wound are evenly distributed over its entire surface, with the goal of achieving equal spacing and bilateral symmetry; then the nylon rope loop string is tightened to completely close the wound, and several pieces of tightened titanium clip piling are found by endoscopy. There is no need for a lot of titanium clips; otherwise there would be a risk of the sutured wound spreading after tightening the nylon cord, which would affect the healing of the wound.

FIG. 4.4 — SMIS suturing method using a metal clip combined with a nylon rope.
It is controversial as to whether purse-string suture can reach the objective of full-thickness suture. The endoscopic metal clip combined with purse-string suturing with nylon rope is an effective method of closing the defects in the wall of the digestive tract, ensuring the suture of the mucosal surface. But, as with metal clip suture, it cannot reach the suture over its full thickness, including the muscle layer and the serous layer.
In addition, in the case of endoscopic metal clip combined with purse-string suturing with nylon rope, postoperative gastrointestinal decompression needs to be supplemented to make the suture more effective. Or there would be a lot of gas in the digestive lumen, and a delayed rupture of the sutured wound and a fall of the metal clip could occur after the tension in the tube wall increases. The double-forceps channel endoscopic metal clip combined with purse-string suture with nylon rope has always been the powerful guarantee and the strength for major hospitals in suturing wounds after endoscopic full-thickness resection. But it is also because of the limitation of the double-forceps channel endoscopic application, making that endoscopic full-thickness resection cannot be widely performed for a long time.
3. Single-forceps channel SMIS purse-string suturing. For the single-forceps channel SMIS purse-string suture, the method consists of using the nylon rope release, after separating the nylon rope loop string from the nylon rope push release device; the nylon rope loop string is first placed in the gastrointestinal cavity through the forceps channel of the single-forceps channel endoscope or with the clamping assistance of the biopsy forceps, then the nylon rope loop string is put in the gastrointestinal cavity first, and then the metal clip release device is inserted through the forceps channel to make the nylon rope to be clamped and fixed on the digestive tract wall of the defect edge with the metal clip. Then the nylon rope push release device is stretched into the forceps channel, the root of the nylon rope is hooked and tightened, and the metal clip is centered by lifting it slightly. This method has solved the issue of the single-forceps endoscopic purse-string suture.
4. Metal clip combined with nylon rope intermittent suturing method (figure 4.5). The nylon rope and the first titanium clip are inserted through the double-forceps channel of the endoscope. The nylon rope and the titanium clip are adjusted to the proper angles and directions, the distal end of the nylon rope is clamped by using the first piece of titanium clip, one tries to press firmly against the wall of the digestive tract over the full thickness of the defective distal end edge to clamp and fix it at a vertical angle. Insert the second titanium clip, clamp and hold the proximal nylon rope, and secure it to the digestive tract wall near the edge of the defect. The nylon rope is closed and narrowed, the distal side of the wound and the proximal side of the defect are pulled together. The above steps are repeated if necessary, the wound is closed completely. The residual wound can also be further closed by simply adding several titanium clips sequentially. Then the gastric tube is placed for decompression.
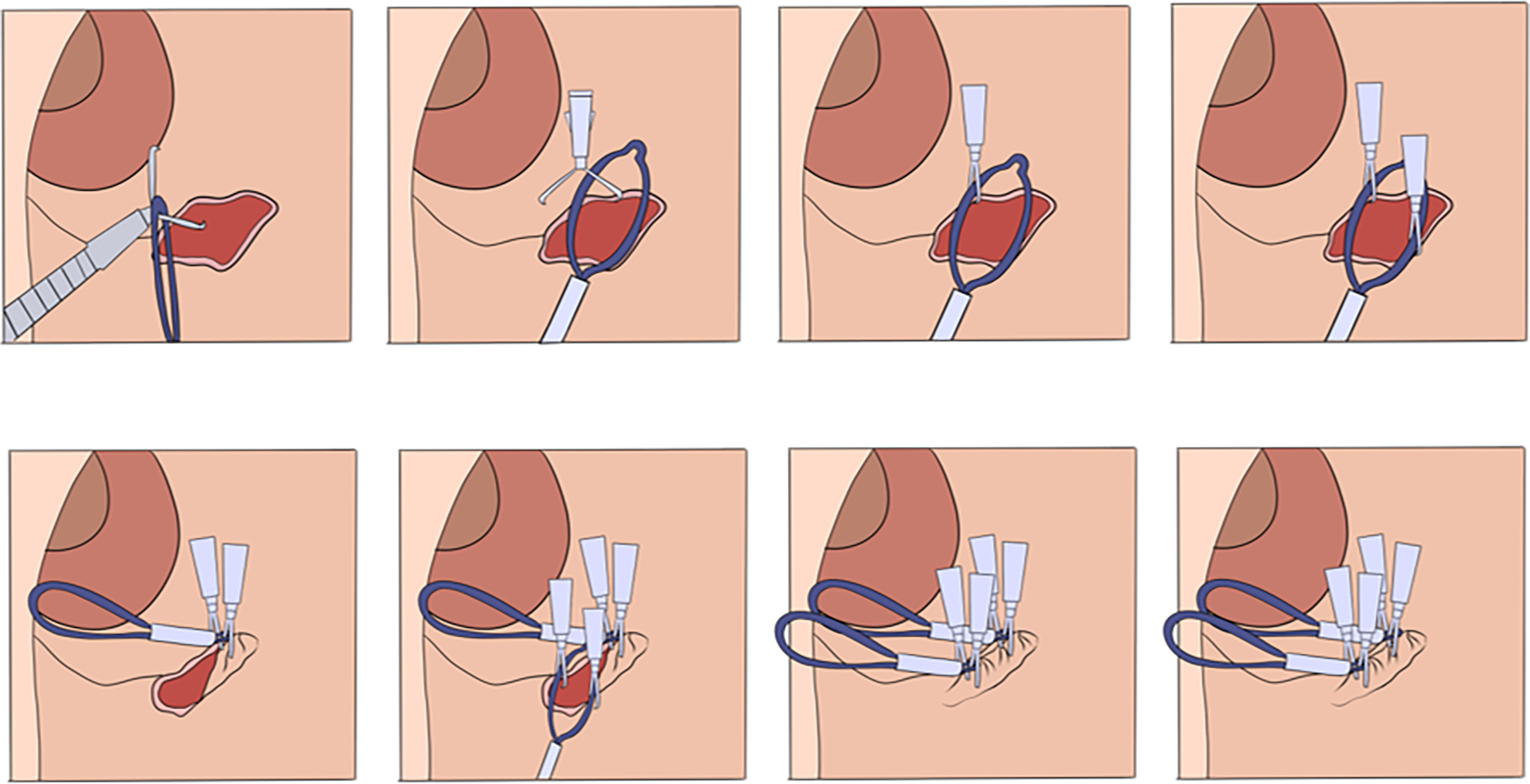

FIG. 4.5 — SMIS method of intermittent suturing using a metal clip combined with a nylon rope.
For patients with more severe pneumoperitoneum, the abdominal puncture needle needs to be inserted into the right lower part of the abdomen in order to evacuate the gases during and after the operation and to reduce postoperative abdominal distension. As this method is similar to intermittent suturing in surgery, it is also called ‘intermittent suture’ or ‘∞-shaped’ suture. In the traditional double-forceps endoscopic purse-string suturing process, starting from the third piece of metal clip, as the two forceps channels are in the same endoscope, it would increase the difficulty of operation. And compared to purse-string suturing, the intermittent suturing with a metal clip combined with a nylon rope is fixed with only two metal clips for each nylon rope, greatly reducing the difficulty of the stitching. So far, with the development of the single-forceps channel endoscopic purse-string suture technique, intermittent suture with a metal clip combined with a nylon rope can also be performed using the single-forceps endoscope.
(IV) Endoscopic closing with fibrin sealant is an endoscopic technique using bioprotein glue to seal smaller fistulas.
There are two kinds of tissue glue most commonly used in clinics: one is biofibrin glue and the other is cyanoacrylate. Depending on the component, biofibrin glue is made of two solutions, one containing human fibrinogen and aprotinin, and the other containing thrombin and calcium chloride. After the two solutions are mixed through the double-cavity channel, a layer of fibrin membrane can be formed on the surface of the fistula, which is similar to the process of hemostasis in the body, so as to achieve the effect of sealing. Cyanoacrylate polymerizes upon contact with water, leading to tissue necrosis and inflammatory reactions. It is not affected by digestive enzymes, and has also antibacterial properties, and can be applied to infected sites. In a recent study, the anastomotic fistula was successfully closed in 61 out of 63 patients by endoscopy using tissue glue, and the closing success rate reached 96.8%, the median amount of bioprotein gel used was 12 ml, the median cyanoacrylate usage was 1.5 ml, the median length of stay after initial treatment was 14 days; of the two patients in whom the treatment failed, one was an elderly female patient with an enterocutaneous fistula after small intestine necrosis, and the other was an obese woman having undergone sleeve gastrectomy; after three fibrin treatments failed, it was decided to treat her surgically. For high-discharge digestive tract fistulas, it is recommended to combine tissue glue with the endoclip or stent to improve the success rate of fistula closure. Victorzon et al. treated six patients with gastrojejunal fistula with tissue glue and stent under endoscopy; the success rate reached 100%, and there was no fistula-related death. Merrifield et al. combined argon plasma coagulation, hemostatic clamp, fibrin glue, stent placement, and distal gastrojejunal stricture; dilation was used to repair chronic gastric fistulas in patients after Roux-en-Y gastric bypass surgery; all three patients saw a complete alleviation of their clinical symptoms, and the treatment was less invasive than with traditional surgery.
(V) Modified double seam suturing method
The modified double seam suturing method is also called “folding paper method,” and is used for the treatment of wound closure in colorectal endoscopic submucosal dissection. It is difficult to use a simple per-endoscopic hemostatic clamp for hemostasis after endoscopic dissection of the colon, especially for larger wounds. So a research team developed a new endoscopic closure technique—modified double seam suturing, used for the treatment of wounds in endoscopic submucosal dissection. This method is also called folding paper method. Double-seam suturing is a technique in which hemostatic clamps are used to clamp the mucous layer while occluding the mucosal muscle layer.
The folding paper method generally applies to the large defects cases of more than 20 mm, especially for patients with a high risk of bleeding and perforation.
The specific operating steps are as follows:
A reopenable clip is used to grip the oral side of the muscle layer along the long axis of the wound, while attracting air to reduce muscle tension and folding the muscle layer along the long axis.
Two reopenable clips grip the anal side of the mucosal defect, and the other side of the muscle layer is also folded.
Reopening clamps are used to connect these folded muscle layers to reduce wound contraction.
A reopenable clip is used to connect the folded muscle layer to the normal mucosa at the edge of the wound along the long axis; notice that the clip should not be buried under the wound. Studies have shown that this closure technique is clinically feasible, with good closure effect, and that large wounds can be closed with a single hemostatic clamp, thus reducing trauma.
(VI) Submucosal reopenable-clip-over-the-line pocket method to perform full-thickness closure
Colorectal endoscopic submucosal dissection can be generally clinically used for performing the complete resection of the mass. However, some tumors, due to their special shape and location, will be stripped of the muscle layer during dissection, resulting in perforation. Therefore, some researchers innovatively adopted the reopenable-clip-over-the-line method inside a submucosal pocket (ROLM–SP). This method can be used to close large full-thickness defects in the third space.
The specific operating steps are as follows:
A clip with a wire is inserted through the auxiliary channel and placed on the muscle layer at the edge of the full-thickness defect.
The thread through the auxiliary channel passes through the hole on one side of the clip, and then the clip is placed on the muscle on the opposite side edge of the full-thickness defect.
The clip with the thread is repeatedly placed on the muscle at the edge of the defect through the hole, resulting in gradual closure of the defect. Finally, the full-thickness defect is completely closed in the submucosal pocket, and the remaining lesion mucosa is removed, and finally the tumor is successfully removed. At the moment, this technique still needs more-in-depth clinical studies to confirm its efficacy and safety.
(VII) Reopenable-clip-over-the-line method to perform full-thickness enclosure
This technique is used for wound enclosure after endoscopic full-thickness resection. Presently, the treatment of gastric stromal tumors must focus entirely on the risk of tumor injury and the excision margin. A research team proposed a non-contact endoscopic full-thickness resection (EFTR). During the resection of the gastric stromal tumor, the incisal margin of this technique is 0.5–1.0 cm from the muscularis propria.
The specific operating process is as follows:
Marks are made around the marginal line of the submucosal tumor protrusion, and submucosal injection will not be conducted for circumferential mucosal incisions outside markings.
Deep mucosal/submucosal furrows are formed on the surface of the muscularis propria. The anal side of the lesion must be tracted with a clamp thread (3-0 polyester suture).
The muscular layer is incised from the anal side along the mucosa/submucosal sulcus, and the defect is closed by using a 4-0 nylon suture with a reopenable clip. Finally, the foci are restored.
Research was conducted for the retrospective analysis of endoscopic full-thickness resection without contact of gastric stromal tumors, which finally concluded that this technique is clinically feasible, and can be expected to achieve radical excision. However, some risk factors can affect the success rate of this technique, particularly large size, intracavitary growth, etc.
(VIII) Suture-assisted complete suturing method
This technique is used to close the wound after full-thickness or non-full-thickness resection of wounds, by applying surgical threads and tissue clips to close them. The specific operating steps (figure 4.6) are as follows:
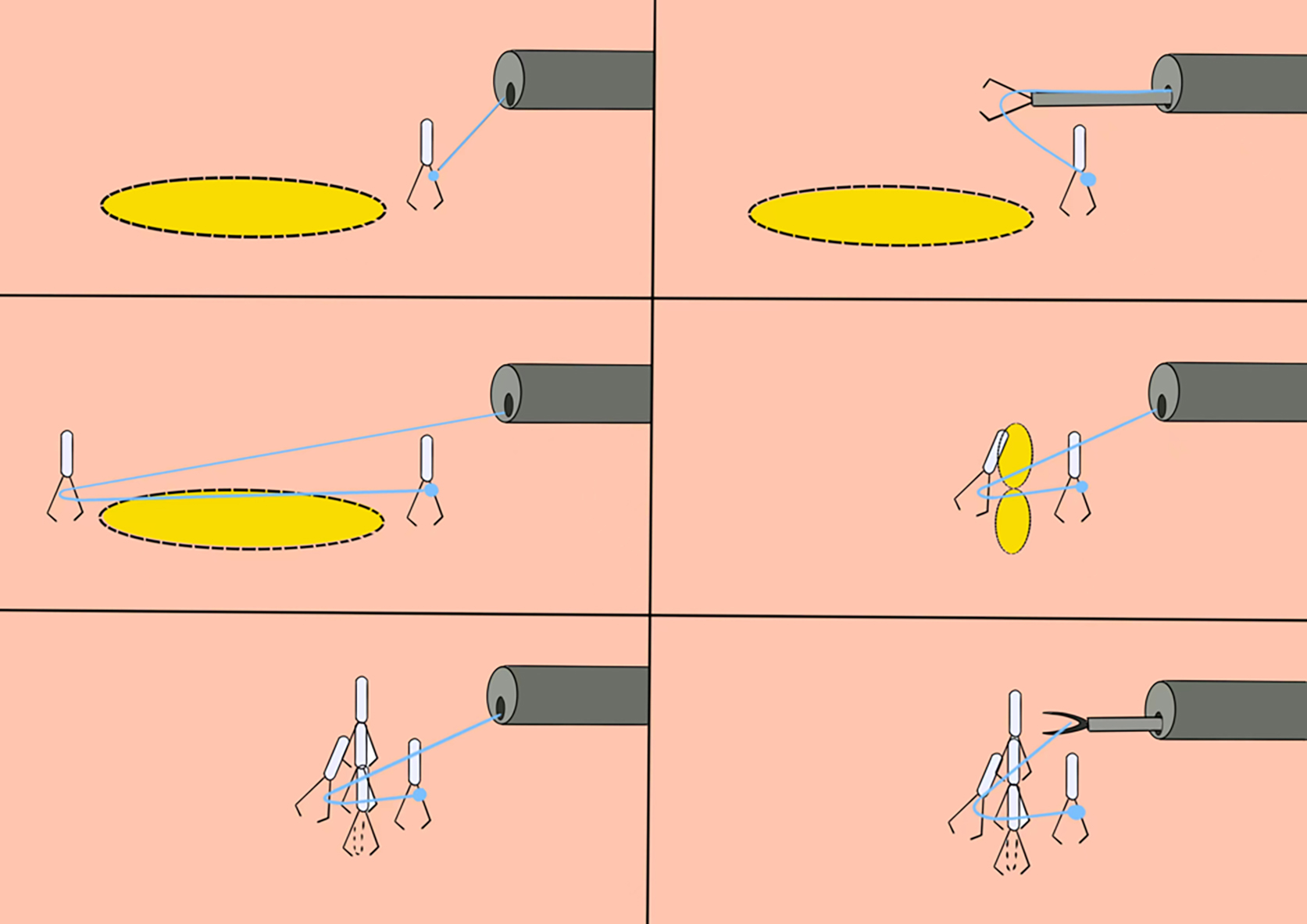

FIG. 4.6 — SMIS suture-assisted complete suturing method.
The tissue-holder with a thread protruding from the endoscopic forceps channel is fixed on the oral side of the mucosa.
The first tissue-holder is fixed with a thread to the anal side mucosa with the second tissue clips (without thread).
The thread is pulled to make the tissue clips closer.
The suture is reinforced, the tissue-holder (without thread) is added for occlusion.
The thread is cut off with endoscopic special scissors after full occlusion.
4.1.2 Hot-Spot Issues and Progress in the Search for a Consensus
(I) An absolutely safe incision closure technique is still the hot-spot topic of study.
With the wide application of the endoscopic treatments like endoscopic submucosal dissection (ESD) and endoscopic full-thickness resection (EFTR), etc., the endoscopic digestive tract local resection technique has become gradually mature. Endoscopic submucosal dissection is an effective endoscopic method for the treatment of gastrointestinal tumors, with high possibility of complete resection. However, adverse events of postoperative bleeding and delayed perforation still exist, one possible reason being that large mucosal defects remain open after ESD. For iatrogenic defects caused by artificial conditions, absolutely safe and effective endoscopic closure is required. To prevent or minimize this, it is needed to focus several techniques for endoscopic mucosal closure.
At present, the endoscopic enclosure techniques mainly include endoscopic metal clip closure, endoscopic suturing, endoscopic purse-string suturing with nylon rope and endoscopic closing with fibrin sealant, etc. Any of the above methods can close a single perforation site fairly easily. However, if two or more instruments pass through the gastric wall at different locations, it can create shear forces and complicate closure. The closing technique for large artificial soft incisions, fistulas and perforations of the digestive tract is still the bottleneck of endoscopic therapy so far. Therefore, the evaluation of the integrity of the closure for the digestive tract must form part of the intraoperative protocol to guarantee the safety of SMIS.
(II) Limitations of the present endoscopic enclosure method
Using through-the scope clips (TTSC) to clamp iatrogenic perforation is the most commonly used and technically mature closure method; it is cheap and simple to operate. But the TTSC is relatively small, the opening distance is limited, and the closing force is insufficient. The European Society of Gastrointestinal Endoscopy (ESGE) recommends that TTSC closure can be used alone for perforations less than 10 mm; for perforations greater than 10 mm, they suggest to consider combining it with the use of nylon rope for closure. TTSC combined with nylon rope to enclose full-thickness defects in the gastrointestinal wall can be carried out by double-forceps channel endoscopy or single-forceps channel endoscopy. The closure of the perforation using a single-forceps channel endoscope is technically difficult, tedious to operate and causes significant interference, which increases the operation time, and there is also a risk of suture failure if the nylon rope fails to release the device for hooking the nylon rope; conversely, treatment with the double-forceps channel endoscope is highly demanding for hospitals and endoscopists, and the ordinary grassroots hospitals are rarely equipped with double-forceps channel endoscope, so the implementation of this technique is restricted. Owing to its strong enclosure force, OTSC can achieve closure of the full thickness of the digestive tract wall. But the price factor limits the wide application of OTSC. Endoscopic suturing devices like Over Stitch, etc., have broad application prospects and huge development space, but they require doctors to have a high professional level and to follow a special training. In addition, further prospective RCT studies are still needed to determine the long-term efficacy and safety of the technique.
In recent years, through the continuous innovation and development of gastrointestinal endoscopy diagnosis and treatment technology, super minimally invasive closure technology has made breakthrough progress, but the existing closure technology still has room for improvement, and relevant devices need to be developed. There is an urgent need for a convenient, safe, and reliable endoscopic artificial incision closure technology that can be applied in clinical treatment, so that the super minimally invasive diagnostic and treatment technology using gastrointestinal endoscopy can reach new heights.
4.2 Super Minimally Invasive Surgery for the Prevention and Therapy of Gastrointestinal Bleeding
Super minimally invasive surgery for the prevention and therapy of gastrointestinal bleeding is a technique of pretreatment or hemostatic treatment at the lesion sites that might have bleeding or are bleeding. At present, it mainly includes endoscopic drug spraying hemostasis, endoscopic hemostasis by injection, endoscopic hemostasis by thermocoagulation, endoscopic electrocoagulation hemostasis, and electroscopic metal clip hemostasis, etc. The latter is the same as electroscopic metal clip closure, and therefore will not be explained here.
4.2.1 Endoscopic Common Super Minimally Invasive Hemostatic Method
(I) Endoscopic drug-spraying hemostasis
Endoscopic drug-spraying hemostasis is a technique of endoscopic direct spraying of norepinephrine, epinephrine, and of other coagulation drugs to stop bleeding in the gastrointestinal tract.
This method has been widely used in clinics. Appropriate hemostatic drugs are sprayed on the bleeding focus through the endoscopic biopsy orifice to stanch the bleeding. Commonly used for cases of mucosal erosion and bleeding, tumor rupture and bleeding, and situations where the area is large but the amount of bleeding is not significant. The commonly used hemostatic drugs include thrombin for injection, norepinephrine solution, epinephrine and medical adhesives. After spraying, the bleeding of the patient’s bleeding focus should be observed again. If there is a lot of blood in the hemorrhagic foci and the surgical field is unclear, rinsing should be carried out to obtain a good visual field.
(II) Endoscopic hemostasis by injection
Endoscopic hemostasis by injection is a technique involving the use of an injection needle to puncture through the mucosa and the submucosal layer to inject drugs for hemostasis.
The injection drugs mainly include diluted epinephrine, hardener, pure alcohol, thrombin, saline, hypertonic saline and hypertonic glucose solutions, etc. This technique is a therapeutic method using an endoscope through a special injection needle to inject the drug into the hemorrhagic foci or their periphery, to reach hemostasis or prevent further bleeding. Endoscopic hemostasis by injection can be applied to the hemorrhagic foci from the esophagus to the rectum. It involves:
tamponade (hemostasis is achieved due to the drugs chosen to have the effect of pressing on the bleeding vessels, such as saline solutions);
vasoconstriction: hemostasis is achieved due to well-chosen drugs (for instance, diluted adrenaline causes the constriction of the arteriole that is bleeding);
sclerosis (the sclerosant carries out venous embolization for hemostasis);
tissue dehydration [high concentrations of alcohol (more than 98%) can dehydrate the tissues, leading to tissue degeneration at the injection site, as well as congestion and redness around the latter, and clotting of blood vessels—in addition, hypertonic salt solution (3%) and 50% dextrose solution act in the same way for hemostasis].
(III) Endoscopic electrocoagulation hemostasis
Endoscopic electrocoagulation hemostasis is an endoscopic technique in which the electrode is placed in contact with the tissue of the bleeding part, and the local tissue protein is coagulated; the blood vessel is blocked by the thermal effect generated by the high-frequency current, so as to achieve the hemostatic effect.
High-frequency electrocoagulation applied to the surface of the bleeding lesion produces a thermal effect, which leads to tissue necrosis, coagulation, and hemostasis. It mainly applies to projectile bleeding, active bleeding or bleeding with exposure of hemispherical vessels, such as peptic ulcer, polyp or wound oozing after electroresection of the submucous mass.
During hemostasis, the lesion area should be washed and cleaned, the liquid in and around the diseased area be fully absorbed, and hemorrhagic foci such as blood vessels be exposed. Then, the electrocoagulator connected with the high-frequency electrical generator is inserted through the endoscopic treatment channel, the electrocoagulation probe is vertically aligned with the blood focus, the probe is gently pressed in its central part, and an appropriate amount of water is injected, and the intensity of the coagulation current is selected. The electrocoagulator probe is energized immediately when it touches the target tissue, until the tissue at the electrocoagulation site turns white. After the hemostatic effect has been obtained, the electrocoagulation probe is withdrawn, and then injecting a proper amount of water into the foci through the water injection hole makes it possible to verify whether hemostasis is complete.
Argon plasma coagulation (APC) is a non-contact electrocoagulation technique in which argon, an inert gas, is ionized into a conductive argon plasma when excited by high-frequency electricity. Argon plasma can achieve the effects of drying, coagulating and inactivating the target tissue through the thermal effect of a high frequency current. When the surface of the diseased tissue is dried and solidified, the electrical conductivity decreases and the impedance increases, and the argon plasma will automatically flow from the place with high impedance to the surrounding tissue with low impedance. Therefore, the solidification range of APC is large and shallow, and the solidification depth can be controlled at about 3 mm through the adjustment of parameters such as time, power, and distance, which is a safe, fast and effective non-contact electric coagulation technique.
(IV) Endoscopic hemostasis by thermocoagulation
Endoscopic hemostasis by thermocoagulation is an endoscopic technique using physical methods as electrocoagulation, laser and microwave application, etc., by generating a thermal effect in the local tissue to realize protein coagulation, blood vessel occlusion, so as to achieve hemostasis.
This technology mainly produces a thermal effect through three forms 1. electrocoagulation, 2. laser and 3. microwave, so as to make proteins and block blood vessels coagulate, so as to achieve hemostasis. An in-depth introduction to the commonly used electrocoagulation hemostasis has been made above, so this part focuses on laser hemostasis and microwave hemostasis.
Endoscopic laser hemostasis is a method using a laser coagulation effect. By using the heating effect induced by light energy, it provokes evaporation of cell moisture, protein coagulation, degeneration, collagen fiber contracture, small blood vessel contraction and closure, which results in mechanical occlusion or intimal thrombosis of small blood vessels. In addition, laser has a certain pressure and impact effect, which can achieve hemostasis. A quartz fiber is inserted through the biopsy hole after the bleeding part has been evidenced during the operation, the laser is diverted and radiated at 0.5–2.0 cm from the lesion site. For patients with a bleeding ulcer, the edge of the ulcer and its bleeding part are irradiated; for patients with hemorrhage of bare blood vessel, the peripheric tissues of the vessels are first radiated, then the vessels; for patients with bleeding tumors, all blood effusion sites and edge tissues on the surface of the tumor tissues should be coagulated. Observation must be continued for 25 min after treatment. Then the endoscope is withdrawn if there is no active bleeding. Until now, this procedure has not been widely used in clinical applications.
Endoscopic microwave hemostasis is a technique of treatment using microwaves to generate heat, making the tissue proteins and vessels coagulate, for the purpose of hemostasis. This surgery is intended for the selective operation of the digestive system, only making the surface mucosa coagulate, without damaging the muscular layer. The hemostatic effect can be achieved in a short time, with a simple operation, and it is not harmful to the human body. It applies to patients with superficial mucosal erosion or ulcer bleeding, small vessel malformation bleeding, and bleeding after duodenal papillectomy. During the operation, the microwave electrode probe is inserted from the endoscopic biopsy hole, it is aligned with the hemorrhagic foci, and the probe is pushed up to 2–3 mm from the front end of the endoscope. The erosive lesion can be treated by the method of plain scan, with spot electric coagulation of the broken and hemorrhagic blood vessels directly performed at the bleeding spots, or it might be directly punctured into the hemorrhagic foci for intramucosal coagulation therapy. Cauterization should be repeated until the tissue of the lesion turns yellow, white, atrophied, and solidified. After endoscopic assessment of the absence of bleeding, the endoscope is withdrawn.
4.2.2 Hot-Spot Issues in the Consensus and Research Progress
The timing of endoscopy and the selection of surgical methods in patients with acute upper gastrointestinal bleeding are still a hot topic of clinical discussion. Acute gastrointestinal bleeding is a common emergency, including acute upper gastrointestinal bleeding (classified into acute non-varicose upper gastrointestinal bleeding and acute esophagogastric varicose bleeding) and acute lower gastrointestinal bleeding. In recent years, with the rapid development of endoscopic treatment techniques, significant efficacy has been obtained in the emergency hemostasis and prevention of bleeding for various cases of patients with acute gastrointestinal bleeding.
Depending on the length of time the patient has been waiting, endoscopy of acute upper gastrointestinal bleeding can be divided into emergency (within 6–12 h), early (12–24 h) and delayed (exceeding 24 h) cases. In 2021, the book Clinical Guideline: Upper Gastrointestinal and Ulcer Bleeding by the American College of Gastroenterology recommended that hospitalization of patients with upper gastrointestinal bleeding (UGIB) be followed by endoscopy within 24 h of their admission (extremely low-quality evidence, conditionally recommended). For low-risk patients (with stable hemodynamics and without serious comorbidities), numerous studies have all confirmed that performing an endoscopy within 24 h of admission reduces the length of stay, lowers mortality as well as hospitalization costs. For high-risk patients (unstable hemodynamics or liver cirrhosis), due to the very limited number of supporting evidence, the 2012 guideline recommended to perform endoscopy within 12 h of admission, which may improve clinical prognosis. According to the present evidence, it shows that if hemodynamic stability and disease control are not carried out in these patients before endoscopy, endoscopy within 12 h of admission may lead to more adverse consequences. Therefore, the 2021 guideline does no longer set the recommended time for endoscopy in high-risk UGIB patients as being within 12 h of admission. Therefore, it is recommended that both high-risk and low-risk patients undergo endoscopy within 24 h of admission. It is important to note that, for high-risk patients, it is recommended to stabilize hemodynamics before endoscopy and pay attention to the management of active comorbidities.
The expert consensus of the acute upper gastrointestinal emergency treatment flow shows that, for high-risk patients with unstable hemodynamics after resuscitation, if the hemodynamics are stable, endoscopy can be performed within 24 h of admission; if hemodynamic instability persists, emergency endoscopy should be considered. At present, for patients with hemodynamic instability, both Chinese and foreign guidelines recommend that emergency endoscopy should be considered after vital signs have stabilized. However, for patients with persistent hemodynamic instability or for whom this is difficult to fix in a short time, there is currently insufficient evidence on whether emergency endoscopy should be performed as soon as possible under active anti-shock therapy, and further clinical research is needed.
For ulcer-induced UGIB patients, endoscopic hemostasis with bipolar coagulation, thermal probe, or injection of ethanol is recommended (medium quality evidence, strongly recommended). A meta-analysis integrating 15 RCTS found that the use of thermocoagulation (bipolar electrocoagulation, thermal probe) was effective in improving the clinical prognosis (rebleeding or death) of ulcer-induced UGIB patients, with no significant difference in efficacy between bipolar electrocoagulation and thermal probe. Therefore, both bipolar electrocoagulation and thermal probe are recommended for endoscopic hemostatic treatment of UGIB patients caused by ulcers, and their efficacy is similar. There is also evidence showing that injection of tissue hardeners (ethanol) is equally effective in ulcer-induced UGIB patients and reduces the risk of rebleeding or death.
In addition, by comparing bipolar electrocoagulation or thermocoagulation with ethanol injection, the evidence showed that thermocoagulation reduced rebleeding, but there was no significant difference in mortality. For UGIB patients at high risk of acute ulcer bleeding, the international consensus group guidelines strongly recommends endoscopic heat coagulation or injection of sclerotherapy. Therefore, at present, thermocoagulation and injection of tissue hardener are the recommended clinical methods for endoscopic hemostasis. However, the different treatment methods have advantages and disadvantages regarding this. The drawback of drug spray hemostasis and endoscopic injection hemostasis is a high rebleeding rate, so they should not be used as a single therapy, but be combined with other forms of hemostasis. Thermal coagulation includes high-frequency electrocoagulation, argon ion coagulation and other methods, with a good effect on hemostasis, but this depends on the equipment, and requires a high level of skill on the part of the surgeon, and it is not without a certain risk of perforation. Mechanical hemostasis uses various hemostatic clips, its effect is reliable, but some foci are difficult to operate, and the technical competence of the surgeon must be high. Therefore, the specific measures should be selected depending on the operating habits of endoscopists and the maximum benefit for patients.
4.3 Super Minimally Invasive Auxiliary Traction Method
The device that traction technique requires is clip + auxiliary device. Presently the clip is the rotatable reusable soft tissue clips, and the auxiliary device includes surgical sutures, dental floss, snare, magnetic beads, gravity hammer, springs, elastic rings, etc. The common auxiliary traction techniques are: clip–dental floss combination, clip elastic ring combination, as well as other combinations.
4.3.1 Clip–Thread Combined Traction Method
This is an external traction technique (figure 4.7). SureClip is basically used nowadays. Auxiliary equipment includes surgical sutures, dental floss, snare, magnetic beads, gravity hammers, springs, elastic rings, etc. But due to its high risk, dental floss is used now. Dental floss is cheap, easy to get, with moderate tension, and reduces the pressure and risks on sites such as the esophageal inlet, the cardia, thus improving safety and operability.
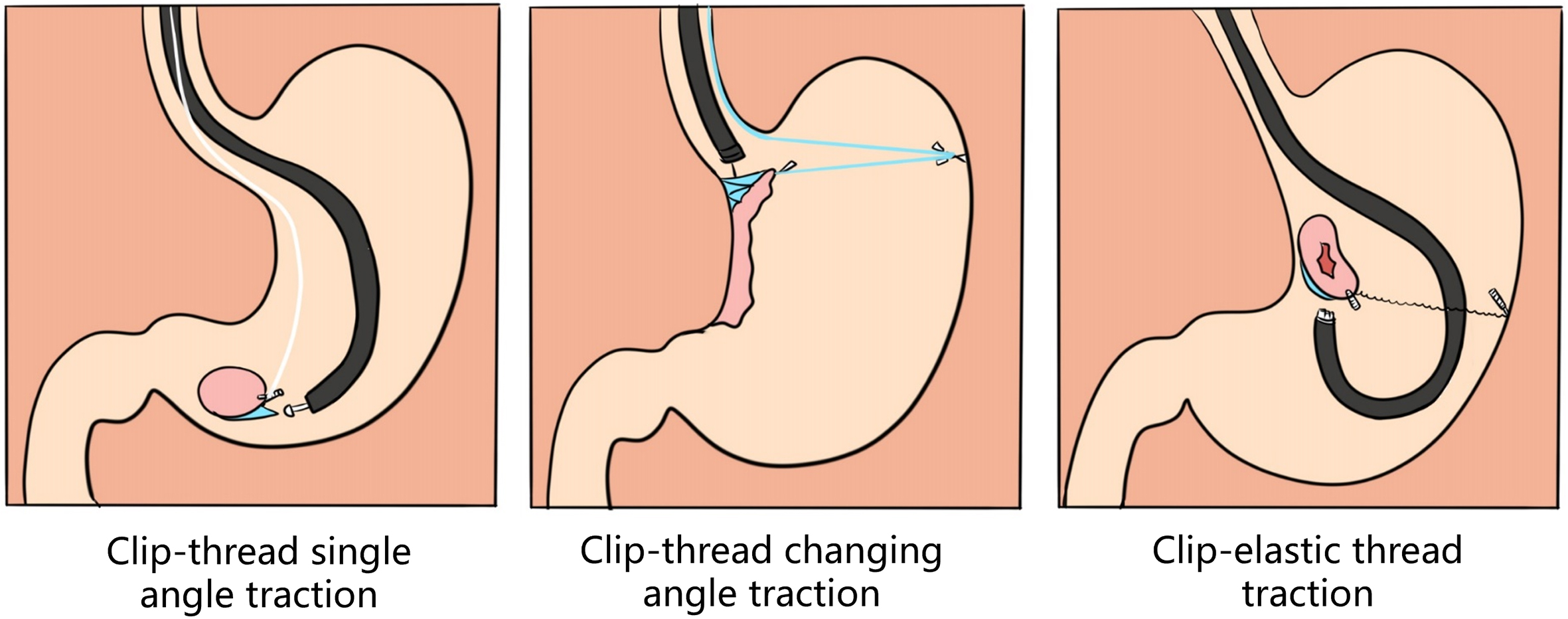

FIG. 4.7 — Clip–thread combined traction method.
Operating method
The clip extends out of the forceps channel, and the mirror body is tied to the root of the corner of the clip with dental floss.
The clip is retracted from the forceps channel.
The endoscope is inserted into the area of the lesion, the edge of the to-be-dissected site is clamped.
The dental floss is externally stretched straight to maintain proper tension.
This method has the advantages of convenience, safety and effectiveness, and the traction force can be adjusted depending on the needs. It can be used in the esophagus, the stomach, and the colorectum, which is currently the mostly used and widely used technique. Its deficits include a single traction direction, and insufficient traction in relation to the requirements of multi-angle traction and ESD for large-area lesions.
4.3.2 Clip–Elastic Ring Combined Traction Method
This is an internal traction technique (figure 4.8). It has been reported in the literature that ESD uses metallic clip–elastic ring combined traction, but as the controllability of the tension of the spring is not good, ESD tension is high at the beginning, and there is no tension when it approaches the end. So this combination is not widely used.
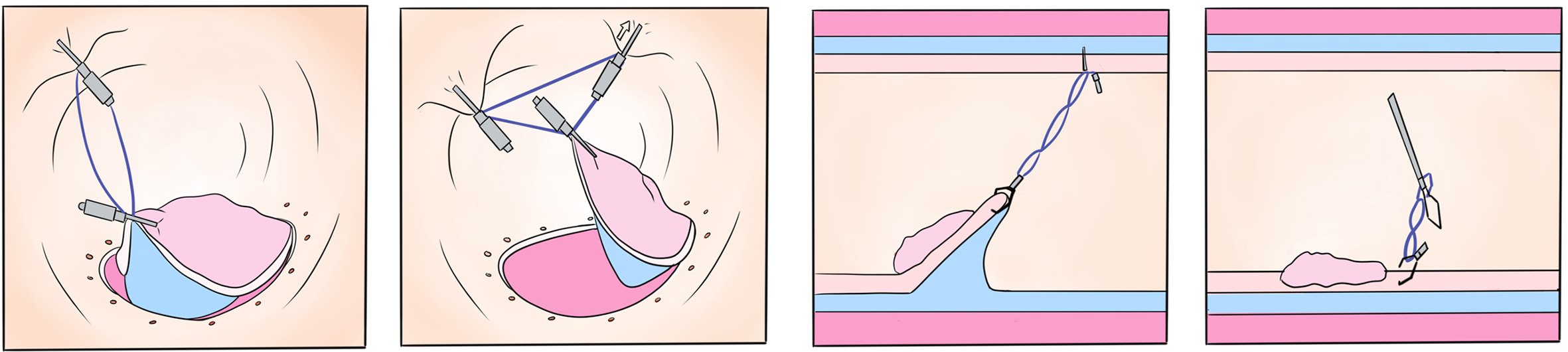

FIG. 4.8 — SMIS clip–elastic ring combined traction method.
The combination of SureClip and the elastic ring has been the hot-spot technique in recent years. This combined technique involves securing the elastic coil at the base of the hemostatic clip, then closing and retracting the assembly into the endoscopic instrument channel. Upon reaching the target lesion, the clip is deployed to grasp the pathological tissue. Subsequently, a second clip is applied to the distal end of the coil, either towards the far end of the lesion or the contralateral side, creating controlled traction on the pathological area.
The advantages of this combination are safety and efficiency, ease of implementation and ease of traction for complicated lesions and complicated sites, with less risk. The drawback is that this combination is often installed personally, so individual errors can occur, affecting the desired effect.
4.3.3 Other Combinations
A relatively less used combined method, but which can be tried, is the internal or external traction technique (figure 4.9). Both methods include clip snare combination, clip gravity hammer combination, clip ‘pulley’ combination, magnetic traction, etc. These traction-enhancing techniques include: Clip-snare combination, Clip-counterweight assembly, Clip-pulley configuration and Magnetic traction systems. These specialized approaches provide targeted traction forces through unique mechanical advantages when conventional methods prove inadequate, particularly in complex anatomical situations requiring precise tissue manipulation. The clip-pulley configuration. This technique demonstrates superior traction mechanics compared to basic clip-dental floss assemblies, offering enhanced control over both traction force magnitude and directional application through its integrated pulley system’s mechanical advantage and vector control capabilities.
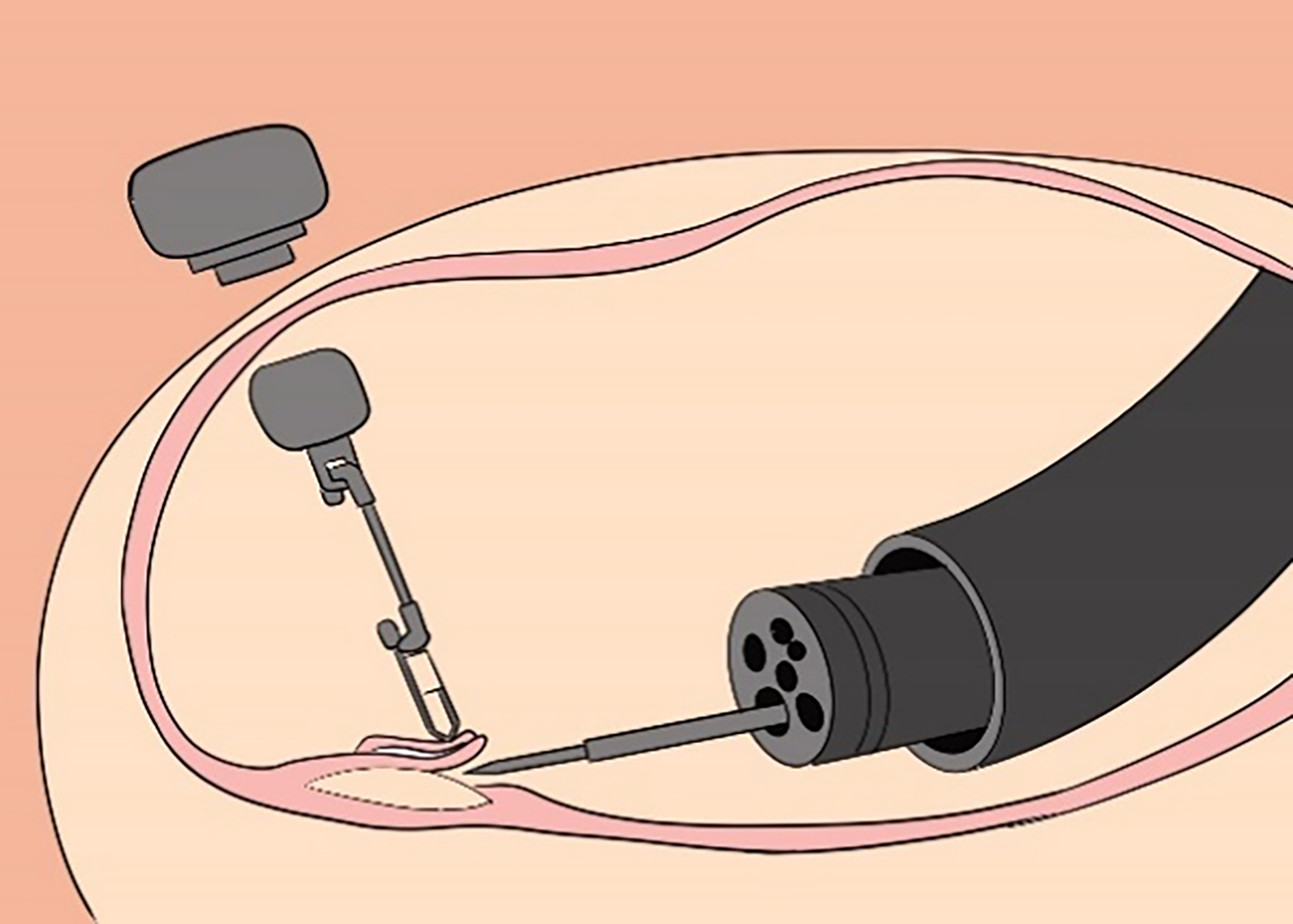

FIG. 4.9 — External magnetic traction method.
4.3.4 Hot-Spot Issues in the Consensus and Research Progress
The technique involving auxiliary traction is widely used in super minimally invasive surgery. After completion of mucosal incision and submucosal preconditioning, different traction techniques have been developed to lift the mucosal flap and better expose the submucosal space, helping us to visualize and accurately identify the layers to be dissected and the blood vessels. Gravity, position changes, and the use of a distal attachment (cap) may be sufficient to complete ESD. However, in some places, this may not be enough. As a result, new traction devices and strategies (external or internal traction) have been developed with the aim of reducing operating time and side effects.
The traction technique in ESD can be divided into two categories that require or do not require additional devices, which can be selected according to the specific situation. Traction provided by the distal cap attachment should always be considered in ESD because of its advantages for exposing the submucosa. Traction technology without device assistance mainly utilizes the tension provided by tissue gravity, tunnels, tunnels and bridges, pockets, etc.
The commonly used device-assisted external traction techniques use clip and thread, clip and snare, external plier, double-cavity endoscopes, etc. The commonly-used device-assisted internal traction techniques include double-clip traction, internal traction thread, magnetic-assisted traction, etc.
In the esophagus, prospective studies have reported the usefulness of the thread clip technique to promote ESD and reduce dissection time. In Western countries, endoscopists with less experience also have a higher success rate using the technique of the thread clip linked to the tunnel. The method combines the traction capabilities of two different techniques and can possibly make ESD safer and faster, but it requires further validation and comparison with traditional techniques. In the stomach, most studies have used clip/floss traction techniques. A randomized control experiment showed a significant reduction in surgical time, but only for lesions located in the upper part of the stomach or in the greater curvature in the middle. Duodenal traction was also reported in a case series involving 29 patients, with shorter surgical time and reduced risk of perforation.
In the colon, some studies have shown that the double-clip and rubber band approach is useful for difficult lesions (fibrotic recurrent adenomas or periappendices) and is associated with reduced surgical time and complete excision. A recent European multicenter, single-group study also showed that the approach can achieve outcomes comparable to those achieved by Japanese experts (96% overall, 88% R 0) with twice the rate of excision (average dissecting speed 39.4 mm
The double-clip traction strategy also helps trainees with ESD, shortening their learning curve and improving their speed and effectiveness. Therefore, this technique could be introduced during the training phase, although the exact timing of its introduction remains to be clarified.
A recent meta-analysis that included only RCTS showed that, although traction was not proven to be more effective for achieving R 0 dissection, the data suggested that traction was associated with shorter surgical times, less side effects, and lower perforation rates. In subgroup analyses, the effects and benefits of esophageal and colorectal ESD were higher: for instance, they shorten surgical time and reduce postoperative complications. Some studies have also found higher overall and R 0 dissection rates for esophageal and colorectal lesions by using the auxiliary traction technique. In the stomach, the benefits of routine traction are unclear, but it could be effective for lesions located in the upper and middle thirds of the stomach, particularly in the greater curvature. Because most traction techniques are cheap with few side effects, these techniques should be considered routinely in esophageal and colorectal ESD, as well as if technical difficulties are encountered in gastric ESD.
References
[1] McCarty T. R., Jirapinyo P. (2023) Endoscopic closure, Gastrointest. Endosc. Clin. North Am. 33(1), 169–182. DOI: 10.1016/j.giec.2022.08.003.
[2] Weiland T., Rohrer S., Schmidt A., Wedi E., Bauerfeind P., Caca K., Khashab M. A., Hochberger J., Baur F., Gottwald T., Schurr M. O. (2019) Efficacy of the OTSC System in the treatment of GI bleeding and wall defects: a PMCF meta-analysis, Minim. Invasive Ther. Allied Technol. 29(3), 121–139. DOI: 10.1080/13645706.2019.1590418.
[3] Chan S., Pittayanon R., Wang H.-P., Chen J.-H., Teoh A. Y. B., Kuo Y. T., Tang R. S. Y., Yip H. C., Ng S. K. K., Wong S., et al. (2023) Use of over-the-scope clip (OTSC) versus standard therapy for the prevention of rebleeding in large peptic ulcers (size ≥1.5 cm): an open-labelled, multicentre international randomised controlled trial, Gut 72(4), 638–643. DOI: 10.1136/gutjnl-2022-327007.
[4] Weiland T., Fehlker M., Gottwald T., Schurr M. O. (2013) Performance of the OTSC System in the endoscopic closure of iatrogenic gastrointestinal perforations: a systematic review, Surg. Endosc. 27(7), 2258–2274. DOI: 10.1007/s00464-012-2754-x.
[5] Goto O. (2023) How should we evaluate the efficacy of endoscopic closure appropriately? A black box between interventions and outcomes, Endoscopy 55(8), 773–774. DOI: 10.1055/a-2073-3608.
[6] Matsuda T., Fujii T., Emura F., Kozu T., Saito Y., Ikematsu H., Saito D. (2004) Complete closure of a large defect after EMR of a lateral spreading colorectal tumor when using a two-channel colonoscope, Gastrointest. Endosc. 60(5), 836–838. DOI: 10.1016/s0016-5107(04)02033-4.
[7] Abiko S., Yoshikawa A., Harada K., Kawagishi N., Sano I., Oda H., Miyagishima T. (2020) New ligation technique using a double loop clip without adhesive for ulceration following endoscopic submucosal dissection of the colon, Endoscopy 52(1), E20–E21. DOI: 10.1055/a-0979-4027.
[8] Motomura D., Toyonaga T., Takayama H., Kodama Y. (2024) Endoscopic double-layered suturing: an updated technique for clip closure of large mucosal defects after endoscopic submucosal dissection, VideoGIE 9(2), 95–98. DOI: 10.1016/j.vgie.2023.09.020.
[9] Masunaga T., Kato M., Sasaki M., Iwata K., Miyazaki K., Kubosawa Y., Mizutani M., Takatori Y., Matsuura N., Nakayama A., et al. (2023) Modified double-layered suturing for a mucosal defect after colorectal endoscopic submucosal dissection (Origami method) (with video), Gastrointest. Endosc. 97(5), 962–969. DOI: 10.1016/j.gie.2023.01.005.
[10] Nomura T., Sugimoto S., Temma T., Oyamada J., Ito K., Kamei A. (2023) Full-thickness resection closure using reopenable-clip over-the-line method inside a submucosal pocket, VideoGIE 8(6), 217–219. DOI: 10.1016/j.vgie.2023.02.011.
[11] Nomura T., Sugimoto S., Matsushima R., Temma T., Oyamada J., Ito K., Kamei A. (2024) Reopenable-clip over-the-line method used inside a submucosal pocket during rectal endoscopic submucosal dissection for a full-thickness defect, Endoscopy 56(S 01), E59–E60. DOI: 10.1055/a-2228-7345.
[12] Mori H., Uedo N., Shichijo S. (2024) No-touch endoscopic full-thickness resection using reopenable-clip over-the-line method for gastric gastrointestinal stromal tumor, Endoscopy 56(S 01), E207–E208. DOI: 10.1055/a-2262-8203.
[13] Chen T., Zhang Y. W., Lian J. J., Zhang H. B., Xu A. P., Li F., Yan X. H., Duan B. S., Zhao Z. Y., Chu Y., et al. (2023) No-touch endoscopic full-thickness resection technique for gastric gastrointestinal stromal tumors, Endoscopy 55(6), 557–562. DOI: 10.1055/a-2013-1902.
[14] Akimoto T., Goto O., Nishizawa T., Yahagi N. (2017) Endoscopic closure after intraluminal surgery, Dig. Endosc. 29(5), 547–558. DOI: 10.1111/den.12839.
[15] Ma L., Linghu E. (2015) Research progress in endoscopic closure techniques for artificial digestive tract incisions, Chin. J. Gastrointest. Endosc. (Electron. Ed.) 2(02), 6–8.
[16] Bowman D. E. (2006) ASGE/SAGES working group on natural orifice translumenal endoscopic surgery, Gastrointest. Endosc. 63(2), 199–203. DOI: 10.1016/j.gie.2005.12.007.
[17] Parsi M. A., Schulman A. R., Aslanian H. R., et al. (2019) Devices for endoscopic hemostasis of nonvariceal GI bleeding (with videos), VideoGIE 4(7), 285–299.
[18] Weilert F., Binmoeller K. F. (2016) New endoscopic technologies and procedural advances for endoscopic hemostasis, Clin. Gastroenterol. Hepatol. 14(9), 1234–1244.
[19] Navaneethan U., Thosani N., Goodman A., et al. (2017) Radiofrequency ablation devices, VideoGIE 2(10), 252–259.
[20] Sachdeva A., Pickering E. M., Lee H. J. (2015) From electrocautery, balloon dilatation, neodymium-doped:yttrium-aluminum-garnet (Nd:YAG) laser to argon plasma coagulation and cryotherapy, J. Thorac. Dis. 7(Suppl 4), S363–S379.
[21] Laine L., Barkun A. N., Saltzman J. R., et al. (2021) ACG clinical guideline: upper gastrointestinal and ulcer bleeding, Am. J. Gastroenterol. 116(5), 899–917.
[22] Cho S. H., Lee Y. S., Kim Y. J., et al. (2018) Outcomes and role of urgent endoscopy in high-risk patients with acute nonvariceal gastrointestinal bleeding, Clin. Gastroenterol. Hepatol. 16(3), 370–377.
[23] Xu J., Dai J., Yin L. (2021) Expert consensus about the emergency diagnosis and treatment of acute upper gastrointestinal hemorrhage, Chin. J. Crit. Care Med. 41(01), 1–10.
[24] Barkun A. N., Almadi M., Kuipers E. J., et al. (2019) Management of nonvariceal upper gastrointestinal bleeding: guideline recommendations from the international consensus group, Ann. Intern. Med. 171(11), 805–822.
[25] Kinoshita J., Iguchi M., Maekita T., et al. (2022) Traction method versus conventional endoscopic submucosal dissection for gastric epithelial neoplasms: a randomized controlled trial, Medicine (Baltimore) 101(13), e29172.
[26] Fraile-López M., Parra-Blanco A. (2020) Double-tunnel circumferential endoscopic submucosal dissection with double clip-band-line traction for an esophageal squamous neoplasm, Endoscopy 52(8), E303–E305.
[27] Sakamoto N., Osada T., Shibuya T., et al. (2008) The facilitation of a new traction device (S-O clip) assisting endoscopic submucosal dissection for superficial colorectal neoplasms, Endoscopy 40(Suppl 2), E94–E95.
[28] Hasatani K., Yoshida N., Aoyagi H., et al. (2022) Usefulness of the clip-and-snare method using the pre-looping technique for endoscopic submucosal dissection of gastric neoplasia: a randomized controlled trial, Ann. Gastroenterol. 35(1), 48–55.
[29] Nagata M. (2022) Advances in traction methods for endoscopic submucosal dissection: what is the best traction method and traction direction? World J. Gastroenterol. 28(1), 1–22.
[30] Pimentel-Nunes P., Libânio D., Bastiaansen B., et al. (2023) Endoscopic submucosal dissection techniques and technology: European Society of Gastrointestinal Endoscopy (ESGE) Technical Review, Endoscopy 55(04), 361–389.
[31] Jacques J., Legros R., Rivory J., et al. (2017) The “tunnel + clip” strategy standardised and facilitates oesophageal ESD procedures: a prospective, consecutive bi-centric study, Surg. Endosc. 31(11), 4838–4847.
[32] Yoshida M., Takizawa K., Suzuki S., et al. (2018) Conventional versus traction-assisted endoscopic submucosal dissection for gastric neoplasms: a multicenter, randomized controlled trial (with video), Gastrointest. Endosc. 87(5), 1231–1240.
[33] Goda Y., Mori H., Kobara H., et al. (2018) Efficacy of sufficient operation view by ring-shaped thread counter traction for safer duodenal ESD, Minim. Invasive Ther. Allied Technol. 27(6), 327–332.
[34] Bordillon P., Pioche M., Wallenhorst T., et al. (2021) Double-clip traction for colonic endoscopic submucosal dissection: a multicenter study of 599 consecutive cases (with video), Gastrointest. Endosc. 94(2), 333–343.
[35] Albouys J., Dahan M., Lepetit H., et al. (2021) Double-clip traction could be superior to the pocket-creation method with cylindrical cap for colonic ESD: a randomized study in an ex vivo model, Surg. Endosc. 35(3), 1482–1491.
[36] Su Y. F., Cheng S. W., Chang C. C., et al. (2020) Efficacy and safety of traction-assisted endoscopic submucosal dissection: a meta-regression of randomized clinical trials, Endoscopy 52(5), 338–348.
Chapter 5 Conditions for Performing Super Minimally Invasive Surgery
5.1 Site and Anesthesia Rules for Super Minimally Invasive Surgery
With the advancement of medical technology, gastrointestinal endoscopy plays an increasingly vital role in the diagnosis and treatment of digestive system disorders. However, the widespread implementation of this technology coupled with rising patient demand, has highlighted the importance of the environment and layout of gastrointestinal endoscopy centers. To enhance the construction and management standards of these centers, the Consensus opinion on the environment and layout of digestive endoscopy centers and the Interpretation of the“Consensus opinion on the environment and layout of digestive endoscopy centers” were published.
The setup of super minimally invasive operating rooms must adhere to the consensus guidelines (figure 5.1). Apart from the standard diagnostic and treatment tools or gastrointestinal endoscopy, specific attention should be given to the unique requirments of super minimally invasive surgery, which requires a variety of accessories and equipment for endoscopic treatment that should be in line with the basic requirements of surgical anesthesia, and should include routine monitoring, oxygen supply and inhalation devices, independent negative pressure suction devices, routine airway management equipment, and intravenous infusion devices, as well as emergency medications and relevant medical apparatus.


FIG. 5.1 — Organization of super minimally invasive surgery centers.
5.1.1 Site Facility
(1) Super minimally invasive surgery operating rooms must be set in a quiet, clean place that is away from polluting sources and dangerous items.
(2) The functional area must include a preparation area, an operation area, and an auxiliary area. The preparation area is comprised of an anesthesia storage cabinet, an anesthesia preparation table, a medicine cabinet, an equipment cabinet and so on; the operating area is furnished with an operating table, an anesthesia machine, a monitor, etc. The auxiliary area includes a recovery room, a washroom, etc.
(3) The super minimally invasive operating room should be equipped with a clean air-conditioning system, the indoor air must be kept clean, with appropriate temperature and humidity.
(4) The floor of the super minimally invasive operating room must be made of anti-slip, easy-to-clean and corrosion-resistant materials. The wall must be smooth, without cracks, and easy for cleaning and disinfection.
(5) The super minimally invasive surgery operating room must be equipped with multi-functional power plugs to meet the power requirements of various medical equipment.
(6) The super minimally invasive surgery operating room must be equipped with firefighting equipment to ensure the safety of the operating room.
5.1.2 Anesthesia Rules for Super Minimally Invasive Surgery
Anesthesia is a crucial task in super minimally invasive surgery, which not only contributes to the smooth running of the operation, but also to the safety of the patient and to his/her postoperative recovery. The Expert Consensus on Anesthesia Management of Common Digestive Endoscopic Surgery (hereinafter referred to as ‘Consensus’), published in January 2019 by the Anesthesia Group, Chinese Society of Digestive Endoscopology, is the cornerstone for improving the surgical safety of surgery and optimizing the anesthesia process to ensure that the operation goes smoothly. It is of considerable significance, and below are the main requirements for anesthesia during the surgical process.
(I) Ensuring safety
The paramount goal of anesthesia is to ensure both patient safety and surgical safety. The anesthetist needs to develop appropriate anesthesia protocols depending on the patient’s physical condition, type of surgery, duration and operating procedure. Operating procedures must be followed to ensure stability of the patient's heart function, other vital organs, and respiratory function. Consensus (3) clearly outlines the norms for pre-anesthesia evaluation, the selection of the anesthesia and monitoring modes during anesthesia, which are necessary for reducing surgical risks and for the safety of patients. The anesthetist must pay close attention to the patients’ vital signs, such as blood pressure, heart rate, respiratory rate, and other indicators, as well as changes in the electrocardiogram and blood oxygen saturation, and identify and treat possible adverse reactions and complications in a timely manner.
(II) Quick recovery
At the end of the operation, the anesthetist needs to ensure the patient can recover quickly, so as he/she can leave the operating room and return to the ward in good time. In order to achieve this goal, the anesthetist needs to choose appropriate anesthetic drugs and dosages to reduce the risk of anesthetic residue in the organism and of delayed recovery. Meanwhile, the anesthetist must also promptly stop the infusion of anesthetic drugs at the end of the procedure to promote the patient’s recovery.
(III) Painlessness
Another important requirement for anesthesia is good results in terms of painlessness. During the surgical process, the patient should be in a painless state to ensure that the operation goes smoothly. In order to achieve this goal, the anesthetist needs to choose appropriate anesthetic drugs and dosages to reduce the patient’s feeling of pain. Meanwhile, the anesthetist also needs to deal with possible pain situations in a timely manner during the procedure to ensure the patient’s comfort. The Consensus (3) is based on scientific anesthesia theory and practice, but also takes full account of the flexibility of the actual operation. For example, in terms of choice of the anesthesia mode, the consensus provides a variety of feasible options, and makes recommendations depending on the specific conditions of the operation and the actual situation of the patient. This combination of science and flexibility makes the anesthesia program more practical and easier to implement.
(IV) Cooperation in operation
The Consensus (3) emphasizes the need for close cooperation between the anesthetist and the surgical team to ensure the smooth running of the operation and improve its chances of success. Before the operation, the anesthetist needs to communicate with the surgeon to understand the specific conditions and needs of the surgery, and develop the appropriate anesthesia program. During the operation, the anesthetist needs to work closely with the surgeon to deal quickly with possible emergencies and ensure the smooth running of the operation.
(V) Focus on the individual differences between patients
As physical condition, the age, the medical history, etc., of each patient are different, the anesthesia program must fully consider individual differences. The Consensus (3) emphasizes the need to conduct a comprehensive assessment of patients prior to anesthesia and to monitor their status throughout the process, including, but not limited to, vital signs such as consciousness, pupil, respiration, heart rate, blood pressure and body temperature. Respecting and paying attention to individual differences of patients helps in developing a more personalized anesthesia program. During surgery, changes in these indicators may show adverse reactions or complications in patients, so it is necessary to detect and deal with them in time. In addition, the anesthetist also needs to carry out intraoperative monitoring of patients, such as blood gas analysis, electrocardiogram, etc., in order to fully understand changes in the physiological state and disease of patients, so as to adjust the anesthesia program and drug dosage in a timely manner.
(VI) Monitoring after anesthesia
With the rapid development of modern medicine, operation and anesthesia techniques are becoming increasingly sophisticated, but the monitoring and treatment after anesthesia cannot be ignored either. In 2021, the report Expert Consensus on Post-Anesthesia Monitoring and Treatment issued by the Chinese Society of Anesthesiology of the Chinese Medical Association provided comprehensive and in-depth guidance in this field, and emphasized the importance of post-anesthesia monitoring and treatment. Due to the common influence of surgical trauma, anesthesia and disease, patients in the recovery period of anesthesia are faced with unique pathophysiological characteristics and potential life risks. Therefore, post-anesthesia monitoring and treatment become the key link in ensuring patient safety and reducing the incidence of complications. The introduction of this consensus undoubtedly provides more clear and specific operational guidelines for the medical personnel. Furthermore, the consensus specifically describes the main aspects of post-anesthesia monitoring and treatment, including restoring the patient’s protective reflexes, monitoring and treating emerging physiological disorders, and identifying and timely managing post-anesthesia and post-surgical complications. The recommendations are comprehensive and detailed, covering various problems that may occur to patients after anesthesia, and provide a strong reference for the medical personnel. However, there are some limitations to this consensus. For example, the consensus does not provide detailed recommendations for strategies of post-anesthesia monitoring treatment for different types of surgeries and populations of patients. It may be refined in the future to improve its practicality and relevance.
In summary, during super minimally invasive surgery, anesthesia is a vitally important task. In order to ensure the safety of patients and the smooth progress of the operation, the anesthetist needs to have professional skills and a rich experience. Meanwhile, the anesthetist also needs to work closely with the surgeon to fully monitor the patient’s condition and deal with possible adverse reactions and complications in a timely manner. Only in this way can the patient receive high-quality anesthesia services, ensuring the success of surgery as well as his/her safety.
5.2 Manning and Requirements for Performing Super Minimally Invasive Surgery
During the operation, the collaborative work of professionals from multiple areas is needed to ensure the smooth running of the operation and the safety of the patient. The main personnel involved in the surgical process and what is expected of them are listed below.
5.2.1 Surgeon
The surgeon is the main person in charge of the operation, who is responsible for the planning of the operation, its implementation and the postoperative treatment. He/she needs to have solid medical knowledge, rich clinical experience and good surgical skills. At the same time, the surgeon needs to demonstrate good communication skills and team spirit in order to effectively communicate and coordinate with patients, anesthesiologists, nurses, and other assistants.
5.2.2 Anesthetist
The anesthetist is one of the important members in the operation process, who is responsible for the implementation and management of anesthesia. The anesthetist needs to have professional anesthesia knowledge and skills, to be able to develop appropriate anesthesia programs depending on the patient’s physical condition and surgical needs, and to strictly monitor the patient’s vital signs. Meanwhile, the anesthetist must have good adaptability and cooperation skills to deal with sudden situations that might arise.
5.2.3 Nurse
The nurse is responsible for patient nursing and monitoring during surgery, including, but not limited to, preoperative preparation, intraoperative nursing, and postoperative management. The nurse needs to have professional nursing knowledge and skills, and to be able to master various nursing operations and techniques. Meanwhile, the nurse also needs to have good communication and observation skills in order to detect and deal with possible adverse reactions and complications in a timely manner.
5.2.4 Operating Room Assistant
The operating room assistant is the assistant to the surgeon who assists the doctor during the operation. He/she needs to have certain medical knowledge and surgical skills, and to be able to skillfully cooperate with doctors to complete surgical operations. Meanwhile, the operating room assistant also needs to have good observation and response ability in order to discover and deal with possible problems in a timely manner. At the same time, he/she also needs to do the postoperative specimen preliminary treatment of super minimally invasive surgery; in recent years, it has played a more and more important role in the treatment of digestive tract early cancer and precancerous lesion. With the continuous progress of technology and the accumulation of clinical experience, the perioperative management of super minimally invasive surgery has become increasingly standardized and refined. In addition, the treatment of postoperative specimens after super minimally invasive surgery is also one of the indispensable tasks, which is directly related to the accuracy of the pathological diagnosis. It is thus also necessary to maintain close contact with clinicians, to share the results of pathological examination and treatment suggestions in a timely manner, so as not to affect the continuation of the treatment and the prognosis of patients. The Guidelines for Perioperative Gastric Endoscopic Submucosal Dissection provided guidance and suggestions for the treatment of surgical specimens. Several aspects need to be paid attention to: the first is the fixation of the specimen. One should make sure that the dissected tissue is properly treated in the first place to maintain its original morphological characteristics and facilitate subsequent pathologic examination. The second aspect is the marking of specimen, including the marking of the incisal margin and at the base, which is vitally important for the pathologist to judge the depth and scope of the tumor infiltration. The final aspect is the filling of a pathological application sheet. The patient-related data, the surgical process and the intraoperative findings must be recorded in detail to provide the necessary clinical background for pathological diagnosis.
5.2.5 Operating Room Head Nurse
The operating room head nurse is the person in charge of operating room nursing and responsible for developing nursing plans and management systems. The head nurse of the operating room needs to have extensive nursing experience and organizational management skills, so as to be able to effectively lead and guide nurses in their nursing work. Meanwhile, the operating room head nurse must also demonstrate good communication and cooperation abilities to work in collaboration with the doctors, the anesthetist, and other personnels involved.
5.2.6 Cleaner
Operating room cleaners play a key role in keeping the environment clean, sterilizing and maintaining surgical safety. They need to clean and disinfect the floor, walls, ceiling, medical equipment, etc. in accordance with regulations to ensure that the surgical environment is sterile and dust-free. Meanwhile, they also need to be responsible for the disposal and transportation of medical rubbish as well as the medical equipment wash and maintenance work. In order to ensure the safety of the operation, the cleaners should strictly abide by the disinfection and sterilization norms as well as the safety and hygiene system, improve their own protection awareness, and avoid the risk of cross infection.
5.2.7 Operation Room Technician
Operating room technicians are mainly responsible for the maintenance of medical equipment. They need to be skilled in the operation and maintenance of various medical equipment, including surgical instruments, monitors, anesthesia machines, etc. During the operation, the technicians must pay close attention to the equipment to ensure that it functions normally and that the data is recording accurately. Meanwhile, they must also manage equipment maintenance and repair work, as well as fix equipment breakdowns in good time to ensure the smooth running of the operation.
5.2.8 Pathologist
Pathologists are responsible for the pathological examination and analysis of surgically removed tissue specimens. They observe the structure and cell morphology of tissue specimens through a microscope to diagnose and type diseases. During surgery, the pathologist needs to make a quick and accurate diagnosis and advise the doctor on the treatment. In order to ensure the accuracy and reliability of the diagnosis, the pathologist must have a solid knowledge and extensive experience in pathology, and follow strict pathology examination procedures and quality standards.
The success of super minimally invasive surgery largely depends on the incisal margin of the surgical specimen and on the depth of infiltration of the resected (dissected) lesion. In order to evaluate these factors more accurately, multiple authoritative academic organizations of the Chinese Medical Association, Branch Pathology, etc. jointly issued the Expert consensus on Endoscopic Submucosal Dissection/Routine Production of Endoscopic Mucosal Resection Specimens [2], aiming at providing more accurate and standardized pathological diagnostic basis for clinical practice. The consensus first emphasizes the importance of the pathological evaluation of ESD/EMR specimens. Since the diagnosis drawn from these specimens directly affects the choice of the subsequent treatment, pathological evaluation must be rigorous and precise. This requires not only high professional skills from the pathologist, but also standardized operating procedures and production methods to ensure the accuracy of the diagnosis. The consensus specifies in detail the handling and routine preparation of ESD/EMR specimens. It gives specific operating guidelines and quality requirements for every step, from sampling, fixation, dehydration, transparency, wax dipping to embedding, slicing and staining, etc. These rules not only help to improve the quality of film-making, but also to preserve the morphology of the tissues and information to the greatest extent possible in order to provide solid support for pathological diagnosis. In addition, the consensus also highlights the importance of multidisciplinary collaboration. The successful implementation of ESD/EMR technology requires the close cooperation of a multidisciplinary team, including gastroenterologists, pathologists, and nurses. Especially when dealing with complex cases, the joint discussion and decision-making of multidisciplinary teams can significantly improve the accuracy of the diagnosis and the effectiveness of the treatment. It is worth noting that the consensus also puts forward requirements for healthcare facilities and operators carrying out ESD/EMR techniques. Medical institutions must maintain a considerable scale of endoscopic diagnosis and treatment workload, and be equipped with perfect facilities and devices. Operators need to undergo professional training, have rich practical experience as well as the ability to deal with complications. These requirements help to ensure the safety and effectiveness of surgery and improve the cure rate and the quality of life of patients.
Through the understanding and analysis of these personnel, we can draw conclusions regarding the manning and requirements for performing the operation: in the operation process, it is necessary for professionals of multiple areas to work together and perform their respective duties to ensure that the operation runs smoothly and that patient safety is guaranteed; personnel in different positions must have the corresponding professional knowledge and skills, and to comply with relevant regulations and norms to ensure the quality and efficiency of work; meanwhile, it is also necessary to strengthen team cooperation as well as communication and coordination to improve the overall level of medical service and provide patients with better-quality medical services.
5.3 Instruments and Parameters of Super Minimally Invasive Surgery
According to the requirements necessitated by the patient’s condition, the digestive endoscope, main machine and apparatus, are prepared, and super minimally invasive surgery preoperative preparations are conducted. The assistant sets all equipment at standby conditions and verifies the connection of the digestive endoscope’s main machine with the digestive endoscope, the connection between the water supply pipe and the water supply bottle, water and gas supply, the connection of the suction apparatus, whether the gastrointestinal endoscope pipeline has suction, the connection between the image processing system and the digestive endoscope, whether the apparatus required by operation is sufficient and correct, whether the connection of each part of the apparatus is correct.
To maximize the advantages of super minimally invasive surgery, it is necessary to carry out sufficient preparation work well before performing the operation. Selecting the good super minimally invasive surgical device is a prerequisite work for the success of the subsequent operations. Therefore, gastrointestinal endoscopy with water supply function is selected (figure 5.2). Meanwhile, matching the instrument with the cap allows sufficient exposure of the visual field, increases operating space and reduces operating time. The equipment required for super minimally invasive surgery includes an electrosurgical device, a digestive endoscope and its accessories, etc.


FIG. 5.2 — With water supply equipment and cap.
5.3.1 Electrosurgical Device
The electrosurgical device, also known as electric knife or electrosurgical instrument, is an important tool of modern medical treatment, which is widely used in modern medical procedures. It incises and stops bleeding by the effect of the high temperature generated by the high-frequency electrical current, which makes it a faster and more precise surgical tool for super minimally invasive surgery. We will discuss each aspect of the electrosurgical device below (figure 5.3).


FIG. 5.3 — Electrosurgical device.
(I) Introduction of the electrosurgical device
The electrosurgical device is a high-frequency current generator, which generates a high temperature under the effect of the generated high-frequency current, enabling incision and hemostasis. It is widely used in various surgical procedures, such as cardiovascular, neurological, thoracic surgeries, general surgery, etc.
(II) Types of electrosurgical devices
Depending on the application scenarios and functions, there is a variety of types of electrosurgical device, including laser knife, radiofrequency knife, ultrasonic knife, cryoprobe, high-frequency electric knife, bipolar electric knife, etc. These devices play an important role in surgery: they improve the efficiency of surgery, reduce bleeding, relieve pain, etc. The types of electrosurgical devices and the products of some major manufacturers will be detailed below (figure 5.4).


FIG. 5.4 — Some common instruments and articles for super minimally invasive surgeries.
1. Laser knife. The laser knife is a kind of surgical instrument that uses laser energy to cut tissues. Its advantages include fast cutting speed, good hemostatic effect and little damage to surrounding tissues. Some mainstream manufacturers include: Lumenis (USA), SIG (Germany), Nanjing Jinling (China), etc.
2. Radiofrequency knife. The radiofrequency knife is a surgical instrument that uses radiofrequency energy to cut and ablate tissues. Its advantages include little damage to surrounding tissues, good hemostatic effect, etc. Some major manufacturers are Boston Scientific (USA), Biostat (Germany), Shenzhen Anke (China), etc.
3. Ultrasonic knife. The ultrasonic knife is a surgical instrument that uses ultrasonic energy to cut and coagulate tissues. Its advantages include fast cutting speed, good hemostatic effect, and little damage to the surrounding tissues. Some mainstream manufacturers are Johnson & Johnson (USA), Ettel (Germany), Guangzhou Huanan (China), etc.
4. Cryoprobe. The cryoprobe is a surgical instrument that uses a refrigerant such as liquid nitrogen to freeze tissues, which can then be cut or ablated. Its advantages include little damage to surrounding tissues, good hemostatic effect, etc. Some major manufacturers are Coulter (USA), Lehmann (Germany), Shanghai Jingke (China), etc.
5. High-frequency electric knife. The high-frequency electric knife is a surgical instrument using a high-frequency current to produce a high temperature enabling incision and hemostasis. Its advantages include little damage to the surrounding tissues, good hemostatic effect, etc. Some mainstream manufacturers are Boston Scientific (USA), ERBE (Germany), Nanjing Jinling (China), etc.
6. Bipolar electric knife. The bipolar electric knife is a surgical instrument that uses a bipolar current for clotting and cutting tissues. Its advantages include little damage to the surrounding tissues, good hemostatic effect, etc. Some major manufacturers are Johnson & Johnson (USA), Ettel (Germany), Guangzhou Huanan (China), etc.
(III) Principle of operation of electrosurgical devices
The electrosurgical device uses the high temperature generated by a high-frequency current to cut and stop bleeding. By heating the knife’s head to a high temperature under the effect of the current passing through it, the instrument cuts and stops bleeding in the tissues. Meanwhile, the device also has a hemostatic function, as it reduces bleeding during surgery by sealing the blood vessels.
(IV) Application field of electrosurgical devices
The electrosurgical device is widely used in various domains, such as cardiovascular, nervous, thoracic surgeries, general surgery, etc. It has the advantages of higher cutting speed, good hemostatic effect, and easiness of operation, better accuracy, etc. Meanwhile, the electrosurgical device, by setting correctly the parameters, can be used for endoscopic procedures such as hemostasis, polypectomy, myotomy, as well as for advanced procedures such as endoscopic ultrasound-guided cystogastrostomy, biliary drainage, and endoscopic ampullaectomy. Endocut is the most commonly used mode in electrosurgical devices for endoscopic surgery, such as sphincterotomy and polypectomy. The Endocut I mode (Effect 1–2, duration 3, interval 3) is used for endoscopic sphincterotomy, while the Forced Coag mode (effect 2, 60 W) is used for hemostasis after sphincterotomy. The Endocut Q mode (effects 2–3, duration 1, interval 3) is used to make polyp stems coagulative when using the Forced Coag mode (effect 2, 60 W).
(V) Advantages and disadvantages of electrosurgical devices
The advantages of the electrosurgical device are fast cutting speed, good hemostatic effect, convenient operation, etc. However, it also has some disadvantages, such as high equipment price, high cost of use, and the need for regular care and maintenance, etc. In addition, the safe use of electrosurgical devices is also very important. It may lead to serious consequences like scalding, burns, etc. if it is improperly used or if there is an operation error. Therefore, the operating procedures and safe-use guidelines need to be strictly observed when using the electrosurgical device.
(VI) Future development trend of electrosurgical devices
With the continuous development of science and technology, electrosurgical devices are constantly improving and perfecting. In the future, electrosurgical devices will be developed to be more intelligent, precise, and minimally invasive. At the same time, with the continuous progress of medical technology, the application field of the electrosurgical device will continue to expand, contributing more to the development of medical science.
(VII) Safe use of the electrosurgical devices
Operating procedures and safety guidelines must be strictly followed when using electrosurgical devices. Firstly, one must ensure that the power supply voltage is stable to avoid breakdown of the equipment or dangerous conditions caused by voltage fluctuations. Secondly, too much heating or scalding the patient’s tissues during the operating process must be avoided, whereas the equipment’s sanitation and hygiene must be kept, so as to avoid the occurrence of infections, etc. In addition, the operator must be professionally trained and certified to manipulate electrosurgical devices.
(VIII) Maintenance of electrosurgical devices
In order to ensure the normal operation and service life of electrosurgical devices, it is necessary to perform regular maintenance. Generally speaking, the electrosurgical device must be regularly checked to ensure that the circuit and the components are working normally and that the cable lines are intact, etc.; the equipment’s surface and sites like knife head, etc., must be cleaned. While using it, attention should also be paid to the occurrence of collisions or falls, which must be avoided so as to prevent damage to the equipment or safety accidents.
(IX) The price of electrosurgical devices and purchasing channel
The price of electrosurgical devices varies according to the brand, the model and the range of use. Generally speaking, hospitals or medical institutions will choose to purchase from classic sales channels of electrosurgical devices to ensure quality and after-sales service. Electrosurgical devices can also be purchased through other channels, such as online shopping malls. But the purchase should be made after careful consideration and comparison of models to guarantee value for money, quality, and reliability.
During the operation, the following measures can be taken to determine whether the electrosurgical device is working properly.
(1) Observe the appearance of the equipment: check whether the equipment has obvious damage or abnormal conditions, e.g., whether the knife head is intact and the cable is damaged.
(2) Check the power supply and the cable: ensure that the power supply and the cable are properly connected, and that here is no bad contact or disconnection, etc.
(3) Test whether the equipment is functioning correctly: before the operation, the electrosurgical device must be tested according to the operating procedures to check whether it is working normally. For example, the power and working time must be tried to observe whether the equipment can work normally, and adjusted if necessary.
(4) Monitor the patient’s response: during the operation, one must pay close attention to the patient’s response, especially the vascular condition of the surgical area. If the patient is found to have discomfort or abnormal reactions, the operation must be stopped in time and the equipment must be checked to ensure it is in good working order. It must be noted that in addition to checking the status of the equipment, the Consensus of Chinese Experts on Clinical Management during Perianaesthesia of Non-Cardiac Surgery for Cardiac Patients (2020) provides comprehensive and detailed guidance for intraoperative patient management, which is helpful to improve the safety of this type of surgery. In practice, the anesthetist should conduct personalized management based on the specific situation of the patient to ensure his safety. The consensus highlights the complexity of non-cardiac surgery and the risks for patients with combined heart disease, noting that perianaesthesia management is a key link in ensuring patient safety. With the aging of the population and the trend of heart disease at a younger age, the number of patients with heart diseases undergoing super minimally invasive surgery is increasing year on year, so it is vitally important to develop and implement scientific perianaesthetic management strategies. This consensus provides valuable guiding suggestions for perianaesthetic management of non-cardiac surgery for patients with heart diseases. It highlights the key role of the anesthesiologist in the operation, including the selection of appropriate anesthesia, close monitoring of hemodynamic indicators, rational medication, and timely multidisciplinary collaboration. These suggestions are helpful to improve the safety of surgery, and reducing the intraoperative and postoperative risks for the patients. However, this consensus also points out that the perioperative treating principles of each type of heart disease are not the same, and the effects of anesthesia and drugs on different heart diseases are also different. Therefore, in practice, the anesthetist needs to conduct personalized management according to the specific condition of the patient, which raises higher requirements for the clinical experience and professional skills of the anesthetist.
(5) Special attention should be paid to the debugging of the pacemaker and the removal of the metal objects worn by the patient. When performing high-frequency electric coagulation and electrical incision treatment on a patient wearing a pacemaker, the electromagnetic interference generated by the high-frequency electric knife can change the pacemaker’s frequency and pacing mode, and the pacemaker can identify the current as cardiac electric activity, inducing malignant arrythmia, ventricular fibrillation, and even cardiac arrest. Therefore, the pacemaker must have been set to VOO mode (fixed-frequency ventricular pacing mode) with the cooperation of the cardiovascular physician and the electrophysiologist before the operation, and an electrocardiogram must be performed to monitor the patient. In order to prevent intraoperative tissue sunburn, the patient must have removed the metal objects he/she is wearing.
(6) Regular maintenance and overhaul: regular maintenance and overhaul of the electrosurgical device must be carried out, including cleaning the knife head and checking whether the cable is intact, etc. Meanwhile, the equipment should be regularly maintained and overhauled to extend the service life of the equipment and ensure safety.
In summary, during the operation, one must pay attention to the working condition of the electrosurgical device at any time as well as to the reactions of the patient, so as to be able to take necessary measures to ensure the normal working of the equipment and guarantee the safety of the patient.
5.3.2 Surgical Equipment for Endoscopic Therapy
The endoscopic surgical equipment includes upper gastrointestinal endoscopy, colonoscopy, double-channel endoscopy, multi-curvature endoscopy, biliary endoscopy, pancreatic duct endoscopy, mechanical circumference endoscopy, electronic circumference endoscopy, circumference ultrasonic probe, linear array ultrasonic probe, soft ultrasonic probe, high-frequency electrical device, thermal probe device, and ultrasonic puncture device (table 5.1).
5.4 Common Instruments and Articles for Super Minimally Invasive Surgery
1. Spray tube. It is used for lesion staining, which makes lesion in the digestive tract more prominent; Lugol’s solution staining is generally used for esophageal lesions, methylene blue staining or indigo rouge staining for gastric lesions, indigo rouge staining for large intestine lesions; the above different dyes can be used to stain the suspected lesions using spray tubes, which protrude out of the biopsy hole channel and align with the lesion and then spray; after dyeing is complete, the gas should be sucked back into the spray tube to prevent the dye from dripping on other mucous membranes.
2. Endoscopic injector. The endoscopic injector is comprised of a needle, a head, an infusion inner tube, an external tube, a protection tube, a booster front handle, and an injection handle. Depending on the liquid injected and the location, the length of the needle tip can be 4 mm, 5 mm, 6 mm.
3. Cutting knife. It is used for mucosal incision. It is associated with the latest high-frequency power supply devices, all with intelligent control systems. But different cutting modes will have different cutting effects, so they will be selected depending on the patient’s condition and his/her needs. The types of cutting knives are varied, and the most commonly used at present are: IT knife (electric knife with ceramic insulation tip), DualKnife, hybrid knife, VS knife, flush knife, etc. Each knife has its own advantages and disadvantages, so the choice should be made according to the needs and the disease.
(1) IT knife: it is a tip-insulated tool designed to prevent accidental damage and perforation of the submucosa and deep layer by the electric knife. It is a valuable aid for the safe dissection of ESDs.
In the first generation of IT knifes, a hemispherical ceramic cap was added to the front end of the tool head, which not only prevents it from sliding flexibly, but also prevents deep mucosal damage while cutting. The length of the cutter’s head can be controlled and the length of the guide wire is 4 mm.
Based on the first generation of electric knife, the second-generation IT knife features three additional electrodes within its hemispherical ceramic cap, which enables faster transverse cutting. The length of the cutter head can be controlled and the length of the guide wire is 4 mm + 0.7 mm × 3.
In IT-Nano, the ‘cap’ of the first-generation IT knifes has been reduced. As the insulation cap becomes ‘thin’, it moves more flexibly in the submucosa and can ‘drill’ into the submucosa more easily.
Flex knife and FlushKnife/BT are not available on the market today.
(2) DualKnife: this knife has a tiny tip that can cut in small spaces, pierce, cross-cut, and is easy to control accurately. But at the same time, because the cutter’s head is small, the cutting burden is smaller, and the surgical task becomes time-consuming. In addition, as the front end of the tool head can conduct electricity, it can also cause damage to tissues. And if its stability is out of control, it presents a higher risk of damage to muscles than the IT knife.
(3) ERBE (hybrid knife): this knife combines four functions (marking, injection, incision, coagulation) into one. Before cutting, an adequate safe cutting space is constructed through submucosal injection to reduce muscle injury. During the operation, the time of instrument exchange and angle adjustment can also be reduced, as reducing the exchange time of the needle and knife tools in building tunnels is suitable for the STER and POME techniques. In addition, the hybrid knife has both the precise pointing force of the DualKnife and the sufficient cutting force of the IT knife, whereas the length of the tool’s body can also be expanded, and it can be changed freely in the limited space. It is suitable for delamination of submucosal fibrosis.
(4) Scissors-like endoscopic knife (VS knife): the recently released disposable high-frequency endoscopic exsector uses the VS knife model MD-47703 Jr from Sumitomo Bakelite Co., Ltd. The cutter’s head is scissor-like, with both electrical cutting and blood clotting functions. When performing ESD treatment, the operation of the VS knife is similar to that of the biopsy forceps, which can pick up the tissue to be incised; after confirming that the muscularis proper or even deeper tissue is not involved, the high-frequency electrical incision is made. The entire incision procedure is like biopsy, which greatly reduces the risk of ESD perforation. In addition, when great vessels are found during excising the lesion, the vessels can be clamped for electrocoagulation and then cut open. This step can replace the use of hot biopsy forceps and reduce the risk of bleeding during the procedure. The advantages of the above VS knife can be utilized for difficult ESD therapies, whereas the tool is easier to master than traditional endoscopic mucotomy knife, and with greater safety.
(5) Flush knife: the Flush knife has a unique water injection function, which allows the head of the knife, the bleeding part, the mucous surface to be rinsed during the operation, and to make an intraoperative submucous injection; the head of the Flush knife is blunt, and its tip is an insulated outer sleeve; the current density is low, which not only effectively prevents perforation, but also hemorrhage. The head of the Flush knife is available in four models of different lengths (1.5, 2, 2.5, 3 mm), which can be selected depending on the operator’s needs.
4. Snare. It consists of an electric coil, a tube sheath, a dragline, a protective sleeve and a handle; it is commonly used for gastrointestinal polypectomy and the removal of gastrointestinal foreign bodies. There exist a variety of models: for instance, oval-shaped, hexagon, half-moon, etc., as well as of types of support.
5. Commonly used instruments for surgical wound hemostasis. Hemostasis plays an essential role in medical work, and is key to protecting the life and safety of patients. In recent years, with the development of surgical refinement, the demand for hemostatic techniques is growing. Intraoperative hemostasis is also crucial for the smooth progression of the operation. During the surgical process, hemorrhage not only affects the clarity of the operative field, but also interferes with the surgeon’s work.
When hemorrhage is effectively controlled, the doctor is able to see the operative field more clearly and to perform the procedure more accurately, thus improving the success rate of the surgery. In addition, intraoperative hemostasis also reduces the need for blood transfusion. Although blood transfusion saves lives, there are also certain risks, such as transfusion reactions, infections, etc. Therefore, through effective intraoperative hemostasis, the need for blood transfusion can be reduced, thereby lowering the related risks. Also, intraoperative hemostasis has important significance to the patient’s postoperative recovery. Heavy bleeding during surgery can lead weakness and slow recovery of the patient after surgery. But effective intraoperative hemostasis reduces blood loss, maintains the patient’s physical power, which is beneficial for quick postoperative recovery. In addition, intraoperative hemostasis also reduces the incidence of postoperative complications. Some of them, related with hemorrhage, such as hematoma, infection, etc., may have adverse effects on the patient’s postoperative recovery. Through the effective control of intraoperative hemostasis, the incidence of these complications can be reduced and the patient’s quality of life can be improved.
The issuance of Expert Consensus on Hemostatic Material Management in Medical Institutions provides a set of scientific and systematic hemostatic material management plans for medical institutions, further highlighting the importance of hemostasis in medical work. Hemostasis is crucial for patient safety. This is particularly true in super minimally invasive surgery, where the risk of intraoperative hemorrhage increases in cases such as lesions near the anal verge, recurrent tumors within scars, type-II lesions, or early gastrointestinal cancer in patients with complicating cirrhosis. Frequent treatment of bleeding will prolong the operation time and increase the risk of surgery invisibly, whereas improper handling may lead to poor surgical results and even threaten the patient’s safety and life. Effective hemostasis techniques can quickly control bleeding and reduce blood loss, thereby reducing the need for transfusion and the associated risk of complications. This is of great significance for improving the success rate of operation and ensuring the patient’s safety. With the continuous development of medical technology, new hemostatic materials and hemostatic related instruments continue to emerge. The application of these instruments and materials provides more options and possibilities for super minimally invasive surgery. However, how to rationally select and apply these new technologies to ensure their safety and effectiveness is a challenge that medical institutions need to face. The Expert Consensus on Hemostatic Material Management in Medical Institutions provides scientific evaluation and management methods for medical institutions, which is helpful to promote the innovative development of hemostatic techniques and further upgrading the medical level.
In order to achieve effective intraoperative hemostasis, doctors need to have a wealth of professional knowledge and operation skills. They need to understand the characteristics and scope of application of various hemostatic methods in order to quickly choose the appropriate one during the operation. Meanwhile, doctors also need to pay close attention to changes in the patient’s vital signs and timely adjust the hemostatic strategy to ensure the patient’s safety. The hemostatic related instruments commonly used in operations by Professor Linghu Enqiang, Department of Gastroenterology, PLA General Hospital, are as follows.
(1) Hot hemostat: mainly composed of the operating part, tip part, insertion part and cable plug, it is compatible with the diameter of more than 2.8 mm of the endoscopic tube, and the maximum opening amplitude of its front-end forceps cup reaches 5–6.5 mm, and is often used in the hemostasis of the endoscopic lesions of tissues and the pre-bleeding treatment of small vessels exposed by wounds.
(2) Hemostatic clamp: it is mainly composed of two parts, the titanium clip and the titanium clip end. Among the hemostatic clamps, one can mention SureClip, Boston Scientific clamp, over-the-scope clip (OTSC), etc. for the treatment of gastrointestinal bleeding, perforation and wound closure.
6. Multi-band ligator. The ligator comprises ligation elements and the handle element equipped with a traction guide wire and an endoscopic fixator. It is commonly used for endoscopic ligation of esophageal varices and hemorrhoids.
7. Foreign body forceps. Composed of a forceps head, an external tube, a dragline and a handle; it is used for the removal of gastrointestinal foreign bodies or auxiliary placement of jejunum nutrition tube, stent removal. The commonly used types in clinical practice are: alligator foreign body forceps, rat tooth foreign body forceps, gannet beak foreign body forceps, three-claw forceps, etc.
8. Mesh basket and net bag. The mesh basket can be used to remove stones smaller than 1.5 cm from the bile duct or foreign bodies from the upper digestive tract; the net bag can be used to remove the foreign bodies from the digestive tract; it can have dimensions of 20 × 50 mm, 20 × 55 mm, etc.
9. Digestive tract obstruction bougie or balloon. Used for the dilation of benign obstructions and stenosis lesions. Savary dilation bougies are commonly used, their moderate softness and the smooth cone at their front enable stenosis to be dilated. The diameters of the dilating bougies can be, for example, 5 mm, 7 mm, 9 mm, 11 mm, 13 mm, 15 mm.
There are two types of balloon dilatating catheters: type A (balloon dilatation catheter) and type B (balloon dilatation catheter with an embedded guide wire).
10. Various accessories used in ERCP. Papillary sphincter cutter, angiographic catheter, biliary guide wire, nasobiliary duct, lithotomy basket, lithotomy balloon, and biliary stent.
11. Endoscopic ultrasound for puncture. The longitudinal-axis ultrasound endoscope can clearly display the passage of the needle and is mainly used for puncture and interventional therapy. At present, the longitudinal-axis ultrasound endoscopes of Olympus, Fujifilm, and Pentax are commonly used.
12. Puncture needle for ultrasound. It is divided into three parts: needle core, needle sheath, and handle. Manufacturers include Wilson-Cook, Boston Scientific, Medi-Globe ultrasound puncture. The surface of the front end of the puncture needle is usually rough so that the tip and the whole puncture needle can be clearly displayed on the ultrasonic image. The puncture depth of the common puncture needle can reach 65 mm. Models are 19G, 22G, 25G. The needle is 4–8 cm long and has a cylinder with negative pressure suction. 19G applies to the suction of cysts/pancreatic pseudocysts. Other cutting ProCore puncture needle with grooves can make tissue strips, which can be used for the puncture in order to establish a clear diagnosis of the pathological tissues to improve the effectiveness of the puncture; these include 19G, 22G and 25G models.
13. Endoscopic ultrasound cell brush. After the 19G puncture needle is inserted into the lesion under the guidance of an ultrasound endoscope, the cell brush in the center is extended into the lesion repeatedly to collect cells. This procedure is mostly used for the diagnosis of cystic diseases.
5.5 Standardized Operation and Management of the Cleaning and Disinfection of Gastroendoscopes
With the continuous innovation and iteration of medical technology, the application of soft gastrointestinal endoscopy is increasingly widening in the medical field, providing great convenience for the diagnosis and treatment of digestive tract diseases. Meanwhile, the problem of cleaning and disinfecting digestive endoscopes has also aroused general concern in the medical field. Due to the complex structure of gastrointestinal endoscopy and the special material used, it is difficult to clean and disinfect, so it is particularly important to formulate a detailed and practical operating guide. In this context, the Practical Operation Guideline for Cleaning and Disinfection of Endoscopes came into being, providing a comprehensive guidance for the cleaning and disinfection of endoscopes.
As precision instruments, digestive endoscopes, such as electronic gastroscopes and electronic colonoscopes, have a complex structure with many accessories, and reach expensive prices. To address this, the guideline puts forward the requirements for cleaning and disinfection, emphasizing the importance of thorough cleaning and high-level disinfection or sterilization.
During the preparation phase before cleaning and disinfection, the guideline details the preparation requirements for personnel, the environment, and materials. Staff must have certain professional knowledge and training to guarantee their competence; for the environment, a special cleaning workbench must be set up, and a clean air flow must be maintained, with an independent ventilation system; appropriate cleaning brushes and a special cleaning enzyme for endoscope, etc., must be chosen for materials cleaning.
For the cleaning process, the guideline emphasizes the importance of pre-treatment. That is, after the endoscope is taken out from the body of the patient, external dirt should be immediately wiped out with a wet gauze containing a cleaning fluid. In addition, the guideline also describes in detail the steps of receiving, cleaning, disinfection, rinsing and drying of endoscopes, as well as the selection and configuration of the cleaning fluids and disinfectants.
In view of the common problems in the cleaning and disinfection process of digestive endoscopes, the guideline also gives corresponding countermeasures. Regarding areas that are particularly challenging to clean—such as the endoscope’s lumen, flexion sites, and junctions—the guideline specifies that thorough manual cleaning must be performed. This entails pre-treatment, leak detection, cleaning, and rinsing, which is vital for achieving the required cleaning standard. Meanwhile, the guideline also emphasizes the importance of monitoring the concentration of cleaning liquids and disinfectants to ensure their effectiveness.
In addition, the guideline also puts forward relevant requirements for automatic cleaning and disinfection machine for digestive endoscopes, which should meet the specifications of Hygienic Requirements for Automatic cleaning and disinfecting Machine for Endoscopes, GB30689-2014 for cleaning, disinfection, rinsing, and self-disinfection, etc. For the problems of secondary contamination in the transport and storage process of sterilized soft endoscopes, the guideline also gives specific suggestions, such as using a special transport vehicle for digestive endoscopes, closed transport bags, and storage management in accordance with the storage requirements for sterile items, etc.
The following requirements must be met according with the guideline.
1. Determine the disinfection process
(1) According to the type and to the specificities of the endoscope, the appropriate disinfection process must be selected. Generally speaking, the disinfection of digestive endoscope should include the steps of enzymatic washing, cleaning, disinfection, rinsing and drying, etc.
(2) According to the actual condition of the hospital or department, detailed operating procedures and precautions should be formulated to ensure the accuracy and effectiveness of endoscopic disinfection.
2. Select the disinfectant
(1) According to the relevant regulations of the National Health Commission, choose the disinfectant that meets the requirements. Disinfectants such as glutaraldehyde and hydrogen peroxide are commonly used for gastrointestinal endoscopy.
(2) According to the material and to the specificities of the endoscope, the appropriate concentration of disinfectant and action time will be selected.
3. Carry out the disinfection process
(1) According to the operation procedure, the endoscope will be immersed in the enzyme lotion, fully soaked and then cleaned.
(2) A cleaning brush will be used to thoroughly clean the endoscope, including pipes, buttons, and operation parts.
(3) After cleaning, the endoscope will be immersed in the disinfectant and disinfected according to the instructions given by the product’s manufacturer.
(4) The endoscope will be flushed with sterile water to ensure that there are no residual disinfectant and contaminant.
(5) The endoscope will be dried with a dry towel or a sterile cloth to ensure that no water stains or residues remain.
4. Cleaning of the endoscope
(1) The endoscope should be cleaned immediately after each use to prevent residues from impairing the effect of the disinfection.
(2) A dedicated endoscopic cleaning brush and cleaner must be used, following the instructions.
(3) The endoscope should be handled with care during cleaning to avoid damaging the endoscope and its accessories.
(4) The endoscope must be rinsed with sterile water after cleaning to ensure that no residual cleaning agents and contaminants remain.
5. Test of the disinfection result
(1) The effectiveness of the disinfection of the endoscope must be tested by bacterial cultivation or with a chemical indicator.
(2) Based on the test results, the disinfection process or the disinfectant must be adjusted to ensure that the disinfection process has met the requirements.
(3) If the disinfection effect is not qualified, remedial measures should be taken immediately to ensure the cleanliness and safe use of the endoscope.
6. Recording of disinfection information
(1) The disinfection process of each endoscope must be recorded in detail, including information like disinfectant name, concentration, action time, operator, etc.
(2) The record should be kept for a long time, in order to trace and evaluate the disinfection result.
(3) The record must be checked and analyzed, experiences and lessons learnt must be summarized, and disinfection quality must continue to be improved.
7. Storage of the endoscope
(1) Endoscopes must be stored in a dry, well-ventilated storage environment to avoid moisture and contamination.
(2) Endoscopes must be classified and stored according to their type, specification, and disinfection date for easy access and management.
(3) Endoscopes and their accessories must be kept clean and intact to avoid cross infection and damage.
(4) Endoscopes and their accessories must be inspected and maintained regularly to ensure their normal operation and safe use.
8. Regular checks
Under the regulations of the National Health Commission and operating requirements of the hospital or department, inspections should be carried out regularly. Inspections should be recorded in a format that operators can easily consult. If results do not meet standards, immediate action should be taken to ensure that quality and safety of endoscope meet the requirements.
In summary, the Practical Operation Guideline for Cleaning and Disinfection of Endoscopes provides comprehensive guidance for the cleaning and disinfection of digestive endoscopes, not only covering all aspects of cleaning and disinfection, but also providing specific solutions for common problems. The introduction of the guideline will help improve the quality of the cleaning and disinfection of digestive endoscopes, reduce the risk of iatrogenic cross infection, and ensure the patient’s safety. Meanwhile, it also provides a scientific, standardized and operable cleaning and disinfection method for medical institutions, which deserves to be carefully studied and put in practice by health professionals.
TAB. 5.1 — Super minimally invasive surgery equipment mode and output power selection.
| Surgical method | Instrument | Mode | Device power (W) | Standard electric coagulation index | Argon flow control (l/min) | Treatment time control (second/time) |
| Marking | DualKnife | Forced | 20–30 or 20 | |||
| Arc knife | APC | 0.5 | ||||
| Mucosal resection | DualKnife | Endocut effect 3 | 80–120 | |||
| Mucosal dissection | DualKnife or IT or triangle knife | Endocut effect 3 | 80–120 | |||
| Vascular pretreatment | IT knife | Forced | 30 –50 | |||
| Hemostat | Soft | 50–80 | ||||
| Intraoperative hemorrhage | Hemostat | Soft | 50–80 or 30 | |||
| Arc knife | APC | 1.0 | ||||
| Wound treatment | Hemostat | Soft | 50–80 or 25 | |||
| Arc knife | APC | 1.0 | ||||
| Endoscopic argon ionization coagulation | Arc knife | Endocut | 45–60 | A40-50 | 2–3 | 1–3 |
References
[1] The Nursing Collaboration Group of the Chinese Society of Gastrointestinal Endoscopy. (2023) Consensus opinion on the environment and layout of the digestive endoscopy center, Chin. J. Gastrointest. Endosc. (Electron. Ed.) 10(4), 240–247.
[2] Qiu X., Linghu E. (2024) Interpretation of expert consensus recommendations on the environment and layout of digestive endoscopy centers (2023, Beijing), Chin. J. Gastrointest. Endosc. (Electron. Ed.) 11(1), 4–6.
[3] The Anesthesia Collaboration Group of the Chinese Society of Gastrointestinal Endoscopy. (2019) Expert consensus on anesthesia management for common gastrointestinal endoscopic procedures, J. Clin. Anesthesiol. 35(2), 177–185.
[4] Anesthesiology Branch of the Chinese Medical Association. (2021) Expert consensus on post-anesthesia monitoring and treatment, J. Clin. Anesthesiol. 37(1), 89–94.
[5] National Clinical Research Center for Digestive Diseases, Chinese Society of Digestive Endoscopy, Digestive Physicians Branch of the Chinese Medical Doctor Association. (2017) Clinical guidelines for gastric endoscopic submucosal dissection during the perioperative period, Chin. J. Pract. Intern. Med. 52(12), 12–24.
[6] Digestive Pathology Group of the Pathology Branch of the Chinese Medical Association, Pathology Technical Expert Group of the Medical Technician Committee of the Chinese Medical Doctor Association, Standardization Department of Pathology Equipment Branch of China Medical Equipment Association, Group of Digestive Tract Diseases, Specialized Committee of Ultrastructural and Molecular Pathology, Chinese Research Hospital Association, Pathology Technology Group of Pathology Professional Committee of Chinese Research Hospital Association, Pathology Technology Group of the Tumor Pathology Professional Committee of the Chinese Anti-Cancer Association. (2023) Expert consensus on routine preparation of endoscopic submucosal dissection/endoscopic mucosal resection specimens, Chin. J. Pathol. 52(10), 989–994.
[7] Non-Cardiac Surgery Anesthesia Branch of the Chinese Society of Cardiothoracic and Vascular Anesthesiology. (2021) Chinese expert consensus on perioperative anesthetic management for non-cardiac surgery in patients with heart disease (2020), Perioper. Saf. Qual. Assur. 5(2), 63–77.
[8] Cho K. B., Jeon W. J., Kim J. J. (2011) Worldwide experiences of endoscopic submucosal dissection: not just Eastern acrobatics, World J. Gastroenterol. 17(21), 2611–2617.
[9] Medical Consumables Evaluation and Management Project Team, Institute of Hospital Management, National Health Commission. (2021) Expert consensus on hemostatic materials management in medical institutions, Chin. Hosp. Archit. Equip. 22(7), 18–27.
[10] Yamashina T., Takeuchi Y., Nagai K., et al. (2017) Scissor-type knife significantly improves self-completion rate of colorectal endoscopic submucosal dissection: single-center prospective randomized trial, Dig. Endosc. 29(3), 322–329.
[11] Ngamruengphong S., Kalloo A. N. (2017) Scissor-type needleknife for colorectal endoscopic submucosal dissection, Endoscopy 49(9), 846–847.
[12] Takeuchi Y., Uedo N., Ishihara R., et al. (2010) Efficacy of an endo-knife with a water-jet function (Flushknife) for endoscopic submucosal dissection of superficial colorectal neoplasms, Am. J. Gastroenterol. 105(2), 314–322.
[13] Fang L. (2023) Publication of the Practice Operation Guidelines for Flexible Endoscope Cleaning and Disinfection: application of the PDCA cycle in centralized cleaning and disinfection of flexible endoscopes, J. Interv. Radiol. 32(1), 5.
[14] Iwakiri R., Tanaka K., Gotoda T., et al. (2019) Guidelines for standaridizing cleansing and disinfection of gastrointestinal endoscopes, Dig. Endosc. 31(5), 477–497.
[15] Beilenhoff U., Biering H., Blum R., et al. (2018) Reprocessing of flexible endoscopes and endoscopic accessories used in gastrointestinal endoscopy: Position Statement of the European Society of Gastrointestinal Endoscopy (ESGE) and European Society of Gastroenterology Nurses and Associates (ESGENA)-Update 2018, Endoscopy 50(12), 1205–1234.
Chapter 6 Standard Process of Super Minimally Invasive Surgery
6.1 Informed Consent Prior to Super Minimally Invasive Surgery
With the rapid development of medical technology, the super minimally invasive theory was first proposed by Professor Linghu Enqiang. It has emerged in the medical field as an innovative operation method that preserves the function of organs without changing their structure. However, any medical procedure involves certain risks. Therefore, before performing super minimally invasive surgery, it is particularly important to ensure that the patient or his/her family is fully informed about the surgical information, the associated risks and the alternatives, so as he/she can make his/her own decisions about whether to undergo surgery; this process is known as informed consent. It is comprised of three steps: the notification by the physician, understanding of the content by the patient, and his/her decision, agreement or refusal. In this chapter, an in-depth discussion will be conducted on the issue of informed consent in super minimally invasive surgery, as well as a comprehensive analysis from multiple points of views such as law, ethics, and practice, with a focus on the practical details, in order to provide more specific guidance for the operative practice.
6.1.1 Legal Basis and Moral Consideration of the Informed Consent
(I) The informed consent is based on the law
As one of the basic principles of medical procedure, informed consent is not only the basic requirement of medical ethics, but also one of the obligations clearly stipulated by laws and regulations, which is clearly reflected in many laws and regulations. For example, it was clearly stipulated in the Civil Code of the People’s Republic of China that medical staff must explain the condition and medical measures to patients during diagnosis and before treatment activities, and obtain their explicit consent. This provision provides a solid legal basis for informed consent. Meanwhile, laws and regulations like the Law of the People’s Republic of China on Medical Practitioners, Regulations on the Administration of Medical Institutions also have specific provisions on informed consent, requiring medical personnel to fully inform the patient or the families of the relevant information and obtain their written consent before performing medical acts such as surgery. Prior to the informed consent procedure, ensure to provide appropriate and understandable information to each patient and assess his/her ability to understand it. The following legal points must be emphasized.
(1) The importance of the physician’s information duty about the diagnosis and treatment process, and of the patient’s right to informed consent. See the detailed stipulation in Article 26 of the Law of the People’s Republic of China on Medical Practitioners: “Doctors must tell the patients or their relatives the truth about the patients’ condition while avoiding any bad news effect. Doctors conducting experimental clinical treatments must obtain approval from the hospital and the consent of the patient himself or his family.”
(2) The information duty of medical institutions and health professionals, so as to ensure that the patient’s right to informed consent is respected.
See in detail Article 11 of Medical Malpractice Management Regulation which points out that “in medical activities, medical institutions and their medical personnel must truthfully inform patients of their conditions, medical measures, medical risks, etc., and answer their inquiries in a timely manner;as further, adverse consequences for patients must be avoided.”
(3) The procedure for informed consent during surgery, special examination or special treatment is specified in detail. See Article 33 of the Regulations on the Administration of Medical Institutions which stipulates specifically that “when the medical institution performs an operation, special examination or special treatment, it must obtain the consent of the patient and/or the consent and signature of his family or related persons; if it is not possible to obtain the opinion of the patient, the consent and signature of the family members or related persons must be obtained; when it is impossible to obtain the opinion of the patient and there is no family member or related person present, or in other special circumstances, the managing physician should propose a medical treatment plan and implement it after obtaining the approval of the person in charge of the medical institution or the person authorized to be in charge.”
(4) The right to informed consent is clearly written into the civil code, which strengthens its legal status. See in detail Article 1219 of the Civil Code stipulation: “Medical personnel must explain their condition and medical measures to patients during diagnosis and treatment activities. When surgery, special examination or special treatment is required, medical personnel should promptly explain the medical risks and alternative medical plans to patients and obtain their explicit consent; if it is not possible or inappropriate to explain this to the patient, it should be explained to the patient’s close relatives and their explicit consent must be obtained. When medical personnel fail to fulfill their obligations mentioned in the preceding article and cause damage to patients, the medical institution should be liable for compensation.”
(5) The patients’ right to informed consent must be reiterated when he/she is receiving renewed medical healthcare services. See in detail the stipulation in Article 32 of the Law of the People’s Republic of China on Basic Medical and Health Care and the Promotion of Health: “When receiving medical and health services, citizens must have the right to informed consent in accordance with the law on matters such as medical conditions, diagnosis and treatment prescriptions, medical risks and medical expenses. If surgery, special examination or special treatment is required, the medical and health personnel must explain the medical risks and alternative medical plans to the patient in a timely manner and obtain his/her consent. If it is not possible or inappropriate to explain to the patient, the patient’s close relatives must be given explanations and their consent obtained.”
(II) The informed consent takes morality as the consideration
In addition to its leagal foundtion,informed censent carries profound ethical implications. ‘Free will of choice means’ that patients with understanding capacity must be able, in the process of seeking healthcare services in a medical relationship, after independent thinking, to make rational and value-oriented decisions about their own diseases and health problems, and take responsible actions and decisions. This right not only upholds the individual dignity and freedom of patients, but also represents a critical requirement of centemporary medical ethics and law. The patient’s right of choice means that patients have the right to know their own condition, the various examination methods, the treatment plans and the medical risks, and that, on this basis, they can freely choose to agree or disagree with the proposed surgery, examination or treatment plans. This right is an important part of the patient’s free will, which aims to ensure the subjective status and autonomous decision-making ability of patients in the medical treatment process. In the implementation of ultra-minimally invasive surgery, the medical staff must fully respect the patient’s wishes and choices, and ensure that patients fully understand the surgical information, the risks and the alternatives, and can decide whether or not to undergo surgery. This not only reflects the respect for the patient as a person, but is also an important guarantee for the legitimacy and justification of medical behavior.
6.1.2 The Contents and Requirements of Super Minimally Invasive Surgery
(I) Name of operation, operation method and purpose
In the written informed consent document, the patient must be specifically informed about the name of the operation, the operation method and its therapeutic purpose, and must fully understand them. This helps patients or their families to accurately understand the specific procedure and the expected results, so that they can make wise choices.
(II) Possibly existing risks and complications
Although super minimally invasive surgery has the advantages of less trauma and faster healing, it still involves certain surgical risks and complications.
Therefore, in the information given for consent, the possible risks, complications and adverse consequences of the operation should be listed in detail, including but not limited to the risks associated with anesthesia and with the surgical operation, of postoperative infection, of hemorrhage, etc. Meanwhile, the probability of these risks and complications and the associated prognosis must also be explained to patients or their families so that they can make a comprehensive assessment.
(III) Alternative or other treatment plan
In the process of informed consent, the medical staff must present other treatment options besides surgery to patients or their families, including drug therapy, physical therapy, etc., and explain the advantages and disadvantages of each option. This helps patients or their families to fully understand the treatment options and make the most appropriate choice for their situation.
(IV) Postoperative notice and nursing
In the process of informed consent, patients should be fully and solemnly informed of the matters that need to be paid attention to after super minimally invasive surgery to promote recovery and reduce the occurrence of complications, such as diet, rest, medication, recheck, etc. Meanwhile, it is also important to explain to the patient or his/her family the particular circumstances that may arise and the measures to be taken to deal with them, so that they can anticipate them psychologically and fully prepare themselves.
(V) The patient’s rights and duties
In the process of informed consent, the patient’s right during surgery must be clearly defined, such as right to be informed, right of choice and right to privacy, etc. Meanwhile, it is also necessary to emphasize the duties with which the patient must comply, such as truthfully informing about his/her condition and actively cooperating with the treatment, etc. This helps to ensure that the patient’s rights and interests are effectively protected during surgery, and further promotes trust and cooperation between doctors and patients.
6.1.3 Practical Implementation of Informed Consent in Super Minimally Invasive Surgery
(I) Preoperative conversation and communication
(1) Special time must be arranged: to ensure the adequacy of the pre-operative conversation, the medical personnel should arrange a special time to communicate with the patient or his family, avoiding hasty or emergency conditions.
(2) Auxiliary tools must be used: during the conversation, the medical staff can use auxiliary tools like pictures, videos or models to help the patient or the family understand the surgical process and the possible risks more instinctively.
(3) Questions and discussions must be encouraged: the medical staff should encourage the patient or the family to raise questions, and answer them patiently. To address the patient’s or his/her family’s concerns and worries, they must discuss them in detail and provide all the explanations necessary to dispel their suspicions.
(II) The informed consent form must be signed
(1) Sufficient time must be provided: to ensure that the patient or his/her family fully understands the contents of informed consent, they should be given sufficient reading time, and answers must be given to any doubts that they may have.
(2) Language should be simplified: a concise and easy to understand language must be adopted, using too professional terms and complicated expressions must be avoided, to ensure that the patient or the family understand easily.
(3) The understanding level must be confirmed: before signing the informed consent form, the medical personnel should ask the patient or the family repeat the surgical information, etc., by asking questions, in order to confirm whether they really understand the procedure, the risks and the alternatives.
(III) File for investigation and privacy protection
(1) The security of the information must be guaranteed: the signed informed consent form should be kept as part of the medical records for future reference and should be left out of sight from unauthorized personnel.
(2) The patient’s privacy must be protected: during the operation and postoperative follow-up visits, the medical staff must strictly protect the patient’s private information from being disclosed, including physical privacy and personal information.
6.1.4 Postoperative Follow-Up Visit and Feedback
(1) Regular follow-up visits must be planned: the medical personnel should develop a regular postoperative follow-up visit plan to ensure to get timely information about the patient’s recovery and the occurrence of complications.
(2) The establishment of a feedback mechanism: the patient or the family must be encouraged to provide feedback opinions on the undergone surgery and on the informed consent process, so that medical personnel can continuously improve and refine the related services. Meanwhile, the medical personnel should also actively ask the patient or the family whether he/she is satisfied with the surgery and whether he/she has suggestions to make.
(3) Feedback must be recorded and handled: the medical personnel should record in detail and handle timely the patient’s or the family’s feedback opinions. As to the problems or deficits arisen, the medical personnel should think about improving the quality of the medical service.
6.1.5 Difficulties and Strategy of Informed Consent in Super Minimally Invasive Surgery
(I) Difficulties associated with informed consent in super minimally invasive surgery
(1) Unsymmetrical information: due to the professionalism and complexity of medical knowledge, it is often hard for the patient or the family to comprehensively and accurately understand the surgical information. This may lead them to make unwise choices during the process of informed consent.
(2) Communication handicap: some patients or the families may have handicaps in terms of language, culture, or psychological ones, etc., leading to difficulties of communication with medical personnels, and to their misunderstanding of surgical information, affecting the effectiveness of the process of informed consent.
(3) Trust crisis: in a context of tense doctor–patient relationship, some patients or the family may be tempted to distrust health professionals. This may lead to suspicions or resistances during the process of informed consent.
(II) Informed consent about the strategy of super minimally invasive surgery
(1) The education of the patients must be improved: easy-to-understand education materials, as illustrated, concise surgical information booklets or videos to help them better understand the procedures, risks and alternatives, should be provided. Patient education lectures on super minimally invasive surgery must be held regularly: experts should be invited for interpretation, interaction possibilities should be provided, so that patients or their families can have opportunities to raise questions and discuss.
(2) The communication mode should be optimized: translation tools or the intervention of professional translators can be used for patients or families who have language barriers to ensure the accurate passing of information. Multipole communication modes should be adopted: depending on the patient or the family’s demands and preferences, the medical personnel can adopt multiple modes like face-to-face communication, telephone consultation, e-mail, etc., to keep contact with them and answer questions in a timely manner.
(3) Building relationships of trust: the medical staff can demonstrate their professional ability and experience in the field of super minimally invasive surgery by sharing their professional background, surgical experience and successful cases, so as to win the trust of patients or of their families.
Emphasizing emotional communication: during the process of informed consent, the medical personnel should emphasize the emotional communication with patients or their families, care about their needs and feelings, give full support and comfort, so as to build good relationships of trust.
In summary, informed consent in super minimally invasive surgery is a complex issue involving multiple dimensions like law, morality, and practice, etc. Before performing super minimally invasive surgery, it is important to guarantee the patients’ rights and promote doctor–patient trust and cooperation by making sure that patients or their families do fully understand the surgical information, the risks and the alternatives, and are able to decide by themselves whether or not to receive surgery. However, in practice, there still exists challenges like unsymmetrical information, communication handicap and trust crisis, etc. To deal with these challenges, patient education, communication modes and the establishment of relationships of trust should be improved. Looking forward, with the continuous development of medical technology and the constant improvement of the medical environment, we have reasons to believe that the process of informed consent in super minimally invasive surgery will become more refined and effective, and will bring better medical experience and treatment effects for patients. Meanwhile, we should also continue to focus on and study the new problems and new challenges in the process of informed consent, so as to push forward the continuous development and progress of medical ethics and laws.
6.2 Preoperative Conversation Skills in Super Minimally Invasive Surgery
In the ever-changing field of gastrointestinal endoscopy and the maturation of the treatment technologies, super minimally invasive surgery has become one of the main means to be considered for treating diseases of the digestive tract, like early cancer or submucosal tumors of the digestive tract, etc., allowing complete organs can be preserved, achieving the ideal of eliminating diseases and improving the patients’ prognosis and life quality. However, as the patient receives the treatment, he/she has often strong physiological and psychological stress reactions, manifesting themselves by anxiety, panic and nervousness to different degrees. These emotional changes often bring changes to the patient’s psychology and vital signs, bringing stress to the patient to different levels, which also generates a negative impact on the examinations and on the treatment to different degrees, affecting the smooth progression and effective results of diagnoses and treatments. As the guardian of public health, the physician’s main responsibility is not only to relieve the patients’ clinical symptoms, but, more than that, to provide them with mental comfort through language and behavior in order to try to reduce their mental burden.
6.2.1 Patient Psychological Profiling and Intervention of Super Minimally Invasive Surgery
(I) Reasons why patients having undergone super minimally invasive surgery may experience psychological stress reactions
Different degrees of psychological stress reactions can be generated as the patient receives pre-, intra, post-diagnosis and treatment by super minimally invasive surgery. Before the treatment, the patient worries about the pains and risks brought by diagnosis and treatment, worrying about unsmooth diagnosis and treatment or bad treatment results; during the diagnosis and treatment phases, due to waiting for diagnosis and treatment, facing with the strange surgical environment, and without his/her relative’s support, anxiety, loneliness, and sense of insecurity arise; after diagnosis and treatment, due to the discomforts brought by surgery, such as abdominal distension from air inflation, digestive tract perforation, hemorrhage, etc., the patient’s feeling of discomfort is aggravated, whereas anxiety increase due to concerns about the unknown postoperative pathological results.
(II) To address the patient’s psychological stress, psychological conditions conducive to the intervention must be put in place
(1) Building a good doctor–patient relationship. The patient’s trust must be obtained. Listening to the patient’s and his/her family’s questions patiently and answering them, using verbal words in communications, being amicable in attitude, encouraging the patient to express his/her worries and uneasiness will reduce the patient’s psychological stress reactions and increase his/her confidence in the treatment.
(2) Helping the patient to understand the disease and correct some erroneous ideas. An erroneous judgement due to insufficient understanding of super minimally invasive surgery and of his/her digestive tract disease by the patient is the more important causing uneasiness, anxiety and panic in him/her. So the patient should be fully informed of the surgical purpose and of the methods prior to the examinations. In terms of cognitive therapy, it makes the patient have psychological support to a very large extent, overcome anxiety and panic, and adapt his state of mind as well as possible in order to allow the diagnosis and the treatment to be carried out smoothly.
(3) Behavioral therapies like multimedia courseware can be used to make the patient fully understand the operation as well as relax physically and mentally. For clinical workers, empathy is a skill of prime importance, as it allows the doctor to care for the patient in a more comprehensive and complete way, making him/her enter the personal environment of the patient, and finally helps the health professional to rebuild the patient’s health state.
6.2.2 Doctor–Patient Communication in Super Minimally Invasive Surgery
Doctor–patient communication is a powerful tool for surgical diagnosis and treatment. The basic principle in communication is to listen and inform.
(1) The physician should start his/her communication from the patient’s perspective, in order to increase the latter’s and his/her family’s trust in him/her.
(2) He/she must listen patiently to the events that the patient and the family worry about.
(3) The physician must have a correct assessment of the patient’s condition. In addition to the prognosis of the disease, he/she should also be familiar with medical expenses, particularly those for which the patient will be charged or high costs, in order to know the level of expenditure that is acceptable for the patient and the family, and to avoid having unnecessary troubles.
(4) He/she must take note of the patient’s emotional state and of that of his family, of their educational background, of their awareness of medical topics, and of their expectations.
(5) He/she must try to prevent his/her own emotional reactions while communicating, learn for self-control, use plain vocabulary and avoid using extreme words, guide patiently the patient and the family through their discomfort, and make them accept the facts.
(6) During the treatment, he/she should describe the situation objectively, including the advantages and disadvantages of the different treatment routes and their curative effects. He/she will also list the risks associated with each method and the possible complications in order of severity so that the patient can choose. He/she will also explain the prognosis, the unpredictability of the surgical treatment, and will predict treatment costs, etc.
6.2.3 Information About Super Minimally Invasive Surgery (Surgical Purpose, Surgical Risk and Special Condition Inform)
During the diagnosis and treatment by super minimally invasive surgery, the incidence of postoperative complications on the prognosis is relatively high. Therefore, the patient should be informed cautiously before diagnosis and treatment.
1. Purpose of super minimally invasive surgery. It is to excise the lesion as early as possible to improve the prognosis or specify the pathogen to improve the basis for subsequent treatment.
2. Surgical risks associated with super minimally invasive surgery
(1) General risks: gastrointestinal bleeding, perforation; unpredictable cardiac and breath arrest, thoracoabdominal aortic rupture or sudden death; excessive vomiting, esophageal and cardiac mucosa torn, asphyxia, aspiration, aspiration pneumonia; throat injury, laryngospasm, parotid swelling, mandibular dislocation, operation difficulty due to the patient’s own abnormal conditions or if the endoscopic examination cannot be completed due to the patient’s lack of cooperation with the examination; a large number of clots found in the gastrointestinal cavity, unsatisfactory observation making diagnosis impossible; amnesia may occur after anesthesia and sedation, with possibility of anesthetic accident; other unpredictable accidents.
(2) Diagnosis and treatment risks presented by special groups: elderly patients with cardiovascular and cerebrovascular diseases are prone to cardiovascular and cerebrovascular events. Patients with lung or chest disease, or reduced respiratory compensatory function, have a significantly increased chance of developing intraoperative hypoxemia or postoperative pneumonia. Patients who have been taking anticoagulant and antiplatelet drugs for a long time are prone to intraoperative bleeding or endoscopic hemostasis difficulty.
Severe liver dysfunction leads to obvious abnormal coagulation function. Patients who have previously undergone gastrointestinal reroute surgery, in addition to the difficulty of access, have an increased chance of bleeding and perforation at the surgical anastomosis and the newly created passageway.
3. Special condition informing
(1) Uncertainties in Super-Minimally Invasive Surgical Management: Including Communication Regarding Positive Resection Margins and Potential Necessity for Secondary Surgery.
(2) Understanding Relative and Absolute Contraindications in Super-Minimally Invasive Surgery: While some patients may meet endoscopic indications, clinical judgment must account for critical limiting factors (e.g., advanced age, multiple comorbidities, critical condition). Proceeding with the intervention under such circumstances may pose life-threatening risks, necessitating: Thorough informed consent consultation regarding procedural uncertainties; Postponing the procedure until hemodynamic/metabolic stabilization; Strategic modification of treatment plans based on multidisciplinary risk–benefit analysis.Then the patient must be fully informed and the surgery can be carried out if he/she fully consents; otherwise, the treatment plan must be changed.
(3) The patient must be informed that in case of limited gastrointestinal endoscopy or of complication, he/she will need to be transferred for classical surgery.
(4) It is necessary to inform that there are multiple treatment methods for a disease.
6.2.4 Example of Informational Conversation on Super Minimally Invasive Surgery
Super minimally invasive surgery is one of the means of traumatic diagnosis and treatment, and the written procedure of informed consent must be followed. Both the physician and patient sign the informed consent form in writing. When the patient does not have full capacity to exercise his/her civil rights, the form will be signed by his/her legal representative; when the patient cannot sign due to illness, he/she must authorize one relative to sign in his/her place; to rescue the patient, when the legal representative or the authorized person cannot sign in time, the medical department must be informed, and the form can be signed by a manager or by an authorized director of the medical institution.
6.2.5 Complication Management Principle
Complications are often hemorrhage, perforation, infection, etc. Fasting and venous nutrition should be prescribed if necessary. In case of obvious hemorrhage, hemostasis and wound closure must be performed under endoscopic direct vision, a gastric tube and a naso-jejunal nutrition tube must be placed, and further treatment should be given in the hospital. Symptomatic treatments like fasting, gastrointestinal decompression, antibiotics and nutrition, etc. must be prescribed, and vital signs such as temperature should be closely monitored. In addition, patients with serious complications must be transferred to the surgery department for quick treatment.
6.3 Standards for the Surgical Record of Clinical Cases
As the front-edge field in modern medical technology, super minimally invasive surgery has been widely used in clinical practice. As important medical instruments, surgical records not only write down the entire process of the operation, but also are an important basis for the evaluation of medical quality, the treatment of medical disputes and scientific research and teaching. Previous studies have shown that there are widespread violations of the procedure of informed consent in China, such as abuse of informed consent, excessive requirements for informed consent and non-standard filling. The rate of such breaches of the rule in some general hospitals has even reached more than 14%. It is therefore very important to develop and follow the registration standards for super minimally invasive surgery to guarantee patient safety and improving the quality of medical care.
6.3.1 Basic Requirements for the Surgical Record
(1) Punctuality: the surgical record must be completed by the surgeon within 24 h after the operation, and he must ensure the accuracy and timeliness of the information.
(2) Completeness: the surgical record should be comprehensive and specific, covering the steps of preoperative preparation, the surgical process, the intraoperative findings, and the postoperative treatment and notes, etc.
(3) Accuracy: the record should be objective and truthful, avoid subjective assumptions and vague descriptions.
(4) Normative: the unified writing format and requirements should be observed, medical terms should be used to ensure the record’s compliance with norms and readability.
6.3.2 Specific Contents of the Surgical Record in Super Minimally Invasive Surgery
1. The patient’s basic information
(1) Name, gender, age: ensure accuracy and absence of errors, for subsequent follow-up visits and management.
(2) Admission No., section, bed No.: for medical record checking and data filing.
(3) Occupation, marriage, ethnicity, living place: have certain reference value to medical evaluation and the preparation of treatment plans.
(4) Contact mode: for the contact and follow-up visit in case of emergency.
2. Preoperative diagnosis and surgical indications
(1) Preoperative diagnosis: specifically list the patient’s main diagnosis and secondary diagnosis.
(2) Surgical indications: describe why this kind of surgery has been chosen for the treatment, including factors like condition severity, failure of conservative treatments, etc.
3. Name of the operation and surgeon’s information
(1) Name of the operation: accurately record the name of the kind of super minimally invasive surgery performed.
(2) Surgeons, assistants, anesthetists: specifically record the names of the members of the operating team for responsibility tracing.
4. Anesthesia mode and preoperative preparation
(1) Anesthesia mode: data such as general anesthesia, local anesthesia, etc., the anesthesia process and its effect should be briefly described.
(2) Preoperative preparation: data such as skin preparation, intestinal preparation, premedication, etc.
5. Surgical process
(1) Position and disinfection: the patient’s position and the disinfection in the surgical area must be recorded.
(2) Incisal location and length: the specific location, length and direction of the incision should be specifically described.
(3) Surgical steps: every step of the procedure in the sequence of time in detail, including tissue separation, foci dissection, hemostasis, suture, etc., must be recorded. For super minimally invasive surgery, the minimally invasive instruments used, the technical features and the advantages should be clearly specified.
(4) Intraoperative findings: foci’s location, size, morphology, color, texture and the relation with the surrounding tissues, etc., must be specified, according to the rules of surgical record.
6. The patient’s reaction
The changes in the patient’s vital signs, the effects of anesthesia and the possible abnormal events during surgery must be recorded.
7. Specimen treatment
Check and record whether the specimen incised is integral. For specimens that need to be fixed, it should be fixed on the plate with the scale in the direction of the long axis of the specimen parallel to the scale line. Measure the size of the specimen. Record the treatment conditions, such as pathological inspection, family members checking the specimen, etc.
6.3.3 Postoperative Treatment and Medical Order
(1) Postoperative vital signs monitoring: the patient’s postoperative vital signs change must be recorded.
(2) Gastric tube or anorectal decompression tube and negative pressure suction: its location, the numbers and the amount and properties of the drainage liquid if available; the dressing change should also be recorded specifically.
(3) Drug therapy, including types, dosages of antibiotics, pain-relieving drugs, nutritional support drugs, etc., and how they were administered.
(4) Rehabilitation guidance: the personalized rehabilitation plans envisaged based on the patient’s conditions, including diet, exercise, rechecking time, etc.
6.3.4 Notice and Follow-Up Visit
(1) Notices: the patient and the family should be reminded about matters that need to be respected postoperatively, such as avoiding intensive exercise, etc.
(2) Follow-up visit plans: detailed plans of follow-up visits, including time, contents, etc., so as to timely know the patient’s recovery and adjust the treatment plans, must be prepared.
6.3.5 Writing Norm and Notice
(1) Use medical terms: the accuracy and professionalism of the recorded contents must be ensured.
(2) Clear handwriting: obliteration and wrongly written characters should be avoided; the record should be kept clean.
(3) Objectiveness and truthfulness: the contents should be based on facts, and avoid subjective assumptions and exaggerations.
(4) Privacy should be protected: the principles of medical confidentiality must be strictly followed, in order to protect the patient’s right to privacy.
(5) Timely signature: the surgical record should be signed, dated (with the time) in good time after it has been completed.
(6) Specificity and accuracy: the contents should be specific, medical terms should be used, the accuracy of the information must be verified. Specific elaborations or important findings or special conditions must be mentioned. Pictures can be attached if necessary.
(7) The document must be well organized: the intraoperative findings must be recorded for each surgical step or observation sequence, the record should be kept in a rational and logic manner.
6.3.6 Specific Writing Skills
(1) Medical terms should be used: when recording the intraoperative findings, standard medical terms should be used to ensure the professionalism and accuracy of the records.
(2) Quantitative description: for quantifiable indicators such as lesion size and depth, specific numerical values should be used as much as possible to improve the comparability and reference value of records.
(3) Drawing aids: for complex anatomical structures and procedure processes, explanation by drawing can be supplemented to make the records more visual and easier to understand.
(4) Abnormalities should be noted: abnormalities in the intraoperative findings, such as inconformity of the foci with the preoperative diagnosis, too much intraoperative hemorrhage, etc., must be specially noted and the corresponding treatment measures recorded.
In summary, normative recording of the operative processes in super minimally invasive surgery is a key measure for ensuring the patient’s safety and upgrade medical quality. By preparing and following the standard surgical record requirements, comprehensive, accurate and timely passing of surgical data can be ensured to provide a powerful support to the evaluation of the medical quality, to the handling of medical dispute and to scientific research and teaching. Meanwhile, it is also helpful to promote trust and cooperation between doctor and patient, building a harmonious doctor–patient relationship.
6.4 Perioperative Diagnosis and Treatment Criteria
Postoperative diagnosis and treatment standards are an important element in ensuring the smooth rehabilitation of the patient who has undergone surgery and preventing the onset of complications. This section will specifically detail the requirements of postoperative diagnosis and treatment criteria based on multiple aspects of preoperative preparation, intraoperative operation, postoperative nursing, rehabilitation guidance, prevention of complications and management of follow-up visits, etc.
6.4.1 Preoperative Preparation
1. Patient evaluation and examination. Before super minimally invasive surgery, a comprehensive physical evaluation of the patient should be carried out, including three major routine examinations (blood, urine, stool), blood biochemical examination, ECG, abdominal ultrasound B, etc. Patients who need surgery require special screenings for hepatitis B, AIDS, syphilis as well prothrombin tests, etc. Depending on the patient’s specific condition, other screenings for CT, MRI, PET–CT, for instance, can also be necessary.
2. Nutrition evaluation and support. Nutrition habits directly affect postoperative recovery. Therefore, a nutritional screening should be carried out within 24 h of the patient’s admission and nutritional intervention must be provided for patients with nutritional risks. Patients with diabetes should pay special attention to perioperative blood glucose assessment and regulation to ensure that blood glucose levels are controlled within a safe range.
3. Psychological counseling and education. Preoperative communication should be carried out with the patient and the family to inform them of the purpose of the surgery, the risks, the expected results and the postoperative consequences. This can be done through diversified and multi-mode education methods, such as oral education, advertising brochures, multimedia video, etc., to help patients relieve their anxiety and improve treatment compliance.
6.4.2 Intraoperative Operation
1. Surgical safety and risk assessment. The requirements in Operation Safety Inspection System, Operation Risks Evaluation System and Operation Site Marking System must be strictly implemented to ensure the safety of the surgical operation. Before the operation starts, comprehensive preoperative assessment should be carried out on the patient to determine the surgical indicators and prepare detailed operation plans.
2. Infection prevention. During the operation, aseptic procedures must be strictly observed, and qualified surgical instruments and consumables should be used. For patients who require full-thickness resection, the prophylactic use of antimicrobials in the perioperative period is recommended to reduce the risk of infection.
3. Delicate operation and hemostatic management. The operation must be delicate and precise to reduce damage to the surrounding tissues. Hemorrhage should be effectively controlled with reasonable hemostasis during the operation. Rescue measures like blood transfusion should be adopted in time for patients with massive hemorrhage.
6.4.3 Postoperative Nursing
1. Condition monitoring and evaluation. A health assessment must be regularly carried out after the operation, including monitoring of vital signs such as temperature, pulse and blood pressure, etc., as well as evaluating the degree of pain. Abnormal conditions should be treated in time and reported to the head physicians.
2. Diet arrangement and nutrition support. Reasonable diet plans adapted to the patient’s condition and type of surgery must be prepared. The patient must be encouraged to intake high-protein, high-vitamin and easy-to-digest foods to ensure sufficient nutrition. For patients who cannot have oral feeding, nutrition can be supplemented by means of intravenous infusions, etc.
3. Exercise guidance and rehabilitation exercise. Provide appropriate activity guidance to patients according to postoperative rehabilitation requirements. Exercises in bed, like flexion and extension exercises, etc., may be recommended in the early times; with the betterment of the condition, the number of exercises performed out of bed may be gradually increased. For patients with a fracture that needs immobilization, rehabilitation exercises should be conducted under the guidance of a doctor to avoid joint stiffness and myophagism.
4. Drug use management and complication prevention. Medicines should be administered on the basis of medical prescriptions and ensure that the patient takes his/her medication on time. Meanwhile pay attention to the side effects and interactive effects of drugs so as to adjust medication plans timely. Preventive measures to avoid complications such as infections, embolism, etc., must be anticipated for the different types of surgery and patient conditions.
6.4.4 Prevention of Complications
1. Infection prevention. Attention should be closely paid to indicators like changes in the patient’s temperature; screening for blood infection should be carried out postoperatively in order to timely discover and treat the signs of infection. For patients at risk of infection, the management of antibiotic use and wound care should be improved.
2. Embolism prevention. For patients at risk of deep vein thrombosis, necessary preventive measures should be taken, such as anti-muscle contraction activities, wearing thrombotic elastic stockings, plantar soles, and intermittent inflatable compression devices. Also attention should be paid to changes in the patient’s symptoms, such as swelling and pain in the lower limbs, etc., in order to proceed with the appropriate examinations and treatments in good time.
3. Prevention of other complications. Depending on the type of the surgery and the patients’ conditions, preventive measures should be implemented to prevent other complications, such as pulmonary embolism and respiratory insufficiency, etc.
6.4.5 Management of Follow-Up Visits
1. Regular follow-up visit and evaluation. Regular follow-up should be arranged after surgery to monitor the patient’s recovery and timely find and solve any problem. Follow-ups include wound curing, function recovery, and management of complications, etc.
2. Discharge guidance and subsequent treatment. Comprehensive evaluations must be conducted before the patient is discharged. Discharge standards must be specified and detailed discharge guidance plans must be prepared, including the names of the discharge medications, their method of use, their dosage and recheck time, etc.
3. Follow-up records and data analysis. During follow-up visits, the changes in the patient’s condition and treatments should be specifically recorded; data should be regularly analyzed to evaluate treatment results and improve treatment plans. Communication and contact with the patient and the family will help to improve satisfaction and confidence.
In summary, the postoperative diagnosis and treatment norms are important guarantees of a smooth recovery for the patient who has undergone surgery and for the prevention of complications. By strictly observing the requirements for the steps of preoperative preparation, intraoperative operation, postoperative nursing, rehabilitation guidance, complication prevention, and follow-up visit, the success rate of the surgery and the quality of the patient’s recovery will be significantly improved. In the future, with the continuous development of medical technology and the update and iteration of the diagnosis and treatment philosophy, the postoperative diagnosis and treatment norms will be continuously improved and optimized to meet the changes in clinical needs.
References
[1] Pawlik T. M., Schwartz T. A., Itani K. M. F. (2023 Feb 1) Practical guide to ethical considerations in clinical trials in surgery, JAMA Surg. 158(2), 204–205.
[2] Giampieri M. (2012 Feb) Communication and informed consent in elderly people, Minerva Anestesiol. 78(2), 236–42.
[3] Kshirsagar M. M., Dodamani A. S., Dodamani G. A., Khobragade V. R., Deokar R. N. (2021) Impact of covid-19 on mental health: an overview, Rev. Recent Clin. Trials. 16(3), 227–231.
[4] Zheng C., Zhang X. (2020) Positive effect of narrative medicine on preoperative conversation of cancer patients, Chin. Med. Ethics 33(01), 85–91.
[5] Mutebi M. (2019 Feb 9) Africa rising: gendered journeys of women patients and providers, Lancet 393(10171), 517–518.
[6] Lu G. (2013) The art and skill of preoperative communication in obstetrics and gynecology, Guide China Med. 11(11), 372.
[7] Xu L., Pan S., Xia P., et al. (2018) Present situation and countermeasures of medical records writing in Zhejiang Province, Chin. Med. Rec. 19(4), 6–10.
[8] Liu L., Liu R. (2023) Analysis on the implementation status of informed consent in level 3 and 4 surgical medical records in a hospital, Chin. Med. Rec. 24(3), 17–19.
[9] Chinese Society of Organ Transplantation of Chinese Medical Association. (2019) Diagnosis and treatment specification for postoperative complications after liver transplantation in China (2019 edition), Chin. J. Transplant. (Electron. Ed.) 13(4), 269–272.
[10] Cao M., Wang Q., Tao H., et al. (2023) Interpretation of evidence-based guideline for the prevention and management of perioperative infection, Clin. Med. J. 21(06), 19–25.
[11] Interventional Physicians Branch of Chinese Medical Doctor Association, Interventional Medicine Professional Committee of Chinese Research Hospital Association, Ni F, et al. (2022) Expert consensus on the use of antibiotics during perioperative period of interventional diagnosis and treatment, J. Interventional Radiol. 31(04), 319–327.
[12] Li Z., Yan C., Li S. (2020) Consensus of Chinese expert panel on perioperative nutrition therapy of gastric cancer (2019 edition), Chin. J. Pract. Surg. 40(02), 145–151.
[13] Liu F., Lou W., Miu C., et al. (2021) Shanghai expert consensus on perioperative management of antithrombotic patients undergoing non-cardiac surgery (2021 Edition), Shanghai Med. J. 44(08), 537–544.
Chapter 7 Preoperative Preparation and Treatment of Complications in Super Minimally Invasive Surgery
7.1 Preparation Before the Endoscopic Operation
7.1.1 Preparation of the Endoscopic Operation of the Upper Digestive Tract
1. Defoaming agent. Currently, the defoaming agents routinely used before gastroscopy in hospitals include dimethylsilicone oil powder and simethicone powder.
(1) Dimethicone oil powder is one of the most commonly used defoamer. It has stable properties and no physiological activity. It is not absorbed by the digestive tract and is harmless to the human body. In 1954, it was reported to have an antifoaming effect in endoscopy. Studies have shown that the clarity of the mucosal field of view under gastroscopy after taking dimethylsilicone oil powder is significantly better than with a lubricant, and that its defoaming effect is fine, which makes it possible to identify precancerous lesions, to diagnose EGC, to reduce the duration of gastroscopy as well as the incidence of adverse reactions, and to improve patient tolerance.
(2) Simethicone powder is a compound of dimethyl silicone oil and silicon dioxide, which is a non-ionic surfactant with a banana flavor. It is an inert substance in physiology and pharmacology. It is not absorbed after oral administration, no toxicity has been reported, and it is a good defoaming agent. Studies have shown that different doses of simethicone powder (200, 400, 600 mg) are beneficial for gastroscopy. With increasing the dosage of simethicone powder, the clarity of the gastroscopic visual field gradually increases, and the dosage of propofol as well as the duration of gastroscopy decreases accordingly. But large doses of simethicone will increase the incidence of adverse reactions. So the most suitable dose of simethicone powder is 400 mg, at which dose the clarity of the visual field is improved and the risk of adverse reactions is reduced.
2. Oral anesthetics
(1) Dyclonine hydrochloride gel is a compound preparation for anesthesia, defoaming and lubrication, which blocks nerve impulse and stimulation, suppresses touch, and relieves discomfort. The drug has the characteristics of little toxicity, little influence on the central system, strong anesthetic effect, fast acting, long-acting time and good defoaming effect. Some studies have found that the operation time, operation satisfaction rate and mucosal visibility of gastroscopy after the application of 10 ml of dyclonine hydrochloride mucilage are better than one of 8 ml.
(2) Lidocaine hydrochloric jelly is a light yellow, viscous liquid compound preparation; its main components are lidocaine, dimethylsilicone oil, etc.; it makes the field of vision clear, conducive to observation; it also has the effect of local anesthesia, which reduces the irritation to the throat when entering the endoscope. Some studies have found that compared to a stock solution of lidocaine hydrochloric jelly, the anesthetic effect of 10 ml of lidocaine hydrochloric jelly (spec 10 ml: 0.2 g) diluted in 50 ml of water was similar, but the efficiency of the defoamer and the total effective rate of the comprehensive treatment were higher, whereas no adverse reactions occurred in either group.
3. Slime remover
Streptomyces protease is a powerful proteolytic enzyme isolated from a Streptomyces culture in 1962. It has been used as a pre-endoscopy drug since 1991 because of its ability to break glycoprotein–peptide bonds in gastric mucus, thereby dissolving it. Many studies have shown that Streptomyces protease has the advantages of mucosal clarity, high detection rates of small lesions, of precancerous lesions and of early gastric cancer, and short inspection operation time. Moreover, the incidence of undesirable effects is low with Streptomyces protease and its safety is high.
N-acetylcysteine (NAC) is often used to remove respiratory mucus. It can also split mucin peptides in gastric mucus to reduce viscosity and facilitate aspiration. When combined with dimethicone, it also improves the clarity of gastroscopic vision. Some studies found that compared to dyclonine hydrochloride jelly alone, acetyl cysteine combined with dyclonine hydrochloride jelly effectively improves the clarity of gastroscopy field, increases the detection rate of minor lesions, and shortens the examination time.
Chymotrypsin is a decomposing enzyme that acts on the peptide chain to break it into small molecules, or acts on the end of the peptide chain to break it into amino acids. Chymotrypsin breaks down the mucus on the gastric wall into a liquid form, which is beneficial for drainage. Studies have shown that chymotrypsin effectively improves the clarity of the gastroscopy field, shortens the operation time, and improves the detection rate of minor lesions, precancerous lesions, and EGC.
Before gastroscopy, defoaming agents or mucolytic agents are commonly used, but it is difficult to achieve the ideal clinical effect with a single drug, so clinical combination of drugs is needed to synergize the effect. There is no unified standard for the treatment with a defoamer combined with a slime remover. Some studies have found that the addition of Streptomyces protease to dimethicone powder and dyclonine hydrochloride mucilage is advantageous for improving mucosal clarity and visual field clarity, and the detection rate of small lesions.
Before gastroscopy, different administration times of defoamer and slime remover can have different effects. Numerous studies have shown that the best cleanliness and visualization were obtained with water, simethicone and Streptomyces protease 30–60 min before inspection. Some studies have shown that with the continuous extension of the application time of the chain enzyme protease, the clarity of the visual field will be reduced to varying degrees, and the clarity of the gastroscopy visual field will be the lowest after 60 min. The reason may be related to the decrease of Streptomyces protease activity with the prolongation of the action time, and the dilution of the drug solution caused by the ingestion of oral endocrine substances in the stomach of the patient.
7.1.2 Preparation of the Endoscopic Operation of the Lower Digestive Tract
(I) Diet preparation
Low-residue/low-fiber foods help to improve the effectiveness of intestinal preparation. It is recommended to stop consuming high-residue, high-fiber foods within 24 h before colonoscopy. The types of diets that can be prescribed before examination include:
Clear liquid diet, i.e. a clarified liquid or semi-liquid diet, not including milk and orange juice, etc. Colored liquids and foods affect the results of a colonoscopy. Although these foods are quickly, eliminated, they do not satisfy hunger and often reduce the patients’ willingness to undergo colonoscopy again.
Low residue/low-fiber diet, i.e. a diet that minimizes residue after digestion. Clinically, the low-fiber diet is commonly used to replace the low-slag diet, and the more uniform standard for fiber intake is less than 10 g.
(II) Common drugs for cleaning intestines
(1) Polyethylene glycol (PEG) electrolyte powder: PEG is a polymer formed by inert ethylene oxide, and it is mixed with water to form an isotonic solution. It is a volumetric laxative, which does not absorb or metabolize. It has no obvious effect on intestinal absorption and secretion function by oral cleansing of a large amount of liquid, and it does not easily cause water and electrolyte disturbance. Compared to other cathartic agents in intestinal preparation, PEG has advantages in terms of quality of intestinal preparation, incidence of adverse effects, patient tolerance, etc. Therefore, PEG electrolyte powder is the most commonly used intestinal cleanser in China. Studies have compared PEG and magnesium sulfate cleansing regimens, and the intestinal cleansing efficiency of the magnesium sulfate group is significantly lower than that of the PEG group, while the incidence of adverse reactions is higher. The compliance of compound polyethylene glycol electrolyte powder greatly affects the effect of intestinal preparation. Single PEG regime’s routine use: the patient starts taking it 4–6 h before colonoscopy, and completes the taking within 2 h; during treatment, an appropriate amount of exercise and abdominal massage can be taken to accelerate the excretion of intestinal peristalsis. Generally, after 1 h of taking the drug, bowel movement accelerates and defecation gradually begins. Patients may feel abdominal distension before defecation, so they can postpone taking the medicine until the symptoms have disappeared and continue to take the medicine until the excretion of clear watery stool. If the defecation characteristics do not meet the above requirements, PEG solution or water can be added, but the total amount is generally not more than 4 L.
(2) Magnesium sulfate is a hyperosmolar solution, which increases the osmotic pressure in the intestinal cavity, stimulates the secretion of intestinal fluid, prevents the absorption of intestinal water, and promotes intestinal peristalsis, so as to clean the intestinal cavity. Therefore, magnesium sulfate can be used as a cleaning agent for intestinal preparation. But due to the risk of intestinal mucosal inflammation, ulceration and hypermagnesemia induced by dehydration, magnesium ion aggregation is not recommended for use in patients with inflammatory bowel disease, suspected inflammatory bowel disease and abnormal kidney function. Routine usage: 50 g of magnesium sulfate diluted in 100 ml of clear water is taken in one go, then the patient drinks 2 L of water. If clear watery stool is observed, water intake may no longer be continued.
(3) The advantage of the sodium phosphate cleansing regimen is that only a small amount (about 1.6 L) of a lemon-flavored solution is ingested orally. Chinese and foreign studies have shown that compared with the oral 4 L PEG solution, the oral sodium phosphate solution has the same intestinal preparation effect, and the patient compliance is better, and there are fewer gastrointestinal adverse reactions such as nausea, vomiting, abdominal distension, etc. So it can be used for specific conditions of intestinal preparation, especially for patients who cannot tolerate oral high-dose intestinal cleansers. But since sodium phosphate preparation is a hypertonic solution, it may induce epilepsy, phosphate nephropathy, and arrhythmia related to electrolyte disturbance during intestinal preparation. According to relevant studies in China and abroad, the elderly population should be asked about their disease history in detail before using sodium phosphate for intestinal preparation to prevent complications. It should be avoided by those with conditions such as intestinal obstruction, arrhythmia, heart failure, chronic kidney disease, electrolyte disorders, or those taking medications such as angiotensin-converting enzyme inhibitors (ACEI) or angiotensin-receptor blockers (ARBs). Routine usage: most patients adopt the sodium phosphate subdose regimen, that is, the first dose is taken 10–12 h before the examination, by dissolving the sodium phosphate powder in at least 800 ml of warm and cool water or diluting the oral solution, and the taking is repeated once 3–5 h before the examination.
(4) Appropriate amount of defoaming agent to improve the intestinal preparation quality: currently, the defoaming agents commonly used for intestinal preparation are mainly simethicone or dimethicone. The appropriate application of a defoaming agent during intestinal preparation not only reduces the number of bubbles that interfere with vision, but also relieves bloating and nausea, thereby improving the quality of the intestinal preparation. Usage recommendations: 15–30 ml of silicone oil can be taken with the last part of the laxative, or within 30–60 min after the laxative is taken; 12 ml of a dimethylsilicone oil emulsion (or 5 g of dimethylsilicone oil powder) is mixed with 100–200 ml of water at room temperature; the mixture must be shaken evenly, and ingested after the last cleanse is taken.
7.1.3 Preparation Before an Enteroscopic Operation
(I) Decide the access path
Generally speaking, for patients with suspected jejunal lesions (black stool as the main manifestation, or capsule endoscopy prompt time index ≤0.6, small intestine three-dimensional CT/MRI indicating that the lesions are located in the jejunum), it is recommended to choose the oral approach for the first colonoscopy. For patients suspected of ileal disease (with hematochezia as the main manifestation, or capsule endoscopy prompt time index >0.6, small intestine three-dimensional CT/MRI indicating that the lesion is located in the ileum), it is recommended to choose the peroral approach for the first colonoscopy. Meanwhile, the endoscopic approach can be selected based on the common site of the disease. For example, when Crohn’s disease is suspected (it is likely to occur in the ileum), peroral examination is preferred, and P–J syndrome (it is likely to occur in the jejunum) can be selected for oral examination.
(II) Anesthesia or sedatives
Enteroscopy is recommended to be performed under anesthesia or sedation. Intravenous anesthesia is usually used, and propofol and other drugs are injected/pumped intravenously slowly. Sedation can be achieved with drugs like midazolam, but all require an ECG and blood oxygen monitoring. During peroral channel examinations, tracheal cannula anesthesia is recommended to avoid aspiration and reduce the complication rate of aspiration pneumonia after examination. During transanal channel examinations, usually intravenous anesthesia is enough. However, a trachea cannula is also required when patients suffer from gastric retention or intestinal obstruction. Therefore, before the endoscopy, the anesthetist should do the relevant assessment work, and anesthesia can be implemented when the patient’s condition meets the requirements of anesthesia. When patients have anesthesia contraindications, in special circumstances, such as patients with strong indications of endoscopy (continuous digestive tract bleeding, capsule endoscopy or routine imaging clearly indicating small intestine lesions, etc.), And it is estimated that lesions can be detected quickly, under the premise of full communication with patients and their families, enteroscopy can be performed by sedation (pethidine, diazepam).
(III) Intestinal preparation
The low-fiber diet must be started one day before the examination; fasting must be observed after dinner. Peroral examinees will be fasting for 8–12 h, without drinking water for 4–6 h; the intestinal preparation plan of the transanal examinees is the same as preoperative preparation for colonoscopy. For patients with incomplete intestinal obstruction, endoscopy should be performed after the intestinal obstruction is relieved and the corresponding intestinal preparation is completed as much as possible.
(IV) X-ray equipment
X-ray is not a necessity for endoscopy, but it improves the efficiency of the endoscope entrance and the penetration depth. For units that first implement endoscopy, the operation should be arranged to be performed in an operating room equipped with X-rays, because it is useful to observe the entrance depth and location of the endoscope and thus aids in resolving the loop formation. In case of suspected local fistula or obstruction of the small intestine, an intraoperative angiography can also be performed (negative preoperative contrast agent allergy test).
(V) CO
Nowadays, there is sufficient evidence showing that the use of CO
(VI) Verification of the integrity of the equipment
The operator must carefully check the integrity of the apparatus before the operation, such as machine equipment, outer casing, air bag, air pump, etc. In particular, it is necessary to pay attention to whether the outer casing or the balloon at the front of the endoscope has air leakage or cannot complete gas injection/venting. The abnormal functioning of the airbag is usually caused the blockage or incorrect installation method of the gas injection pipe of the endoscope or the outer casing, which needs to be rechecked and replaced.
7.1.4 Preparation Before an Operation by Duodenoscopy
Esophageal-gastro-duodenoscopy (EGD): pre-medicated simethicone or simethicone combined with n-acetylcysteine significantly improve esophageal and stomach visibility. When undergoing gastroscopy, patients are usually advised to fast for 4–6 h to prevent aspiration. However, the latest data suggests that endoscopy, which allows clear fluids to pass through for up to 2 h, improves the comfort in anxious patients without increasing the regurgitation of gastric contents.
7.1.5 Preparation Before the Ultrasonic Endoscopy Examination
The preoperative preparation for ultrasound gastroscopy is often the same as that of gastroscopy. However, in addition to helping to observe the mucosal surface, water should also be used as the medium to observe the lesion’s level during ultrasound gastroscopy. If the medium is mixed with gas or mucosal surface liquid, the ultrasound gastroscopy observation will be inaccurate. Therefore, compared to ordinary gastroscopy, ultrasonic gastroscopy has higher requirements for mucosal cleanliness. Currently, it is recommended to use Streptomyces protease and dimethylsilicone oil as the preoperative preparation for ultrasonic gastroscopy. The results of adjuvant drugs such as tetracaine hydrochloride, belladonna tincture, etc., combined with traditional preoperative drugs have improved the preoperative gastroscopy, but the appropriate proportion is not clear at present. The preparation of combined drugs is a long and tedious process, requiring specialized personnel, lack of research with large sample sizes. Even if multi-center, prospective studies have made it possible to develop a new clear and transparent oil-in-water (O/W) DM nanoemulsion (DMNs) to improve the problems of matching and proportional distribution, its practical clinical applicability has yet to be confirmed.
7.1.6 Preparation Before the SpyGlass Operation
Peroral cholangioscopy is a high-risk endoscopic procedure that requires deeper sedation than conventional peroral endoscopic procedures. The preoperative evaluation involves clinical features, patient baseline, and preoperative imaging, etc. However, no uniform conclusion can be reached on whether a preoperative EGD surgery is necessary. Currently, no studies have shown that preoperative EGD can help avoid postoperative and intraoperative complications. For patients without dysphagia, presumed stenosis, or anatomical changes in the upper GI tract, there is no indication for performing a preoperative EGD. Preoperative EGD guarantees the safety and success of peroral cholangioscopy in the event of anatomic changes in the upper gastrointestinal tract resulting in stenosis or in other dynamic problems.
7.1.7 Hot-Spot Issues in the Consensus
(I) Personalized preoperative preparation for colonoscopy and novel intestinal preparation drugs
Before colonoscopy, it is necessary to fully evaluate the patient’s physical condition and conduct adequate bowel preparation. The success rate of intestinal preparation could be higher than 90%, which can considerably improve the detection rate, diagnosis and treatment of colorectal cancer and early colorectal lesions by colonoscopy. Boston Bowel Preparation Scale (BBPS) is recommended for preoperative preparation assessment. A BBPS score ≥ 6 is considered as good, and can further reduce the rate of missed diagnoses. Except for the conventional artificial medical structure preoperative scoring, the artificial intelligence scoring system assists endoscopic operators for further analyses. In the colonoscopy process, deep learning-based AI can already achieve real-time feedback on the quality and cleanliness of bowel preparation. The evaluation by the artificial intelligence system is relatively objective and uniform, thereby reducing evaluation deviations. It may now be necessary to further train and validate AI comprehensive bowel preparation analysis in multi-center, large sample, RCT studies.
For the use of cleansers required for bowel preparation, the selection of the most appropriate drug and the frequency of medication intake must be determined by taking into account the patient’s general state of health, his/her medical history, his/her history of medication intake, his/her preferences in terms of flavors, his/her past bowel preparation conditions, etc. The most commonly cleanser used in China is PEG. PEG has advantages in terms of quality of bowel preparation, lower incidence of adverse reactions, patient tolerance, etc. For patients without risk factors in the event of inadequate bowel preparation, a single taking of 2 L of PEG may be used because there is no difference in outcomes between a single dose of 2 L PEG and a dose of 4 L in this population. For patients with risk factors in the event of inadequate bowel preparation, a segmented regimen of PEG should be used for bowel preparation. Currently the latest Chinese expert consensus points out that the risk factors for inadequate bowel preparation include so far: chronic constipation, bowel preparation not strictly conducted according to the requirements (e.g., preoperative high-fiber diet, inadequate PEG dosage), BMI > 25 kg/m
The advantage of the sodium phosphate cleansing regimen is that only a small amount (about 1.6 L) taken orally of lemon-flavored solution is needed. Studies in China and abroad have shown that the sodium phosphate solution taken orally has the same effect of bowel preparation as the PEG solution taken orally, and that patient compliance is better. Therefore, it can be used for bowel preparation in specific situations, especially for patients who cannot tolerate large doses of oral bowel cleansers. Beware of adverse reactions such as epilepsy. At present, the herbs commonly used in China in bowel preparation include senna leaf and castor oil, etc. Studies have shown that after senna leaf medicine, the cecal intubation rate during the endoscopic procedure reaches more than 90%, the disease detection rate reaches 40%, and the patient’s satisfaction is improved somehow. However, the occurrence of complications such as diarrhea limits its application and its role is generally limited to that of an adjuvant. In addition to this, patients with risk factors and with inadequate bowel preparation can continue to be assisted by other drugs, such as rubiprostol. It acts as a chloride channel (type-2 chloride channel, CIC-2) activator and increases bowel secretion and transport, thereby promoting defecation. Multiple RCT studies have confirmed that PEG combined with rubiprostol provide better bowel cleaning. Linalotide is a guanylate cyclase C receptor activator, which promotes the secretion of bowel fluids, increases the water content in the feces, and promotes the opening of chloride ion channels, increasing bowel operation and promoting defecation; existing studies have shown that preoperative preparation combined with PEG has a good cleaning effect. Lactulose is decomposed into low-molecular-weight organic acids such as lactic acid and acetic acid in the colon, resulting in a pH decrease in the bowel, stimulating bowel peristalsis and producing mild catharsis. Compared to the oral administration of PEG alone, the combined use significantly increases defecation frequency in patients suffering from constipation and improves the bowel cleansing effect. For enemas, there is no clear background allowing to improve the effectiveness of the preparation.
For elderly patients, their gastrointestinal motivity is significantly decreased, the colon pouch is relaxed, and bowel preparation is generally inadequate, so the preparation efficiency can be greatly improved by a segmented oral PEG plan. For children younger than 2 years of age, a saline enema is generally recommended for cleansing purposes; in patients with active lower gastrointestinal bleeding undergoing emergency colonoscopy, PEG can be added orally if tolerated.
(II) Evaluation indicators for preoperative examination of small intestine capsule endoscopy
Related preoperative small-bowel endoscopy: small-bowel capsule endoscopy (SBCE) and device-assisted enteroscopy (DAE), air bubbles, food substances in the small-bowel, multiple factors of transport time between stomach and small-bowel affecting small-bowel visualization quality (SBVQ), diagnostic yield (DY) and cecum rate (CR), the preoperative preparation uses PGE to enhance SBVQ and DY, but has no effect on CR. Fasting or PGE combined with siloxane enhances SBVQ but has no effect on CR. The use of pro-kinetic bowel preparations is generally not recommended. The optimal timing of bowel preparation for defecation has not been determined. The European Society for Gastrointestinal Endoscopy (ESGE) recommends fasting without solid foods for 8–12 h and fluids for 4–6 h before anterograde DAE. For retrograde DAE, a colonoscopy preparation regimen is recommended; conscious sedation, deep sedation, and general anesthesia are all acceptable alternatives: the choice between them must be controlled by surgical complexity, clinical factors, and local organizational protocols; if the location of the small-bowel lesion is unknown or uncertain, the ESGE recommends that the anterograde approach be generally preferred, and in case of significant bleeding, ESGE recommends the initial anterograde approach; if the location of the small-bowel lesion is unknown or uncertain, the ESGE recommends that the anterograde route be generally preferred.
For esophageal-gastro-duodenoscopy EGD: pre-administered simethicone or simethicone combined with n-acetylcysteine significantly improves esophageal and gastric visibility. Adding protease to simethicone improves the gastric visibility score; the use of helicase in endoscopic irrigation during the biopsy reduces mucus thickness, biopsy depth, and improves diagnostic evaluation during endoscopic biopsy. During gastroscopy, patients are usually advised to fast for 4–6 h to prevent aspiration; however, the latest data suggest that endoscopy, which allows clear fluids up to 2 h, improves the comfort of anxious patients without increasing the regurgitation of gastric contents.
(III) Novel preoperative magnetic-controlled capsule endoscopy
The emerging magnetic-controlled capsule endoscopy does not require intubation, which significantly reduces the pain of patients with good tolerance, and is expected to further replace the traditional gastroscope in the future and to become a more convenient and comfortable examination method. Traditional capsule endoscopy cannot actively observe the gastric mucosa, and external training using the in vitro magnetic field control allows for much better use capsule endoscopy in clinical practice; this generally applies to populations who cannot tolerate traditional gastroscopy methods. The stomach preparation plan is somewhat different. It not only requires full filling of the gastric cavity to reduce the folding of the gastric wall, but also needs the elimination of mucus and bubbles from the gastric wall and improvement of the gastric visual field. Fasting for more than 8 h is required before surgery; an appropriate amount of defoamer (10–30 ml of simethicone) must be ingested 40–60 min before the examination, and 20 000 U of Streptomyces protease can be added at the same time; repeated turning over exercises to change position must be performed during about 15 min; starting 10 min before the examination, the patient should drink water in a few times until there is a feeling of fullness in the abdomen. The specific recommendations are as follows:
(1) Stomach preparation must be routinely performed with water and defoamer, and protease preparations can be added;
(2) After taking the defoamer orally, the patient should repeatedly turn over to change position, which can improve the cleanliness of stomach preparation;
(3) If the visual clarity for stomach preparation is not good, measures such as ingesting an appropriate quantity of drinking water, changing position, preparing again after stomach emptying, etc., may improve the situation.
(IV) Special preoperative examination of inflammatory bowel disease
For some patients with special diseases, such as inflammatory bowel disease, a slag-free semi-liquid or liquid diet is recommended one to two days before endoscopy. The preferred bowel cleanser is polyethylene glycol electrolyte powder, meanwhile taking silicone defoamer. Salty cleansers that may cause mucosal inflammation or ulcers must be avoided. Ingesting 3 L of polyethylene glycol is recommended for transanal colonoscopy; peroral endoscopy can be performed by fasting for 12 h or orally taking 1–2 L of polyethylene glycol. For the particular case of IBD patients, especially with gastrointestinal bleeding, incomplete pyloric or intestinal obstruction, hypoproteinemia, severe anemia, water and electrolyte disorders or elderly patients, bowel preparation should be adjusted accordingly.
7.2 Preoperative Preparation of the Patient for Super Minimally Invasive Surgery
7.2.1 Medical Preparation
The medical prescriptions should be followed, fasting and not drinking for more than 8 h before the gastroscopy; bowel preparation should be made before the colonoscopy.
7.2.2 Nursing Preparation
(I) Psychological nursing
By explaining in detail the purpose, the method, the basic operation process and the advantages of super minimally invasive surgery, the effect of anesthetic drugs, the possible complications, the intraoperative precautions and the nursing effects, the patient’s psychological preoperative nervousness, uneasiness and anxiety will be alleviated, concerns will be eliminated, and a favorable mental state will be maintained. If the patient is nervous and has difficulty falling asleep the night before surgery, sedative and sleeping drugs can be given. The patient should fully understand the necessity of surgery, understand his/her condition through communication with the medical staff in order to eliminate feelings of fear.
The patient should be encouraged and comforted, so as to maintain his/her emotional stability and to build up his/her self-confidence to overcome the disease. Sharing successful medical histories with the patient will arouse his/her enthusiasm and encourage him to cooperate with the treatment, to pay attention to his/her rest and to maintain a good mental state.
(II) Routine preparation
(1) Preoperative routine examination of blood routine, blood biochemistry, serum four items, coagulation four indices, blood type, electrocardiogram, chest X-ray, etc.; blood samples should be prepared if necessary.
(2) If taking aspirin, NASID and antiplatelet agglutination drugs, the patient should contact the doctor and stop taking them for seven to ten days before surgery, depending on his/her condition.
(3) The operation informed consent should be routinely signed. If the patient needs to undergo anesthesia, he/she must be told of the operating process of sedation and anesthesia, and be explained the purpose and risks of sedation anesthesia before consenting and signing the anesthesia informed consent.
(4) For smokers, smoking should be banned one day before operation; the elderly and physically weak patients should be accompanied by their family, and female patients should avoid to have surgery during their menstrual period.
(III) Diet nursing
1. In case of peroral super minimally invasive surgery. Fasting should routinely begin 8–10 h before the operation, and fasting on water 2 h beforehand. Patients undergoing peroral endoscopic super minimally invasive cardiac sphincterotomy should not have eaten or drunk anything 48 h before the operation to ensure en empty stomach.
2. In case of transanal super minimally invasive surgery. The patient should eat slag-free or low-slag diet two days before surgery, and staple food is not limited. He/she should eat vegetables containing less fibers, e.g., eggs, bean products, etc. Do not eat or eat less fruit and meats.
Bowel preparation: Bowel cleansers, most commonly polyethylene glycol electrolyte powder must be taken orally. The day before the examination, 2 h after dinner, a box of polyethylene glycol electrolyte powder 82.2 g dissolved in warm water should be drunk within 30 min; 4 h before the operation, three boxes of polyethylene glycol electrolyte powder 246.6 g dissolved in warm water should be drunk within 2 h. While taking the medication, patients who are able to move should walk sufficiently, and gently rub their abdomen until the stools become liquid and clear. The patient should drink 15 ml of simethicone and then take laxatives for 15 min. For patients with constipation and elderly, whose peristalsis is poor, if bowel preparation is poor, a cleansing enema can be administered if necessary. The use of mannitol for catharsis must be forbidden, so as to avoid the occurrence of inflammable gas in the bowel, leading to an explosion charged with electricity. For patients who must use it, inert gas should be used to thoroughly replace flatulence gas.
(IV) Drug nursing
1. In case of peroral super minimally invasive surgery. Intake of drugs like anticoagulants, antiplatelet agglutination drugs, hypoglycemic medications, etc., must be stopped before surgery, whereas antihypertensive drugs are to be taken orally on the day of the surgery. The diabetic patient can keep colorless sugar bars to hand and take them if necessary.
2. In case of transanal super minimally invasive surgery. Intake of drugs like anticoagulants, antiplatelet agglutination drugs, hypoglycemic medications, etc., must be stopped before surgery, whereas antihypertensive drugs are to be taken orally on the day of the surgery. After clear purging. The diabetic patient can keep colorless sugar bars at hand and take them if necessary.
(V) Other nursing care
(1) A preoperative nursing diagnosis must be established based on the patient’s own condition as well as the corresponding nursing plans and nursing goals based on the nursing diagnosis. Improving the patient’s general condition, establishing a good nurse–patient relationship so that the latter trusts the medical staff and is confident in his/her ability to overcome the disease will all contribute to better cooperation with the doctor for effective treatment.
(2) One day before treatment, do personal cleaning, bathing, washing hair, shaving, cutting nails, etc. well. Before surgery, women should not wear heavy makeup. All accessories should be removed on the day of surgery, including glasses, contact lenses, jewelries, watches, hearing aids, movable teeth and other personal items. Patients wearing pacemakers should ask the cardiology department for consultation.
(3) Complete medical records should be compiled and verified, including all laboratory records and data such as examination documents. Data like the patient’s name, gender, age, surgical item, weight, etc., must be checked, so as to know his/her medical history and examination in detail, whether there are contraindications, etc. Change loose clothing and enteroscopy pants for patients undergoing transanal super minimally invasive surgery.
(4) Establish the intravenous channel; the first choice is the right upper limb. Monitor and record vital signs. Evaluate the duration of super minimally invasive surgery, and if it takes a long time (usually more than 3 h), catheterization should be performed. To adapt to the postoperative changes, adaptive exercise should be done preoperatively, in particular to practice defecating and urinating in bed. Beddings with urinal, WC pan, and urine pad, etc., should be prepared.
7.2.3 Hot-Spot Issues in the Consensus
For patients on oral antiplatelet agents or anticoagulants, timely communication must be made to inform them about the risk of thrombosis associated with stopping antiplatelet or anticoagulant treatments and the risk of hemorrhage associated with the continuation of the treatment. Individual specificities should be considered before discontinuing antiplatelet and anticoagulant therapy. The risk after discontinuation of the treatment and the possible risks of hemorrhage from continued oral antiplatelet medication must be assessed.
For low-risk super minimally invasive endoscopic procedures, it is recommended to continue to use P2Y12 receptor antagonists as single or dual antiplatelet therapies (DAPT), and to continue anticoagulant therapies such as warfarin. It should be ensured that the International standardized ratio (INR) does not exceed the treatment range during the week before surgery, meanwhile it is recommended to omit the morning dose of direct oral anticoagulants (DOACs) on the day of surgery. For high-risk super minimally invasive endoscopic surgery in patients at low risk of thrombosis, it is recommended to discontinue P2Y-12 receptor antagonists seven days before surgery. For people with DAPT, it is recommended to continue taking aspirin; meanwhile it is recommended to stop warfarin five days before surgery, and to recheck the INR before surgery to ensure it is lower than 1.5. And, on the contrary, in the case of high-risk ultra-minimally invasive endoscopic surgery in patients at high risk of thrombosis, it is recommended to continue taking aspirin while consulting the cardiology department to discuss the risks/benefits of discontinuing the use of P2Y-12 receptor antagonists. In patients using anticoagulants, warfarin should be temporarily discontinued and replaced with low-molecular-weight heparin (LMWH); for patients with oral anticoagulants undergoing high-risk super minimally invasive surgery, the last dose of anticoagulants is recommended three days before surgery. For patients who take dabigatran orally and have an estimated glomerular filtration rate of 30–50 ml/min, the last dose is recommended five days before surgery.
For all patients who discontinue anticoagulant and antiplatelet drugs before surgery, it is recommended to resume treatment two to three days after surgery, and to fully inform patients of the increased risk of postoperative hemorrhage, and pay close attention to the results of postoperative routine stool analysis. The coagulation dysfunction must be corrected according to the severity of postoperative hemorrhage and to the patient’s risk of thrombosis. For hemodynamically unstable patients taking vitamin K antagonists, intravenous vitamin K and factor IV prothrombin complex concentrate (PCC) are recommended. Generally, when the hemoglobin level drops by more than 20 g/l, so that a blood transfusion or unplanned hospitalization is necessary, the hemorrhage has a clinical significance.
7.3 Wound Pre-Treatment and Infection Prevention in Super Minimally Invasive Surgery
The digestive lumen is not a sterile environment, and the normal digestive mucosa is an important barrier against infection. Super minimally invasive surgery destroys the normal gastrointestinal mucous membrane, and endoscopic treatment involves all layers of the digestive tract or even the extraluminal, which lead to postoperative infection and complications.
Common bacteria in the esophagus are Firmicutes (70%), Bacteroidetes (20%), Actinobacteria (4%) and Proteobacteria (2%), and the main bacteria genera are Staphylococcus, Prevotella and Veyronella; due to the effect of factors like gastric fluids, the number of bacteria in the stomach is small. Common bacteria include: Actinobacteria, Bacteroidetes, Firmicutes and Proteobacteria (including Helicobacter pylori); the main group of bacteria is Streptococcus; for common colorectal bacteria, Enterococci are more common at the proximal end, but at the distal end, Pseudomonas aeruginosa and Escherichia coli/Shigella are more abundant; others are Proteus, Albus or Staphylococcus aureus; the most common bacteria involved in infections of the biliary tract are mainly: Escherichia coli, Klebsiella, Pseudomonas aeruginosa and Enterococcus, while Staphylococcus epidermidis, Streptococcus and Enterococcus faecium are uncommon; the most common pancreatic infections are caused by genera like Enterobacteriaceae (E. coli is the most common), Staphylococcus, Enterococcus, Streptococcus, anaerobic bacteria, Pseudomonas and Acinetobacter. In infectious pancreatic necrosis, Gram-negative bacteria are more common in biliary pancreatitis, Gram-positive bacteria are more common in alcoholic pancreatitis, and multimicrobial infection is more likely to occur in pancreatic abscesses. Firmicutes and Proteobacteria are the most abundant in the small intestine, while Bacteroidetes are the least abundant. And Firmicutes are mainly represented by lactic acid bacteria, including Streptococcaceae, Lactobacillaceae and Sarcobacillaceae. Proteobacteria mainly include Neisseraceae, Pasteurelliaceae and Enterobacteriaceae.
The risks of exposure to infection are multiple. During endoscopy, secretions are easy to regurgitate into the endoscopic cavity, causing opportunistic bacteria to shuttle back and forth between the patient’s digestive tract and the endoscopic cavity, increasing the risk of infection. Other causes may be the retention of food debris and fluids in the digestive tract, as well as causes related to the surgical procedure, like intraoperative infection, postoperative massive hemorrhage and perforation, hydrothorax and ascites, infection, etc.
7.3.1 Wound Pre-Treatment Plan in Super Minimally Invasive Surgery
1. SMIS through a natural cavity channel presents risks of infection, and preoperative treatment of the wound must be conducted
(1) The preoperative treatment of the upper digestive tract (including esophagus, stomach, duodenum) is as follows:
The patient must fast for 12 h and not drink for 2 h before the operation. Sterile water should be used to wash the esophagus and the gastric cavity under the preoperative gastroscope to ensure the upper digestive tract is clean so as to reduce the number of bacteria.
It is recommended that patients take an oral mucus remover (such as Streptomyces protease, etc.) 30 min before surgery to improve the visibility in the surgical field, shorten the operation time, and reduce the risk of complications.
Before anesthesia begins, the patient must rinse his mouth repeatedly with sterile water or a normal saline solution.
Isolation measures must be taken, gastroscopes should be strictly disinfected, and disposable sterile instruments should be used during the operation. In addition, the preoperative pre-treatment for the surgery of the natural cavity of the biliary tract and of the pancreas can be applied to that of the upper digestive tract.
(3) The preoperative treatment of the lower digestive tract (including rectum, colon) is as follows:
A low-slag diet must be given after admission and before treatment, with routine preoperative fasting for 12 h.
Bowel preparation should be performed preoperatively to fully empty and clean the bowel tract, the procedures for bowel preparation include oral laxatives or warm saline enemas, oral defoaming agents (such as siloxane), etc..
Isolation measures must be taken, gastroscopes should be strictly disinfected, and disposable sterile instruments should be used during the operation.
During the operation, the accumulation of bowel juice should be fully cleaned. In addition, the preoperative pre-treatment for the surgery of the natural cavity of the appendix can be applied to that of the lower digestive tract.
The situation for the surgery of the natural cavity of the middle digestive tract is a special case, and it is necessary to choose and refer to the procedure for the lower digestive tract depending on the site of the lesion, and to refer also simultaneously to the procedure for the upper and lower digestive tracts.
2. SMIS through a tunnel channel presents risks of infection, and preoperative treatment of the wound should be conducted
In general, the preoperative pretreatment of wounds caused by endoscopic surgery through a tunnel channel is the same as for those caused by endoscopic surgery through a natural cavity channel. However, peroral endoscopic myotomy (POEM) is used for the retention of the digestive tract caused by dynamic diseases such as achalasia and gastroparesis, and the preoperative fasting time needs to be extended to no less than 48 h.
3. SMIS through a digestive tract puncture channel presents risks of infection, and preoperative treatment of the wound should be conducted
Currently, the technique is mainly performed by puncturing the stomach and the duodenum, establishing channels through the gastro-pancreas, the gastro-gallbladder, the gastro-jejunum, the duodeno-bile duct, etc. to complete surgery. From a technical point of view, it is divided into endoscopic ultrasound-guided fine needle aspiration/biopsy (EUS–FNA/B) and lumen-apposing metal stent (LAMS). In general, the preoperative pretreatment of the wounds caused by endoscopic surgery through a digestive tract puncture channel is the same as for those caused by endoscopic surgery through a natural cavity channel.
4. SMIS through an extracorporeal skin puncture channel presents risks of infection, and preoperative treatment of the wound should be conducted
Currently, the technique is mainly performed by the puncture of extracorporeal skin, establishing channels like in vitro – bile duct to complete the operation. Clinically, percutaneous transhepatic cholestroscopy is more mature, and its complications are mainly infection, bleeding, biliary fistula, etc. Therefore, it is recommended to strictly disinfect the external skin puncture area before the operation according to the surgical disinfection criteria. Patients must fast for 6–8 h before the operation and not drink for 2 h beforehand.
5. SMIS through a multiple-cavity channel presents risks of infection, and preoperative treatment of the wound should be conducted
Currently, SMIS through a multiple cavity channel mainly uses gastrointestinal endoscopy combined with thoracoscopy or laparoscopy, which are combined to dissect the giant submucosal tumors and giant polyps of the digestive tract, etc. Therefore, in addition to following the routine skin disinfection procedure for thoracoscopy or laparoscopy, multiple-cavity channel surgery should also follow the methods of pretreatment of wounds used in endoscopic surgery.
Thoracoscopic or laparoscopic skin disinfection should be performed after surgical anesthesia. The traditional way of disinfecting the skin consists of rubbing the surgical area with 2.5%–3% iodine tincture, leaving it to dry and rubbing twice with 70% alcohol to remove the tincture. Disinfection specifications mainly as follows:
The disinfectant, should be applied from the center of the clean operating area to the periphery;
The area of skin disinfection must be generally 15 cm in diameter around the surgical incision. If the incision needs to be extended, the skin disinfection area should be enlarged.
7.3.2 Antibiotic Application in Super Minimally Invasive Surgery
(I) SMIS per natural cavity channel
1. Per-natural-cavity surgery of the upper digestive tract. In SMIS of the upper digestive tract through a natural cavity, antibiotics should be used appropriately in low-risk patients with non-full-thickness defects of the upper digestive tract; it is recommended to use antibiotics for high-risk patients with non-full-thickness defects of the upper digestive tract; it is recommended to use antibiotics in case of full-thickness defects of the upper digestive tract.
Routine prophylactic use of antibiotics is not recommended in case of surgery of the upper digestive tract through a natural cavity. The incidence of bacteremia after local resection/dissection of esophageal lesions is low (1%), and fever is not associated with bacteremia, so routine prophylactic antibiotics are not required; The risk of bacteremia after local resection/dissection of gastric lesions is low and transient, and routine prophylactic use of antibiotics is not recommended. Therefore, routine prophylactic use of antibiotics may not be necessary. However, the use of antibiotics should be considered when the following conditions exist: long operation time, large area of lesion resection, complicated gastrointestinal perforation, massive gastrointestinal bleeding, diabetes, poor immune function (especially in organ transplant recipients) and malnutrition, etc.
2. Biliary and pancreatic surgery through a natural cavity. For surgeries entering the biliary and pancreatic natural cavity, the preoperative use of antibiotics is not recommended if there is no specific infection; antibiotics are recommended if infection has existed. Antibiotics are recommended both before and after surgery for procedures where drainage is expected to be difficult and the risk of infection is high.
Patients treated with endoscopic retrograde cholangio-pancreatography (ERCP) do not need to use antibiotics before surgery. However, when any of the following are present, the prophylactic use of broad-spectrum antimicrobials should be considered:
Sepsis with biliary tract infection has developed;
There is bile duct stenosis in the hepatic hilar;
In case of endoscopic interventional therapy of pancreatic pseudocysts;
In patients who have received an organ transplant or who are immunosuppressed;
Primary sclerosing cholangitis;
On patients with moderate to high risk of heart disease, such as heart valve disease; the antimicrobial profile of the drug should cover Gram-negative bacteria, Enterococcus and anaerobic bacteria.
3. Operation of the lower digestive tract through a natural cavity channel. Antibiotics are not recommended for colorectal SMIS with non-full-thickness defect and wounds <2 cm;
Antibiotics are recommended for colorectal SMIS with non-full-thickness defect and wounds less than to 2 cm wide, and for all colorectal SMIS defects.
After local resection/dissection of colorectal lesions, the risk of bacteremia is relatively low if the wound is small, and antimicrobial agents are usually not recommended; if the wound is large, it will become a window for bacterial invasion, which leads to infection, and the full-thickness defect leads to secondary peritonitis, which makes that the use of antibiotics can be considered. Antibiotics may be quinolones, second- or third-generation cephalosporins, and nitroimidazole, as appropriate. The total duration of postoperative medication should generally not exceed 72 h, but can be extended appropriately.
4. Appendix natural cavity surgery. Routinely use antibiotics after ERAT surgery.
After endoscopic retrograde appendicitis therapy (ERAT) has fully cleared the appendiceal cavity and established a smooth drainage system, third-generation cephalosporins combined with nitroimidazoles are usually recommended. Piperacillin-tazobactam should be used in severe complications, and carbapenems can be used if the patient is allergic to these drugs. The specific use time of antibiotics should be adapted to the patient’s clinical symptoms, body temperature and blood infection indicators.
(II) SMIS through a tunnel channel
1. Tunnel surgery of the upper digestive tract. It is recommended to use antibiotics for SMIS of the upper digestive tract through a tunnel channel. It is recommended by the Gastrointestinal Endoscopic Tunnel Technique Consensus that antibiotics be used empirically 30 min before surgery to prevent infection, and be maintained for no more than 48 h after surgery. The choice of antibiotics can be betalactam against tunnel-technique-related infecting organisms (including Pseudomonas aeruginosa and Acinetobacter), such as second- and third-generation cephalosporin (ceftriaxone, cefuroxime, cefazolin) and amoxicillin-clavulanate, etc. If the patient is allergic to penicillins, aztreonam combined with clindamycin and third-generation quinolones can be used.
2. Tunnel surgery of the lower digestive tract. SMIS of colorectal lesions through a tunnel channel should routinely use antibiotics. Infection around the incision after colorectal surgery is mainly caused by misplacement of the intestinal flora. It is recommended that antibiotics covering Gram-negative bacteria be applied intravenously from 30 min before surgery to 24–48 h after it. Prophylactic use of antibiotics should not be too long, since prolonged use may lead to the production of drug-resistant strains, and even to pseudomembranous enteritis.
(III) SMIS per puncture channel
(1) EUS–FNA/Type B SMIS surgery through a puncture channel has a low probability of infection in case of gastric puncture, and antibiotics should be used appropriately; antibiotics are recommended for per-duodenal puncture. However, in the presence of therapeutic procedures such as ablation, antibiotics are recommended.
Studies have shown that the incidence of infection during per stomach endoscopic ultrasound-guided (EUS) puncture is very low, with the incidence of solid lesions ranging from 0.4% to 1%; the incidence of cystic infections is only 0.44%, so routine prophylactic use of antibiotics is not recommended. The incidence of cholangitis with EUS-guided transcholedochoduodenostomy is 1.5%, so postoperative antibiotics are recommended to minimize the incidence of infection. For the above conditions, prophylactic antibiotic use is recommended in the presence of ablation and for other therapeutic procedures. Third-generation cephalosporins and nitroimidazole are recommended for antibiotics.
(2) In the case of LAMS stent placement by SMIS surgery through a puncture channel, strict intravenous antibiotics are required.
The common complications after LAMS stent implantation include secondary infection. With the use of antibiotics, the incidence of secondary infections has decreased. For pancreatic pseudocysts or encapsulated necrosis, broad-spectrum antibiotics, such as second- and third-generation cephalosporins or quinolones, should be used according to experience before LAMS stent implantation. Then the selection of the antibiotic for treatment must be made depending on the results of specimen culture and drug sensitivity. It is necessary to consider the tissue permeability and side effects of antibiotics at the same time, and the application time extends until five to seven days after surgery. Studies have shown that the most common cultures of infective peripancreatic effusions were Gram-positive Enterococcus (45%) and Gram-negative Enterobacteriaceae (42%).
(3) Antibiotics are recommended for percutaneous SMIS.
Percutaneous transhepatic cholangiography can be used to evaluate the nature of biliary stricture, biliary biopsy, lithotripsy and lithotomy, etc. There are risks of cholangitis and sepsis after surgery. For malignant biliary obstruction without obvious signs of infection, the prophylactic use of antibiotics covered with Gram-negative bacteria and metabolized by bile is recommended prior to interventional therapy; benign biliary obstruction, such as biliary calculus, is often combined with infection, and if antibiotics were administered before the operation, no additional ones are required. Escherichia coli is the main Gram-negative bacteria in infections of the biliary tract, and Enterococcus is the main Gram-positive bacteria, so third-generation cephalosporins, carbapenems and vancomycin, etc., can usually be selected for the treatment.
(IV) SMIS through a multiple cavity channel
(1) Gastroscopy combined with thoracoscopic surgery requires the strict use of intravenous antibiotics.
Pneumonia, pleural cavity infection and surgical site infection may occur after thoracoscopic surgery, so empirical antibiotic therapy is very important. As the anoxic nature of the pleural space is conducive to the growth of anaerobic bacteria, so the routine intravenous antibiotics should be used within 60 min before the skin incision. Infections caused by a variety of microorganisms commonly found in the skin and respiratory flora (such as Staphylococcus aureus, coagulase negative Staphylococcus, Streptococcus pneumoniae, and Gram-negative bacilli) can be adequately prevented by cephalosporin antibiotics, and vancomycin or teicoplanin should be used in patients allergic to penicillin or to cephalosporin antibiotics. The specific choice of antibiotics should also be based on local epidemiology and the antibiotic sensitivity pattern of the microorganisms causing hospital surgical infections.
(2) Gastroscopy or endoscopy combined with laparoscopic surgery requires the strict use of intravenous antibiotics.
Pulmonary infection and intraperitoneal infection are common postoperative infections after laparoscopy. Studies have shown that pulmonary infection cases after laparoscopic surgery amount to 9.67%, and corresponding preventive measures must be taken to reduce the incidence of infection. Meanwhile, the invasive procedure of abdominal surgery increases the risk of bacterial contamination of the peritoneal cavity, so strict intravenous antibiotics are required. Endoscopic surgery combined with laparoscopic surgery includes traditional exposed resection and newly emerged non-exposed resection, among which non-exposed resection includes non-exposed endoscopic gastric wall inversion, the clean non-exposed technique, and the non-exposed full-thickness resection technique. The degree of abdominal infection caused by exposed and non-exposed resection is different, and antibiotics should be selected according to the specific severity of abdominal infections.
7.3.3 Hot-Spot Issues in the Consensus
In endoscopic surgery, due to the communication between various natural channels, some external bacteria and bacteria with a fixed value in different channels are dispersed towards the operation sites, damaging the operation sites or the mucosa, with the risk of bringing a local bacterial infection. Gastrointestinal endoscopy-related bacteremia is also associated with the risk of localized infection of distant tissues; in such kind of infection, drug-resistant flora is also not rare.
Bacteremia is comparatively more dangerous in cases of SMIS-induced infections. But comparatively, the risk of bacteremia caused by SMIS interventions does not significantly increase; and routine interventions through a natural cavity do not really increase the risks. Prospective studies in patients with solid or cystic tumors with EUS–FNA have shown that the incidence of surgery-related infection complications such as bacteremia is relatively low, and routine use of antibiotics is not recommended. In contrast, patients who received antibiotics three to five days before surgery had a higher incidence of postoperative infection combined with antibiotic adverse reactions. Patients with severe neutropenia and advanced hematological malignancies are at increased risk of bacteremia and sepsis after gastroenteroscopy, and appropriate preoperative antibiotics can be selected for prevention depending on the conditions.
Patients must fast overnight prior to the PEG procedure (6 h for solid liquids, 2 h for clear liquids, longer if the gastric peristaltic function is impaired such as gastroparesis). ESGE recommends a prophylactic single dose of an intravenous lactam antibiotic (or an appropriate alternative antibiotic) when performing an endoscopic procedure to reduce the risk of postoperative wound infection. A number of RCT studies have shown that prophylactic single administration of lactam antibiotics during percutaneous catheterization (PEG-J/DPE-J) is important for the prevention of postoperative infection in pericutaneous or internal stomy wounds. A meta-analysis that included 10 randomized controlled trials (1059 patients) showed that prophylactic penicillin or cephalosporin treatment reduced the risk of postoperative wound infection. Risk reduction by cephalosporin is relatively less than with penicillin. There is still a lack of large-scale clinical trials to verify the timing of the preoperative antibiotic treatment for PEG, but it is generally believed that 30 min before PEG operation is reasonable. For patients allergic to penicillins, the anti-infective treatment may be given orally with cotrimoxazole in PEG procedures.
For ERCP procedures with a high risk of both in situ infection and secondary blood-borne infection, the current guidelines suggest that preoperative use of antibiotics is not necessary for all patients who are to undergo ERCP. Some factors that easily cause PEC or may increase its severity are indications of antibiotic prophylaxis, such as primary sclerosing cholangitis, hilar obstruction, and peroral cholangiography. In addition to that, complete biliary drainage is difficult to achieve for some patients with cholangiocarcinoma or others who are expected to undergo ERCP surgery. Or, for patients with severe autoimmune function impairment, quinolone or cephalosporin antibiotics taken one day before surgery are relatively safe and effective. The antibiotics selected should be considered based on local epidemiology. Some researchers have analyzed the results of adding antibiotics (gentamicin, etc.) to ERCP contrast media; the results are not satisfactory. In addition to the antibiotics commonly used after ERCP surgery, the generation of some drug-resistant bacteria should also bring concern. In Chinese studies, most of the bacteria isolated in the blood after an ERCP were resistant to ciprofloxacin and ceftriaxone after the bacterial drug sensitivity test in the blood of patients having undergone ERCP surgery. Antibiotic prophylaxis of ERCP may increase the proportion of antibiotics-resistant bacteria isolated from the bile. For MDROS infection caused by ERCP interventional trauma operation, it is generally believed that preoperative application of antibiotics cannot be avoided, and that MDROS infection is mostly caused by incomplete cleaning of the ERCP operating system, and multiple drug-resistant bacteria infection may lead to adverse clinical results. Carbapenem-resistant enterobacteriaceae are carbapenem-producing MDROs, i.e. enzymes that hydrolyze lactam rings and inactivate lactam antibiotics. Patients with low immunity, especially those with bile duct cancer and biliary stents, are at increased risk of infection. The gastric mucosa is relatively protective, so some surgical techniques for gastric mucosa, such as gastric ESD/EMR surgery, present a relatively low risk of infection, and the use of routine preoperative antibiotics is not recommended. However, for patients with high risk factors (old age, large operating area, low immunity, etc.), the first and second generation of cephalosporin antibiotics can be administered transiently before surgery, as appropriate.
Currently, the relatively controversial part of antibiotic prophylaxis before endoscopic surgery lies in the treatment of postoperative infections caused by inadequate disinfection and cleaning of digestive endoscopic instruments, and in the resolution of serious adverse reactions caused by secondary infection of some carbapenem-resistant bacteria; furthermore, for some of the characteristic cases of SMIS surgery, there is now sufficient evidence showing that the application of preoperative antibiotics can lead to the best prognosis, the timing of the application antibiotics remaining an issue to be solved. The prophylactic use of antibiotics is not only related to the chosen method of SMIS, but also to the patient’s baseline data. It is recommended to use more individualized means, to pay attention to the patient’s particular condition, and to comprehensively evaluate the use of preoperative antibiotics to obtain greater benefits.
7.4 Prevention and Treatment of Intraoperative Complications in Super Minimally Invasive Surgery
7.4.1 Complication Prevention and Treatment
(I) Intraoperative hemorrhage
Intraoperative hemorrhage is an hemorrhage that causes the patient’s hemoglobin to drop by more than 20 g/l. In order to prevent massive hemorrhage during the operation, the submucosal injection should be sufficient during the operation, which exposes more vessels and is conducive to electrocoagulation and hemostasis. Intraoperative hemorrhage can be treated with various kinds of incision knives, hemostat or metal clips, and preventive hemostasis can be performed on the exposed vessels during the dissecting process.
(II) Emphysema, pneumothorax and pneumoperitoneum after intraoperative perforation
Intraoperative subcutaneous emphysema (manifested as emphysema of the face, neck, chest wall and scrotum) and mediastinal emphysema (swelling of the epigastric area that can be evidenced by gastroscopy) usually do not require special treatment, and the emphysema usually subsides on its own. Patients with severe intraoperative pneumothorax [airway pressure over 20 mmHg (1 mmHg = 0.133 kPa), SpO
(III) Intraoperative mucosal layer injury in the tunnel technique
Mucosal injury in the tunnel technique adopts two grades of trichotomy depending on the mucosal injury: (MI): grade MI-0, no mucosal layer injury; grade MI-p (MI perforation), mucosal layer injury. Grade MI-p is divided into two subgrades: grade MI-pc (MI controlled)—the injury in the mucosal layer can be occluded with metal clamps or closed with fibrin glue; grade MIpunc (MI uncontrolled), the mucosal layer injury cannot be endoscopically closed. Intraoperative repeated submucosal injection to maintain a good liquid pad and timely hemostasis to maintain a clear endoscopic field of vision not only help to reduce the defect in the muscularis propria, but also help to reduce the risk of mucosal injury. If tunnel mucosal injury occurred during the operation, it can be occluded with metal clips or closed by spraying fibrin glue in the tunnel.
(IV) Hypoglycemia
Anesthesia hides hypoglycemic tremor, weakness, syncope, altered mental status, epileptic seizure, etc. Therefore, the diagnosis of intraoperative hypoglycemia is very difficult, as is the appropriate intraoperative reconstitution of intravenous fluids for energy.
(V) Cardiorespiratory arrest
Continuous ECG monitoring and a good intravenous channel must be established before surgery. The operation must be immediately stopped and the patient rescued if there is heart beat and respiration arrest.
(VI) Epileptic seizure
Operation must be immediately stopped if the patient has an epileptic seizure during the operation. He/she must be moved to a safe places, excessive intervention in the seizure process should be avoided, as well as any falls or injuries; he/she should be placed on flat ground, in a lateral position, and the collar or loose objects should be loosened so that he/she can breathe easily. During seizure, the time, duration, and symptoms of the seizure should be observed and recorded in order to help the specialist assess the type and severity of the seizure to determine appropriate treatment plans.
7.4.2 Hot-Spot Issues in the Consensus
(I) Intraoperative complications for patients with achalasia in super minimally invasive surgery
For patients with achalasia, general drug therapy only applies to patients who cannot tolerate surgery or have great risk of surgery, and is only effective in the short term; the treatment plan and results are not satisfactory. But super minimally invasive endoscopic treatment for achalasia is relatively mature, including endoscopic balloon dilation, which is performed at the esophageal stenosis site by extending the guide wire from the endoscopic lateral hole through direct vision under the endoscope. The most common intraoperative complication during this period is esophageal perforation, which has an incidence of 1.9%, and is related to the diameter of the balloon selected for primary dilation. Generally a conservative treatment can be selected for ordinary small perforation; patients with significant perforation, leading in particular to mediastinal infection, should be fitted with titanium clips or self-expanding metal stents as alternative means of treatment.
Intraoperative complications of POEM surgery. As the representative surgical operation in the field of super minimally invasive surgery, in POEM the near posterior wall of the esophagus as the safety site of the submucosal tunnel is mainly chosen, and different treatment schemes can be selected depending on different conditions during the operation. The common intraoperative complications include mucosal injury and perforation; the occurrence of mucosal injury is 2.8%–4.8%; the occurrence of perforation is about 0.2%–0.7%, which easily happens at sites like cardia. After completion of myotomy in the tunnel and sufficient hemostasis in the operation, the metal clips are used to close the defect wounds, and fibrin glue can be sprayed to hold the gastrointestinal decompression tube in place; delayed perforation after surgery can lead to mediastinal and abdominal infection. Gas-related complications include subcutaneous emphysema, pneumomediastinum, pneumothorax and pneumoperitoneum, etc. The intraoperative use of carbon dioxide will reduce the occurrence of related complications. Closed thoracic drainage must be performed timely for patients with massive pneumothorax, mediastinal emphysema and oxygen saturation below 90% to avoid the onset of critical diseases like respiratory distress. Delayed hemorrhage: for patients with severe hemorrhage due to POEM tunnel surgery, timely stop the endoscopic treatment and pull out the metal clips at the tunnel entrance. The endoscope is reintroduced into the tunnel and intra-tunnel irrigation is performed, as well as electrocoagulation of the bleeding spots with a hemostat. The hemostatic clamp is close again after successful completion.
(II) ERCP intraoperative complications
The most common one is intraoperative hemorrhage. ERCP intraoperative hemorrhage is slightly different from hemorrhages occurring in other endoscopic operations, which are hematemesis and/or black stool or Hb reduction to more than 20 g/l. Patients are considered at increased risk of intraoperative and postoperative hemorrhage after ERCP sphincterotomy if at least one of the following factors is present: preoperative application of antiplatelet drugs, platelet count < 50 × 10in vitro anatomical study concluded that the nipple should be cut in the 10–11 o’clock area, and that this incision only covers 10% of all papillary arteries. In addition to that, compared to pure cutting current, using mixed current reduces the occurrence of hemorrhage, whereas not increasing the risk of PEP; The ESGE also recommends treating persistent or delayed sphincterotomy hemorrhage with a local injection of epinephrine (1:10 000). When epinephrine injection alone fails, the hemostatic clamp can be added and delivered via a capped gastrointestinal endoscopy or duodenoscope, which is relatively difficult to do when the papillary muscle is bleeding. The ESGE also recommends temporary placement of the self-dilating metal stent with complete biliary duct coverage for sphincterotomy hemorrhage treated with standard hemostasis. Given the high incidence of bleeding from papillary sphincter during ERCP surgery, some experts have proposed to insert a nasobiliary drainage tube after PSB hemostasis to prevent clots in the biliary tract from forming bile duct obstruction.
7.5 Prevention and Treatment of Postoperative Complications in Super Minimally Invasive Surgery
7.5.1 Complication Prevention and Treatment
(I) Postoperative hemorrhage
Postoperative hemorrhage is shown as hematemesis, black stool or hematochezia, etc. Severe cases can lead to hemorrhagic shock, which generally occurs within one week after surgery, but it can also appear in two to four weeks. If symptoms like hematemesis, much black stool, with bright color, and obvious hemoglobin reduction occur after surgery, endoscopy should be timely performed and the wounds must be carefully observed. In the event of active bleeding, hemostasis must be carried out using a thermoelectric clamp or metal clamp occlusion. Postoperative hemorrhage is often related to factors like poor blood pressure control and corrosion of residual blood vessels by gastric acid. In addition, postoperative hemorrhage is also related to the site of the lesion to some extent, mainly in the gastric antrum and the lower rectum.
(II) Postoperative hemorrhage in the tunnel
If the delayed hemorrhage in the tunnel cavity is serious, endoscopic hemostasis is required, involving the removal of the metal clips at the tunnel’s entrance and the insertion of the endoscope into the tunnel to rinse and clean the cavity, while electrocoagulating the bleeding spots with a hemostat and occluding the tunnel entrance again after hemostasis has been achieved.
(III) Mediastinal emphysema, pneumothorax, and pneumoperitoneum after postoperative perforation
Delayed perforation usually manifests itself as abdominal distension, increased abdominal pain, signs of peritonitis, fever, pneumatosis or an increase in pneumatosis compared to the initial situation, as evidenced by imaging. Delayed postoperative perforation is mostly related to factors of poor wound suture, excessive electrocoagulation, moving out of bed too early, eating too early, poor blood sugar control, and gastric acid corrosion of wounds. In order to reduce the occurrence of postoperative delayed perforation, if there is large, deep wounds, or wounds with fissures, the period of bed rest and fasting will be appropriately extended after surgery, and also gastrointestinal decompression (placement of anal canal drainage for patients after lower gastrointestinal surgery); blood glucose levels will be strictly controlled for patients with diabetes; for patients with smaller perforation and mild pleuroperitoneal cavity infection, treatments like fasting, anti-infection cares and acid suppression, etc. will be given. Closed thoracic drainage may be performed in patients with effusion, and smooth drainage maintained for peritoneal puncture catheterization; if the infection cannot be limited through a conservative treatment or if it is associated with severe chest and abdominal infection, laparoscopic surgical exploration should be performed as early as possible for perforation repair and abdominal drainage.
(IV) Postoperative effusion in the chest, abdomen and pelvis or abscess
Mediastinal abscess is mostly due to postoperative eating too early; at this time the tunnel entrance is not healed, and chyme enters the tunnel through the muscle incision in the mediastinum and causes an infection. The esophageal tunnel and mediastinal dirt must be cleaned endoscopically by inserting a drainage tube in the mediastinum and placing it outside the body through the nose, without closing the tunnel entrance. The fasting food and non-drinking period should be extended, in combination with the administration of intravenous antibiotics, while performing enteral nutrition therapy through the lower jejunal nutrition tube. The drainage tube will be removed after the mediastinal abscess has been completely drained.
(V) Postoperative fistula of the digestive tract
A digestive tract fistula is a passage for digestive fluid into the chest or the abdominal cavity through a leak created by endoscopic surgery. Esophagomediastinal and esophagothoracic fistulas are common. Once the fistula has appeared, close thoracic drainage should be performed to maintain smooth drainage, and adequate nutritional support must be given. Metal clips and various closing instruments can be used if necessary, as well as a recoverable stent and other ways to plug the fistula. In severe cases, a surgical intervention is required.
(VI) Serious postoperative infection
The patient must fast for food and not drink for 72 h after the operation. Treatments like intravenous acid suppression and nutrition support, etc., must be administered regularly, and broad-spectrum antibiotics must continue to be administered by intravenous drip for two days. If the patient’s temperature rises after surgery, as well as infection indicators like white blood cells, neutrophils, IL-6, etc., it must be considered that the patient suffers from a postoperative infection, and empiric anti-infective therapy must be administered, while blood cultures will be performed. The administration of antibiotics will be adjusted after the results are available.
(VII) Postoperative electrocoagulation syndrome
Patients with early colorectal disease have a higher risk of electrocoagulation syndrome after ESD. Age > 60, lesion diameter ≥ 5 cm, colonic disease, malignant tumor, broad-based polyp, and mass resection are all high-risk factors for electrocoagulation syndrome after ESD in patients with early colorectal disease. Therefore, clinical attention should be paid to the aforementioned high-risk factors, and active targeted prevention and control measures (such as the application of postoperative antibiotics) should be adopted to reduce the occurrence of electrocoagulation syndrome and improve the prognosis of patients.
(VIII) Postoperative esophageal stent displacement or detachment
Stent placement has been widely used in the treatment of benign and malignant esophageal diseases, with more and more indications, such as esophageal stenosis and esophageal fistula. It quickly relieves the symptoms of patients and improves their quality of life. However, various complications may occur after stent implantation, including chest pain, foreign body sensation, stent obstruction, reflux esophagitis, hemorrhage, displacement, etc., among which the most common is stent displacement with its incidence of 20%–60%. Endoscopic suture fixation can be a reasonable method to solve the problem, but its safety remains the subject of discussions. Titanium clamp fixation is another effective method, but the titanium clamp fixed by endoscopy is too small and easy to separate by itself, and its safety needs to be further verified; in addition, it can also be fixed by a snare, by placing the snare for polyp dissection at the proximal end of the stent, which is lasso-linked and adducted at the other end, making finally the stent relatively well fixed.
(IX) Postoperative esophageal stenosis
As the esophageal lumen is relatively narrow, the incidence of stenosis after endoscopic resection of small lesions is low. But for the endoscopic treatment of large lesions, postoperative stenosis is the common complication. With the continuous proposal and application of multiple preventive methods, including oral or local injection of hormones, esophageal metal stents, prophylactic endoscopic balloon dilation (EBD) and poly glycolic acid (PGA), the incidence of postoperative stenosis also gradually decreases. Previous studies have confirmed that mucosal injuries covering ≥3/4 of the circumference of the esophagus is an independent risk factor for the formation of postoperative stenosis, and that the incidence of postoperative esophageal stenosis increases to 66%–100%. In addition, the length (if ≥3 cm) and the depth of postoperative histological immersion (when infiltration depth > lamina propria (M2)) are also independent risk factors for stenosis. Intraoperative injury of the muscularis propria of the esophageal mucosa could increase the risk of incidence of stenosis, but there is no relevant data to support this. The incidence of stenosis after endoscopic resection of non-circumferential lesions was 56%–76%, while the incidence of stenosis after endoscopic resection of circumferential lesions was as high as 100%. In the process of endoscopic resection of esophageal mucosal diseases, circumferential resection should be avoided and the normal mucosa should be preserved as much as possible. There is great difference in the incidence of endoscopic stenosis between non-circumferential and circumferential lesions, mainly because of their different healing processes. The peripheral esophageal mucosal epithelium can only crawl from the oral and anal sides of the wound, whereas the non-peripheral mucosa with lesions can crawl and grow from the oral, anal, and lateral sides simultaneously. Therefore, in the process of ESD surgery for lesions of the esophageal mucosal, the area of the lesions must be accurately evaluated and marked before surgery, and circumferential resection should be avoided as much as possible during surgery, which can significantly reduce the incidence of postoperative stenosis. After surgery, ingestion of hormones is currently the most important method to prevent stenosis. But metal stents are not recommended for patients after endoscopic resection of large areas of non-circumferential esophageal lesions. In patients having undergone total resection of the esophageal mucosa, hormones are insufficient to prevent stenosis, and autologous skin transplantation must be considered. For patients with mucosal injury length ≤ 10 cm after endoscopic resection of large esophageal lesions, self-dilating balloons can be used in vitro to prevent stenosis. Endoscopic dilation therapy is currently the first-line treatment for benign esophageal stenosis. The possibility of esophageal perforation should be highly considered in patients with clinical manifestations of fever, persistent chest pain, shortness of breath, and tachycardia after esophageal dilation. If perforation occurs, immediate placement of a covered metal stent is recommended. Esophageal stent placement is not recommended as the first-line treatment for benign esophageal stenosis. Endoscopic dilation or incision combined with local hormone injection can be used to treat refractory esophageal stenosis.
(X) Postoperative pancreatitis
NSAID is a classic preoperative preventive medicine. The latest guidelines of the American Society for Gastrointestinal Endoscopy (ASGE) also clearly indicate that in case there is no contraindication, performing NSAID prior to ERCP significantly reduces the incidence of PEP (OR = 0.49). Rectal administration significantly reduces the risk of PEP compared to oral administration. Rectal administration has the advantages of taking effect quickly and of good bioavailability, bypassing most of the first pass elimination, shortening the peak and durations, and reducing gastrointestinal adverse reactions. Rectal administration of 100 mg of indomethacin or diclofenac prior to ERCP reduces the risk of PEP.
Venous hydration should be considered. The ASGE guideline points out that intravenous hydration with lactated Ringer’s solution prevents PEP (OR = 0.60). Studies have shown that a single injection or a long-term (>12 h) infusion of somatostatin has a better preventive effect on PEP, but that a short-term infusion has no significant benefit. Nitrates help the drainage of the pancreatic duct after ERCP and relieve the contraction of the sphincter of Oddi.
The insertion of a stent in the pancreatic duct should also be considered for the prevention of PEP in the case of endoscopic surgery. For high-risk patients, such as those for whom intubation is difficult, accidental insertion of the pancreatic duct, injection of a contrast agent into the pancreatic duct, or pancreatic sphincterotomy, prophylactic placement of a pancreatic duct stent reduces the incidence of PEP because the stent reduces the internal pressure in the pancreatic duct. But pancreatic duct stents may also lead to complications, such as stent occlusion, pancreatitis, stent displacement, pancreatic duct perforation, invasion of the duodenum, stent-related pancreatic duct structural changes, infection, etc.
The ASGE guideline suggests using guide wire-guided intubation to reduce PEP incidence. In the event of anatomical changes, malignant lesions or failing of guided intubation after more than two or three attempts, other methods may be considered, such as the double wire technique, presphincterotomy, wire-guided intubation along the stent, or per-pancreatic papillary sphincterotomy. But it is worth noting is that these methods may also increase the risk of PEP.
(XI) Postoperative stent obstruction or displacement of bile duct or pancreatic duct
The average patency period of general plastic biliary stents is from three to six months. The average interval between replacements of a pancreatic duct stent is from eight to twelve months. Once the plastic stent is obstructed or displaced, quick replacement should be considered; nevertheless, the stent can also be replaced every three to six months if necessary.
7.5.2 Hot-Spot Issues in the Consensus
(I) POEM postoperative complication
POEM postoperative infection mainly manifests itself as an in-tunnel or a mediastinal infection. In the event of an abscess, it should be drained promptly; among these, mediastinal abscesses are the most serious. If the patient starts eating too early after surgery (within three to four days), wound closure is incomplete, chyme could enter the tunnel and the mediastinum through the incision in the muscle, and ultimately cause infection. In this case, it is necessary to endoscopically enter the tunnel again in good time for irrigation and for retaining mediastinal diversion, the tunnel entrance being kept open, and to rinse repetitively. The incidence of postoperative digestive tract fistula is low for POEM. Maintaining the integrity of the esophageal mucosa of the tunnel during operation is key to prevent fistulas of the digestive tract. Once esophageal fistula occurs, an esophageal metal support can be used to close the fistula, whereas thoracic closed drainage can be performed and the antibiotic treatment upgraded.
Among the noteworthy complications after peroral endoscopic myotomy (POEM), gastroesophageal reflux disease (GERD) has a relatively high incidence rate (8.5%–19%). The treatment strategy includes a tiered approach: oral proton pump inhibitors combined with gastrointestinal motility promoting drugs are preferred. If drug control is poor, interventional treatments such as endoscopic cardiac constriction surgery can be performed.
(II) Complications of Ulcerative Colitis Treatment
In the treatment of ulcerative colitis, postoperative hemorrhage is the common complication. Among the treatments available for ulcerative colitis, capsule endoscopy can be chosen as a relevant means of treatment. When performing capsule endoscopic therapy, the common complications is capsule retention, when the capsules have not been eliminated from the body for two weeks or more. For patients at risk of intestinal obstruction, it is recommended to perform firstly an imaging examination for evaluation. For patients with capsule retention, observation and follow-up can be performed if they do not present any symptoms; in case of acute obstruction of the small intestine or intestinal perforation, emergency postoperative treatments can be administered.
(III) ERCP postoperative complication
ERCP occupies a relatively important position in super minimally invasive endoscopic surgery. Comparatively speaking, ERCP is an invasive interventional operation, and postoperative complications are relatively more and more complicated. The most serious and common postoperative complication is post-ERCP pancreatitis (PEP). In about 90% of the cases, it is mild or moderate. The ESGE defines it as pancreatitis with more than three times the normal amount of amylase or lipase that occurs more than 24 h after ERCP with abdominal pain of recent onset or significantly more advanced, worsening. More exactly, the diagnosis of PEP requires two of the following three criteria:
Abdominal pain consistent with acute pancreatitis (persistent, severe, often radiating to the back);
Serum lipase or amylase activity is at least three times the normal value;
The characteristic findings of acute pancreatitis are present on enhanced CT and MRI or transabdominal ultrasound.
The risk factors for PEP include dysfunction of the sphincter of Oddi, female patient with past PEP history, difficult intubation, pancreatic injection, biliary tract and endoscopic pancreatic myotomy, etc. As current research focuses on various fields of the prevention of post-ERCP pancreatitis, the ESGE consensus recommendations are as follows:
For patients with non-steroidal anti-inflammatory drug contraindications, conventional rectal treatment with 100 mg diclofenac before ERCP can effectively reduce the probability of PEP; and prophylactic stenting has also been confirmed to have a corresponding role in terms of preventing PEP.
For some patients at high risk of PEP, the ESGE recommends that short 5-Fr pancreatic stents be used. In case of pigtail tube on the duodenal side, stent patency will be evaluated again five to ten days later. The efficiency of stent replacement after salvage ERCP has not been confirmed by large-scale RCT studies. Currently there are still no large-scale observational studies evaluating the risk factors for hemorrhage after endoscopic sphincterotomy showing that proton pump inhibitors are protective.
For patients with postoperative abdominal pain who will be discharged on the day of the ERCP, serum amylase and/or lipase will be tested 2–6 h after the ERCP.
Patients with serum amylase and lipase values less than 1.5 and 4 times the normal upper limit, respectively, can be discharged without the concern of developing pancreatitis after ERCP.
For postoperative cholangitis, the ESGE defines it as a newly occurring high fever, with temperatures higher than 38 °C for more than 24 h combined with cholestasis; when biliary drainage is incomplete, i.e. in case of primary hilar obstruction and sclerosing cholangitis, the risk of cholangitis after ERCP is high.
Post-ERCP perforation is evidenced when imaging examinations reveal the presence of gas in the cavity outside the GI tract, whether or not the patient has the corresponding symptoms of abdominal pain or high fever. Generally speaking, perforation occurs after duodenal papillary sphincterotomy. But, in practical operations, perforation can also occur because of risk factors like biliary stricture and balloon dilation, operation by a very thin guide wire, improper operation of the tip of the endoscope, dysfunction of the sphincter of Oddi. Perforation can be divided into four types according to Stapfer’s typing, including duodenal wall perforation, periampullary perforation, biliary tract or pancreatic duct perforation with only single retroperitoneal gas, of which the most common is ampullary perforation due to sphincterotomy. The treatment for periampullary perforation is relatively more problematic. Periampullary perforation is more insidious and difficult to detect, and is often accompanied by an increase in amylase, with a high risk of being misdiagnosed as PEP. Subsequent worsening of the infection and abdominal pain occurs gradually, with obvious tenderness in the right hypochondriac, and the infection is difficult to be controlled in time, with a poor prognosis. If massive subcutaneous emphysema is discovered during surgery and circulatory failure occurs, surgery is mandatory. For some perforations of the duodenal wall at the level of the non-duodenal papillary part, the endoscopic suture technique is generally recommended for wound closure. Hypoxemia after ERCP is generally uncommon and is clinically defined as blood oxygen saturation <85%, usually due to the sedation operation. Hypotension or hypertension may occur during and after ERCP surgery.
One of the important and hard-to-handle complication is in-stent restenosis. In addition to clinical surgical common infections due to pathogenic bacteria such as G-Bacillus or G+Coccus, multidrug-resistant infections after ERCP are also common due to incomplete cleaning of the duodenoscope or of the ERCP endoscope and to the complex structures of these devices.
7.6 Postoperative Nursing in Super Minimally Invasive Surgery
7.6.1 Peroral Super Minimally Invasive Surgery
(I) Routine nursing
1. Observation of the condition. Changes in the patient’s condition should be closely observed, such as vital signs, abdominal pain, abdominal distension, hematemesis, hematochezia, etc., and whether there are complications. Low-flow oxygen must be administered continuously via a nasal tube. Patients having undergone general anesthesia must lie supine for 6–8 h without a pillow with their head tilted to one side. During airway nursing, inhalation of vomit into the trachea causing suffocation must be avoided. If the patient has respiratory and circulatory disorders, such as hypotension, hypoxemia, arrhythmia, etc., or if there is a delay in awakening sense recovery, the anesthetist should be asked to check and deal with it in time. Patients are not allowed to sign important documents or to make important decisions on the day of the anesthesia.
2. Drug use nursing. After the operation, medical orders must be followed with regard to the administration of intravenous anti-inflammatory drugs, acid suppression, enzyme inhibition, hemostasis, gastric mucosal protective agents and treatments with fluid nutritional supplements, the healing of surgical wounds must be monitored, as well as the effectiveness of the medication and any adverse reactions. If oral medication is prescribed, the patient must be told the correct method and time for taking it.
3. Diet nursing. After following the medical instructions to fast from food and drink for about three days, while receiving intravenous supplementation, the gradual transition to fasting can consist of drinking a small amount of water, then eating a light liquid, then gradually moving on to a semi-liquid diet, then a full diet.
4. Airway nursing. When a patient undergoing super minimally invasive surgery with tracheal intubation suffers from postoperative throat pain and the sensation of foreign body, he/she must be asked not to cough forcefully; oxygen atomization inhalation can be practiced, and the symptoms can be alleviated after a few days.
5. Tube nursing. If the patient keeps the drainage tube after the operation, it should be properly secured before the nurse returns to the ward, and folding, twisting or compression of the tube should be avoided. The drainage must be kept smooth, the color, nature and quantity of the drainage liquid should be observed, the drainage liquid must be poured and the drainage bag replaced timely. The patient and his/her family should be explained notifications that may during intubation, and how to prevent tube extruding.
6. Movement. Follow medical order after the operation to educate the patient’s movement range. If there is no complication, ask the patient who has regained consciousness following anesthesia to stay strictly in bed for rest, to defecate and urinate in bed; after awakening from anesthesia, ask the patient if he/she has feelings of dizziness, if his/her hands and feet become limp, etc. Once he/she is able to move out of bed as usual, he/she can go to the toilet with the family’s support. Medical instructions must be followed to gradually change the movement range, little by little; sudden changes of position must be avoided, as well as risks of falling out from bed due to postoperative physical weakness.
(II) Complication nursing
1. Hemorrhage
(1) Evaluation: hematemesis and black stool are the characteristic manifestations of upper digestive tract bleeding. The patient’s bleeding condition must be evaluated according to the amount of digestive tract bleeding: if the fecal occult blood test is positive, it means that the bleeding is greater than 5 ml; if there is black or tarry stools, it means that the bleeding is greater than 50–70 ml; if there is hematemesis, the blood accumulated in the stomach generally exceeds 250–300 ml; if there are systemic symptoms, for example dizziness, fatigue, palpitation, perspiration, etc., maybe the bleeding exceeds 400–500 ml; if a shock appears, such as manifestations of lowered blood pressure, shallow and rapid breathing, thready and rapid pulse, cold moist limbs etc., it means that the bleeding exceeds 1000 ml.
(2) Measures: ECG monitoring must be continued, the patient must be given continuous oxygen at low flow, his/her vital signs, as well as the occurrence of palpebral conjunctiva, nail color and hematemesis, black stool, should be observed. If the patient is on a gastric tube, the color and amount of drainage fluid must be observed and recorded in detail. In case of massive hemorrhage, the patient should lie down and raise the lower limbs slightly to ensure cerebral blood supply. He/she must tilt the head to one side when vomiting, suffocation must be avoided, secretions should be aspired from the respiratory tract using a vacuum extractor if necessary, the respiratory tract should be maintained clear. Two or more intravenous channels must be established timely, blood prepared, blood transfusion prepared, medical instructions should be timely followed to provide hemostasis, blood transfusion and fluid supplementation to maintain effective blood circulation. In/out must be accurately recorded, the colors, natures and amounts of vomits and stool must be observed. Medical instructions must be followed to timely send vomit and stool specimens; lab results of stool occult blood, hemoglobin, red blood cell count and red blood cell volume must be rechecked, anemia levels, whether bleeding stops must be recorded. Emergency endoscopic hemostasis or surgery should be performed if necessary.
2. Perforation. The perforation of the wall of the gastrointestinal tube results in a communication between the gastrointestinal cavity and the abdominal cavity or chest cavity. If it is not treated in time, severe pneumoperitoneum, mediastinal emphysema or retroperitoneal emphysema and diffuse peritonitis may occur.
After the operation, the patient should strictly lie in bed for rest; vital signs and consciousness change, occurrence of blood pressure drop, faster pulse, pale face, aggravated abdominal pain and tense abdominal muscle, tenderness, rebound tenderness should be closely observed. If the patient experiences severe abdominal pain or if the physical examination reveals a rigid abdomen, weakened or absent bowel sounds, the doctor must be immediately informed; he/she must be cooperated with to ensure prompt treatment. In the meantime ECG monitoring should be made, an ambulance and a negative pressure suction device prepared; an abdominal X-ray or a chest and abdominal CT scan at the patient’s bedside must be carried out urgently; the presence of free gas under the diaphragm at varying degrees must be checked. Medical instructions should be followed as regards fasting for food and water, gastrointestinal decompression, intravenous anti-inflammation, acid suppression and nutrition fluid supplementation therapy. Emergency surgery must be performed if necessary.
(III) Infection
We must insist on the concept of asepsis, including the care of the various drainage tubes. Operating routines should be strictly observed. The medical staff should strictly observe hand hygiene rules to avoid any cross-infection. Pipe withdrawal indications should be strictly followed, the intubation time must be reduced as much as possible. The patient’s temperature fluctuations must be monitored; the doctor should be notified in good time if the temperature is abnormal. Antibiotics should be administered according to medical prescriptions. Antibiotics should be prepared just before use to ensure their efficacy. Identify a caregiver, reduce visits, ensure beds are tidy, ensure personal hygiene, particularly in case of dirt: clothes should be changed timely. The patient’s mouth and skin should be kept clean to prevent infection.
(IV) Pain
The commonly used pain assessment method is based on a numeric rating scale (NRS) from 0 to 10: mild pain (1–3), the pain can be tolerated, not affecting sleep; moderate pain (4–6), the pain is obvious, affecting sleep, requiring analgesic control; severe pain (7–9) the pain is intense, requiring a strong analgesic, seriously affecting sleep and autonomic nerve function; intense pain (10), the pain is hard to tolerate and requires a high dose of a powerful analgesic.
Measures. The nurse evaluates the pain levels and informs the doctor. Medical prescriptions must be followed for the application of an analgesic, increased monitoring of patients is required, as well as basic care for bedridden patients, while creating a comfortable environment for patients, providing psychological care, distracting them, giving advice on maintaining good position and postures. When the patient feels discomfort due to upper abdominal distention, the doctor should be informed in good time, the nature and scope of abdominal pain must be strictly observed, while monitoring the patient’s vital signs. Analgesics should not be administered at random until the diagnosis has been clearly established, and abdominal pain can be relieved by appropriate movements, a change of posture, of position, distraction of attention, etc. Below a score of 4, mainly in psychological nursing, the patient must be comforted, his mood maintained stable, his/her confidence in overcoming the disease must be established; above a score of 4, medical instructions must be followed before using painkillers; the conditions of use of the medication and the reactions after its use must be observed.
(V) Esophageal stenosis
Due to the narrow esophageal lumen and many circumferential lesions, stenosis is a common complication after the operation of esophageal lesions. After the operation, the patient’s vital signs and the condition after the diet is resumed must be observed. During the dietary transition period, if the patient shows manifestations of esophageal stenosis like difficulty in swallowing, substernal discomfort, such as heartburn, retrosternal or subxiphoid burning sensation, stabbing pains, the doctor must be informed in god time. The patient should be asked to sit or lie down in a semi-lying position, with the head of the bed raised 15–30° during sleep. To avoid complications as reflux esophagitis, esophageal ulcer, bleeding due to gastric acid regurgitation, medication to suppress acidity and anti-inflammatories must be administered according to the doctor’s recommendations. In the meantime, oral care must be carried out to keep the mouth clean and avoid the occurrence of stomatoglossitis and oral ulcer.
(VI) Pancreatitis
Postoperative pancreatitis is the most common complication after performing ERCP. The occurrence of postoperative pancreatitis is related to pancreatic parenchyma impairment, mostly mild pancreatitis, and its common reasons include intubation injuring the sphincter of Oddi, too fast contrast agent, overdose injection, functional disturbance of the sphincter of Oddi; the original biliopancreatic diseases cause high pressure in the pancreatic bile duct. After endoscopic retrograde cholangiopancreatography, the increase in blood amylase to four to five times the normal level for 2–24 h is considered to be post-operative hyperamylasemia. After postoperative prophylactic application of antibiotics and drugs to inhibit pancreatic secretion, and general treatment such as fasting, the patient can make a full recovery. When elevated blood amylase is accompanied by persistent severe abdominal pain, nausea, vomiting and other symptoms, it should be considered it is combined with acute pancreatitis and it must be actively treated with it.
7.6.2 Psychological Nursing
After the operation, the patient may worry about whether the lesion sites have been dissected cleanly. Psychological support should be given to help the patient relax and relieve anxiety. The patient and his/her family must be helped to understand the knowledge relating to the disease and the prognosis. Nursing plans and measures must be prepared in line with the concerns that the patient may have postoperatively, in order to solve his/her problems, and self-care training should be provided to help him/her gradually move from the role of patient to his/her original social role and resume a normal life as soon as possible.
7.6.3 Health Education
1. Exercise. Intensive exercises like long-distance travel, running, etc., must be avoided in the first month after surgery. Use of abdominal force, hot compresses, bathing in hot water and hot spring should be avoided. Heavy manual labor must be forbidden.
2. Diet. The patient and family must be informed about the diet. Emphasis must be placed on the quality of the diet and the importance of following the diet regularly for recovery from the illness. Within the first month after surgery, the patient must pay attention to diet, ban cigarettes and alcohol, ban drinking strong tea and coffee, follow a regular diet, avoid eating and drinking too much, and avoid raw, hard and stimulating foods. The diet should consist of multiple small meals, with less semifluid slag, less fiber-rich foods like leek, celery, etc., and the patient should avoid taking oral drugs stimulating the esophagus and gastric membrane like NSAIDs. Bowel movements should be kept smooth and oral laxative drugs be taken if necessary.
3. Rest. The patient should pay attention to rest, to keep a pleasant mood, to avoid nervousness, to use sedatives appropriately, to combine rest with work in a reasonable fashion.
4. Take medicine. Patients should be explained the importance of taking medicine if needed. Medical instructions must be followed: the patient must take the medication as prescribed, at the prescribed time and dose, not reduce the dose by himself/herself, not stop the treatment, and follow again medical instructions to reduce the dose.
5. Recheck. Patients must be informed of certain related medical facts in order to avoid recurrence and prevent complications. All this must be regularly rechecked according to the postoperative pathological results and the surgical condition. Patients should be advised to undergo a follow-up endoscopy every three months, six months and twelve months after the operation, then carry out a follow-up endoscopy once a year and imaging tests and tumor marker tests, while being attentive to the risk of tumor recurrence. Timely visit if there is abdominal pain, hematemesis and blood in stool, etc.
7.6.4 Transanal Super Minimally Invasive Surgery
(I) Routine nursing
1. Condition observation. Changes in the patient’s condition should be closely observed, including vital signs, abdominal pain, vomiting, and hematochezia, etc., as well as any complication. A nasal catheter must be inserted for a continuous low-flow supply of oxygen. The patient must lie down for 6–8 h, without a pillow for patients under general anesthetic, with the head tilted to one side, taking care to keep the airways clear, and avoiding the risk of vomits being aspired into the trachea and causing suffocation. If the patient is found to have respiratory and circulatory disorders, etc., such as hypotension, hypoxemia, arrythmia, etc., or a delayed recovery from the anesthetic, the anesthetist should be asked to check and take the necessary measures in good time. Patients are not allowed to sign important documents or to make important decisions on the day of the anesthesia.
2. Drug use nursing. After the operation, medical prescriptions must be followed to administer intravenous treatment with anti-inflammatory, gastric acid inhibitors, hemostasis agents, drugs and liquid nutritional supplements, to promote healing of surgical wounds; the effectiveness of the medication and adverse effects must be monitored. If oral medication is administered, the patient should be told the correct method and the right time to take it. Patients with severe abdominal distention can take simethicone orally following the medical prescription, whereas patients must be told to lie on their side to pass gas, depending on the treatment.
3. Diet nursing. After following a medical prescription for a food and water fasting regime or a fasting diet with a small amount of water, while supplementing with intravenous fluids, the patient should gradually switch to a light liquid diet, then gradually to a semifluid and full diet.
4. Airway nursing. When a patient having undergone tracheal intubation has postoperative throat pain and the sense of foreign body, he/she must be asked not to cough forcefully, whereas oxygen atomization inhalation can be used so that the symptoms are alleviated after a few days.
5. Movement. After the operation, medical orders must be followed to educate the patient’s movement range. If there is no complication, the patient who does not awake from anesthesia must be asked to stay strictly in bed for rest, to defecate and urinate in bed; after awakening from anesthesia, he/she should be asked whether he/she has feelings of dizziness, and hands and feet becoming limp, etc.; after the off-bed movement is as usual, he/she can go to the toilets with family support. Medical orders must be followed to gradually change the movement range little by little, avoiding sudden changes of position, as well as the risks of falling from bed due to postoperative physical weakness.
(II) Complication nursing
1. Hemorrhage
(1) Evaluation. Hematochezia is the characteristic manifestation of bleeding of the lower digestive tract. The patient’s bleeding condition must be evaluated according to the amount of blood lost: if it only appears in the form of a positive fecal occult blood test, this indicates that the bleeding is greater than 5 ml; if it appears in the form of black or tarry stools, this indicates that the bleeding is greater than 50–70 ml; in the case of hematemesis, the blood accumulated in the stomach generally exceeds 250–300 ml; if there are systemic symptoms, for example dizziness, fatigue, palpitation, perspiration, etc., the bleeding might exceed 400–500 ml; if shock appears, such as manifestations of lowered blood pressure, shallow and rapid breathing, thready and rapid pulse, cold moist limbs, etc., the bleeding exceeds 1000 ml.
(2) Measures: ECG should be monitored continuously; a continuous low flow of oxygen should be administered; the patient’s vital signs, as well as palpebral conjunctiva, nail bed color and hematemesis, black stool, must be strictly observed. If the patient is on a gastric tube, the color and amount of drainage fluid must be observed and recorded in detail. In case of massive hemorrhage, the patient should lie down and slightly raise the lower limbs to ensure cerebral blood supply. He/she should tilt the head to one side when vomiting, avoid suffocation and aspiration, and secretions in the airway must be cleared with a vacuum extractor if necessary, whereas the airway should be maintained smooth. Two and more intravenous channels should be timely established, blood and blood transfusion should be prepared. Medical order must be timely followed to allow for hemostasis, blood transfusion, and fluid supplementation to maintain effective blood circulation. In/out must be accurately recorded, the colors, natures and amounts of vomits and stool must be observed. Medical instructions must be followed to timely send vomit and stool specimens; lab results of stool occult blood, hemoglobin, red blood cell count and red blood cell volume must be rechecked, anemia levels, whether bleeding stops must be recorded. Emergency endoscopic hemostasis or surgery should be performed if necessary.
2. Perforation. After the operation, the patient should strictly lie in bed for rest; vital signs and consciousness change, occurrence of blood pressure drop, faster pulse, pale face, aggravated abdominal pain and tense abdominal muscle, tenderness, rebound tenderness should be closely observed. If the patient experiences severe abdominal pain or if the physical examination reveals a rigid abdomen, weakened or absent bowel sounds, the doctor must be immediately informed; he/she must be cooperated with to ensure prompt treatment. In the meantime ECG monitoring should be made, an ambulance and a negative pressure suction device prepared; an abdominal X-ray or a chest and abdominal CT scan at the patient’s bedside must be carried out urgently; the presence of free gas under the diaphragm at varying degrees must be checked. Medical instructions should be followed as regards fasting for food and water, gastrointestinal decompression, intravenous anti-inflammation, acid suppression and nutrition fluid supplementation therapy. Emergency surgery must be performed if necessary.
3. Infection. We must insist on the concept of asepsis, including the care of the various drainage tubes. Operating routines should be strictly observed. The medical staff should strictly observe hand hygiene rules to avoid any cross-infection. Pipe withdrawal indications should be strictly followed, the intubation time must be reduced as much as possible. The patient’s temperature fluctuations must be monitored; the doctor should be notified in good time if the temperature is abnormal. Antibiotics should be administered according to medical prescriptions. Antibiotics should be prepared just before use to ensure their efficacy. Identify a caregiver, reduce visits, ensure beds are tidy, ensure personal hygiene, particularly in case of dirt: clothes should be changed timely. The patient’s mouth and skin should be kept clean to prevent infection.
(III) Psychological nursing
After the operation, the patient may worry about whether the lesion sites have been dissected cleanly. Psychological support should be given to help the patient relax and relieve anxiety. The patient and his/her family must be helped to understand the knowledge relating to the disease and the prognosis. Nursing plans and measures must be prepared in line with the concerns that the patient may have postoperatively, in order to solve his/her problems, and self-care training should be provided to help him/her gradually move from the role of patient to his/her original social role and resume a normal life as soon as possible.
(IV) Health education
1. Exercise. Intensive exercises like long-distance travel, running, etc., must be avoided in the first month after surgery. Use of abdominal force, hot compresses, bathing in hot water and hot spring should be avoided. Heavy manual labor must be forbidden.
2. Diet. The patient and family must be informed about the diet. Emphasis must be placed on the quality of the diet and the importance of following the diet regularly for recovery from the illness. Within the first month after surgery, the patient must pay attention to diet, ban cigarettes and alcohol, ban drinking strong tea and coffee, follow a regular diet, avoid eating and drinking too much, and avoid raw, hard and stimulating foods. The diet should consist of multiple small meals, with less semifluid slag, less fiber-rich foods like leek, celery, etc., and the patient should avoid taking oral drugs stimulating the esophagus and gastric membrane like NSAIDs. Bowel movements should be kept smooth and oral laxative drugs be taken if necessary.
3. Rest. The patient should pay attention to rest, to keep a pleasant mood, to avoid nervousness, to use sedatives appropriately, to combine rest with work in a reasonable fashion.
4. Take medicine. Patients should be explained the importance of taking medicine if needed. Medical instructions must be followed: the patient must take the medication as prescribed, at the prescribed time and dose, not reduce the dose by himself/herself, not stop the treatment, and follow again medical instructions to reduce the dose.
5. Recheck. Patients must be informed of certain related medical facts in order to avoid recurrence and prevent complications. All this must be regularly rechecked according to the postoperative pathological results and the surgical condition. Patients should be advised to undergo a follow-up endoscopy every three months, six months and twelve months after the operation, then carry out a follow-up endoscopy once a year and imaging tests and tumor marker tests, while being attentive to the risk of tumor recurrence. Timely visit if there is abdominal pain, hematemesis and blood in stool, etc.
7.6.5 Hot-Spot Issues in the Consensus
The Enhanced recovery after surgery (ERAS) program provides a form of multidisciplinary care and has been shown to predictably improve short-term outcomes associated with procedures such as ERCP and EMR. The original ERAS program focused on the importance of multidisciplinary teamwork to apply concepts that will maximize the efficiency of surgical recovery. The specific goals include the use of multimodal systems to reduce complications. And the proposal of the accelerated rehabilitation surgical care concept encourages medical care workers to further provide quality care and reduce the occurrence of complications. Compared to routine nursing, accelerated rehabilitation surgical nursing is patient-centered, starting from psychological intervention, advance and individual analgesic nursing, strengthening medical cooperation, etc.; it is close to the patients’ clinical needs, establishes rapid rehabilitation pathways, actively prevents postoperative complications, and effectively improves the clinical effect of surgical treatment.
For ERCP postoperative care, it is recommended to rest quietly in bed for one to two days, and to exercise in the department after three days, and to prohibit frequent and vigorous activities within one week. Blood amylase and routine postoperative blood tests must be checked three hours after the operation and the following morning until they are normal. Changes in vital signs should be closely observed, attention should be paid to consciousness changes, careful observation is needed for nausea, vomiting, abdominal pain, and black stool. After the patient has fasted food for two to three days, a decision will be made as to whether the diet should be restore diet according to clinical symptoms, blood amylase and blood routine results. The diet will consist of liquid and soft foods for one week, followed by a gradual return to a normal diet. With regard to common postoperative complications, bleeding should be monitored, as it is frequent in patients with blood clotting disorders or who are taking aspirin or steroids. Hemorrhage can occur in most patients with an incision that is too large or a cut that is too small; the process of extracting large stones can cause tear and bleeding of the nipple and papillary vascular variation within 24 h. Pancreatitis, which is easily induced from repetitive multiple intubations, congestion and edema of pancreatic duct opening, results from excessive electrocoagulation while cutting if the opening of the pancreatic duct is erroneously excised. Attention should be paid to the occurrence of abdominal pain, nausea, vomiting, and fever; timely check for blood amylase. Patients with only an increase in blood amylase can make a full recovery in two to three days after a simple treatment like fasting. For patients developing pancreatitis, active management according to acute pancreatitis protocols should be initiated promptly. In cases of intestinal perforation, the patient’s abdominal symptoms and vital signs, mental state should be closely observed. If perforation is suspected, an X-ray examination must be carried out immediately to determine whether there is free gas under the diaphragm, and whether or not there is perforation; if so, first a conservative treatment should be administered, with fasting from food and water; in case of continuous gastrointestinal decompression, intravenous fluid supplementation should be given.
When colorectal polyps are resected using endoscopic mucosal resection (EMR), they can be completely resected as a whole, which guarantees the integrity of the histopathological specimens, with the dual role of accurate examination and treatment. EMR is thus a representative surgical method in the field of super minimally invasive surgery. But EMR belongs to the category of traumatic operations, can lead to complications to some extent, and this is not helpful for the patient’s postoperative recovery. Therefore, it is vital to improve nursing and combine nursing with super minimally invasive surgery. General postoperative nursing care consists of instructing the patient to rest in bed for 6 h, to try not to speak for 24 h after the operation, to avoid intensive exercises like holding one’s breath and cough, and not to bathe in hot water. The examination of the patient’s sign must be improved, whether the patient has abdominal discomfort must be observed. Some patients may have pharyngeal edema symptoms like sore throat, hoarseness, he/she can be guided to orally take Watermelon Frost Throat-Clearing Buccal Tablets, rinse his/her mouth with an icy normal saline solution to alleviate discomfort, and the symptoms can be alleviated in one to two days. For EMR postoperative diet nursing, the patient can be advised to fast for food and water for 72 h after the operation; light foods and liquids can be given after 72 h. Depending on the patient’s recovery, semifluid and full diet can be gradually resumed. The patient’s diet consists of eating several small meals, nutritive foods enriched in proteins and vitamins. EMR postoperative complications are mostly postoperative infection and postoperative hemorrhage. But postoperative pain and discomfort should also be paid attention to. If active bleeding occurs, that is, when nursing, when the patient has symptoms like black stook, blood in stool, lowered blood pressure, pale face, etc., the doctor should be immediately notified so that symptomatic treatment can be prescribe quickly. Postoperative perforation is manifested as by worsening abdominal pain. In case of abdominal tenderness and pain on pressure on physical examination, the doctor should also be notified for prescribing a treatment. Once the diagnosis is confirmed, surgery will be performed immediately. On day before the patient discharges, he will be given discharge advices and required to stay at home for recuperation. Within 30 days after the operation, intensive exercise and heavy manual labor should not be conducted. Once the patient has improper symptoms like abdominal pain, black stool, etc., he/she must be admitted for treatment immediately, drugs must be used according to the medical prescription, emphasizing the importance of regular rechecking. A decision will be made on a subsequent return to the clinic depending on the state of health.
References
[1] Digestive Endoscopy Committee of the Endoscopists Branch, Chinese Medical Doctor Association; Professional Committee of Tumor Endoscopy of China Anti-Cancer Association. (2019) Chinese guideline for bowel preparation for colonoscopy (2019, Shanghai), Chin. J. Intern. Med. (7), 485–495.
[2] Colorectal Group of the Chinese Medical Association Branch of Gastrointestinal Endoscopy. (2023) Consensus on bowel preparation for colonoscopy (2023, Guangzhou), Chin. J. Dig. Endosc. (06), 421–430.
[3] Wu Y., Zhang S., Yu Z., et al. (2013) Multicenter study of premedication of pronase granules to improve visibility during gastroendoscopy, Chin. J. Dig. Endosc. (12), 671–674.
[4] Chen X., Feng Y., Chu G. (2016) Research progress in preprocedural preparation for gastroscopy, Chin. J. Dig. Endosc. (2), 133–136.
[5] National Clinical Research Center for Digestive Diseases; National Digestive Endoscopy Quality Control Center; Capsule Endoscopy Collaborative Group of Chinese Society of Digestive Endoscopy; Capsule Endoscopy Group of Digestive Endoscopy Branch of Shanghai Medical Association. (2021) Chinese guideline on magnetically controlled capsule gastroscopy (2021, Shanghai), Chin. J. Dig. Endosc. (12), 949–963.
[6] Wang J. (2014) Evaluation on efficiency of dyclonine mucilagines and lidocaine mucilagines in gastroscopy, Chin. J. Pract. Med. (21), 63–65.
[7] De Francesco V., Alicante S., Amato A., et al. (2022) Quality performance measures in upper gastrointestinal endoscopy for lesion detection: Italian AIGO-SIED-SIGE joint position statement, Dig. Liver Dis. 54(11), 1479–1485.
[8] Feng S. M., Zhao Y., Xu Q., et al. (2020) Development and characterization of A new dimethicone nanoemulsion and its application for electronic gastroscopy examination, Int. J. Nanomed. 15, 5405–5416.
[9] Zeng X., Wu Y., Tan R. (2023) Sedation/anesthesia management in the perioperative period for endoscopic submucosal dissection for upper gastrointestinal lesions, Int. J. Anesthesiol. Resusc. (10), 1100–1104.
[10] Small Bowel Endoscopy and Capsule Endoscopy Group of the Chinese Society of Digestive Endoscopy. (2018) Chinese guideline for clinical application of enteroscopy, Chin. J. Dig. Endosc. (10), 693–702.
[11] Rondonotti E., Spada C., Adler S., et al. (2018) Small-bowel capsule endoscopy and device-assisted enteroscopy for diagnosis and treatment of small-bowel disorders: European Society of Gastrointestinal Endoscopy (ESGE) Technical Review, Endoscopy, 50(4), 423–446.
[12] Super Minimally Invasive Surgery Collaboration Group of Chinese Society of Digestive Endoscopy; Endoscopic Physicians Branch of Chinese Medical Doctor Association; Beijing Branch of Chinese Society of Digestive Endoscopy. (2021) Consensus of Chinese experts on the management of achalasia (2020, Beijing), Chin. J. Dig. Endosc. (04), 256–275.
[13] Dumonceau J. M., Kapral C., Aabakken L., et al. (2020) ERCP-related adverse events: European Society of Gastrointestinal Endoscopy (ESGE) guideline, Endoscopy 52(2), 127–149.
[14] Hassan C., East J., Radaelli F., et al. (2019) Bowel preparation for colonoscopy: European Society of Gastrointestinal Endoscopy (ESGE) guideline - update 2019, Endoscopy 51(8), 775–794.
[15] Bisschops R., Areia M., Coron E., et al. (2016) Performance measures for upper gastrointestinal endoscopy: a European Society of Gastrointestinal Endoscopy quality improvement initiative, United Eur. Gastroenterol. J. 4(5), 629–656.
[16] Song H. J., Moon J. S., Do J. H., et al. (2013) Guidelines for bowel preparation before video capsule endoscopy, Clin. Endosc. 46(2), 147–154.
[17] Kaminski M. F., Thomas-Gibson S., Bugajski M., et al. (2017) Performance measures for lower gastrointestinal endoscopy: a European Society of Gastrointestinal Endoscopy (ESGE) quality improvement initiative, Endoscopy 49(4), 378–397.
[18] Colorectal Group of the Chinese Society of Digestive Endoscopy. (2023) Consensus on bowel preparation for colonoscopy (2023, Guangzhou), Chin. J. Dig. Endosc. (06), 421–430.
[19] Veitch A. M., Radaelli F., Alikhan R., et al. (2021) Endoscopy in patients on antiplatelet or anticoagulant therapy: British Society of Gastroenterology (BSG) and European Society of Gastrointestinal Endoscopy (ESGE) guideline update, Gut 70(9), 1611–1628.
[20] Chen Q., Ning B., Wu Q., et al. (2023) Chinese expert consensus on wound pretreatment and antibiotic application in endoscopic super minimally invasive surgery (2023, Beijing), Chin. J. Gastrointest. Endosc. (Electron. Ed.) 10(2).
[21] Li D., He R., Hou G., et al. (2020) Characterization of the esophageal microbiota and prediction of the metabolic pathways involved in esophageal carcinoma, Front. Cell Infect. Microbiol. 10, 268.
[22] Ianiro G., Molina-Infante J., Gasbarrini A. (2015) Gastric microbiota, Helicobacter 20(Suppl 1), 68–71.
[23] James K. R., Gomes T., Elmentaite R., et al. (2020) Distinct microbial and immune niches of the human colon, Nat. Immunol. 21(3), 343–353.
[24] Leite G., Weitsman S., Parodi G., et al. (2020) Mapping the segmental microbiomes in the human small bowel in comparison with stool: a REIMAGINE study, Dig. Dis. Sci. 65(9), 2595–2604.
[25] Hastier-De Chelle A., Onana-Ndong P., Olivier R., et al. (2022) Impact of antibiotic prophylaxis and conditioning modalities in per-oral endoscopic myotomy for esophageal motor disorders, Scand. J. Gastroenterol. 57(12), 1522–1530.
[26] Eichel V. M., Jabs J. M., Unser S., et al. (2021) Does the reprocessing of endoscopes have to take place immediately after pre-cleaning? A first evaluation, Clin. Endosc. 54(4), 526–533.
[27] China experts consensus on the protocol of early gastric cancer screening (2017, Shanghai), Chin. J. Dig. (2018) 38(2).
[28] Nelson R. L., Hassan M., Grant M. D. (2020) Antibiotic prophylaxis in colorectal surgery: are oral, intravenous or both best and is mechanical bowel preparation necessary? Tech. Coloproctol. 24(12), 1233–1246.
[29] Zhao Y., Li B., Sun, Y., et al. (2022) Risk factors and preventive measures for anastomotic leak in colorectal carcinoma, Technol. Carcinoma Res. Treat. 21, 15330338221118983.
[30] Li Z., Linghu E. (2019) Chinese guideline for bowel preparation for colonoscopy (2019, Shanghai), Chin. J. Intern. Med. 58(7).
[31] Zhang Z., Yan X., Duan B., et al. (2022) A new preparation method for peroral endoscopic myotomy in patients with achalasia can effectively reduce the esophageal residual contents: a comparative retrospective study, Gastroenterol. Res. Pract. 2022, 6953166.
[32] Li L., Spandorfer R., Qu C., et al. (2018) Gastric per-oral endoscopic myotomy for refractory gastroparesis: a detailed description of the procedure, our experience, and review of the literature, Surg. Endosc. 32(8), 3421–3431.
[33] Chinese guideline for the clinical practice of endoscopic ultrasound-guided fine-needle aspiration, Chin. J. Dig. Endosc. (2017) 34(1).
[34] Joshi G. P., Abdelmalak B. B., Weigel W. A., et al. (2023) American Society of Anesthesiologists Practice Guidelines for preoperative fasting: carbohydrate-containing clear liquids with or without protein, chewing gum, and pediatric fasting duration-a modular update of the 2017 American Society of Anesthesiologists Practice Guidelines for preoperative fasting, Anesthesiology 138(2), 132–151.
[35] Kawata N., Tanaka M., Kakushima N., et al. (2016) The low incidence of bacteremia after esophageal endoscopic submucosal dissection (ESD) obviates the need for prophylactic antibiotics in esophageal ESD, Surg. Endosc. 30(11), 5084–5090.
[36] Liu Y., Chen Y., Shu X., et al. (2020) Prophylactic antibiotics may be unnecessary in gastric endoscopic submucosal dissection due to the low incidence of bacteremia, Surg. Endosc. 34(9), 3788–3794.
[37] Linghu E. (2019) Expert consensus on standardized endoscopic resection of early gastric cancer (2018, Beijing), Chin. J. Dig. Endosc. 36(6).
[38] Izumi K., Osada T., Sakamoto N., et al. (2014) Frequent occurrence of fever in patients who have undergone endoscopic submucosal dissection for colorectal tumor, but bacteremia is not a significant cause, Surg. Endosc. 28(10), 2899–2904.
[39] Chinese guidelines for screening and endoscopic diagnosis and treatment of early colorectal cancer (2014, Beijing), Natl. Med. J. China (2015), (28).
[40] Yang B., Kong L., Ullah S., et al. (2022) Endoscopic retrograde appendicitis therapy versus laparoscopic appendectomy for uncomplicated acute appendicitis, Endoscopy 54(8), 747–754.
[41] Chinese Medical Association Branch of Gastrointestinal Endoscopy; Chinese Medical Doctor Association Branch of Endoscopic Physicians; Beijing Medical Association Branch of Gastrointestinal Endoscopy; China Medical Device Association Branch of Gastrointestinal Endoscopy Innovation. (2018) Consensus on digestive endoscopic tunnel technique (2017, Beijing), Chin. J. Dig. Endosc. (1), 1–14.
[42] Jenssen C., Brkljacic B., Hocke M., et al. (2016) EFSUMB guidelines on interventional ultrasound (INVUS), part VI - ultrasound-guided vascular interventions, Ultraschall Med. 37(5), 473–476.
[43] Ran W., Shan J., Sun X. (2020) Advances in endoscopic ultrasound-guided drainage and the use of novel mushroom-tipped covered metal stents for pancreatic pseudocyst treatment, World Chin. J. Digestol. 28(5).
[44] Teoh A. Y. B., Bapaye A., Lakhtakia S., et al. (2020) Prospective multicenter international study on the outcomes of a newly developed self-approximating lumen-apposing metallic stent for drainage of pancreatic fluid collections and endoscopic necrosectomy, Dig. Endosc. 32(3), 391–398.
[45] Turan A. S., Jenniskens S., Martens J. M., et al. (2022) Complications of percutaneous transhepatic cholangiography and biliary drainage, a multicenter observational study, Abdom. Radiol. (NY) 47(9), 3338–3344.
[46] Wu Z., Wu X., Yao W., et al. (2021) Pathogens’ distribution and changes of antimicrobial resistance in the bile of acute biliary tract infection patients, Chin. J. Surg. 59(1).
[47] Del Toro López M. D., Arias Díaz J., Balibrea J. M., et al. (2021) Executive summary of the Consensus Document of the Spanish Society of Infectious Diseases and Clinical Microbiology (SEIMC) and of the Spanish Association of Surgeons (AEC) in antibiotic prophylaxis in surgery, Cir. Esp. (Engl. Ed.) 99(1), 11–26.
[48] Ren J., Wu X. (2020) Chinese guideline for the diagnosis and management of intra-abdominal infection (2019 edition), Chin. J. Pract. Surg. 40(1).
[49] Khashab M. A., Chithadi K. V., Acosta R. D., et al. (2015) Antibiotic prophylaxis for GI endoscopy, Gastrointest. Endosc. 81(1), 81–89.
[50] Beilenhoff U., Biering H., Blum R., et al. (2017) Prevention of multidrug-resistant infections from contaminated duodenoscopes: Position Statement of the European Society of Gastrointestinal Endoscopy (ESGE) and European Society of Gastroenterology Nurses and Associates (ESGENA), Endoscopy 49(11), 1098–1106.
[51] Arvanitakis M., Gkolfakis P., Despott E. J., et al. (2021) Endoscopic management of enteral tubes in adult patients - part 1: definitions and indications. European Society of Gastrointestinal Endoscopy (ESGE) guideline, Endoscopy 53(1), 81–92.
[52] Gkolfakis P., Arvanitakis M., Despott E. J., et al. (2021) Endoscopic management of enteral tubes in adult patients - part 2: peri- and post-procedural management. European Society of Gastrointestinal Endoscopy (ESGE) guideline, Endoscopy 53(2), 178–195.
[53] Dumonceau J. M., Andriulli A., Elmunzer B. J., et al. (2014) Prophylaxis of post-ERCP pancreatitis: European Society of Gastrointestinal Endoscopy (ESGE) guideline - updated June 2014, Endoscopy 46(9), 799–815.
[54] Yu Y., Shi D., Zhang D., et al. (2019) Research progress on local medication for wound management after endoscopic mucosal resection, Chin. J. Dig. Endosc. (1), 70–73.
[55] Pimentel-Nunes P., Libânio D., Bastiaansen B. A. J., et al. (2022) Endoscopic submucosal dissection for superficial gastrointestinal lesions: European Society of Gastrointestinal Endoscopy (ESGE) guideline - update 2022, Endoscopy 54(6), 591–622.
[56] Manes G., Paspatis G., Aabakken L., et al. (2019) Endoscopic management of common bile duct stones: European Society of Gastrointestinal Endoscopy (ESGE) guideline, Endoscopy 51(5), 472–491.
[57] National Clinical Research Center for Digestive Diseases; Chinese Society of Digestive Endoscopy; Chinese Medical Doctor Association Branch of Gastroenterologists. (2017) Perioperative guidelines for gastric endoscopic submucosal dissection, Chin. J. Dig. Endosc. (12), 837–851.
[58] Surgical Group of Chinese Society of Digestive Endoscopy; Natural Orifice Transluminal Endoscopic Surgery (NOTES) Group of Chinese Society of Digestive Endoscopy; Digestive Endoscopy Committee of the Endoscopists Branch, Chinese Medical Doctor Association; Gastrointestinal Surgery Group of the Chinese Society of General Surgery, Chinese Medical Association. (2023) Chinese consensus on endoscopic diagnosis and management of gastrointestinal submucosal tumors (version 2023), Chin. J. Dig. Endosc. (04), 253–263.
[59] Chinese Society of Digestive Endoscopy; Branch of Endoscopic Physicians of Chinese Medical Association; Gastrointestinal Endoscopy Branch of Beijing Medical Association; Digestive Endoscopy Innovation and Development Branch of China Medical Device Industry Association. (2018) Expert consensus on digestive endoscopic tunnel technique (2017, Beijing), Chin. J. Dig. Endosc. (1), 1–14.
[60] Linghu E. Q. (2019) New classifications of intraoperative bleeding and muscularis propria injury in endoscopic resection, Chin. Med. J. (Engl.) 132(15), 1856–1858.
[61] National Center for Quality Control of Digestive Endoscopy; National Clinical Research Center for Digestive Diseases; Gastrointestinal Early Cancer Alliance (GECA); Branch of Endoscopic Physicians of Chinese Medical Doctor Association; Chinese Society of Digestive Endoscopy (CSDE); Professional Committee of Tumor Endoscopy of Chinese Anti-Cancer Association. (2020) Chinese expert consensus on endoscopic submucosal dissection-related adverse events (2020, Wuxi), Chin. J. Dig. Endosc. (06), 390–403.
[62] Al-Haddad M. A., Elhanafi S. E., Forbes N., et al. (2023) American Society for Gastrointestinal Endoscopy guideline on endoscopic submucosal dissection for the management of early esophageal and gastric Carcinomas: methodology and review of evidence, Gastrointest. Endosc. 98(3), 285–305.e238.
[63] Peng Z., Li Y., Wu Y., Liu X. (2023) Complications and management status of endoscopic therapy for esophagogastric varices, Chin. J. Dig. Endosc. (01), 72–78.
[64] ERCP Group of Chinese Society of Digestive Endoscopy; Biliary and Pancreatic Group of Chinese Medical Doctor Association Branch of Gastroenterologists; National Clinical Research Center for Digestive Diseases. (2018) Chinese Guidelines for ERCP (2018), Chin. J. Dig. Endosc. (11), 777–813.
[65] Zhu L., Li J., Liu Z., Cai M., Qin W., Chen W., Zhang Y., Zhong Y., Yao L., Zhou H. (2023) Evaluation and management of gastrointestinal fistula after upper gastrointestinal tunnel endoscopic surgery, Chin. J. Dig. Endosc. (12), 1006–1010.
[66] Chinese Society of Digestive Endoscopy (CSDE); Branch of Endoscopic Physicians of Chinese Medical Doctor Association; Beijing Branch of Chinese Society of Digestive Endoscopy; Innovation and Development Branch of Digestive Endoscopy of China Medical Device Industry Association. (2018) Consensus on digestive endoscopic tunnel technique (2017, Beijing), Chin. J. Dig. Endosc. (1), 1–14.
[67] Surgical Group of Chinese Society of Digestive Endoscopy; Natural Orifice Transluminal Endoscopic Surgery (NOTES) Group of Chinese Society of Digestive Endoscopy; Digestive Endoscopy Committee of the Endoscopists Branch, Chinese Medical Doctor Association; Gastrointestinal Surgery Group of Chinese Society of General Surgery. (2023) Chinese consensus on endoscopic diagnosis and managment of gastrointestinal submucosal tumors (version 2023), Chin. J. Dig. Endosc. (04), 253–263.
[68] ERCP Group of Chinese Society of Digestive Endoscopy; Branch of Endoscopic Physicians of Chinese Medical Doctor Association; Beijing Branch of Chinese Society of Digestive Endoscopy. (2021) Chinese expert consensus on the endoscopic diagnosis and treatment of pancreatic fistula (2020, Beijing), Chin. J. Dig. Endosc. (04), 276–287.
[69] National Clinical Research Center for Digestive Diseases; Chinese Society of Digestive Endoscopy; Chinese Medical Doctor Association Branch of Gastroenterologists. (2017) Clinical guidelines for perioperative management of gastric Endoscopic Submucosal Dissection (ESD), Chin. J. Dig. Endosc. (12), 837–851.
[70] Dumonceau J. M., Kapra C., Aabakken L., et al. (2020) ERCP-related adverse events: European Society of Gastrointestinal Endoscopy (ESGE) guideline, Endoscopy 52(2), 127–149.
[71] Wang J., Ma L., Fan Z. (2016) Research progress in measures to prevent esophageal stent migration, Chin. J. Dig. Endosc. (01), 62–65.
[72] Irani S., Baron T. H., Gluck M., et al. (2014) Preventing migration of fully covered esophageal stents with an over-the-scope clip device (with videos), Gastrointest. Endosc. 79(5), 844–851.
[73] Gastroenterology Group of the Society of Pediatrics, Chinese Medical Association; Editorial Board of Chinese Journal of Pediatrics. (2023) Expert consensus on diagnosis and treatment of esophageal stricture in Chinese children, Chin. J. Pediat. (07), 588–593.
[74] Wang Z., Zheng H., Li S., Li Z. (2023) Progress of prevention and treatment for esophageal stricture after endoscopic resection, Chin. J. Dig. Endosc. (03), 237–242.
[75] General Thoracic Surgery Group of the Society of Pediatric Surgery, Chinese Medical Association; Cardiothoracic Surgery Committee of the Branch of Pediatric Surgeons, Chinese Medical Doctor Association. (2023) National expert consensus on surgical management strategies for esophageal stenosis in children (2023 edition), Chin. J. Pediat. Surg. (09), 774–783.
[76] Wang Y., Ma L., Liu C., Wei Y., Zhang Y., Li J., Wu T. (2023) Prevention and treatment strategies for esophageal stenosis after thoracoscopy for type III esophageal atresia, Chin. J. Pediat. Surg. (09), 784–788.
[77] Chandrasekhara V., Khashab M. A., Muthusamy V. R., et al. (2017) Adverse events associated with ERCP, Gastrointest. Endosc. 85(1), 32–47.
[78] Dietrich C. F., Bekkali N., Burmeister S., et al. (2022) Controversies in ERCP: indications and preparation, Endosc. Ultrasound 11(3), 186–200.
[79] van Halsema E. E., Kappelle W. F., Weusten W., et al. (2018) Stent placement for benign esophageal leaks, perforations, and fistulae: a clinical prediction rule for successful leakage control, Endoscopy 50(2), 98–108.
[80] Inflammatory Bowel Disease Group of the Chinese Society of Gastroenterology, Chinese Medical Association. (2020) Experts guideline on digestive endoscopy techniques in the diagnosis and management of inflammatory bowel disease in China, Chin. J. Inflammatory Bowel Dis. (04), 283–291.
[81] Digestive Endoscopy Committee of the Endoscopists Branch, Chinese Medical Doctor Association; Pancreatology Committee of CMDA; Chinese Journal of Digestion; Chinese Journal of Digestive Endoscopy; Chinese Journal of Pancreatology; Chinese Journal of Gastroenterology; Chinese Journal of Practical Internal Medicine. (2018) Expert consensus on perioperative medication for endoscopic retrograde cholangiopancreatography, Chin. J. Dig. Endosc. (10), 704–712.
[82] Roveron G., Antonini M., Barbierato M., et al. (2018) Clinical practice guidelines for the nursing management of percutaneous endoscopic gastrostomy and jejunostomy (PEG/PEJ) in adult patients: an executive summary, J. Wound Ostomy Continence Nurs. 45(4), 326–334.
[83] Lin Q., Zhang Q., Zhou N., et al. (2020) Application of accelerated rehabilitation surgical nursing in perioperative nursing of ERCP patients with choledocholithiasis, Int. J. Nurs. (10), 1829–1832.
[84] Tian H., Yue L. (2021) The impact of early cold compress nursing based on the concept of Enhanced Recovery After Surgery (ERAS) on pain scores and complications in patients with ankle periarticular fractures, Int. J. Nurs. (17), 3157–3159.
[85] Low D. E., Allum W., De Manzoni G., et al. (2019) Guidelines for perioperative care in esophagectomy: Enhanced Recovery After Surgery (ERAS(®)) society recommendations, World J. Surg. 43(2), 299–330.
[86] Yu A., Liu J., Kong Q., Zhu G. (2016) Nursing cooperation of ERCP interventional therapy in elderly patients, Chin. J. Mod. Nurs. (27), 3977–3979.
[87] Ji Y., Xu L. (2023) Analysis of the coordination of care for colon polyps treated by Endoscopic Mucosal Resection (EMR), Guide China Med. 21(18), 5–8.
[88] Liang S., Lin Z., Chen L. (2023) Precise nursing of Endoscopic Submucosal Dissection (ESD) in the treatment of broad-based colorectal polyps, Guide China Med. 21(12), 139–141.
Part Two Specific Discussion of Super Minimally Invasive Surgery
Chapter 8 Super Minimally Invasive Surgery of Esophageal Diseases
8.1 Early Esophageal Carcinoma and Super Minimally Invasive Surgery
8.1.1 Introduction
(I) Introduction to early esophageal carcinoma
Early esophageal carcinoma (EEC) is a lesion restricted to the mucous layer or the submucosal layer, with or without lymph node metastases. EEC and precancerous lesions (including intraepithelial neoplasia) are evidenced by endoscopic white light imaging (WLI) when the color of the mucosa appears patchy, red or white, the mucosa being morphologically slightly raised or depressed; few lesions are totally flat with a rough surface; they may be accompanied by erosion or nodular changes, and the submucosal blood vessels are blurred or disappeared. Carcinoma can be detected in narrow-band imaging can be shown as dark brown area, iodine staining can be shown as colorless and/or positive pink sign, the arrangement of the capillary loops in the esophageal epithelial papilla are disordered, dense, dilated and distorted. Patients with early esophageal carcinoma may not have obvious symptoms, and reflux and indigestion are the most common clinical symptoms in early patients.
(II) Diagnosis of early esophageal carcinoma
1. Endoscopic diagnosis. Upper gastrointestinal endoscopy combined with histopathology is the gold standard for the diagnosis of early esophageal carcinoma and precancerous lesions. WLI is a basic technique for the detection of early esophageal lesions, but most EEC and precancerous lesions are not obvious under WLI, which affects the detection rate. Thus, currently, in the population at high risk of esophageal carcinoma, it is mainly necessary to resort to chromoendoscopy, and one can also resort to electronic staining endoscopy and magnifying endoscopy when conditions permit.
2. Ultrasonic endoscope. EUS allows the evaluation of the change in the hierarchy of the esophageal wall, tumor infiltration depth, the presence of abnormally enlarged lymph nodes in the vicinity and the invasion of peripheral organs in patients with early esophageal carcinoma. Clinical staging of early esophageal carcinoma by EUS is recommended before treatment.
3. Imaging examination
(1) CT is the most commonly used imaging method for clinical staging of esophageal carcinoma, which is mainly used to determine the location, the infiltration depth, the relative relationship between the tumor and surrounding structures and organs, lymphatic metastasis and distant metastasis.
(2) MRI generates artifacts due to interferences caused by the heart, great vessels and breathing movement, which may affect the staging of esophageal carcinoma. So it is usually not the first-choice examination for early esophageal carcinoma.
(3) A systemic ultrasound of the superficial lymph nodes can determine the status of the lymphatic metastases.
(4) When a patient presents with suspected or distant lymphatic metastasis, Positron Emission Tomography/Computed Tomography(PET/CT) is recommended.
(III) Super minimally invasive surgical technique for the treatment of early esophageal carcinoma
Early esophageal carcinoma can be treated endoscopically by super minimally invasive surgery (SMIS), and mostly through a peroral natural cavity channel (peroral endoscopic complete partial resection of the early esophageal carcinoma) or a per-tunnel channel (per-tunnel endoscopic complete partial resection of the early esophageal carcinoma).
The techniques applied currently to treat early esophageal carcinoma by super minimally invasive surgery and their past names are displayed in table 8.1.
TAB. 8.1 — List of the techniques and past names applied currently to the treatment of early esophageal carcinoma by super minimally invasive surgery.
| No. | Super Minimally Invasive Surgery (SMIS) | Technique | Past Name |
| 1 | Peroral super minimally invasive resection of an early esophageal carcinoma | Super minimally invasive non-full-thickness resection | Endoscopic submucosal dissection |
| Endoscopic submucosal dissection with traction | |||
| Cap-assisted endoscopic mucosal dissection | |||
| 2 | Per-tunnel super minimally invasive resection of an early esophageal carcinoma | Super minimally invasive non-full-thickness resection | Endoscopic tunnel submucosal dissection |
| Endoscopic submucosal dissection with traction | |||
| 3 | Peroral super minimally invasive stenosis treatment for early esophageal carcinoma | Super minimally invasive surgery of stenosis | Gastrointestinal endoscopic super minimally invasive esophageal surface skin transplantation |
| Endoscopic esophageal stenting |
8.1.2 Peroral Super Minimally Invasive Surgery with Non-Full-Thickness Resection for the Treatment of Early Esophageal Carcinoma
Peroral super minimally invasive surgery by non-full-thickness resection for early esophageal carcinoma can achieve precise excision of the mucosal layer, the mucosal muscle layer and the submucosa of the esophageal carcinoma foci by endoscopic mucosal dissection, endoscopic submucosal dissection with traction, and cap-assisted endoscopic mucosal dissection.
(I) Indication and contraindication
1. Indications
(1) Absolute indications: when the lymphatic metastasis rate of M1 and M2 lesions in the esophagus is only 0%–5%, and when there is no suspicious lymphatic metastasis in preoperative evaluation.
(2) Relative indications: when the lymphatic metastasis rate of M3 and SM1 stage esophageal carcinoma is between 10% and 20%, and when there is no suspicious lymphatic metastasis in preoperative evaluation.
2. Contraindications
(1) Negative lifting sign of lesion tissues after submucosal injection.
(2) Patients with coagulation disorders and taking anticoagulants are not suitable for surgery before the coagulation function is corrected.
(3) Patients with poor general condition who cannot tolerate endoscopic surgery.
(4) Patients with esophageal varices or varicosity, without effective bleeding prevention countermeasures.
(5) In case of lymphatic metastasis detected during the preoperative evaluation.
(II) Preoperative preparation
(1) Master the indications and contraindications of painless endoscopy, ask about the patient’s age, whether he has heart or lung diseases and hypertension history, allergies to drugs, conduct routine examinations or the necessary preoperative examinations: blood, coagulation function, blood biochemistry, ECG, chest film, etc.
(2) Conduct active and effective communication with the patient and family, fully evaluate the patient’s condition and psychological state; for patients with negative moods like anxiety, depression, worry, psychological support must be provided, improve the communication with the patient, try to alleviate his/her negative mood and establish confidence.
(3) Advise the patient to eat mild and easy-to-digest food one night before, fasting for food and water for 12 h before surgery.
(III) Postoperative nursing
(1) The patient should be strictly lying in bed for rest for 24 h after the operation, avoid big amplitude activities. The occurrence of complications, fever, palpitations, cold sweat, abdominal pain, black stool, infection and hemorrhage, etc., should be monitored.
(2) After the operation, the patient should be fasting for food for 48–72 h, and then switch to a light liquid diet (rice soup, noodle soup, milk, etc.). Then he/she should gradually switch to a semifluid diet (soft noodle, porridge, etc.), be forbidden to eat rough and spicy foods, avoid heavy manual labor within half a month. The fasting period should be extended for patients who are hemorrhaging and those with large wounds. Nutritional supplementation and anti-inflammation as well as acid suppression drugs should be prescribed.
(3) To rid the patient of his/her concern, he/she should be explained that the metal clip used during the operation will fall off and be spontaneously eliminate with the healing of the wound. If the surgical wound forms an ulceration, it can be generally totally cured after one to two months, so the patient must be told to check-back regularly and undergo a follow-up endoscopy.
(4) Postoperative pathologies may include high-grade intraepithelial neoplasia, intramucosal carcinoma, and submucosal superficial carcinoma, which present a risk of local residual and recurrence. So close follow-up is needed, with an enhanced abdominal CT scan if necessary; if there is local recurrence, endoscopic resection can be performed in good time.
8.1.2.1 Endoscopic Submucosal Dissection (ESD)
Endoscopic submucosal dissection (ESD) is an endoscopic technique for the determination of lesion boundaries, involving marking, submucosal injection, full lifting of lesions, gradual dissection of the tissue between the muscle layer of the lesion’s mucosa and the muscularis propria using an electric knife to completely dissect the membrane of the lesion and the submucosal layer, adduction of the lesion, and finally treatment of the wounds. It is one of the therapeutic methods for treating early esophageal carcinoma. Compared to other endoscopic dissections, it has a higher en bloc resection rate, a higher complete resection rate and a lower local recurrence rate.
(I) Surgical operation and skills
1. Anesthesia and position. All patients undergo general anesthesia with tracheal intubation, lying on their left side; attention should be paid to keep them warm, adjusting the room temperature and the infusion temperature, and avoiding pressure sores.
2. Operating steps (figure 8.1)
(1) The extent and depth of the lesion must be determined. First, a routine endoscopy should be performed to determine the foci’s site, size and shape, which should be combined with staining and magnifying endoscopy to determine the foci’s extent, nature and infiltration depth.
(2) Marking: electrocoagulation marking should be directly conducted by using argon plasma coagulation (APC) and an electric knife. For lesions that lack clear boundaries, Lugol’s iodine solution should be used first to stain the lesion; after determining the lesion’s extent under NBI observation, electrocoagulation marking at 2–5 mm outside of the lesion should be made, the interval for every marking spot being about 2 mm. The oral side of the lesion is marked using a double-layer labeling method to distinguish it from the anal side of the lesion.
(3) Submucosal injection: a submucosal injection should be conducted outside the marking spot by using an endoscopic injection needle, the quantity of liquid to be injected each time being 2–5 ml; the focus must be lifted and separated from the muscle layer, which is helpful for the complete resection of the lesion by ESD without injuring the muscularis propria and for reducing the occurrence of complications as perforation and hemorrhage, etc.
(4) Edge incision: incising the lesion’s peripheral membrane is ESD is the key step for ESD therapy. The mucosa is incised with the electric knife along the outside of the marks, the first incision is usually made at the distal end of the lesion, after that a circumferential incision is made. The depth of the mucosal incision must be enough to cut the mucosal muscle and expose the blue submucosa.
(5) Dissection: the major process is dissection. When the tumor around is fully incised, the submucosa is dissected by the incision knife beneath the lesion.
(6) Treatment of the wound: when the tumor is completely resected, preventive hemostatic treatment must be carried out. A hemostat and APC are used to treat the possible exudating sites. The greater exposed vessels should be occluded with the hemostat. Finally, the membrane-protective solution agent is sprayed to protect the ESD wounds and prevent hemorrhage. For patients whose local dissection is deep, with muscle layer cracks, the metal hemostatic clamps are necessary to suture the cracks.
(7) Specimen treatment: the sediment attached to the specimen’s surface must be gently removed with the gauze and placed on a sponge board for fixation. The surface of the membrane must be placed upward, the spread of the specimen must be consistent with the size of the tumor observed by endoscopy; the sample is then pinned to the board, and the oral and anal sides of the lesion are marked. Lugol’s iodine is applied to stain again in order to confirm whether the lesion is completely resected.

FIG. 8.1 — Operating steps of the resection of early esophageal carcinoma by peroral super minimally invasive surgery. A. The lesion refuses to stain after iodine staining. B. The marked lesion. C. Circumferentially incised membrane. D. Submucosal dissection. E. The wound after resection. F. Lesion specimen after resection.
(II) Hot-spot issues in the consensus and progress of the study
1. The effectiveness of ESD in the treatment of peripheral esophageal carcinoma has not been confirmed by prospective studies. Presently the Japanese esophageal ESD/EMR guideline (2020) suggests: “for cT1a-EP/LPM superficial esophageal squamous cell carcinoma involving the circumesophageal ring, the length ≤50 mm, ER therapy is recommended when preventive measures for stenosis are available. (Recommendation level weak. Modified Delphi score: moderate = 7, low = 3, high = 9; evidence strength: C).”
2. Prevention and treatment of esophageal stenosis after early esophageal carcinoma surgery is still the hot-spot issue of the study. Researches have shown that the prevention and treatment methods of ESD postoperative esophageal stenosis mainly include:
physical dilation: endoscopic esophageal dilation, external self-dilating balloon, esophageal stent, endoscopic radial incision;
glucocorticoids, including oral and local injections of glucocorticoids;
autogenous tissue grafting;
anti-inflammatory or antifibrotic drugs: mitomycin C, botulinum toxin type A, tranilast;
regenerative medicine: extracellular matrix scaffold, cell sheet transplantation;
other methods: polyglycolic acid, carboxymethyl cellulose, self-assembling peptides.
In addition, avoiding muscular injury during the operation may also reduce the incidence of esophageal stenosis. Although the current methods for the prevention and treatment of esophageal stenosis after ESD surgery are novel and numerous, most of them are still in the exploration and research stage, and there are still their own problems to be solved, such as the side effects of hormones, stent displacement, and clinical validation of regenerative medicine, etc. In the future, with the continuous exploration and improvement of various prevention and treatment methods, especially the rapid development of tissue engineering and regenerative medicine, it is believed that the problem of esophageal stenosis after endoscopic resection can be effectively solved.
8.1.2.2 Endoscopic Submucosal Dissection with Traction
Endoscopic submucosal dissection with traction is a technique for endoscopically determining the boundaries of the lesion, including marking, submucosal injection, full lifting of lesions, obtaining a better surgery field with the traction of the external force on the lesion, gradual dissection of the tissue between the muscle layer of the lesion’s mucosa and the muscularis propria using an electric knife until complete dissection of the lesion’s membrane and the submucosal layer and adduction of the lesion, and finally the treatment of the wounds. A meta-analysis has shown that the R0 resection rate of ESD with traction is similar to that of routine ESD, but that its operation time is shorter, and that both the complication and perforation rates are lower than for routine ESD.
According to the ESGE technical review of ESD, the use of traction methods in esophageal ESD is recommended. The traction technique in ESD can be divided into two categories that require or do not require additional devices, which can be selected depending on the specific situation. Traction provided by the distal cap attachment should always be considered in ESD because of its advantages in exposing the submucosa. Traction techniques that do not require assistance from a device are mainly relying on tissue gravity, tunnels and bridges, as well as on the tension provided by the pouches. Common device-assisted external traction techniques include clamps and wires, clamps and snares, external pliers, double-cavity endoscopes, etc. The commonly used device-assisted internal traction techniques include double-clamp traction, internal traction line, magnet-assisted traction, etc.
(I) Surgical operation and skill (figure 8.2)
1. Clip-floss traction. After dissection of the oral-side end part of the early esophageal carcinoma, tissue clips and dental floss should be attached to the mucous layer separated from the oral-side end, then external traction should be applied to separate the mucous layer from the muscularis layer.

FIG. 8.2 — SMIS operative steps of early esophageal carcinoma: dental floss/tissue clips with traction, endoscopic submucosa dissection. A. Early esophageal carcinoma after iodine staining. B. Circumferentially incised lesions. C. Tissue clips/dental floss are applied for traction at oral-side end. D. After traction, submucosal layer must be fully exposed, whereas submucosal dissection should be continued. E. Postoperative wound. F. Tissue specimen.
2. ‘8’-loop traction (figure 8.3). After partial dissection of the oral-side end of the lesion, eight-loop/tissue clips should be attached to mucous layer at the oral-side end, the other end being attached to the normal membrane at the opposite side (anal side) of the lesion, by applying tissue clips to obtain the traction effect.

FIG. 8.3 — Operating steps of the SMIS endoscopic submucosal dissection of an early esophageal carcinoma using a ‘8’-loop and traction. A. Early esophageal carcinoma after iodine staining. B. Circumferentially incised lesions. C. After submucosal dissection of the oral-side end, the lesion mucosa contractures, and dissection is difficult. D. ‘8’-loop/tissue clips for traction. E. The mucous layer is clearly separated from the muscle layer after traction. F. Postoperative wound.
(II) A classic case
History: the patient, a middle-aged woman, was admitted to the hospital mainly due to the “discovery of high-grade intraepithelial neoplasia of the esophageal mucosa for more than one month.” Gastroscopy showed that, 29–31 cm away from the incisor, the left posterior wall of the mucous membrane was rough, slightly raised, with a brittle texture. Pathological examinations revealed high-grade intraepithelial neoplasia of the esophageal squamous epithelium. In the past, she has suffered from type-1 diabetes, she has been treated with iodine-131 for hyperthyroidism and has a hypertension history, and she is allergic to penicillin and streptomycin.
Diagnosis: High-grade intraepithelial neoplasia of the esophageal mucosa.
After an adequate preoperative evaluation, an improvement in nutritional status, and the signing of informed consent for surgery, the patient underwent a peroral super minimally invasive resection of an early esophageal carcinoma (Refer to video 8.1, Peroral super minimally invasive resection for early esophageal carcinoma (See online resources)).
Surgical process: a 0-IIb lesion can be seen in the esophagus at 30–32 cm from the incisor, the lesion area refused to stain after iodine staining; there is a pink sign (+) on the oral side. APC (0.6 l/min, 30 W) circumferentially marked the extent of the lesion, which was deiodinated with a VitC solution. The lesion mucosa was circumferentially incised with a DualKnife, the lesion was gradually dissected with floss-assisted traction with a triangle knife and an IT knife, and the wound was treated with a hemostat after complete dissection. No obvious active bleeding was found. The wound was at 30–35 cm from the incisor, covering a circle of about 5/6 of the lumen.
(III) Hot-spot issues in the consensus and progress of the study
The various ESD techniques with traction have advantages and disadvantages: Current traction-assist methodologies—including positional adjustment techniques, gravitational traction systems, and suture-anchored clip methods—provide relatively straightforward implementation for routine procedures but demonstrate constrained capabilities in both traction force modulation and directional control precision during endoscopic interventions. The titanium-clipping method with wire is simpler, and applies to simple surgical operations, but the traction force and directions provided are limited; snare traction and internal traction methods are efficient, but a special traction device is needed; the external traction method and the bi-mirror method are direct and efficient, but are greatly affected by space restriction; the magnetic anchoring method can overcome the space limitation for traction, but the operation mode needs further study. In addition, there are some other methods that cannot be widely used due to their high cost and significant risk of wounds.
8.1.2.3 Cap-Assisted Endoscopic Mucosal Resection
Cap-assisted endoscopic mucosal resection (EMR-C) is an endoscopic technique for determining the boundaries of lesions, including marking, submucosal injection, full lifting of the lesion, placement of a snare in the cap, resection of the lesion after having absorbed it in the cap and adducted the lesion, and finally treatment of the wounds. This method does not require special instruments, is simple and easy to do, with high success rate. Small flat lesions can be cleaned at one time, and it can be used to resect lesions of the mucous membrane with a diameter of less than 1 cm. But the resection depth is not easy to master, the muscle layer can be easily damaged, with the risk of causing hemorrhage and perforation, which makes the method unsuitable for larger lesions.
(I) Surgical operation and skills
1. Anesthesia and position. All patients undergo general anesthesia with tracheal intubation, lying on their left side; attention should be paid to keep them warm, adjusting the room temperature and the infusion temperature, and avoiding pressure sores.
2. Operating steps. Prepare according to routine endoscopy. Firstly, comprehensive observe the esophageal condition, and then conduct iodine staining to specify the lesion distance and site. Electrocoagulation marking outside 1–2 mm around the determined lesion site and submucosally inject an adrenaline saline solution at 1–2 cm of the lesion distal/proximal end, to make sure that the submucosal tissue of the lesion is sufficiently separated from the muscularis propria.
(1) The extent and depth of the lesion must be determined. First, a routine endoscopy should be performed to determine the foci’s site, size and shape, which should be combined with staining and magnifying endoscopy to determine the foci’s extent, nature and infiltration depth.
(2) Marking: electrocoagulation marking should be directly conducted by using argon plasma coagulation (APC) and an electric knife. Lesions that lack clear boundaries should be first stained with indigo rouge; after determining the lesion’s extent under NBI observation, electrocoagulation marking at 2 mm outside of the lesion should be made, the interval for every marking spot being about 2 mm.
(3) Submucosal injection: the quantity of liquid to be injected each time is 2–5 ml; the focus must be lifted and separated from the muscle layer, which is helpful for the complete resection of the lesion without injuring the muscularis propria and for reducing the occurrence of complications as perforation and hemorrhage, etc.
(4) Lesion resection: the endoscope is reinserted after the cap has been installed. The snare is placed in the cap and absorbs the lesion tissue into the cap by negative pressure. The snare must be tightened and the appliance can be turned on to resect the membrane. The resected membrane tissues are absorbed into the cap and removed using the endoscope.
(5) Treatment of the wound: after checking that the lesion has been completed resected, the endoscope is reintroduced to observe if the wound is bleeding and is perforated, etc. It is necessary to check whether there are any residues. If there is a significant amount of residues, the membrane must be resected again or the submucosa must be dissected again following the steps above. If there are only small remains, APC therapy can be used.
(II) Hot-spot issues in the consensus and progress of the study
In recent years, there have been few studies on cap-assisted endoscopic mucosal resection for the treatment of esophageal mucosal lesions, but there are many reports on its application to gastric and intestinal mucosal lesions. In a meta-analysis of four endoscopic treatments for early esophageal carcinoma, the overall resection rates were 99.5% for ESD, 26.5% for EMR-C, and 24.1% for MBM, and the local recurrence rates were 95.6% for EMR-C, 42.9% for MBM, and 11.6% for ESD. EMR-C has no advantage. The study conducted by Lu Cuiqin et al. demonstrated that cap-assisted endoscopic full-thickness resection (Cap-EFTR) and endoscopic submucosal dissection (ESD) showed no statistically significant differences in en bloc resection rates and R0 resection rates when treating gastric submucosal tumors with diameters <10 mm. Cap-EFTR is more efficient, less use of consumables and less expensive, and shortens the postoperative recovery time of patients with small gastric submucosal tumors. It has a good control effect on inflammation and fewer complications, and is a safe and effective surgical method. EMR-C therapy is more advantageous than ESD therapy for gastric neuroendocrine tumors of type I that are small in diameter, limited to mucosa and submucosa, and without lymphatic metastasis or distant metastasis. Compared to submucosal injection alone, the combination of transparent cap-assisted EMR (EMR-C) significantly improves the resection effect of large intestine stemless polyps, reduces the risk of postoperative hemorrhage, improves the patient’s prognosis, shortens the length of hospital stay, and reduces the patient’s burden.
The endoscopic cap has become a favored key accessory in various surgeries. Generally speaking, its use may reduce surgical time, improve visualization, and reduce surgical complications; in short, it maximizes the super minimally invasive and rapid potential of endoscopy. Compared to traditional endoscopy, connecting the cap to the endoscope for examination has multiple advantages. It is especially useful in the following respects: it minimizes blind spots during screening colonoscopy, it provides a constant distance from the lesion for clear viewing during magnifying endoscopy, it accurately assesses the size of various gastrointestinal lesions, it prevents mucosal damage during the removal of foreign bodies, it ensures adequate space in the submucosal space during endoscopic submucosal dissection (ESD) or third-space endoscopy, it provides the best approach angle to the target, as well as suction of the mucosa and submucosal tissue with negative pressure for resection. It can thus be speculated that cap-assisted endoscopic mucosal resection will also have broad application prospects in terms of the resection of esophageal mucosal lesions, but the indications, scope, types and advantages of this method need further study.
8.1.3 Non-Full-Thickness Resection of Early Esophageal Carcinoma by Per-Tunnel Super Minimally Invasive Surgery
Per-tunnel super minimally invasive surgery by non-full-thickness resection of early esophageal carcinoma can achieve accurate resection of the mucosal layer, the mucosal muscular layer and the submucosa of the esophageal carcinoma focus by methods like endoscopic submucosal tunnel dissection (ESTD), endoscopic submucosal dissection with traction, etc.
(I) Indications and contraindications
1. Indications
(1) Generally applies to lesion areas greater than 1/2 week of esophagus and is consistent with early esophageal carcinoma and precancerous lesions;
(2) The indication of endoscopic resection is the same as that of ESD.
(3) ESTD includes single-tunnel ESTD and multi-tunnel ESTD. In order to facilitate and expedite the establishment of the submucosal tunnel, a tunnel width is generally about 2 cm, so single-tunnel ESTD is recommended for lesions less than 1/2 week of esophagus, and double-tunnel ESTD is recommended for lesions greater than 1/2 week.
2. Contraindications
(1) When the lesions found by EUS deeper than the submucosa.
(2) With lymphatic or distant metastasis detected by EUS or other imaging examinations;
(3) In case of advanced or undifferentiated cancer.
(4) With coagulation function, cardiopulmonary dysfunction or other endoscopic or anesthesia contraindications.
(II) Postoperative nursing
Symptomatic treatments like postoperative fasting for food and water for 72 h, routine acid suppression, anti-infective prophylaxis must be carried out; later the patient will gradually switch to a liquid, then semiliquid, then full diet; postoperative treatment for one month with PPIs and gastric mucosal protective agents will be prescribed.
I. Endoscopic submucosal tunnel dissection
Endoscopic submucosal tunnel dissection (ESTD) is an endoscopic technique for determining lesion boundaries, marking the lesion’s oral and anal sides, incising the lesion’s oral-side and anal-side membranes after submucosal injection, entering the submucosal to dissect, and establishing a tunnel between muscularis mucosa and muscularis propria, and, in the tunnel, dissecting the entire lesion on both sides and adduct the lesion, and then treating the wounds. Studies have shown that compared to ESD, ETSD reduces muscular injury and improves the healing of artificial mucosal injuries in less time for lesions larger than 1/2 esophageal circumference. Therefore, when the lesion exceeds half the circumference, the tunnel method is recommended for esophageal ESD.
(I) Surgical operation and skills
1. Anesthesia and position. All patients undergo general anesthesia with tracheal intubation, lying on their left side; attention should be paid to keep them warm, adjusting the room temperature and the infusion temperature, and avoiding pressure sores.
2. Operating steps
(1) The extent and depth of the lesion must be determined (figure 8.4). First, a routine endoscopy should be performed to determine the foci’s site, size and shape, which should be combined with staining and magnifying endoscopy to determine the foci’s extent, nature and infiltration depth.
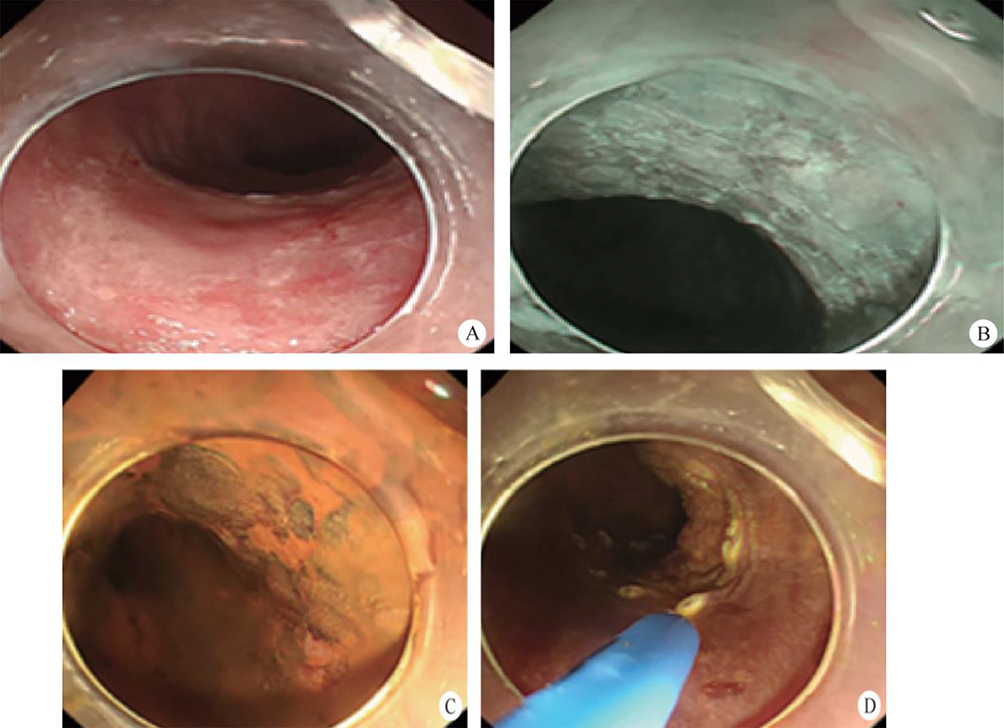

FIG. 8.4 — Super minimally invasive resection of early esophageal carcinoma through a tunnel channel to determine the lesion scope. A. Esophageal flat lesion seen in white light; B. NBI to observe the lesion. C. Dye-repellent zone found after iodine staining; D. Circumferential marking.
(2) Marking: marking should be made at 2 mm from the lesion edge using argon or an electric knife, with an average distance of 2–5 mm. If the lesion is of a diameter close to that of the lumen, it is necessary to mark the oral side and the anal side separately.
(3) Submucosal injection: the quantity of liquid to be injected each time is 2–5 ml; the focus must be lifted and separated from the muscle layer, which is helpful for the complete resection of the lesion by ESD without injuring the muscularis propria and for reducing the occurrence of complications as perforation and hemorrhage, etc.
(4) Anal and oral mucosal margins are incised: the submucosal injection of a mixture of fluids (including epinephrine, methylene blue solution and normal saline solution) must be made using an injection needle, the membrane must be lifted, and then the oral-side and anal-side membranes must be incised at the level of the normal membranes outside the anal marker and the submucous layer should be fully repaired.
(5) Establishment of a submucosal tunnel: the tunnel must be established from the oral side to the anal side. During this process, the cap is used, the dissection must be made downward by gradually using the triangle knife or the IT knife, until the connection with the incision of the anal-side margin. If submucosal vessels appear during the process of tunnel dissection, the electrocoagulation hemostat should be used for pre-coagulation, so as to prevent hemorrhage.
(6) Dissection in the tunnel (figure 8.5): the dissection must be made in the tunnel along the muscularis propria, pushing with the electric knife from the oral to the anal side, until reaching the anal incision.
(7) Side incision of the margin: incise from the tunnel’s oral side to the tunnel’s anal side outside the marks on both sides.
(8) Treatment of postoperative wounds (figure 8.6): hemostasis must be carried out on the wound vessels by using argon knife or electrocoagulation forceps. If the membrane has been dissected deeply or if there are injuries to the muscle layer, one or multiple titanium clips should be used for occlusion to prevent hemostasis. Fibrin glue can be sprayed if needed.
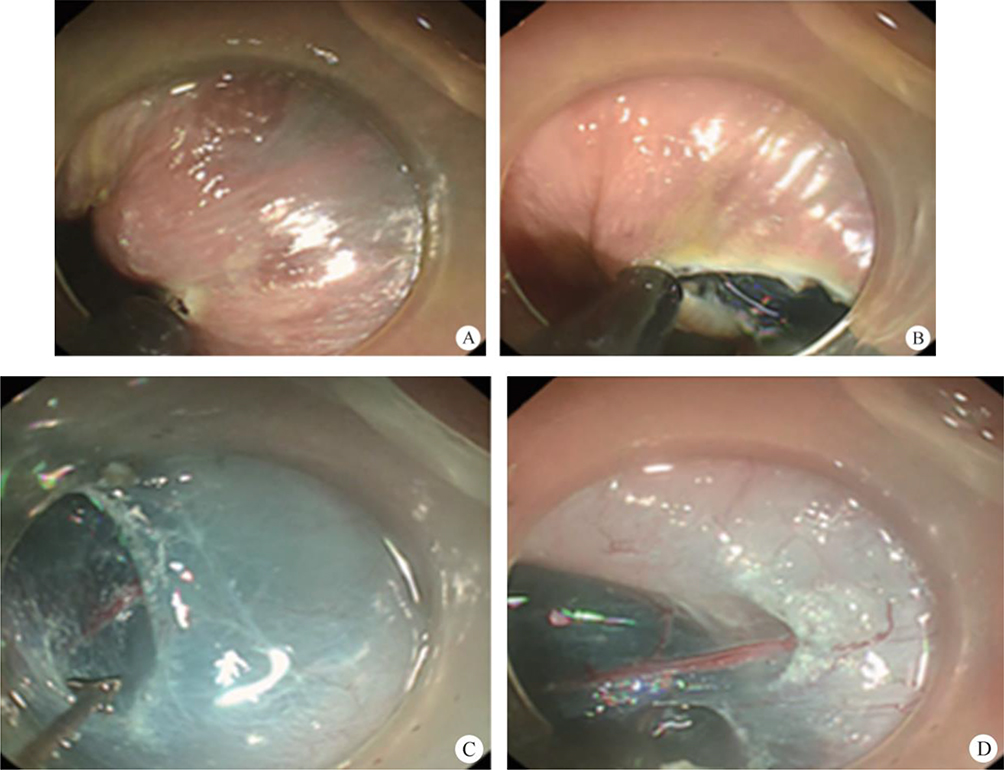
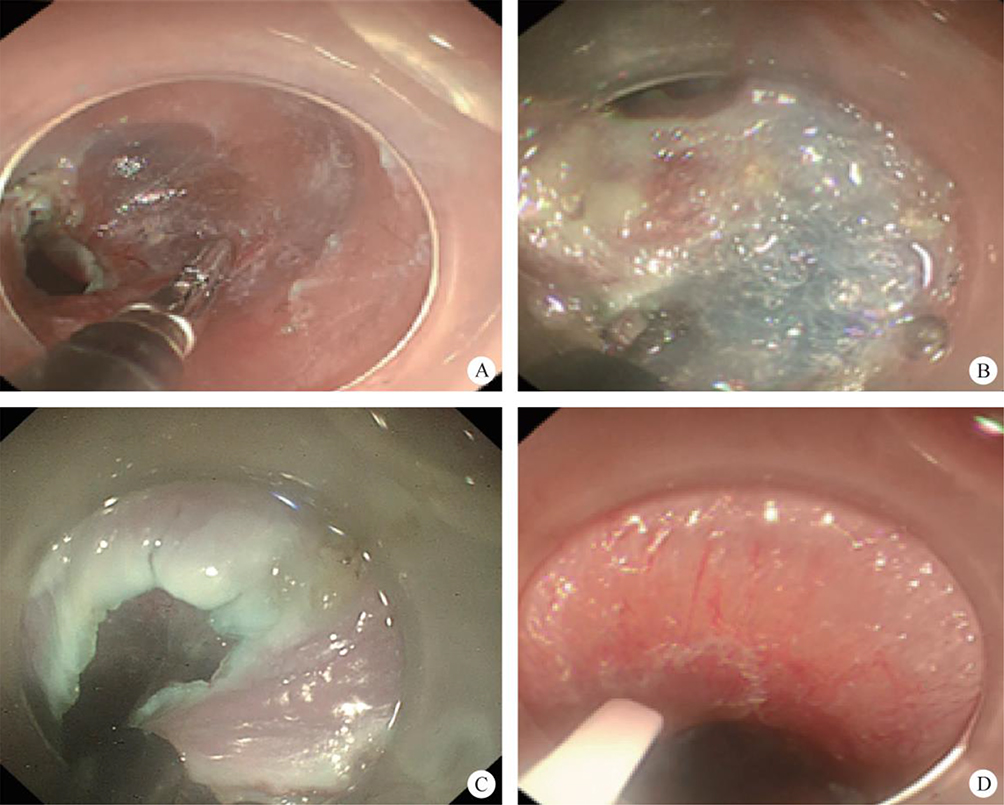

FIG. 8.5 — Steps of the dissection of esophageal early carcinoma in the tunnel by super minimally invasive resection through a tunnel channel. A. Lesion bulge after submucosal injection. B. Marginal incision. C. Submucosal dissection. D. Bare exposed blood vessels.

FIG. 8.6 — Resection of an early esophageal carcinoma lesion and wound treatment by super minimally invasive resection through a tunnel channel. A. Electric coagulation hemostasis of the exposed blood vessels. B. Complete submucosal dissection. C. Incised membranes on both sides of the tunnel. D. Tissue glue is sprayed on the wound to protect it.
(9) Double-tunnel ESTD operating method: it is basically similar to the single-tunnel ESTD operating method. (figure 8.7). After incising the oral and anal margins, create two tunnels approximately 1.5 cm wide. Apply the cap and establish two tunnels to dissect the lesion using the same method (figure 8.8).
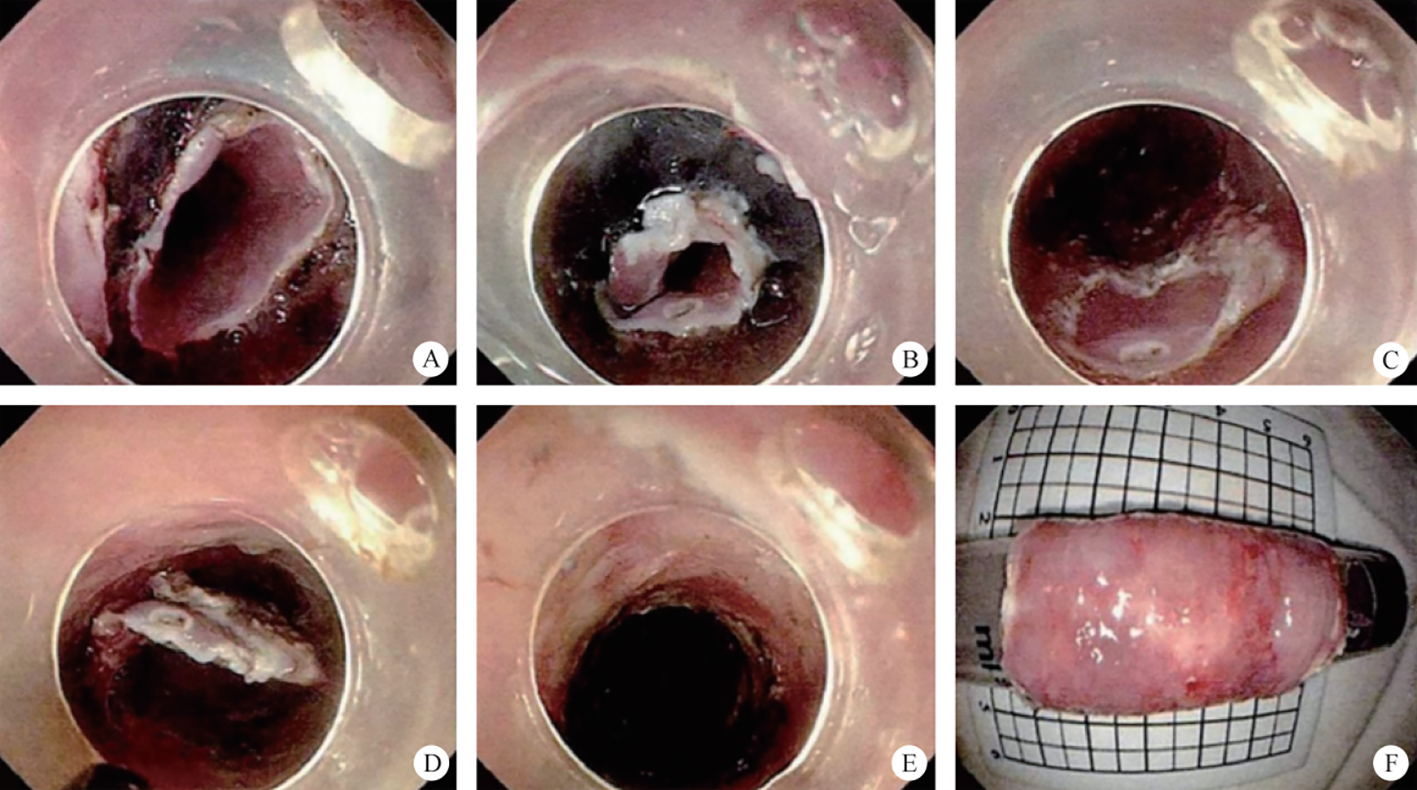
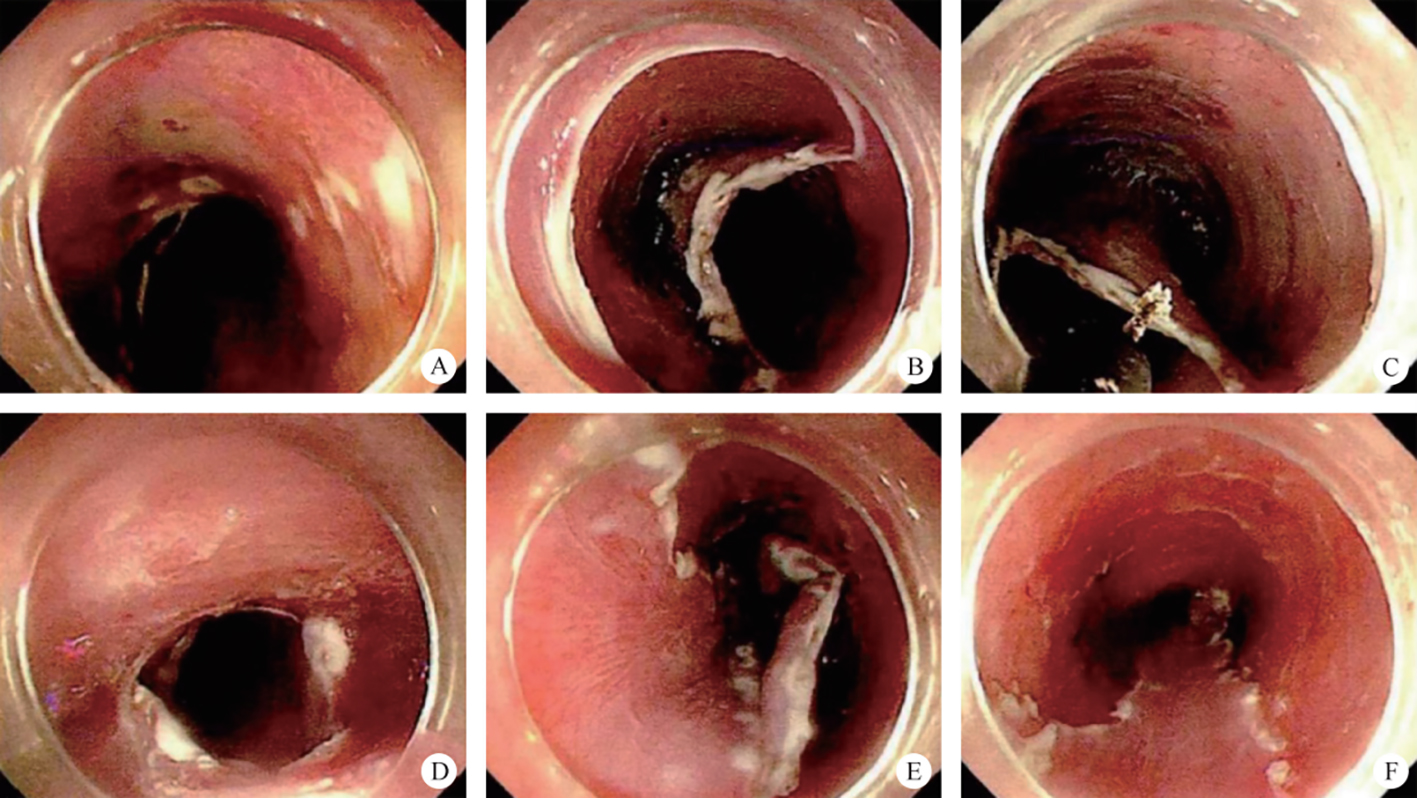

FIG. 8.7 — Operating steps of single-tunnel ESTD surgery therapy. A. Mark the lesion’s extent and incise the anal-side membrane. B. Incise the oral-side membrane. C. Tunnel established. D. Dissection to the anal-side margin. E. Incise the edge of the side. F. After lesion dissection.

FIG. 8.8 — Operating steps of double-tunnel ESTD surgery therapy. A. Incision on the anal side. B. Incision on the oral side. C. First tunnel. D. Second tunnel; E. After dissection of the lesion. F. Resected specimen.
(II) A classic case
History: patient, a middle-aged male, was admitted to the hospital mainly due to the discovery of a “high-grade intraepithelial tumor of the esophagus for more than one month.” Gastroscopy showed that the mucosa of the posterior wall of the esophagus was patchy, rough and erosive at 1–6 o’clock at 30 cm from the incisor teeth. Staining with Lugol’s iodine showed that some mucous membranes were resistant to staining. The pathological results showed that the local epithelium had moderate to severe atypical hyperplasia, without chest pain or pain radiating to other sites. Past history: hypertensive disease, used to performed inguinal hernia repair. No abnormalities were found during the physical examination for admission.
Diagnosis: high-grade intraepithelial neoplasia of the esophagus.
After full preoperative evaluation, improving nutrition condition and signing the surgery informed consent form, the early esophageal carcinoma per-single-tunnel channel SMIS resection was performed on the patient (Refer to video 8.2, Early esophageal carcinoma resection by super minimally invasive surgery through a tunnel channel (See online resources)).
Surgical process: IIb + IIC lesion was found in the esophagus 29–33 cm from the incisor, with a rough and corrosive surface and clear boundaries; the lesion area was unstained after iodine staining. Argon was used to mark the lesion’s margin, and a mixture of methylene blue, epinephrine and saline solution was injected submucosally, after incising the lesion’s anal-side and oral-side membranes successively using a triangle knife, the mixture must be injected on one side of the submucosa on the oral side, while dissecting along the submucosa, and a tunnel established up to the anal side of the lesion; the lesion has been completely dissected when both sides of the tunnel have been incised with the triangle knife, IT knife. Once the operation has been completed, the wounds are treated with the hemostat and fibrin glue is sprayed on the vessels that remain visible.
(III) Hot-spot issues in the consensus and progress of the study
It is difficult to perform ESTD surgery for large-circumference esophageal superficial carcinoma: superficial esophageal carcinomas with large axial length are widespread, and there is not enough operating space for the endoscope, which results in unclear surgical field, risk of injuring vessels and muscle layers, and prolonged operating time. Risks of complications like anesthesia accident, perforation, mediastinal infection, etc., are increased. Finally it is necessary to dissect piece by piece or to resect with a snare, which increases the difficulty of the pathological evaluation. In addition, since the resection cannot be complete, additional surgical treatment is required. Studies have shown that modified multi-tunnel endoscopic submucosal dissection is safe and effective in the treatment of peripheral superficial carcinoma with larger length, which effectively reduces muscle injury and damage to the pathological mucosa, and significantly shortens the operation time.
8.1.4 Esophageal Skin Grafting by Digestive Endoscopic Super Minimally Invasive Surgery
Digestive endoscopic super minimally invasive esophageal skin grafting surgery is a technique that involves transforming the patient’s autologous skin into a skin graft around the esophageal fully coated stent, and then placing the esophageal stent by a peroral endoscopic approach, and fixing the skin graft onto the esophageal wound in order to prevent postoperative stenosis of early circumferential esophageal carcinoma. This technology, developed by professor Linghu Enqiang’s team, is the world’s first super minimally invasive method for preventing esophageal stenosis.
The selection of the donor area for skin graft transplantation should minimize the discomfort and appearance concerns of the patient, while achieving the best outcome for the recipient area. The selection of the donor area depends on the desired skin color, texture and thickness of the recipient area. The specific conditions of the donor tissue are also a determining factor. The site of skin graft extraction is generally the lateral side of the patient’s right thigh. Extraction of a too thick piece of skin can lead to delayed wound healing at the donor site, as well as pathological pigmentation and scarring. The quality of the donor skin should also be assessed to specify whether there is past sunburn (e.g., keratosis) and scarring. Adequate preparation of the site is required for skin grafting. The wound bed must be well supplied with blood, without necrotic and ischemic tissue, and without inflammation or infection. After ESTD endoscopic tunnel resection of early esophageal carcinoma, the basal surface of the surgical wound should be left as much as possible with good blood supply to reduce the burning of the wound blood vessels caused by hemostasis, so as to improve the survival of the skin graft.
(I) Indications and contraindications
1. Indications. Circumferential lesions meeting the endoscopic resection criteria for early-stage esophageal carcinoma and precancerous lesions (refer to the previous section for indications), with absence of autoimmune diseases and preexisting extensive scarring.
2. Contraindications The same as for the therapy of early esophageal carcinoma by super minimally invasive ETSD surgery.
(II) Preoperative preparation
(1) Master the indications and contraindications of painless endoscopy, ask about the patient’s age, whether he has heart or lung diseases and hypertension history, allergies to drugs, conduct routine examinations or the necessary preoperative examinations: blood, coagulation function, blood biochemistry, ECG, chest film, etc.
(2) Conduct active and effective communication with the patient and family, fully evaluate the patient’s condition and psychological state; for patients with negative moods like anxiety, depression, worry, psychological support must be provided, improve the communication with the patient, try to alleviate his/her negative mood and establish confidence.
(3) Advise the patient to eat mild and easy-to-digest food one night before, fasting for food and water for 12 h before surgery.
(III) Surgical operation and skills
1. Anesthesia and position. All patients undergo general anesthesia with tracheal intubation, lying on their left side; attention should be paid to keep them warm, adjusting the room temperature and the infusion temperature, and avoiding pressure sores.
2. Operating steps (figure 8.9)
(1) For esophageal circumferential early carcinoma, double-tunnel ESTD is usually used, which mainly includes the following four steps: firstly, circumferential marking of the lesion’s anal-side and oral-side membranes; secondly anal-side and oral-side opening, successively; then submucosal dissection from the lesion’s oral side to its anal side; creation of two submucosal tunnels; finally, incision of the side edge. After complete resection of the lesion, prophylactic hemostasis must be performed on the wound.
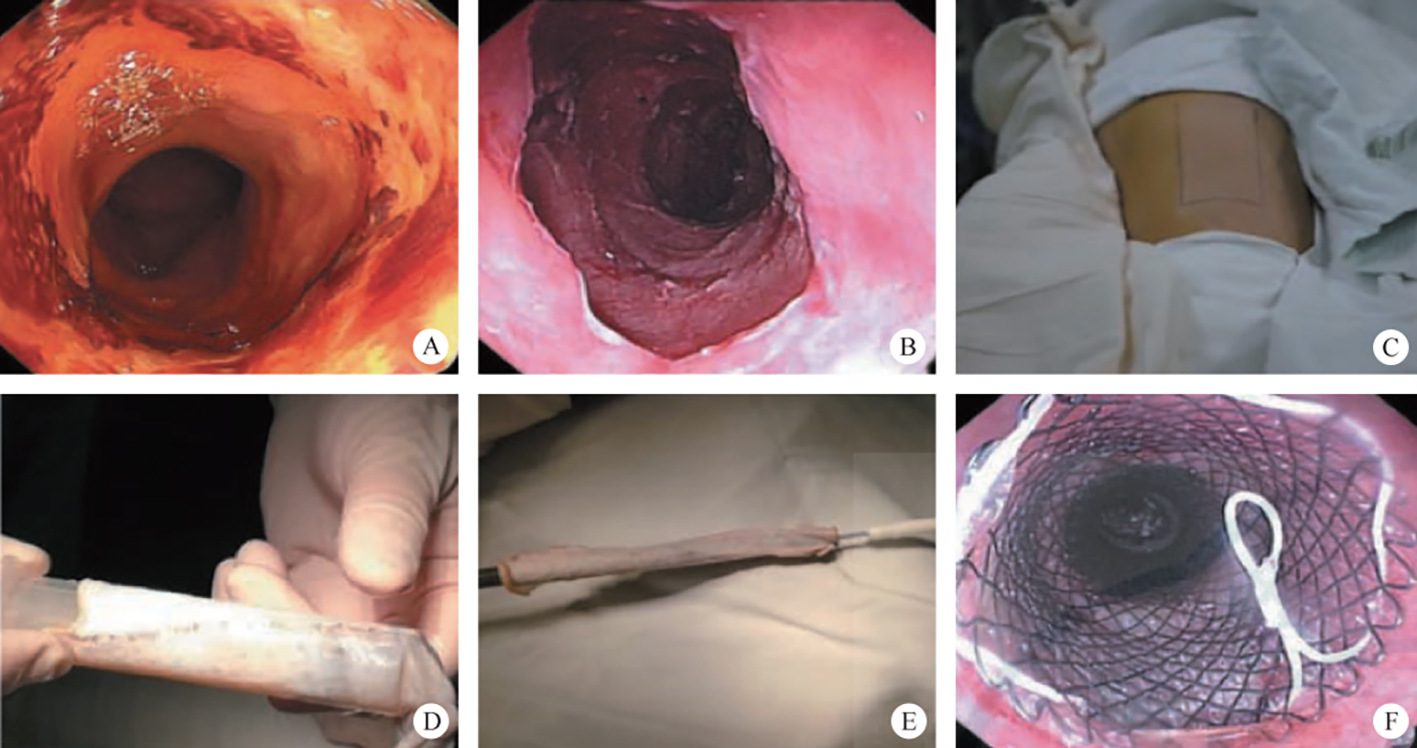

FIG. 8.9 — Schematic diagram of esophageal skin grafting by digestive endoscopic super minimally invasive surgery. A. Endoscopic staining with Lugol’s iodine showed staining rejection of the circumferential esophageal mucosa. B. Esophageal wound after circumferential ESTD surgery. C. The epidermal skin graft should be taken on the external side of the patient’s right thigh. D. The graft is sewn into a cuff shape with absorbable thread. E. Suture of the cuff skin graft onto the esophageal covered stent. F. The transplanted skin graft must totally cover the esophageal wound.
Refer to video 8.3, Digestive endoscopic super minimally invasive esophageal skin grafting surgery-1 (See online resources).
(2) During performing esophageal circumferential ESTD, the plastic surgeon takes the skin, and the size of the transplanted skin graft depends on that of the esophageal ESTD postoperative wound. First, the external side of the epidermal skin graft is taken from the patient’s right thigh with a roller graft knife; absorbable threads must be used to suture the transplanted skin graft to cuff-shaped, and then the cuff-like skin graft is sutured onto the esophageal covered stent. Using a stent-releasing system, the stent must be gradually released under endoscopic observation, making the transplanted skin graft to totally cover the esophageal wound. To prevent the esophageal stent from moving, it must be fixed it onto the esophageal wall with metal clips. Finally, a three-cave-nutrient-canal is placed endoscopically and a pressure bandage is applied to wounds on the thigh.
(3) Application of stents by injection: the placement method is the same as with the routine stent, i.e. under continuous hormone treatment by injection through the drug-injection hole to alleviate stenosis.
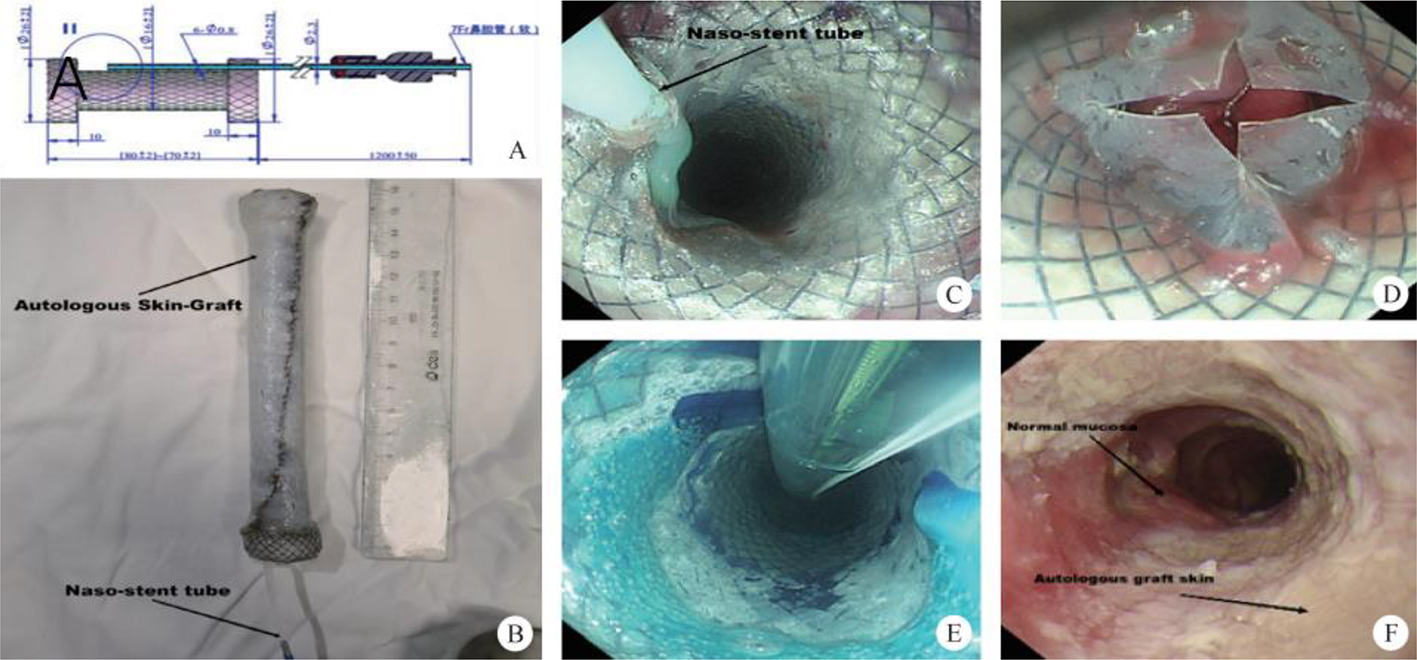

FIG. 8.10 — Structure and application of a drug-eluting stent. A. Design of the drug-eluting stent. B. Stent covered with a skin graft. C. The stent is placed into the wound after esophageal circumferential resection. D. Anti-reflux valve of the drug-eluting stent. E. Injection of the mixed solution of hormones and methylene blue into the storage capsule of the drug-eluting stent. F. Re-checking the state of growth of the esophageal skin graft after the operation.
Refer to video 8.4, Digestive endoscopic super minimally invasive esophageal skin grafting surgery-2 (See online resources).
(IV) Postoperative nursing
After the operation, the patient should fast for food and water for one week, enteral nutrition through jejunum nutrition tube should be given, switching gradually to soft food. Intravenous PPI injections must be administered for at least one week, followed by oral intake for at least eight weeks. Routine postoperative prophylactic antibiotics must be administered for three days. The stent can be removed four weeks after the operation. Before taking it out, recheck by endoscopy once every week to observe whether the skin graft is growing, and whether there are complications like hemorrhage, perforation, infection, etc. For thigh wounds, if there is obvious exudate after the operation, the dressing must be changed timely. If there is no obvious exudate, the dressing must be changed one week after the operation. The stitches can be removed after two weeks.
(V) Hot-spot issues in the consensus and progress of the study
1. Large samples and related controlled studies are still needed to verify the prevention of postoperative esophageal endoscopic stenosis with autologous tissue. At present, there are no standard guidelines for the prevention of stenosis after endoscopic resection of esophageal circumferential lesions. In recent years, tissue engineering methods have been used to prevent stenosis after endoscopic esophagectomy. Some studies have reported that the transplantation of oral epithelium and gastric antrum mucosa has certain effects. The skin tissue of the thigh has good compatibility, and is a laminated flat epithelium, which is more tolerant to a variety of physical and chemical stimuli. Currently, studies on the prevention of postoperative esophageal endoscopic stenosis on autologous tissues are based on small-size samples, and further studies involving large samples as well as related control studies are needed.
2. How to ensure the survival rate of skin graft remains to be studied. Chai et al. performed endoscopic submucosal tunnel dissection (ESTD) on eight postoperative patients. After taking the patients’ moderately thick autologous skin of the lateral thigh, they sutured the skin graft as “cuff-like” with absorbable threads looped on the external side of the metal stent, and finally placed the stent at the site of the injury to the esophageal mucosa. To prevent the stent from moving, the stent was fixed on the esophagus with metal clips; five patients did not eventually develop stenosis at a median follow-up of seven months, three patients had stenosis 12, 13 and 35 days after the stent was removed. Zou et al. conducted a case-matched control study with patients who were prevented from esophageal stenosis using only metal stents. In the experimental group, 19 patients underwent autologous skin transplantation to prevent esophageal stenosis after resection of esophageal lesions. Compared to the control group, the occurrence of esophageal stenosis in the experimental group was obviously lowered (36.8% vs. 78.9%). Autologous skin transplantation significantly reduces the incidence of esophageal stenosis after resection of circumferential esophageal lesions. But how to ensure the survival rate of the skin graft is key to obtain good effects, and larger-scale clinical studies are still needed.
3. A comparative study with regenerative medicine technology and autologous transplantation of esophageal mucosa is lacking. The early re-epithelialization of EMR sites induced by autologous buccal keratinocytes conducted by Sakurai et al. suggests that the implantation of keratinocytes may reduce the risk of stenosis after resection of circumferential lesions. Polyglycolic acid (PGA) combined with autologous esophageal mucosa (AEM) and temporary stent implantation (TSI), PGA combined with AEM, and the combined self-controlled dilation balloon (SDB) effectively prevents ESD postoperative stenosis and significantly reduces the number of endoscopic dilations of ESD postoperative esophageal stenosis. Currently, there is no comparative study between the results of autologous skin transplantation and the methods reported above.
8.2 Super Minimally Invasive Surgery of Esophageal Subepithelial Lesions
8.2.1 General Introduction
(I) Introduction to esophageal subepithelial lesions
Esophageal subepithelial lesions are lesions of the complete mucosal epithelium, which originate from uplifted lesions in the esophageal wall below the epithelium, and can be located in the mucosal myometria, the submucosa, and the muscularis propria. Submucosal tumor is also called subepithelial lesion (SEL) abroad. SELs can appear at any site of digestive tract, and the occurrence rate at each site is different. The stomach is the site with the most frequent occurrence of SELs, followed by the esophagus. Also, analysis of SEL pathological results shows that it is most often benign, with very few malignant cases. In general, when the tumor’s width is less than 2 cm, there are no clinical manifestations. But this also depends on the site where the tumor is growing, and it is discovered in routine gastrointestinal endoscopy examinations in most cases. There are occasionally some sites with clinical symptoms where the tumor is in progression and its size becomes larger, with, for example, abdominal pain, hemorrhage, obstruction. With the popularization of the endoscopic and EUS examination techniques, the detection rate of SELs increases year by year. The types of SELs are varied, and there is a certain correlation between the location of the SEL and its histopathology. Most submucosal tumors of the esophagus are leiomyomas, accounting for 60%–80% of the SELs in the esophagus, and most frequently occur in the middle and lower sections of the esophagus.
(II) Diagnosis of esophageal subepithelial lesions
In routine endoscopy, the routine white light endoscope evaluates the lesion’s size, location, activeness, the color and shape of the mucosa in the raised part, whether there is erosion or hemorrhage. But the related data (nature of the lesion and its sources) cannot be provided. So we need to combine with endoscopy with EUS and on that basis observe the lesion’s internal echoes, origins, heterogeneity, growing mode and the relation with blood vessels, which makes it possible to judge the benignity or malignancy of the lesion.
It is recommended that all patients with SLE undergo routine white-light endoscopy combined with EUS as a preoperative examination on a voluntary basis. For all patients with subepithelial lesions (SELs), conventional white-light endoscopy combined with endoscopic ultrasound (EUS) is strongly recommended as part of their preoperative evaluation, provided informed consent is obtained. This multimodal approach serves as the cornerstone for definitive lesion characterization ad spatial mapping of submucosal pathologies. When suspecting a malignant tumor or if the larger diameter of the tumor exceeds 2 cm, it is recommended to perform CT and MRI examinations, which will provide important information for tumor grading, treatment and prognosis evaluation in clinical practice.
When the tumor can be identified as a benign SEL by routine white-light endoscopy combined with EUS, a biopsy is not relevant. Only when we suspect a malignant or cannot determine whether it is benign or malignant by white-light endoscopy combined with EUS, can we resort to active tissue sampling by EUS–FNA/FNB, MIAB, etc., to perform a pathological evaluation. However, the accuracy of the diagnosis of SEL after biopsy is limited, and the subsequent endoscopic resection is affected. For patients who meet the criteria for endoscopic surgery on the premise that the tumor can be completely resected, this can be done by experienced endoscopists in units with mature endoscopic treatment technology without preoperative pathological diagnosis. In summary, the diagnosis and treatment of SEL should be based on the histological type, location, size and symptoms of SEL as well as the patient’s condition.
(III) Implementation of super minimally invasive resection of esophageal subepithelial lesions
Esophageal subepithelial lesions can be cured by super minimally invasive surgery (SMIS), mostly performed endoscopically through the peroral natural cavity channel, per-tunnel channel and per multiple-cavity channel. This includes peroral super minimally invasive resection of esophageal subepithelial lesions, which is a local technique for complete resection. Per-tunnel super minimally invasive resection of esophageal subepithelial lesions is a technique in which, after inserting perorally a gastrointestinal endoscope, the esophageal submucosal tunnel is artificially created, and local complete resection of esophageal subepithelial lesions by tunnel channel can be performed. Per multi cavity super minimally invasive resection of esophageal subepithelial lesions is a technique combining the use of gastrointestinal endoscopy and thoracoscopy for local complete resection of esophageal subepithelial lesions by subcutaneous and per multiple-cavity channels.
The list of the techniques, with their past names, of super minimally invasive surgery for esophageal subepithelial lesions currently used is shown as table 8.2.
TAB. 8.2 — List of techniques, with their past names, applied to the treatment of esophageal subepithelial lesions by super minimally invasive surgery.
| No. | Super Minimally Invasive Surgery (SMIS) | Technique | Past Name |
| 1 | Peroral super minimally invasive resection of esophageal subepithelial lesions | Super minimally invasive non-full-thickness resection | Endoscopic submucosal mass excavation |
| Endoscopic submucosal dissection | |||
| Endoscopic submucosal dissection with traction | |||
| Cap-assisted endoscopic mucosal resection | |||
| 2 | Peroral super minimally invasive resection of esophageal subepithelial lesions | Super minimally invasive full-thickness resection | Endoscopic subepithelial mass full-thickness resection |
| 3 | Per-tunnel super minimally invasive resection of esophageal subepithelial lesions | Endoscopic submucosal tumor resection by tunnel | Per submucosal tunnel endoscopic tumor resection |
| Resection of esophageal subepithelial lesions with traction | |||
| 4 | Per-multi cavity super minimally invasive resection of esophageal subepithelial lesions | Per multiple-cavity channel super minimally invasive resection | Multiple endoscopic combined therapy |
8.2.2 Peroral Super Minimally Invasive Resection of Esophageal Subepithelial Lesions
There are many endoscopic methods for peroral super minimally invasive resection of esophageal subepithelial lesions, but before using a method, full account must be taken of the tumor’s size and depth, and of the condition of the adjacent peripheral organs. SMIS can be implemented after fully predicting the risks of adverse events of the operation like perforation, hemorrhage, mediastinal emphysema. Now take endoscopic submucosal excavation (ESE) as the main technique for a complete introduction. ESE is an extension of endoscopic submucosal dissection (ESD) in the treatment of SEL. It is usually the circumferentially resection of the membrane above the SEL around the tumor to fully expose it, and then to dissect gradually along the tumor’s edge to achieve complete resection. The rate of complete resection reaches 92% according to documented data.
(I) Indications and contraindications
1. Indications. SELs exhibiting intraluminal protrusion, as confirmed by preoperative imaging modalities such as EUS and CT, which is hard to resect by a snare.
2. Contraindications. Specific lesions with lymphatic or distant metastasis; SELs with partially specified lymphatic or distant metastasis; block biopsy is needed for obtaining pathological results, which can be regarded as a relative contraindication; patients identified after detailed preoperative evaluation as being in poor general health, who cannot tolerate endoscopic surgery.
(II) Preoperative preparation
Routine preoperative examinations must be completed, including coagulation function, CT, EUS and endoscopy, as well as the evaluation of the general physical conditions, which will allow to discard patients with contraindications related to anesthesia or endoscopy. For patients taking anti-coagulant drugs, anti-platelet drugs or other drugs affecting coagulation, a withdrawal period of five to seven days is needed before the operation. The time of preoperative continuous fasting depends on the location of the lesion: for the obstructed lesions of the upper digestive tract without outflow tract, fasting for food for 12 h and fasting for water for 4 h is necessary; for lesions of the obstructed upper digestive tract with outflow tract, the fasting time is longer depending on the time it takes to empty the stomach, and the patient must orally take a defoaming agent and an anti-slime agent 30 min before the operation.
(III) Surgical operation and skills
1. Anesthesia and position. Tracheal intubation and general anesthesia are mostly used. The common position for the patient during surgery is lying on his/her left side.
2. Surgical operating step (figure 8.11)
(1) The lesions should be marked with a Dual Knife.
(2) The mixture must be injected into the submucosa surrounding the lesion with an injection needle, the membranes must be incised at the foci’s margins along the markers by using a DualKnife or a triangle knife.
(3) The submucosal tissues and the fibers of the tumor pericapsular muscle must be gradually dissected with a DualKnife, an IT2 knife or a triangle knife, and the tumor must be dissected with a knife or a snare.
(4) After the lesion is resected, the wounds must be healed by electrocoagulation to prevent delayed hemorrhage. If the condition permits, the wounds must be closed with tissue clips.
(5) As membranes covering the SEL is also resected, the artificial ulceration cannot be closed with clips in many occasions. If possible, artificial ulcerations should be closed to reduce the possibility of perforation, infection and delayed hemorrhage. For lesions with deeper MP layers or thinner artificial ulcer walls, clips must be used to prevent perforation. Fibrin sealant is used to close the wound in some patients.
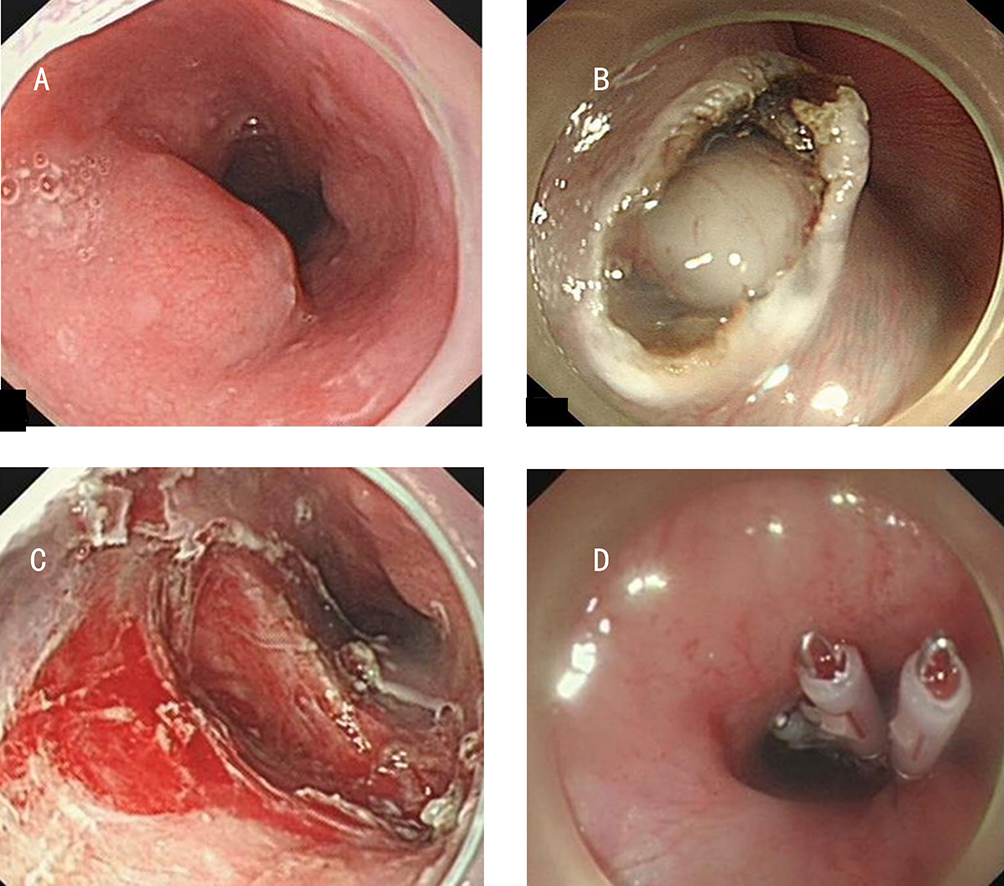

FIG. 8.11 — Operating steps of peroral super minimally invasive endoscopic resection of esophageal subepithelial lesions. A. Esophageal lesions found under white light. B. The membrane is incised to expose the lesion. C. The wound after the lesion is resected. D. Wound closed by tissue clips.
(IV) Postoperative nursing
(1) The patient should strictly lie in bed for rest for 24 h after the operation, avoid big amplitude activities. The occurrence of complications such as fever, palpitations, cold sweat, abdominal pain, black stool, infection, and hemorrhage, etc., must be monitored.
(2) Postoperative fasting for food for 48–72 h, and switching to light liquids (rice soup, noodle soup, milk, etc.), then gradual transition to a semi-liquid diet (soft noodle, porridge, etc.). The patient should be forbidden to eat rough and spicy foods, should avoid heavy manual labor within half a month; the fasting time should be extended for patients who are hemorrhaging and with large wounds; nutritional supplements and anti-inflammatory and acid suppression drugs should be given.
(3) To rid the patient of his/her concern, he/she should be explained that the metal clip used during the operation will fall off and be spontaneously eliminate with the healing of the wound. If the surgical wound forms an ulceration, it can be generally totally cured after one to two months, so the patient must be told to check-back regularly and undergo a follow-up endoscopy.
(4) Postoperative pathologies like high-risk tumors or local residual and recurrence risks require close monitoring or additional treatment if necessary.
(V) Hot-spot issues in the consensus and progress of the study
In contrast to STER, the endoscopic submucosal excavation (ESE) technique is performed by making a longitudinal or circular incision in the mucosa above the lesion and then dissecting the layer until the tumor is found. Once the lesion is completed and removed, the medical personnel will attempt to make closer the edges of the remaining mucosa with the metal endoscopic clamp. ESE is derived from endoscopic submucosal dissection, which is a more common resection technique but does not apply to lesions caused by deeper layers. Previous studies have shown that the ESE technique is safe and effective for the muscularis propria of both esophageal and gastric lesions. One of the previous studies reported that for SELs whose diameter is less than 10 mm (<10 mm), ESE STER was recommended rather than STER, whereas for SELs whose diameter is greater than 10 mm (>10 mm), STER was to be the first choice. Multiband mucosal resection (MBM), endoscopic submucosal dissection (ESD), or endoscopic submucosal excavation (ESE) all resulted in a lower four-year local recurrence rate.
A systematic review and meta-analysis found that STER and ESE resection of esophageal and gastric SELs from the muscularis propria had similar efficacy and safety. However, the duration of surgery is significantly shortened with ESE, and the total resection rate, overall resection rate, recurrence rate and total number of adverse events (including bleeding and perforation) are the same with both techniques. But a retrospective comparison concluded that both STER and ESE effectively treat esophageal SELs originating from the MP layer. STER might be better, because its operating speed is faster, with less complications and shorter length of stay. For the comparison of STER with ESE, more large-scale cohorts and random experiments to explore are still needed.
8.2.3 Per-Tunnel Super Minimally Invasive Resection of Esophageal Subepithelial Lesions
Per-tunnel super minimally invasive resection of esophageal subepithelial lesions is mostly used for the resection of the muscularis propria or of extramural tumors of the esophagus. The most representative endoscopic method is submucosal tunneling endoscopic resection (STER). STER technique is a new technique developed on the basis of peroral endoscopic myotomy (POEM), which is also an extension of the ESD technique. It is a therapeutic method in which, after endoscopically building a submucosal tunnel by STER, the block muscularis propria is resected in the tunnel—it is a branch of the endoscopic tunnel technique for treating lesions of the muscularis propria. The operating steps are: creation of the tunnel opening, creation of the submucosal tunnel, tumor resection, wound treatment and closure of the tunnel opening.
(I) Indications and contraindications of STER surgery
1. Indications Considering that the lesions to be resected will pass through several narrow places and the tunnel width is limited, it is recommended to select STER for lesions with a transverse diameter ≤2.5 cm.
Relative indication: 2.5 cm ≤ transverse diameter ≤ 3.5 cm.
2. Contraindications
(1) Patients with severe cardiopulmonary insufficiency cannot undergo endoscopic surgery.
(2) Obvious blood coagulation function disorder.
(3) There is large area of scar formation or matching in the tunnel area.
(4) Patients with suspicious malignant tumor.
Relative contraindication: (1) SELs with incomplete and ulcerated mucosal surfaces, which eliminates the importance of establishing the tunnel to maintain mucosal integrity; due to inflammation, the ulcer area has submucosal adhesions and it is difficult to create the submucosal tunnel; (2) the tumor is located at the entrance of the esophagus, with no space to form the submucosal tunnel; (3) the tumor’s transverse diameter > 3.5 cm, and cannot be totally resected in the tunnel.
(II) Surgical operation and skill
1. Anesthesia and position. the common positions are the left lying position, the supine position and supine position with right shoulder lifting up. In terms of anatomy, the esophagus is located at the back of trachea and heart, in front of the spine. It is relatively safe to establish a submucosal tunnel at the proximal end rear wall of the esophagus.
The appropriate locations can be helpful for the safety and the simplicity of the operation. The key principle of DETT is to divide the walls of the digestive tract into two layers (mucous membrane and MP) and maintain the integrity of one layer while opening the other layer for treatment or diagnosis. Therefore, the intraluminal and extraluminal spaces are isolated, and intraluminal gas or fluid cannot enter the extraluminal space.
2. Operating steps
(1) Establishment of the tunnel opening (figure 8.12): the gastroscope the inserted up to the target site, and the tunnel entrance is established 5 cm away from the lesion site. A submucosal injection of 6–8 ml of a mixture of methylene blue, normal saline solution and adrenaline is administered, then an inverted T-shaped incision is made to create the tunnel entrance. Inverted T incision: such inverted T-shaped incision was combined with a horizontal incision of 0.8 cm and a vertical incision of 1.0 cm. This tunnel incision was similar to the inverted ‘T’ incision. Thanks to the tunnel’s large space, it is very convenient for the endoscope to enter and exit and for the incision’s postoperative closure, as well as for external gas evacuation and drainage, thus reducing the occurrence of gas-related complications resulting from tunneling.
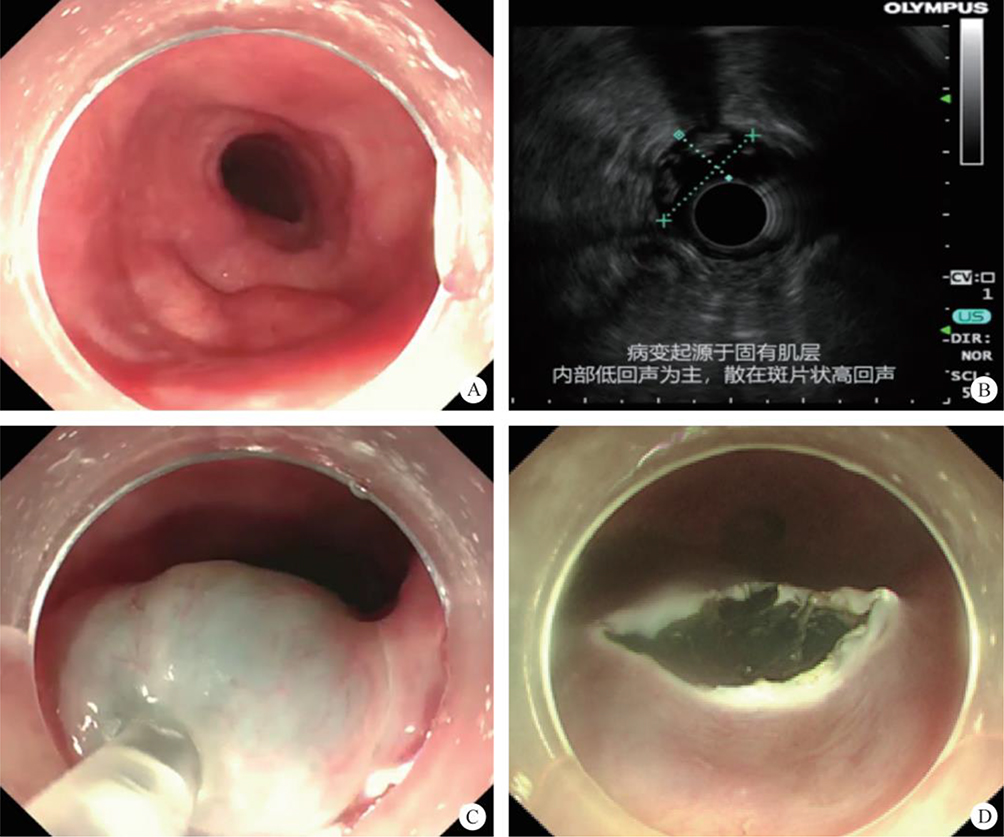

FIG. 8.12 — Positioning and establishment of the tunnel entrance in per-tunnel super minimally invasive resection of esophageal subepithelial lesions. A. Gastroscopy to locate submucosal tumors. B. Endoscopic ultrasound shows the lesion. C. Submucosal injection after determining the tunnel entrance. D. Making the inverted T incision.
(2) Submucosal tunnel formation: from the oral side to anal side, a tunnel must be formed between the membrane and MP layer, and terminated at the distal end of the tumor. Repetitive submucosal injection is helpful to avoid accidental injury to tunnel membranes. The exposed vessels and small vessels should be dealt with timely to ensure to obtain a satisfactory endoscopic field. To keep mucosal integrity and reduce bleeding, submucosal dissection should be performed near an MP without a vascular network.
(3) Tumor resection (figure 8.13): after being totally exposed with a knife or a snare, the tumor is resected with the complete envelope of the MP layer. After the tumor was resected, the submucosal tunnel must be rinsed with a normal saline solution. The incised margins must be carefully treated with a hemostat and APC to reduce hemorrhage and to avoid delayed hemorrhage and postoperative infection.
(4) Closure of the incision site: clips are used for closing the incision. The horizontal incision and the reverse T incision are closed in a vertical fashion. SELs with a diameter of <1.5 cm can be extracted directly by endoscopic suction, whereas a snare or a basket should be used to extract SELs with a diameter of ≥1.5 cm.
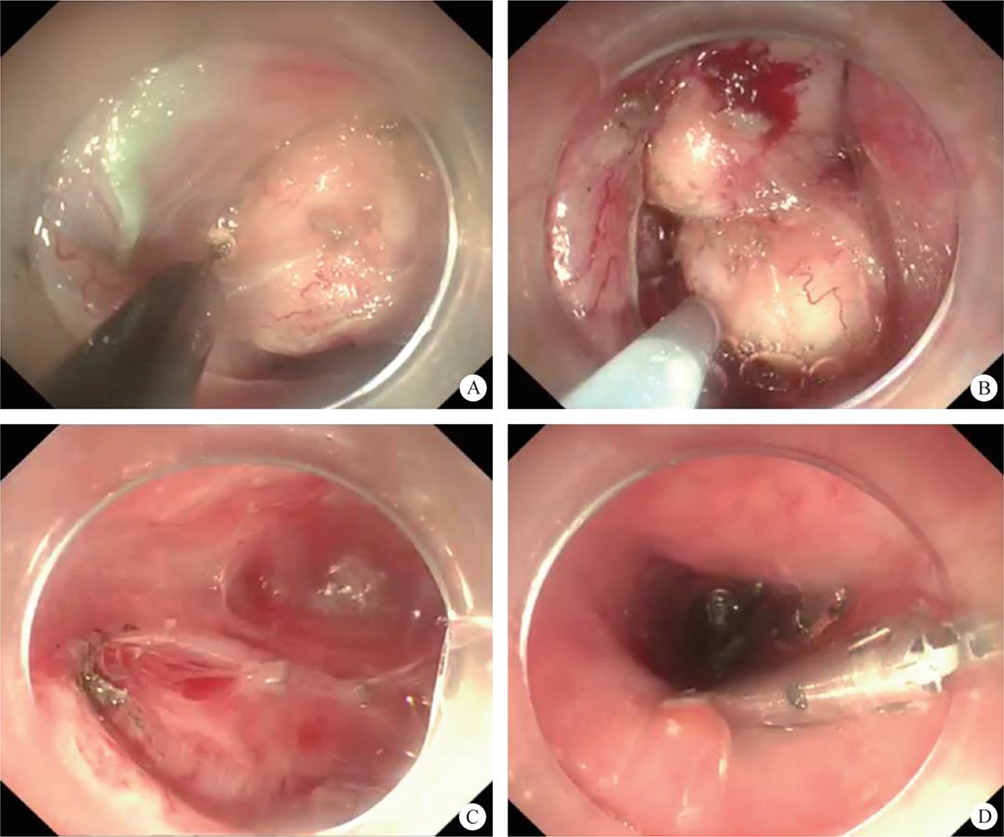

FIG. 8.13 — Resection in the tunnel and wound treatment in per-tunnel super minimally invasive resection of esophageal subepithelial lesions. A. Dissection of a submucosal tumor in the tunnel. B. The tumor is taken out with a snare. C. The wound after tumor resection. D. Closure of the tunnel entrance with tissue clips.
(III) Postoperative treatment
The patient must be fasting for food for two to three days, then take liquid food for three days, and gradually resume to a normal diet within two weeks. Proton pump inhibitors are given intravenously for two to three days, followed by oral proton pump inhibitors for four weeks. If there is no sign of infection, stop intravenous injection of antibiotics after two to three days, otherwise long-term antibiotics or potent antibiotics should be used to control infection. Supervised endoscopy should be performed at three, six, and twelve months postoperatively, and then conducted once every year.
(IV) A classic case
History: the patient, a middle-aged man, was admitted to the hospital mainly due to the fact that “gastroscopy revealed esophageal swelling for more than two months by physical examination.” Gastroscopy indication: esophageal eminence, considering big possibility of leiomyoma, originating from the muscularis propria. CT examinations had revealed an esophageal upper occupying lesion, with slightly bigger lymph nodes around the esophagus and in the left supraclavicular fossa. He had a history of hypertension, gallstones after cholecystectomy and appendectomy. No abnormalities were found for admission physical examination.
Diagnosis: Esophageal submucosal eminence.
After full preoperative evaluation, improvement of the nutrition state and signing of surgery informed consent, the patient was performed peroral super minimally invasive resection of esophageal subepithelial lesions (Refer to video 8.5, Peroral super minimally invasive resection of esophageal subepithelial lesions through a tunnel channel (See online resources)).
Surgical process: there was a submucosal eminence in the esophagus 25 cm from the incisor, covered with normal membrane. After the mucous layer had been incised to establish an inverted T opening 20 cm from the incisor with a triangle knife, the endoscope entered the tunnel to continue to gradually dissect the submucosa and the muscularis propria with the triangle knife and an IT knife to fully expose the tumor. Electrocoagulation was carried out with a snare to incise the lesion. The exposed vessels were treated with a hemostat. The tunnel opening was closed with tissue clips.
(V) Hot-spot issues in the consensus and progress of the study
1. Indications of STER in esophageal subepithelial lesions. Endoscopic submucosal tunnel resection maintains the integrity of the mucosa by establishing a tunnel between the submucosa and the MP layer. Maintaining mucosal integrity reduces perforation, fistula, and secondary infection, and promotes rapid wound healing. Limited studies have shown that STER has some advantages compared to video-assisted thoracoscopic enucleation, namely shorter operation time, reduced blood loss, reduced cost, and reduced postoperative chest pain. At present, STER has become the standard method for treating GI SELs derived from the MP layer, especially in China.
Compared to surgical treatment, STER has the advantages of small invasion, quick postoperative recovery, low hospitalization cost, and high acceptability. However, STER cannot totally replace surgical treatment. The best treatment method should be selected according to the illness itself, so that making the patient benefits from it.
In summary, STER is not suitable for all SELs and the doctor should strictly evaluate the indications. STER applies to benign lesions less than 3.5 cm in diameter without adequate blood supply as determined by EUS, CT, or other imaging tools (evidence level, II; Recommended strength, A).
STER classifies SELs according to the following categories: (1) SELs originating from the MP layer; (2) SELs with complete covering of the superficial mucosa; (3) SELs whose longest diameter is ≥10.00 and horizontal diameter is ≤3.5 cm; (4) SELs located in the esophagus, more than 3–5 cm from the entrance to the esophagus, the cardia, the stomach, and the rectum.
According to the experience of Du et al., as the tunnel cannot be created, STER does not apply to SELs located at the upper part of the esophagus less than 3–5 cm from the entrance to it. The inner diameter of the tunnel must be of about 3.5 cm; therefore, SEL’s horizontal diameter after STER treatment should not be greater than 3.5 cm. Several studies on large SELs greater than 3.5 cm have concluded that STER is feasible even for large tumors. Chen et al. proved that the longest diameter of STER was 7 cm. However, previous studies on STER focused on SELs with diameters not exceeding 5.5 cm. There is still debate as to the upper limit of the tumor’s diameter for successful STER. It should be noted that STER is not recommended for ulcerated SELs for because the integrity of the mucosa cannot be maintained. In addition, SELs with irregular edges have higher risks of malignancy and should not be treated with STER.
2. Comparison between STER and other techniques for esophageal subepithelial lesions. Although STER appears to be a time-consuming procedure compared to non-tunnelling ones, it has a higher overall removal rate compared to ESD and can maintain mucosal integrity. However, in order to confirm this conclusion, a prospective comparative study of STER and ESD is needed. A previous study reported that ESE in addition to STER is recommended for SELs with diameters less than 10 mm, while STER should be preferred for SELs larger than 10 mm. A study comparing endoscopic non-tunnel resection and tunnel resection challenges the advantages of tunnel resection, revealing that both methods are effective and safe, whereas tunnel resection takes longer. Although submucosal tunneling endoscopic resection (STER) demonstrates multiple advantages in preserving mucosal integrity and achieving high en bloc resection rates, additional comparative studies with larger patient cohorts against other established endoscopic resection techniques are required to assess whether STER provides therapeutic superiority for subepithelial lesions (SELs) originating from the muscularis propria (MP) layer.
8.2.4 Super Minimally Invasive Resection Per-Multi Cavity of Esophageal Subepithelial Lesions
Super minimally invasive resection per multiple cavity of esophageal subepithelial lesions mostly applies to treatments of tumors outside the esophageal muscularis propria or mediastinal tumors. Its past name was multiple endoscope combined therapy. It is an endoscopic combined thoracic and laparoscopic technique. The former can, with the assistance of the endoscope, better expose some tumors that cannot be resected due to the limitation of the endoscopic viewing angle through pulling, grasping, and pushing, and timely repair treatment by suturing can be performed in case of complications, thus improving the safety of endoscopic therapy, whereas the latter can position itself precisely with endoscopic assistance and choose the right surgical field, avoiding unnecessary traumas and excessive treatment. Upper and middle esophageal lesions are often combined with thoracoscopy, and lower esophageal lesions are often combined with laparoscopy.
(I) Indications and contraindications
1. Indications
(1) Bigger tumors, hard to resect with the only endoscope, with higher possibility of hemorrhage and perforation.
(2) In endoscopic surgery, the lesions are small and difficult to find or are located in a difficult location.
(3) Patients who, in addition to suffering from digestive tract diseases, have other diseases need combined surgery.
2. Contraindications
(1) Patients with serious underlying diseases, who cannot tolerate surgery and anesthesia.
(2) Patients with a history of complex thoracic and abdominal surgery and severe adhesions.
(II) Surgical operation and skills
The patient must be in supine leg splitting position; tracheal intubation and general anesthesia is used.
(1) The pneumoperitoneum is established on the umbilicus and the endoscope is inserted.
(2) Intraoperative gastroscopy is performed to locate the submucosal tumor, and the location of the tumor inside and outside the esophagus is determined endoscopically.
(3) After complete dissection of the tumor using the endoscope and the cavity endoscope, a specimen is removed.
(4) The wound is endoscopically closed.
(III) Postoperative nursing
Depending on the postoperative conditions, postoperative use of prophylactic antibiotics, water fasting, intravenous infusion of PPIs, etc., gradually transition diet can be prescribed.
(IV) A classic case
History: the patient, a young woman, visited Changzhou No. 2 People’s Hospital due to “belting, difficulty in swallowing for more than one year.” Chest enhanced CT showed an obvious thickening of the lower esophagus and the cardiac tube wall, the thicker ones being about 2.5 cm, whereas mild enhancement was found. Ultrasound gastroscopy revealed a smooth-surface submucosal eminence in the lower esophagus, and a circular eminence was found at the entrance of the diaphragm. A hypoechoic mass occupying space was found 37–43 cm away from the incisor, and the internal echo was uneven. The lesions were pushed towards the esophageal wall with unclear boundary with the esophageal wall, and the cross-section size was 5.8 cm × 3.7 cm. 3D reconstruction showed that a large tumor surrounded the esophagus and was adjacent to the aorta; the tumor body was ‘horseshoe-shaped’ around the junction of the esophagus and the cardia, with the long axis of about 6.4 cm and the short axis of about 3.7 cm.
Diagnosis: Benign tumor of the gastroesophageal junction.
Treatment: the tumor was exfoliated through an endoscopic submucosal tunnel, with resection of the tumor’s root to ensure the integrity of the membrane. Then the tumor was completely separated and resected by laparoscopy. The postoperative pathological findings suggested an esophageal leiomyoma. The final en bloc resection size was 7 cm × 4 cm × 4 cm.
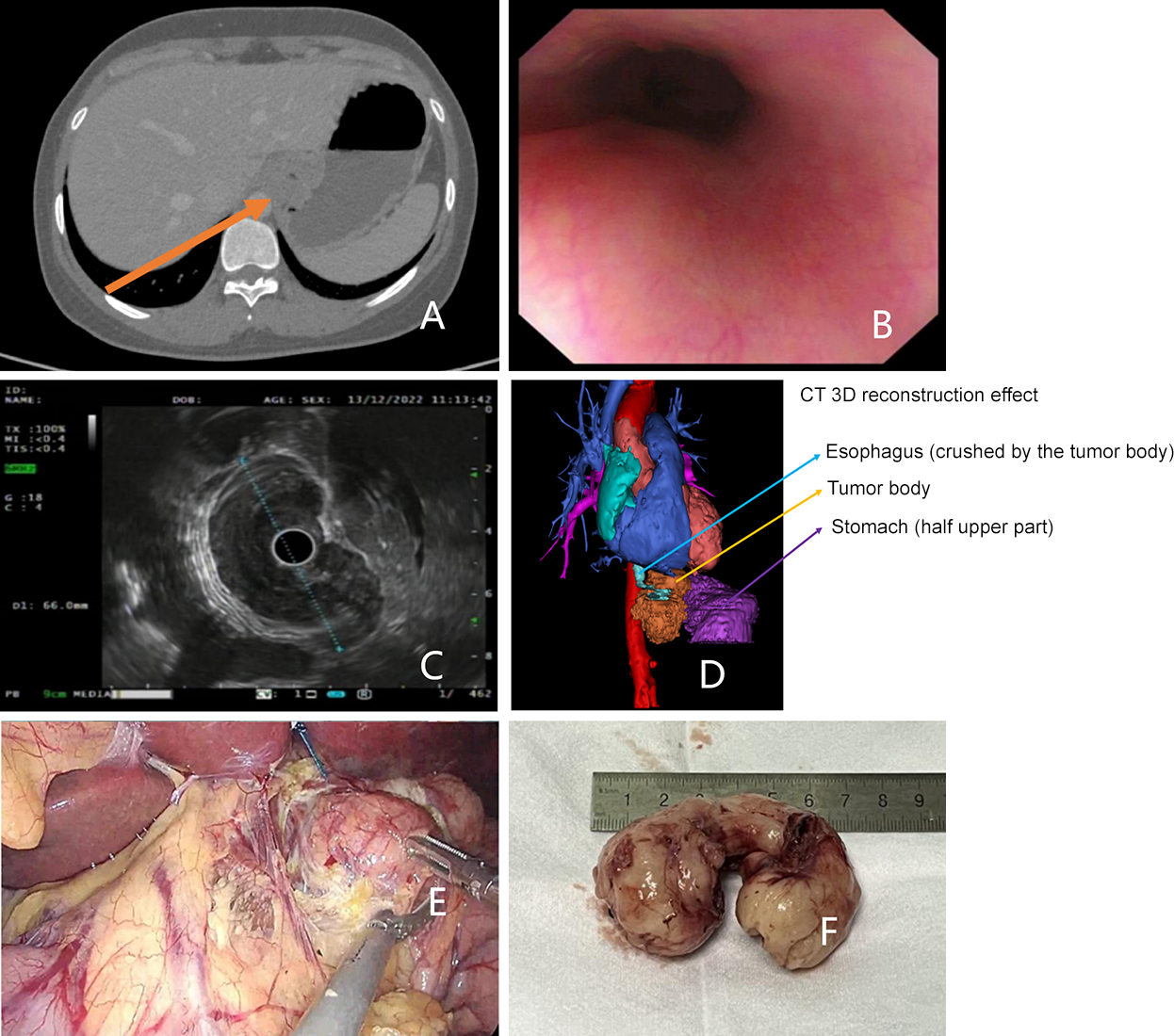

FIG. 8.14 — Operating steps of super minimally invasive resection per multiple cavity of esophageal subepithelial lesions. A. CT showed gastroesophageal junction tumor. B. Esophageal submucosal eminence observed endoscopically. C. Circumferential endoscopic ultrasound image. D. 3D image reconstruction. E. Laparoscopic surgery. F. Completely resected tumor.
(V) Hot-spot issues in the consensus and progress of the study
When the tumor is located at difficult sites like cardia, pyloric or minor curvatures of the stomach, endoscopic surgery can easily damage the esophageal sphincter, resulting in reflux or gastrointestinal obstruction, etc. LECS not only provides the surgeon a clear view, but also reduces the incidence of long-term complications after surgery. Studies have shown that LECS has significant advantages in terms of reducing intraoperative blood loss, reducing postoperative pain score, shortening first postoperative recovery time and the time spent on a liquid diet, reducing the length of stay and the rate of complications.
8.3 Super Minimally Invasive Radiofrequency Ablation for Intraepithelial Neoplasia
Intraepithelial neoplasia (IN) is a pathological diagnostic term, replacing the previous names of heteroplastic hyperplasia, atypical hyperplasia or carcinoma in situ, etc. Various degrees of atypia, differing in cell morphology and tissue structure with the original normal esophageal mucosa. Its characteristic is a precancerous lesion characterized by morphological abnormalities in cells and structures, genetic clonal changes in genes, and biological progression to invasive and metastatic esophageal cancer. After that, the Vienna International Consensus Conference also reclassified and readjusted the diagnostic classification criteria for dysplasia. Depending on the degree of cell atypia, the complexity of tissue structure and the characteristics of biological behavior, mild and moderate dysplasia is classified as low-grade intraepithelial neoplasia (LGIN), whereas severe dysplasia is defined as high-grade intraepithelial neoplasia (HGIN). As one of the precancerous lesions of esophageal carcinoma, esophageal LGIN can be treated by endoscopic radiofrequency ablation (RFA). In recent years, relevant research reports have shown that this operation for the treatment of esophageal LGIN has many advantages such as safety, effectiveness, small complications, simple operation, and can be used in the context of outpatient treatments, which are gradually becoming more popular and are encouraged in clinical applications.
8.3.1 Principle of Radiofrequency Ablation
The therapeutic principle of radiofrequency ablation consists of inducing the movement of charged particles in the tissue to generate heat through the action of a high-frequency alternating current, thus causing coagulation and the necrosis of tissue cells. Its features include uniform action, large coverage area for one-time, low temperature, and the absence of any excitatory neuromuscular effect. Meanwhile, in radiofrequency ablation applied to gastrointestinal mucosal lesions, in its ablation process, the ablation electrode can partially absorb the current reflux, so that the current will not pass through the body; so it will not be necessary to attach the negative electrode plate, avoiding any impact on patients with cardiac pacemakers (figures 8.1–8.16). In addition, relevant animal experimental studies have shown that radiofrequency ablation has little effect on normal tissues around the treated area, and also has little effect on deep base-level tissues. This is why radiofrequency ablation demonstrates unique advantages in certain respects. For instance, radiofrequency ablation of some circumferential esophageal lesions or mottled esophageal mucosal lesions will be of great help to effectively control postoperative luminal stenosis.
8.3.2 Indications and Contraindications of Super Minimally Invasive RFA Surgery
(I) Indications
(1) After strict and standard magnetifying endoscopic screening + biopsy, according to WHO/Vienna assessment standards, the pathological results evidence esophageal LGIN (the pathological diagnosis must be co-diagnosed by two senior physicians from the pathology department)—in case of disagreement, the final confirmation will be made by a third senior physician from the pathology department).
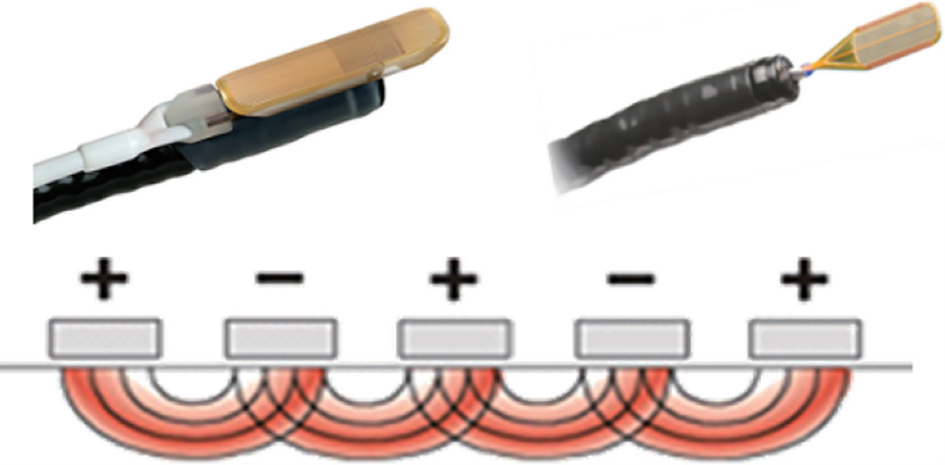

FIG. 8.15 — Device for radiofrequency ablation.

FIG. 8.16 — Electrode plate for radiofrequency ablation (electrode plate to achieve local current reflux).
(2) The surface morphology of endoscopic lesions is consistent with type 0–II in the Paris typing.
(3) The anticoagulant, blood activating drugs were not taken, or the patient has been able to stop taking such drugs for at least seven days prior to treatment, although it has been taken previously.
(4) If there are no other contraindications for endoscopic treatment, and the patient has signed the informed consent form for endoscopic treatment.
(II) Contraindications
(1) The results of a strict standard magnetifying endoscopic screening + biopsy according to WHO/Vienna assessment standards are not consistent, after pathological confirmation, with a diagnosis of esophageal LGIN (the pathological diagnosis must be co-diagnosed by two senior physicians from the pathology department—in case of disagreement, the final confirmation will be made by a third senior physician from the pathology department).
(2) In the presence of the classical contraindications of endoscopic therapy or examination: cardiopulmonary insufficiency, coagulation disorders, hepatic and renal dysfunctions, etc.
(3) In the presence of gastroesophageal varices, giant esophageal diverticulum.
(4) For pregnant or lactating women.
(5) In case of long-term use of anticoagulant, blood activating drugs that cannot be stopped.
(6) Other endoscopic treatment contraindications, or refusal to sign the informed consent form for endoscopic therapy.
8.3.3 Preoperative Preparation
(1) The patient and his/her family should be informed in detail of the multiple possibilities for the progression of esophageal LGIN to HGIN or even of early esophageal carcinoma, of the long-term maintenance status, or natural reversal, and of related matters such as potential clinical benefits, risks, adverse events and postoperative recovery management, rechecking made necessary by super minimally invasive radiofrequency ablation; he/she must sign the informed consent form.
(2) The examination and lab items needed by endoscopy, such as blood routine analyses, assessment of liver and kidney function, electrolyte, coagulation function, serological test, electrocardiogram, chest film (or chest CT), anesthesia evaluation must be refined and checked. In the meantime, the patient should be told that anticoagulant drugs such as aspirin, clopidogrel bisulfate tablets and blood-activating drugs such as salvia miltiorrhizae, safflower, notoginseng and ginkgo biloba must be stopped one week before surgery.
(3) On the day of the treatment, preoperative fasting for food and water for 8 h is needed; 50–60 ml of a defoaming and slime remover containing pronase granules and polydimethylsiloxane must be preoperatively taken. The patient should change position in the bed for 10 min, holding in the mouth 5–10 ml of dyclonine hydrochloride mucilage during 1–2 min, which will be then swallowed.
8.3.4 Surgical Operation and Skills
1. Anesthesia and position All patients undergo intravenous general anesthesia, left lying on their left side; attention should be paid to keep them warm, adjusting the room temperature and the infusion temperature, and avoiding pressure sores.
2. Surgical operation step
(1) Observation and staining: before treatment, a screening by magnifying endoscopy combined with narrow-band imaging (ME + NBI) must be made to reconfirm the site of the lesion, and to determine whether the lesion has any boundaries, as well as to collect information relating to the surface micro structure, microvessels, etc. If the above observation methods are difficult to use to clearly examine the lesion, then using Lugol’s iodine staining should be considered for more in-depth auxiliary observation. After observation, the in-situ biopsy of the lesion area should be performed again. Since pathological tests cannot be performed after radiofrequency ablation, pathological specimens need to be taken again before surgery to provide relevant diagnostic evidence.
(2) Boundary marking: if the extent of the lesion is relatively limited or easy to identify, subsequent radiofrequency therapy can be directly conducted. If the endoscopically evidenced lesion area is relatively large, to avoid the insufficient coverage of the subsequent radiofrequency scope, the lesion boundary could be marked under the magnifying endoscope, and then the subsequent radiofrequency ablation therapy should be carried out.
(3) Endoscopic radiofrequency ablation: under the magnifying endoscope, the therapeutic endoscope (GIF-Q260J/HQ290) should be replaced by the radiofrequency ablation machine (output power: 57 W, energy density: 10–12 J/cm8.17). After that, the second alation must be carried out on the same area and the above steps repeated. After ablation of the esophageal mucosa, scraping the white coagulated necrotic tissue at the lesion’s surface is not necessary. Sometimes the necrotic mucosal tissue can fall off by itself; if it needs to be scraped, this can be completed with the aid of the ablation electrode plate and of the endoscope-connected water function. If the lesion is big and plain ablation cannot cover the total membrane of the lesion, the ablation electrode plate margin will be superposed to the margin of the previous ablation area on 3–5 mm, and then ablation will be conducted again until all lesion areas are effectively covered. The entire ablation extent should be more than 0.5–1 cm beyond the lesion’s boundaries to ensure nothing is missing.
8.3.5 Postoperative Treatment
Postoperative fasting food for 4–6 h, with appropriate fluid supplement, will be prescribed; subsequently, a cool liquid diet will be given, and switching gradually to a semifluid, full diet; the patient will orally take PPIs and a mucosa protector for one month after the operation.

FIG. 8.17 — Super minimally invasive radiofrequency ablation of a low-grade intraepithelial neoplasm. A. After radiofrequency ablation, the lesion surface appears as a white coagulated necrosis. B. Scraping of necrotic mucosal tissues of the lesion’s surface after radiofrequency ablation.
Refer to video 8.6, Super minimally invasive radiofrequency ablation of low grade intraepithelial neoplasia (See online resources).
8.3.6 Postoperative Follow-Up Visit
We recommend close follow-up by telephone and in an outpatient setting after the RFA in order to record the occurrence of adverse events. Meanwhile, patients will be informed and urged to return to the hospital for endoscopic reexamination three months, six months, one year, then every year until five years after surgery, each time using the ME+NBI method for reexamination, and a biopsy should be taken from the original treatment area to be sent for histopathological examination.
8.3.7 Hot-Spot Issues in the Consensus and Progress of the Study
1. About the extension of esophageal mucosa lesion RFA treatment indication. RFA is currently indicated for the treatment of simple esophageal LGIN, but there is no clear conclusion or consensus on its suitability for the treatment of HGIN. At present, some preliminary studies have discussed the therapeutic effect of RFA on partial esophageal HGIN, and certain progress has been made. Prospective, multi-center, large-sample clinical studies in this regard are urgently needed to further expand the scope of the RFA treatment for early esophageal carcinoma.
2. Prospect of the application of RFA in the treatment of early circumferential esophageal carcinoma. As we all know, early circumferential esophageal carcinoma is a special type of early esophageal cancer, and ESTD is considered one of the first options for treatment. However, postoperative stricture becomes an inevitable major complication, which would seriously affect the patient’s quality of life. RFA is less likely to cause postoperative stenosis due to the small injury it causes to deep muscle tissue, but at the same time, its relatively limited therapeutic depth also limits its use in the treatment of esophageal HGIN or of early esophageal carcinoma. Further exploration and studied in this field in the future, such as using RFA to treat the superficial areas of the disease, thereby minimizing injury to the deep muscle layer, could lead to advances in solving the problem of postoperative esophageal stricture.
8.4 Super Minimally Invasive Surgery for Esophageal Stricture
(I) Introduction to esophageal stricture
Esophageal stricture refers to a situation in which a standard endoscope cannot pass through the esophageal stenosis with varying degrees of dysphagia, often accompanied by reflux, retrosternal pain, and weight loss. According to the nature of the stricture, it can be divided into malignant esophageal stricture and benign esophageal stricture. Esophageal malignant stenosis is commonly seen in esophageal carcinoma or is caused by external pressure from malignant tumors of non-esophageal origin. Due to poor general health and distant tumor metastasis, radical resection cannot be conducted on most patients, so conservative therapy can be used to alleviate the symptom of difficulty in swallowing. Esophageal benign stenosis is often caused by large esophageal lesions after ESD surgery, anastomotic stenosis after surgery, ulcerative lesions, chemical corrosion or radiation damage. Large esophageal lesions usually refer to those with an estimated resection circumference of ≥3/4 and a resection length of ≥3 cm, including circumferential esophageal lesions. Previous studies have reported that the incidence of esophageal stenosis after endoscopic resection of non-circumferential lesions is no less than 56%, while the incidence of postoperative stenosis of circumferential esophageal lesions is as high as 100% (figure 8.18).

FIG. 8.18 — Endoscopic observation of a mucosa defect and severe recurrent stricture after ESD. A. Mucosa defect exceeding 3/4 of the circumference. B. Repetitive occurrence of esophageal stricture, shown as needles.
(II) Diagnosis of esophageal stricture
Esophageal stricture is mainly diagnosed through medical history and imaging examination, among which gastroscopy can directly observe the esophagus, and is the ‘gold standard’ for diagnosing esophageal stricture. When endoscopy reveals that the diameter of the stricture is less than 1 cm, or when the usual endoscopes (about 1 cm in diameter) cannot pass, and when the trouble is accompanied by dysphagia and other symptoms, it can be diagnosed as esophageal stricture. Upper gastrointestinal angiography can indirectly reflect the condition of the esophagus and determine whether there is esophageal stricture. The etiology of esophageal benign stricture must be determined before treatment, and an endoscopic biopsy must be performed to exclude malignant tumors and identify specific etiologies such as eosinophilic esophagitis.
Esophageal stricture can be divided into simple stricture and complex stricture. Simple esophageal stricture refers to local, agonic stricture, the length of the stricture being less than 2 cm, usually located in the lower esophagus; generally the stricture can disappear after three to five endoscopic dilation therapies. Complex esophageal stricture refers to angular, irregular stenosis with a length of structure of ≥2 cm, and esophageal stricture still appears repetitively after endoscopic dilation therapy for three to five times. If endoscopic dilation therapy is performed once every two weeks for five consecutive times, if a standard endoscope still cannot pass through the stricture site or if esophageal stricture is diagnosed again, we are dealing with refractory esophageal stricture.
(III) Techniques of implementation of super minimally invasive prevention and treatment of esophageal stenosis
Super minimally invasive prevention and treatment of esophageal stenosis are mostly carried out through a peroral natural cavity channel in order to perform endoscopic therapy, including super minimally invasive incision for gastrointestinal stenosis + endoscopic submucosal injection, endoscopic dilation, endoscopic stent implantation, etc.
8.4.1 Esophageal Stenosis Peroral Super Minimally Invasive Incision + Submucosal Injection
Endoscopic super minimally invasive surgery is the first-choice treatment method of esophageal stenosis, mainly including super minimally invasive incision, endoscopic submucosal hormone injection, endoscopic dilation, etc. Super minimally invasive incision for gastrointestinal stenosis is an endoscopic technique using a high-frequency electric incision knife to radially cut the scar along the narrowing opening to dilate the stenosis’ lumen. It is often combined with endoscopic submucosal injection, that is, an endoscopic technique using injection needle to inject locally drugs like hormones into the mucosa to prevent or treat stenosis of the digestive tract.
(I) Indications and contraindications of super minimally invasive surgery for esophageal stenosis
1. Indications. Benign strictures resulting from various etiologies, including those secondary to endoscopic minimally invasive therapies, surgical interventions, or caustic injuries.
2. Contraindications. (1) In the event of other esophageal diseases. (2) In the event of overall anesthesia contraindications. (3) In the event of esophageal malignant stenosis or other malignant tumors. (4) The patient is allergic to prednisone acetate or triamcinolone acetate. (5) Pregnant and lactating women.
(II) Preoperative preparation
(1) Preoperative evaluation: all patients with esophageal stenosis must undergo endoscopic evaluation before treatment to determine the type, length, and diameter of the stenosis.
(2) Super minimally invasive resection of esophageal stenosis needs to be conducted under general anesthesia, so patients with high anesthesia risk need to undergo a sufficient preoperative risk assessment.
(3) Preoperative fasting for food or liquid: a fat fluid diet should be administered two days before the operation, with fasting for food and water for 8 h at the date of the surgery, so as to be sure that the cavity is completely empty. The patient should avoid using anti-platelet drugs and/or anti-coagulants one week before the operation.
(III) Surgical operation and skills
1. Anesthesia and position. All patients undergo general anesthesia with tracheal intubation, lying on their left side or in supine position in order to facilitate the surgical operation.
2. Surgical operating step (figure 8.19)
(1) Radial incisions should be made longitudinally along the esophageal lumen: the gastroscope enters the esophageal stenosis, observes and evaluates the stenosis site. One to three sites with obvious scars must be chosen, according to stenosis’ condition, in order to perform a radial incision with a hook. The incision should reach the surface of the muscularis propria or the incision base should be located on the plane formed by the narrow mucosal connection at both ends. The choice of the incision depth is crucial: a too shallow incision will have no therapeutic effect, and a too deep incision will increase the risk of hemorrhage and perforation. If the anastomosis is narrow, some anastomosis nails can be pulled out.
(2) The hard fibrotic scars between incisions should be carefully scraped: some scar tissues along the incision must be remove circumferentially, making the endoscope front end smoothly pass through the stenosis site. Hemorrhage from the wound must be observed. If there is a small exudation, conservative therapy like local hot biopsy forceps for hemostasis can be applied.
(3) Hormones must be injected after the stenosis is incised: the injection needle must be placed through the endoscope’s forceps channel, then 1 ml of triamcinolone acetate (10 mg/ml) will be injected into each of the four limits at the narrowest part of the esophageal stenosis. The wound’s hemorrhage condition will be observed.

FIG. 8.19 — Schematic diagram of esophageal stenosis and surgery by injection of submucosal hormones. A. The diameter of the esophageal stenosis is about 3 mm. B. Longitudinal radial incision along the esophageal lumen with a hook. C. Removal of scar tissues after radial incision. D. Injection of hormones after the stenosis is incised.
3. Postoperative treatment. The patient must fast for food and water. The diet will include acid suppression, fluid supplement, prevention of infections for three to five days, and other symptomatic measures; the diet will be gradually modified as follows: liquid diet → semiliquid diet → full diet; treatment with antacid medication and gastric mucous membrane protectors will be administered for two to three months.
(IV) A classic case
History: the patient, an elderly male, was admitted to the hospital due to “difficulty in swallowing for more than six months.” The patient had undergone surgery due to “esophageal carcinoma” eight months ago. He had been having difficulties in swallowing for more than two months after the operation, with past multiple times endoscopic balloon dilation and recurrent symptoms of difficulty swallowing.
Diagnosis: postoperative anastomosis stenosis
After full preoperative evaluation and having signed the surgery informed consent, the patient was administered an injection of esophagostricture release + hormones. See figure 8.20 (Refer to video 8.4, Super minimally invasive surgery for peroral esophageal stenosis) (See online resources).
Postoperative fasting for food and water, intravenous acid suppression, fluid supplement were prescribed for three days, the patient gradually resuming a normal diet. The situation was re-examined by gastroscopy one month after the operation: the esophageal is clearly less significant than before.
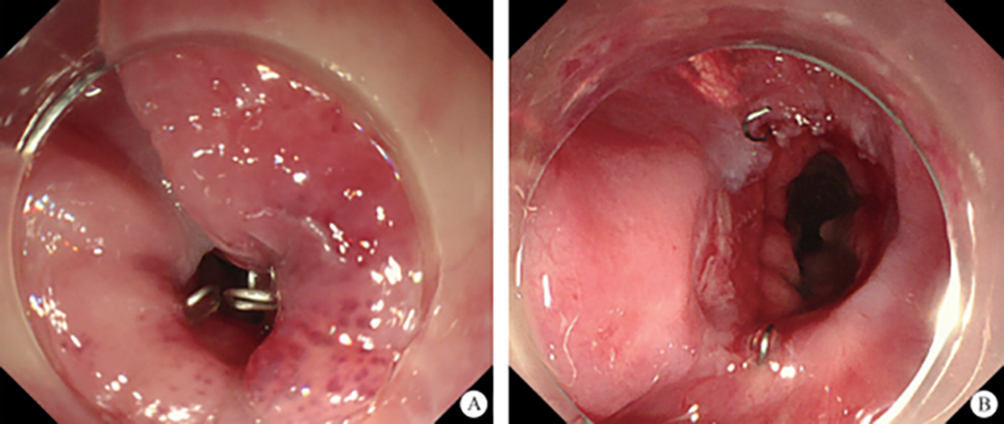

FIG. 8.20 — Contrast image before and after incision of the esophageal stenosis and submucosal injection surgery. A. The postoperative anastomosis of an esophageal carcinoma is narrow and the endoscope cannot pass through it. B. After stenosis incision and injection of hormones for one month, the stenosis appears significant less severe.
(V) Hot-spot issues in the consensus and study progression
1. Refractory benign esophageal stenosis still needs active and effective treatment plans. The treatment of benign refractory esophageal stenosis is still a major clinical problem. The main treatment methods include endoscopic stenosis incision, drug injection, external self-dilating balloon, esophageal stent implantation, regenerative medicine, etc. All methods have certain advantages and disadvantages. The most appropriate treatment method should be selected according to factors like stenosis reasons, degree and patient wish to reach the best curative effect.
2. Regenerative medicine is still awaiting more in-depth studies on the treatment of esophageal benign stenosis. Regenerative medicine techniques have broad prospects for the treatment of esophageal stenosis, as the cell tissues are easy to obtain, the extracellular matrix is retained, is easy to integrate into the deficit site, promoting wound healing and preventing scar narrowing, with low complication occurrence and satisfactory curative effects. But the cell culture cycle is long, and the preparation cost is high. So far, most studies are still at the animal model stage, and the long-term effects and prospective clinical applications of these techniques are yet to be verified.
8.4.2 Peroral Super Minimally Invasive Dilation of Esophageal Stenosis
Super minimally invasive dilation mainly includes balloon dilation and bougie dilation. The mechanical tension generated by the balloon or bougie causes the fibrous scar tissue to be strongly stretched, thus achieving the effect of splitting the narrow mucosal muscle layer. Since bougie dilation cannot be carried out under direct vision, it is not easy to detect bleeding and perforation in time, and the operation is relatively complex, requiring repeated withdrawal of the bougie to dilate layer by layer. Therefore, endoscopic balloon dilation is currently more widely used clinically. In order to further improve the balloon’s efficiency and reduce the patient’s economic burden, Professor Chai Ningli from PLA General Hospital designed an external self-dilating balloon, allowing easy operation, high safety while not affecting the patient’s diet, etc. It can not only prevent large-area esophageal stenosis after ESD surgery, but also be used to treat stenosis, especially for refractory benign esophageal stenosis.
(I) External self-dilating balloon
The balloon’s total length, effective length and diameter are 120 mm, 80 mm and 18 mm, respectively (figure 8.21). The tip of the balloon catheter is fixed with a protective hose, while air (30–35 ml) is used instead of water to inflate the balloon, increasing its safety and convenience. After the balloon is endoscopically placed, the patient can operate it by himself, avoiding repeated visits to the hospital. The syringe must be used to inflate it 4–5 times a day, about 15–20 min each time, and the balloon should be removed according to the degree of wound healing evidenced during reexamination.


FIG. 8.21 — Schematic diagram of an external self-dilating balloon. A. The patient can operate the balloon by himself. B. The parameters associated with the balloon.
(II) Indications and contraindications of the external self-dilating balloon
1. Indications: (1) Benign strictures resulting from various etiologies, including those secondary to endoscopic minimally invasive therapies, surgical interventions, or caustic injuries; (2) in the event the standard gastroscope (9–11 mm) cannot pass; (3) no recurrence under endoscopic observation; (4) stenosis length ≤ 8 cm.
2. Contraindications: (1) patients with coagulation abnormalities; (2) patients with esophageal fistula; (3) patients with mental abnormalities; (4) esophageal malignant stenosis due to pathologies like tumors; (5) patients with serious organ function disorder or insufficiency; (6) poor compliance, inability to operate the balloon as required.
(III) Preoperative preparation
(1) Routine preoperative examinations must be refined; they include mainly: blood routine, liver function, renal function, electrolyte, blood coagulation, serum four items, ECG, etc., to eliminate the contraindications of endoscopic therapy.
(2) Preoperative fasting for food for 8 h, fasting for water for 2 h; the patient should orally take 50 ml defoaming slime remover (containing 10 000 units streptomyces protease, 1 g of sodium bicarbonate, 10 ml of polydimethylsiloxane) 15 min before the preoperative examination.
(IV) Surgical operation and skills
1. Position. All patients are lying on their left side, and the self-dilating balloon is placed under anesthesia or alert state.
2. Operating steps for placing the external self-dilating balloon
(1) Pre-dilation: as the actual pressure of the new balloon is less than that of the traditional dilated balloon, it is impossible to mechanically tear the mucosa in the esophageal stenosis quickly and effectively. Therefore, before placing this balloon, the routine traditional water balloon is generally chosen (figure 8.22), or the technique of endoscopic radial incision (figure 8.23), so as observe and assess whether there is a risk of delayed perforation and hemorrhage from the wounds.
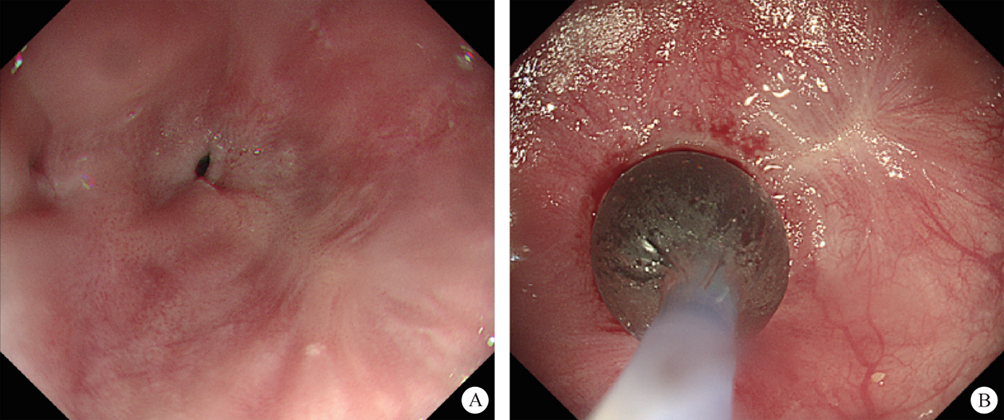
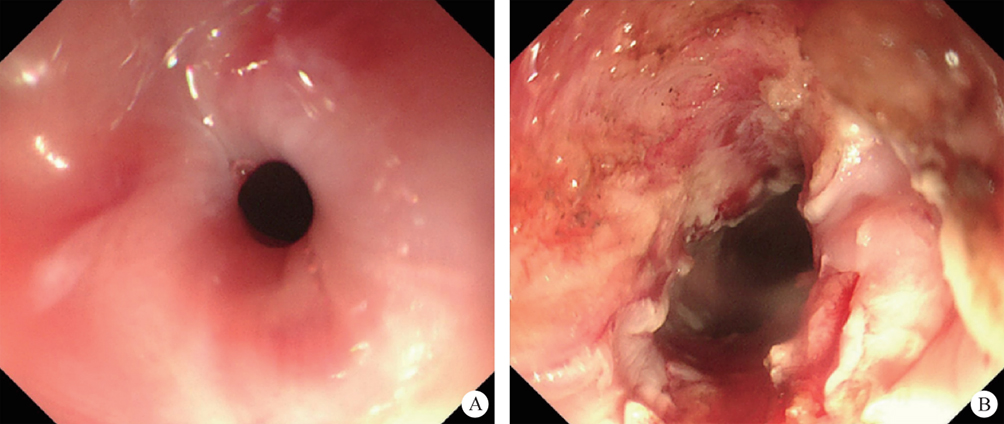

FIG. 8.22 — A. Stenosis diameter about 2 mm. B. Use of traditional EBD to dilate.

FIG. 8.23 — Operating steps of super minimally invasive dilation of esophageal stenosis. A. Stenosis diameter of about 3 mm. B. Endoscopic radial incision.
(2) Place in the balloon: the placement method is similar to that of the gastric retention tube, using liquid paraffin to lubricate the balloon, which must be slowly inserted along the selected nostril, asking the patient to make a swallowing movement. The balloon’s position must be adjusted under endoscopic observation, so as to ensure that the balloon is located in the middle part of the stenosis (figure 8.24A).
(3) Fixing the balloon: the balloon must be attached to the side of the patient’s nose and the attachment place recorded (figure 8.24B). Refer to video 8.7, Super minimally invasive prevention and treatment of peroral esophageal stenosis (See online resources).
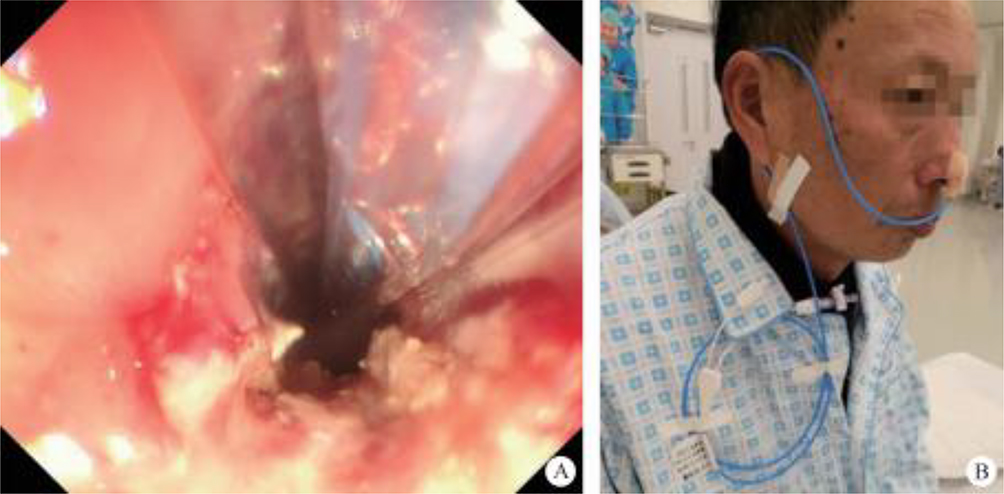

FIG. 8.24 — Operating steps of the super mini-invasive implantation of a self-expanding balloon for the treatment of esophagal stenosis. A. Endoscopy-assisted insertion of a self-dilating balloon. B. External fixation of the balloon.
(V) Postoperative treatment
(1) As this operation can be conducted on an outpatient or inpatient basis, there generally is no needs for antibiotics. Fluid foods can be taken on the day of the surgery and gradually replaced by a normal diet.
(2) The patient must take orally a routine treatment with IPP or a new antacid medication (a potassium antagonist) for one to two weeks continuously, until the self-expanding balloon is removed. The patient should start using the self-expanding balloon on the day of the operation, expanding it four to five times a day, for 15–20 min each time.
(3) The endoscope should be rechecked once every two for weeks to assess the healing of the esophagus, until the wound is totally healed.
(VI) A classic case
History: the patient, a 63-year-old woman, was admitted to the hospital due to “Postoperative stenosis of early esophageal carcinoma ESD for more than two years.” In the past, she has undergone 31 endoscopic balloon dilations (EBD) and 4 endoscopic radial incisions and cuttings (RIC). Examination results after admission: an esophageal lumen stenosis of 24–28 cm from the incisor; the diameter was about 1 mm.
Diagnosis: refractory esophageal stenosis.
After full preoperative evaluation, the patient was explained the benefits and risks of the various treatment methods of refractory stenosis available currently. The patient chose the external self-dilating balloon method. After signing the surgery informed consent form, after performing endoscopic traditional balloon dilation on the patient, the self-dilating balloon was inserted to prevent esophageal stenosis (figure 8.25).
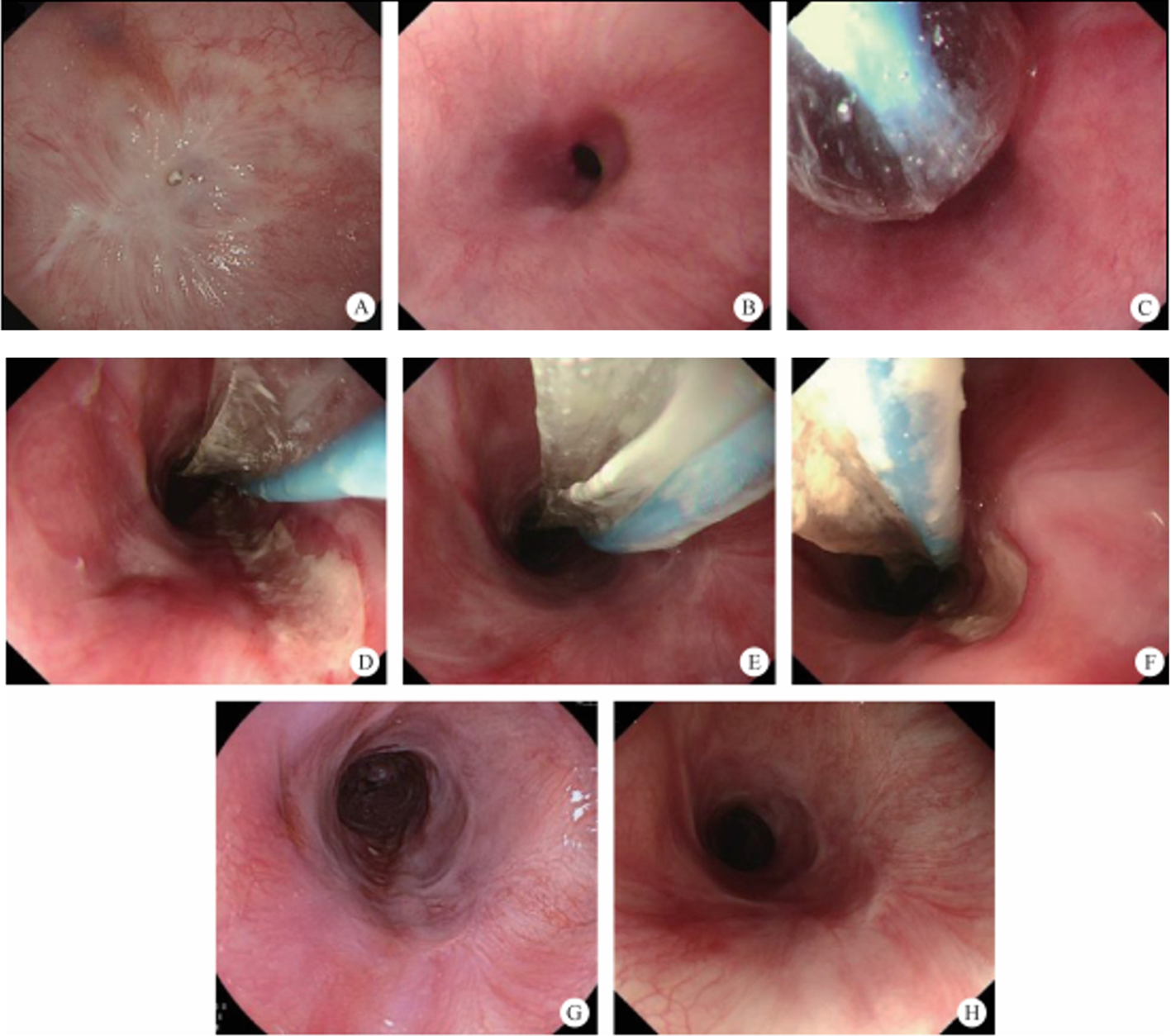

FIG. 8.25 — Treatment of refractory esophageal stenosis by using a self-dilating balloon. A. The diameter is less than 1 mm at the narrowest at the esophageal stenosis. B. When inserting the self-dilating balloon, the stenosis diameter is of about 3 mm. C. After using traditional EBD therapy, the self-dilating balloon is inserted. D–F. When rechecking 20 days, two months, three months after the operation, the standard endoscope passed smoothly, whereas a shallow ulceration formation was found locally. G. After taking out the balloon four months after the operation, the wound was basically cured. H. At the eighth month after the operation, no stenosis formation was found.
A liquid diet has been prescribed on the day of the surgery, after which a normal diet has been gradually resumed. The balloon was removed 16 weeks after the operation; no esophageal stenosis was observed during the postoperative gastroscopic reexamination at eight months.
(VII) Hot-spot issues in the consensus and progress of the study
1. Standard measures for the prevention of stenosis after ESD surgery for large esophageal lesions have not been established. At present, preventive measures mainly include: oral or local injection of hormones, PGA membrane, metal stent implantation, self-dilating balloon, etc. In addition, new technologies such as tissue engineering technology, esophageal mucosa transplantation, autologous skin transplantation + coated metal scaffold, carboxymethyl cellulose membrane, and hydrogel also have certain effects on the pre-prevention of stenosis. In the future, it will be necessary to innovate in prevention strategies and conduct multi-center clinical studies to continually reduce the incidence of stenosis and improve patient’s quality of life.
2. The therapeutic effect of postoperative surgery or corrosive stenosis should be improved. These two types of stenosis are not effectively treated by traditional endoscopic dilation, and can easily develop into refractory benign esophageal stenosis. At present, although the use of external self-dilating balloon improves the quality of life of patients to a certain extent, avoids repeated visits to the hospital, and greatly reduces the economic burden of patients, the problem of restenosis after balloon removal has not been effectively solved. Associating medicine with industry, as well as R&D of biological materials might be the direction for future development.
8.5 Super Minimally Invasive Surgery for Gastroesophageal Reflux Disease
8.5.1 Introduction
(I) Introduction to gastroesophageal reflux disease
Gastroesophageal reflux disease (GERD) refers to symptoms and (or) complications resulting from the reflux of gastric contents to the esophagus and the throat. The most common typical symptoms include heartburn, regurgitation, and it can also bring about related extraesophageal (including throat, trachea) symptoms, seriously affecting the patient’s quality of life. Proton pump inhibitors (PPIs) are the cornerstone of GERD treatment, but about 1/3 of the symptoms still cannot be totally alleviated after PPI therapy, particularly regurgitation symptoms. C-BLART has been reported as one of the new therapeutic methods replacing endoscopy for refractory GERD. The effective rate at six months for the treatment of GERD is 74.6%. And compared to routine PPI medication, symptoms such as sour regurgitation and heartburn, LES pressure and acid exposure times are obviously relieved. C-BLART has the following advantages: ease of use, time saving (the average time of use is only 8 min), low cost, possibility of repetition and high safety, which makes it practical for promotion and clinical use.
(II) Diagnosis of GERD
The presence of typical symptoms such as acid reflux and heartburn, abnormal reflux monitoring and objective examination of reflux such as gastroscopy can be used to confirm GERD. The reflux disease questionnaire can be used as an auxiliary tool for diagnosing GERD. A PPI experimental therapy can be used as a primary diagnostic method in patients with suspicion of GERD. Gastroscopy plays an important role in the detection of GERD complications, the evaluation of anti-reflux anatomy, and the detection of other diseases. Esophageal reflux monitoring, especially pH-reactance monitoring, provides objective reflux parameters and is currently the ‘gold standard’ for diagnosing GERD.
(III) Techniques of GERD super minimally invasive surgery and their past names
GERD can be cured by endoscopic super minimally invasive surgery (SMIS) through a peroral natural cavity channel, including endoscopic radiofrequency ablation, peroral incisionless fundoplication, anti-reflux mucosal resection, peroral endoscopic cardiac constriction, etc.
8.5.2 Peroral Super Minimally Invasive Radiofrequency Ablation to Cure Gastroesophageal Reflux Disease
Super minimally invasive radiofrequency ablation includes methods like physical ablation, chemical ablation, etc. Among them, endoscopic radiofrequency ablation (ERFA), as an intervention for the treatment of GERD, applies radiofrequency energy under the lower esophageal mucosa in order to strengthen the function of the lower esophageal sphincter, thus reducing the incidence of esophageal acid reflux. The principle of ERFA therapy is to enhance the function of the esophageal sphincter by precisely controlling radiofrequency energy at the submucosal level, heating the tissue locally, promoting collagen contraction and regeneration. This treatment can not only effectively relieve symptoms, but also reduce or eliminate the need for long-term use of drugs, especially for patients who do not respond well to drug therapy or have surgical contraindications. The treatment process needs to be performed by an experienced physician or gastrointestinal specialist to ensure the safety and effectiveness of the treatment. The patient’s history, symptoms and imaging examination results should be fully evaluated before the operation. The position and intensity of radiofrequency energy must be precisely grasped during the operation. It is necessary to observe closely and give appropriate nursing care and follow-up after the operation to ensure the stability of the patient’s recovery and the long-term effect. In general, as an innovative treatment option, endoscopic radiofrequency ablation has brought new hopes to patients with GERD, and has gradually become one of the important clinical treatment methods, especially with the advantages of lasting therapeutic effect and high safety.
(I) Indications and contraindications of ERFA surgery
1. Indications: (1) patients 18 and above; (2) patients with frequent symptoms of reflux or (and) heartburn for more than six months; (3) patients diagnosed with gastroesophageal reflux disease by preoperative gastroscopy, after 24 h of gastroesophageal impedance monitoring and high-resolution esophageal manometry; (4) patients with unsatisfactory medication and whose quality of life is obviously affected, unwilling to take medicine for treatment for a long time or cannot take medicine due to side effects; (5) patients refusing surgical treatment.
2. Contraindications: (1) patients with severe heart and lung disease who cannot undergo gastrointestinal endoscopy; (2) patients with suspected perforation of the digestive tract; (3) patients with acute corrosive digestive tract infection; (4) patients refusing this therapy.
(II) Preoperative preparation
The patient must be fully informed of the conditions, procedure and risks, sign the surgery related informed consent form, fast for 8 h before the operation.
(III) Surgical operation and skills
1. Anesthesia and position. The patient is at deep intravenous anesthesia state, care must be taken to keep him/her warm, room and infusion temperatures should be adjusted, pressure sores should be avoided. Refer to the Expert Consensus Opinion on Digestive endoscopy for Sedation/Anesthesia in China.
2. Surgical operating steps (figure 8.26)
(1) Circumferential disinfection
1) The top position of gastroesophageal junction and barrette esophagus must be endoscopically recorded; the endoscope must be withdrawn after insertion of the guide wire.
2) The measuring balloon catheter must be inserted using a guide wire, the inner diameter of the esophagus must be endoscopically measured. The measurement begins 2 cm from the focus oral-side end, the interval is 2 cm, until passing the cardia.
3) The balloon ablation catheter with the minimum diameter according to the measured inner diameter of the esophagus must be selected.
4) The endoscope must be inserted after placing the balloon ablation catheter using the guide wire; it should be endoscopically determined that the ablation catheter is at the lesion’s site. After filling the balloon, the ablation therapy is conducted by a radiofrequency ablation generator that transmits radiofrequency energy to the ablation catheter.
5) After the first ablation therapy, the guide wire, the ablation catheter and the endoscope are withdrawn, the balloon ablation catheter is rinsed so as to remove coagulants.
6) The above steps are repeated to carry out the ablation again.

FIG. 8.26 — Schematic diagram of peroral super minimally invasive radiofrequency ablation for gastroesophageal reflux disease. A. Observe the site of gastroesophageal junction. B. Insert the measuring balloon catheter by guide wire. C. Insert the balloon ablation catheter. D. Conduct the radiofrequency therapy after balloon inflation. E. Treatment by points. F. Withdraw the guide wire after treatment. Pictures come from Internet: Methods of endoscopic therapy of gastroesophageal reflux disease _Stretta (sohu.com).
(2) Focal ablation (figure 8.27)
1) The focal ablation catheter is placed at the endoscopic head end and inserted into the esophagus with the endoscope. The ablation catheter is generally located at 12 o’clock in the endoscopic field.
2) The ablation catheter is closely attached to the lesion membrane to initiate the ablation therapy.
3) The necrotic membranes of the ablation surface are endoscopically rinsed.
4) The above steps are repeated to carry out the ablation again.
5) If there are multiple foci need ablation therapy, the endoscope should be intermittently withdrawn to rinse the ablation catheter so as to remove necrotic coagulants.
(IV) Postoperative nursing
(1) The patient should fast for food for 24 h after the operation, be administered an appropriate intravenous fluid supplement, and gradually resume diet according to the conditions. Postoperative acid suppression therapy will be suggested. The proton pump inhibitor at the routine dose can be used, twice each day, for two weeks.
(2) For patients with postoperative pain, 500–1000 mg of paracetamol can be given according to needs, not exceeding four times each day. For patients whose pain is not relieved by paracetamol, 50 mg of diclofenac can be added, without exceeding twice each day. If the occurrence of other serious complications is suspected, related examinations should be performed timely (e.g., emergency gastroscope, X-ray filming or CT).

FIG. 8.27 — Images of peroral super minimally invasive radiofrequency ablation for gastroesophageal reflux disease. A, B. Endoscopic radiofrequency therapy under direct vision. C. After endoscopic radiofrequency therapy under direct vision. D. Endoscopic image after traditional non-direct radiofrequency therapy.
(V) Hot-spot issues in the consensus and progress of the study
1. Endoscopic radiofrequency therapy has positive effects, with the patient’s postoperative GERD symptoms clearly alleviated. RF endoscopic therapy is a minimally invasive endoscopic treatment approach of GERD that is effective, safe, durable and reproducible, and does not conflict with other GERD treatment methods. It can still be performed after the failure of fundoplication, which improves various clinical indicators in GERD patients in the short term. For example, a meta-analysis that included 28 studies (on a total of 2468 patients) suggested that heartburn scores, incidence of invasive esophagitis, and esophageal acid exposure were all reduced after Stretta radiofrequency therapy: 51% of the patients no longer used PPIs after surgery, and the patients’ quality of life was improved. However, there was no statistically significant increase in LES pressure. Another eight-year follow-up study of 26 patients receiving Stretta radiofrequency therapy showed that all patients had significantly lower heartburn symptoms and GERD HRQL scores, significantly higher scores of quality of life, and that 76.9% of patients stopped using PPIs completely.
2. The long-term efficacy of endoscopic radiofrequency ablation is to be verified. There is currently still a lack of controlled trials of endoscopic radiofrequency therapy on a large sample and of comparisons with other anti-reflux surgeries to determine its long-term efficacy.
8.5.3 Peroral Incisionless Fundoplication for Gastroesophageal Reflux Disease
Transoral incisionless fundoplication (TIF) applies a special in-cavity device to conduct gastroesophageal folding without surgical incisions, which reduces the frequency of TLESR, lowers the dilation degree of the gastroesophageal junction, and effectively controls reflux symptoms. There are two devices for performing TIF, Esophyx and MUSE, respectively.
TIF using the EsophyX® device was approved by the U.S. Food and Drug Administration in 2007 for the endoscopic treatment of GERD, and more than 25 000 procedures have been performed to date. The device is designed to generate the folding of the full serous membrane, and to reconstruct valves with a length of about 3 cm and a circumference of about 270–300 degrees on the basis of the surgical principles of laparoscopic fundoplication. In the current iteration of the technique (TIF 2.0), the procedure is anatomically and physiologically similar to surgical fundoplication. During the procedure, the fundus of the stomach is folded up and around the distal end of the esophagus, which has retracted beneath the diaphragm and is held in place with polypropylene fasteners. This causes the sling fibers to be tightened and strengthened at the proximal stomach (the lower part of the LES), highlighting the incision of the cardia, making the angle steeper, and reestablishing the valve mechanism.
(I) Indications and contraindications for performing TIF by using Esophyx
1. Indications. GERD patients with esophageal hiatal hernia with objective reflux evidence and without obvious abnormalities of the structural anatomy of the esophagogastric junction (e.g. >2 cm), especially patients who respond to PPI therapy but cannot tolerate the long-term use of drugs.
2. Contraindications. (1) Body mass indicator (BMI) >35 kg/m
(II) Surgical operation and skills
1. Anesthesia and position. All patients undergo general anesthesia with tracheal intubation, lying on their left side; attention should be paid to keep them warm, adjusting the room temperature and the infusion temperature, and avoiding pressure sores.
2. Operating step (figure 8.28)
(1) A diagnostic endoscopy must be carried out to specify the anatomical features of the diaphragmatic foot, whether there is a hiatal hernia and its size, and determine whether there are malignant or other abnormal mucosal changes.
(2) The endoscope will be inverted, and the molder will be endoscopically inverted at the device’s front end; the gastroesophageal junction will be fixed with a spiral traction needle, and pulled back to pull the tissues between the molder and the tubular restorer; the molder will be tightened to make the lower esophagus close to the gastric fundus; the puncture needle and the reinforcement will be passed through the side hole of the repair tube, through the muscular layer and the tightly attached esophagus and gastric wall, whereas the H-fixator will be released to form the fold 2–3 cm above the gastroesophageal junction.
(3) The endoscope should be rotated from different angles using the Esophyx device to form the 200°–270° fundus folding. Suction of the air in the stomach during operation will be helpful to clamp the tissue tightly. After the operation is over, an endoscopic observation will be carried out to ensure that the fold is sufficient and to detect any perforation or hemorrhage.
(III) Hot-spot issues in the consensus and progress of the study
1. TIF is an effective method for refractory GERD endoscopic therapy. Fundoplication is currently considered the best anti-reflux surgery for GERD patients who do not want to use PPIs for a long period of time. A meta-analysis integrating 20 RCTS (1892 patients) suggested that fundoplication of different procedures improved heartburn and reflux symptoms better than PPIs. TIF is regarded as the effective means for refractory GERD endoscopic therapy. It is currently admitted that TIF 2.0 is a safe and effective therapeutic method for GERD patients whose Hill grading < grade 3; its efficacy and safety have been confirmed in multiple studies. A meta-analysis integrating randomized controlled trials of 233 patients showed that, compared to pseudo treatment or PPI therapy, the changes in the patients’ condition three years after TIF surgery were significant in terms of esophageal pH, lowered PPI use ratio, and quality of life. Another follow-up study after five years of TIF surgery found that in 63 patients, the reflux symptom had disappeared in 86% of them, whereas 46% patients had totally stopped using PPIs, and that the total score related to GERD had been reduced from 22.2 to 6.8. This demonstrates that TIF is a safe and long-term cure for troublesome GRED symptoms.
2. TIF’s long-term effect is still not validated. Currently, most of the research results on TIF are compared with data relating to PPI treatment, and comparative studies with gold standard laparoscopic Nissen fundoplication and long-term follow-up studies with large samples are still lacking. A clinical study of 151 TIF2.0 patients who underwent nine years of postoperative follow-up showed that GERD symptoms had been continuously alleviated, with 69%–80% of patients having reduced their treatment and proton pump inhibitor intake.

FIG. 8.28 — Peroral incisionless fundoplication (TIF). The gastroscope is inverted to observe the valves at the gastroesophageal junction. B. Fundoplication by using the TIF device. D. Gastroesophageal valves after TIF reconstruction.
8.5.4 Peroral Super Minimally Invasive Mucosa Resection for Gastroesophageal Reflux Disease
For transoral super minimally invasive mucosa resection for gastroesophageal reflux disease, the endoscopic therapeutic method applied is antireflux mucosa resection (ARMS), which has been developed on the basis of ESD, EMR, and the litigation technique. This technique was proposed and named by a Japanese scholar, and is carried out mainly by making a mucosal incision in the antireflux barrier in the cavity (about 3 cm on or off the dentate line, about 2/3 of the circumference), inducing the formation of wound scar tissue, promoting GEFV function reconstruction by hyperplasia and contraction of scar tissues, increasing LES pressure, which effectively plays the role of antireflux, meanwhile avoiding stenosis resulting from mucosal circumferential incision.
(I) ARMS’s indications and contraindications
1. Indications: (1) DeMeester score >15 in 24 h of continuous esophageal pH monitoring; (2) acid exposure time (AET) (PH < 4) > 6%.
2. Contraindications: (1) when the disease is combined with hiatus hernia (>2 cm) and esophageal manometry indicates abnormalities in esophageal motility (e.g., achalasia or LE relaxing insufficiency); (2) pregnant women; (3) patients who have been taking oral anticoagulants for a long time.
(II) Preoperative preparation and postoperative treatment
The patient must be fully informed of the conditions, procedure and risks, sign the surgery related informed consent form, fast for 8 h before the operation.
(III) Surgical operation and skill
1. Anesthesia and position. All patients undergo tracheal intubation and general anesthesia, left-lying position, pay attention to keeping warm, adjusting room temperature and infusion temperature, avoid pressure sores.
2. Operating step (figure 8.29)
(1) Mucosal marking along lesser curvature side of the cardia.
(2) The mixed solution of methylene blue +0.9% NaCl is injected at multiple labeled points along the lateral mucosa to fully lift the mucosa.
(3) The endoscope must be inverted, and EMR or ESD be performed along the lesser curvature of the cardia, the mucosa must be incised in a crescent shape over 1/2–3/4 of the circumference, with the cardiac valves on the side of the greater curvature of the cardia being preserved. The length of the incision is approximately 3 cm, with the EGJ as the limit, including the esophageal side (1 cm) and the gastric side (2 cm).
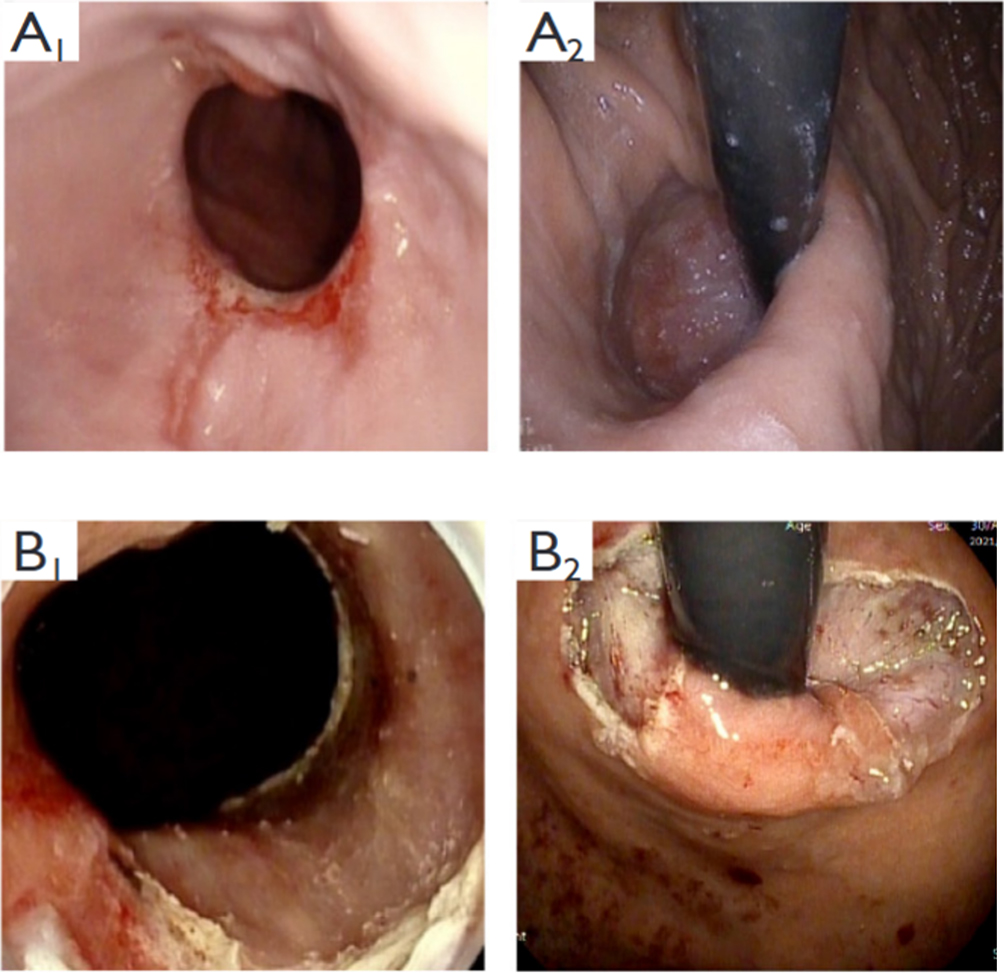

FIG. 8.29 — Peroral super minimally invasive mucosa resection for gastroesophageal reflux disease. A1, A2. ARMS preoperative endoscopic images. B1, B2. ARMS postoperative endoscopic images.
(IV) Postoperative treatment
The patient should fast for food and water for three days, be given treatments like acid suppression, fluid nutritional supplementation, etc.; then he/she must follow a gradual transition diet: liquid diet → semiliquid diet → full diet, eat liquid food for one week; postoperatively, continue to take oral PPIs for 14 days; avoid spicy foods for one month after the operation.
(V) Hot-spot issues in the consensus and progress of the study
1. ARMS is a safe and effective method for treating refractory GERD. ARMS is a safe and effective method for treating refractory GERD, with higher clinical reaction rates, acceptable safety and significantly improved GERD-related quality of life. Inoue’s first study (2014) on 10 patients with refractory GERD having undergone ARMS found that their heartburn scores and reflux scores had been significantly reduced, and that all patients had been able to stop PPI intake without adverse effects. In a meta-analysis including 10 studies (307 patients), Garg et al. found that the technical success rate and clinical response rate of ARMS were 97.7% and 80.1%, respectively. Compared to the situation before the operation, the GERD health-related quality of life (GERD-HRQL), the GERD questionnaire (GERD-Q) and the mean acid exposure time had decreased significantly after ARMS. A prospective study of 23 patients with chronic PPI-dependent GERD receiving ARMIDE (11 ARMS and 12 ARMA) found that 73.9% of patients reported subjective overall improvement. The total reflux symptom index score, esophageal acid exposure time, acid reflux times and DeMeester score were significantly reduced. Three patients (13%) had increased AET (3.4%–6.1%, 6.3%–15.4%, 3.2%–5.6%); however, all patients were reported as having had an overall improvement, two patients had been able to subjectively stop PPI intake. One patient (4.3%) saw his/her corrosive esophagitis and reflux symptoms worsen. 56.5% of patients stopped using PPIs. This suggests that ARMI is generally effective and safe in the treatment of PPI-dependent patients.
2. The long-term efficacy of ARMS is unclear, and there are still some issues that need to be addressed. In case of proper selection of patients, the application prospects of ARMS are optimistic due to its significant therapeutic effects, good safety and surgical simplicity, but its long-term safety and efficacy need to be verified by large-scale prospective studies. There are still some problems to be solved for ARMS, such as determination of indications, the extent of intraoperative mucosal resection and the normalization of the operation process.
8.5.5 Clip Band Ligation Anti-Reflux Therapy for Gastroesophageal Reflux Disease
Clip band ligation anti-reflux therapy for gastroesophageal reflux disease (C-BLART for GRED) is a technique of peroral ligation at the near end of the gastroesophageal junction by fixing the mucosa and the muscle layer using metal clips to form a fold, subsequently causing ischemia, necrosis, exfoliation, repair of the tissues in this area, after which scars are formed, thus decreasing the diameter of the cardia.
C-BLART has been reported as one of the new therapeutic methods for replacing endoscopy for refractory GERD. The six-months effective rate for treating GERD is 74.6%. And compared to routine PPI medication, symptoms like sour regurgitation, heartburn, LES pressure and acid exposure time were obviously relieved. And it has the advantages of simple operation, less time of intervention (average time is only 8 min), low cost, repeatability, high safety, which is favorable to clinical promotion and use.
(I) Indications and contraindications of super minimally invasive C-BLART
1. Indications: (1) Refractory gastroesophageal reflux disease (GERD), it is defined as persistent symptoms of reflux and heartburn showing no significant improvement after 8 weeks of double-dose proton pump inhibitor (PPI) therapy, with objective confirmation of symptom-correlated reflux evidence through endoscopic evaluation, high-resolution esophageal manometry, and impedance-pH monitoring. (2) unwilling to receive medication, with strong wish of surgery.
2. Contraindications: (1) esophageal ulcer or stenosis; (2) early esophageal carcinoma, precancerous lesions of precancerous lesions such as Barrett’s esophagus larger than 3cm, etc.; (3) serious reflux esophagitis (grade LA-C, LA-D); (4) esophageal motility diseases like achalasia diffuse esophageal spasm; (5) when associated with an autoimmune disease; (6) with overall anesthesia contraindication; (7) patients with contraindications to gastroscopy and treatment by gastroscopy.
(II) Preoperative preparation
The patient must be fully informed of the conditions, procedure and risks, sign the surgery related informed consent form, fast for 8 h before the operation.
(III) Surgical operation and skills
1. Anesthesia and position. All patients undergo general anesthesia with tracheal intubation, lying on their left side; attention should be paid to keep them warm, adjusting the room temperature and the infusion temperature, and avoiding pressure sores.
2. Operating steps (figure 8.30)
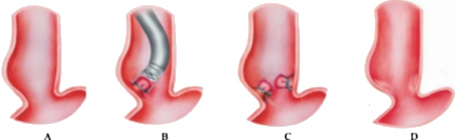

FIG. 8.30 — Schematic diagram of super minimally C-BLART. A. Cardia’s relaxing state before operation. B. The ligation ring is released to contract the cardiac mucosa. C. Clips are used to fix the root of the elevated mucosa. D. Postoperative constriction of the cardia.
(1) Sucking and ligation: the ligation device must be installed at the front-end of the gastroscope, perorally enter the lower esophagus; the distance from gastroesophageal junction to incisor must be specified, then endoscopic suction by negative pressure of the cardiac mucosa in the 6 o’clock and 12 o’clock directions towards the cap must be carried out, with ligation of the rubber ring to the mucosa using an appropriate device (figure 8.31).
(2) Root fixation: disposable metal soft tissue clips must be fixed at the root of the ligation ring (figure 8.32).
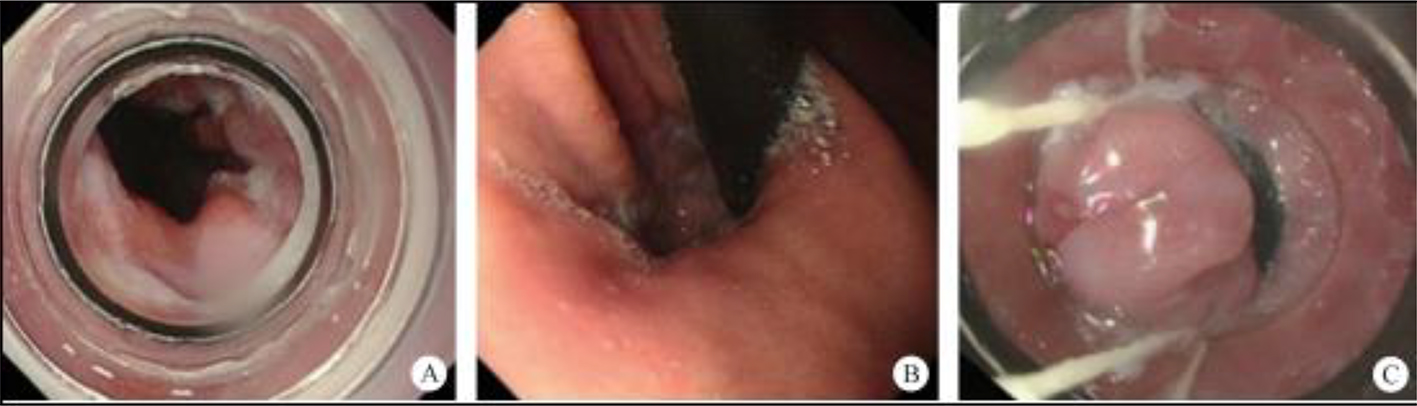

FIG. 8.31 — Sucking and ligation steps of peroral super minimally invasive cardiac constriction for gastroesophageal reflux disease. A. Cardia’s relaxing state before operation; B. Hiatal hernia at the cardia (inverted view); C. The ligation ring is released to contract the cardiac mucosa.
(IV) Postoperative treatment
The patient should fast for food and water for three days, be given treatments like acid suppression, fluid supplement, nutrition support, etc.; then he/she must follow a gradual transition diet: liquid diet → semiliquid diet → full diet, eat liquid food for one week; postoperatively, continue to take oral PPIs for 14 days; avoid spicy foods for one month after the operation.
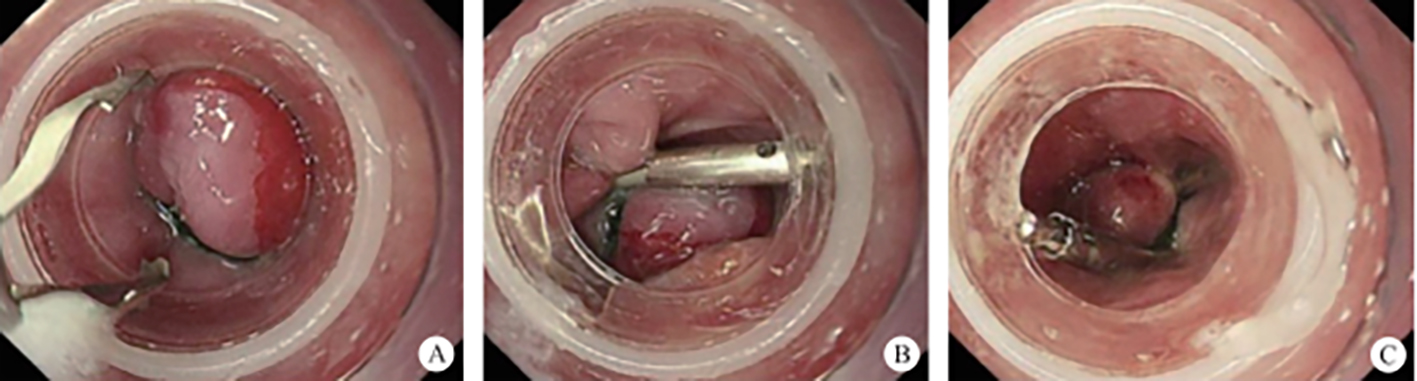

FIG. 8.32 — Root fixation steps of peroral super minimally invasive cardiac constriction for gastroesophageal reflux disease. A. Ready-to-release clips. B. Clips fixing the root of the elevating mucosa. C. Two clips placed at the level of the cardia.
(V) A classic case
History: the patient, a 36-year-old woman, was admitted to the hospital due to “intermittent sour regurgitation, heartburn for more than 10 years.” Ten years before, with no obvious inducing factors, the patient presented with sour regurgitation, heartburn, pain behind the sternum, with occasional cough, foreign body sensation in the pharynx, worsening after eating sweets, leading to visiting a local hospital where considered gastroesophageal reflux disease was considered. After giving omeprazole orally for one day, her symptoms alleviated. After stopping, sour regurgitation, heartburn symptoms reoccurred.
Examination results after admission: gastroscopy showed that the lamellar mucosa was rough, red and borderless on the anterior wall of the cardia; 24-h pH results showed esophageal pH-resistance. The results of the monitoring were consistent with gastroesophageal reflux disease, the possibility of heartburn symptoms was 85.7%, indicating acid regurgitation. DeMeester score 102.8. The motility monitoring results were as follows: LES low pressure, with weakened motor function of the esophageal body; GERD-Q score 23 points, GERD-HRQL score 18 points, HAD scale score 7 points in the past 3 months.
Diagnosis: gastroesophageal reflux disease.
After full preoperative evaluation and having signed the surgery informed consent form, the patient has undergone C-BLART therapy (Refer to video 8.8, Super minimally invasive cardiac constriction for gastroesophageal reflux disease: Cardiac orifice narrowing via Band Ligation and Additional Reinforcement Techniques (See online resources)).
(VI) Hot-spot issues in the consensus and progress of the study
1. Peroral endoscopic cardiac constriction still needs further exploration. According to the present study results, C-BLART significantly improved GERD patients’ sour regurgitation degree, but presently there is no specific detailed technical details yet, particularly the depth and times of ligation, etc. Therefore, the impacts of ligation depth, location, time, etc., still need to be studied in order to improve efficacy.
2. The short-term curative effect is good for endoscopic anti-reflux mucosal treatment, but long-term efficacy still needs verification. Endoscopic anti-reflux mucosal treatment is simple and easy to operate, mainly including anti-reflux mucosal resection, anti-reflux mucosal ablation and C-BLART. A meta-analysis showed that the clinical success rates of anti-reflux mucosal resection, anti-reflux mucosal ablation, and C-BLART were 68.6% (95%CI 62.2%–74.4%), 86.7% (95%CI 78.7%–91.9%), 76.5%, respectively. And acid exposure time, DeMeester score and hiatal hernia degree were significantly improved within one year. But currently research reports on the long-term therapeutic effects and verification on large samples are lacking. So far, the PLA General Hospital has taken the lead in the implementation in China of a study on the treatment of multi-centerperoral endoscopic cardiac constriction.
8.6 Super Minimally Invasive Peroral Myotomy of Esophageal Diverticula
An esophageal diverticulum is one layer or the entire layer of the cystic protrusion of the esophageal cavity on the wall of the esophagus which is identical to the esophageal cavity. Esophageal diverticulum is a rare disease that causes difficulty in swallowing, regurgitation, and chest pain as it progresses. Esophageal diverticula can be divided into three categories according to its location in the esophagus: (1) Zenker’s diverticula, which are the most common esophageal diverticula, located at the pharyngeal-esophageal junction; (2) diverticula of the esophageal midpiece, located at median esophagus; (3) paradiaphragmatic diverticula, just above the diaphragm.
Peroral super minimally invasive myotomy of esophageal diverticula is carried out by submucosal tunneling endoscopic septum division (STESD). STESD is the latest super minimally invasive surgery based on peroral endoscopic cardiomyotomy (POEM), a technique that allows full exposure and an incision in the diverticular ridge of the normal muscular layer of the esophagus while maintaining mucosal integrity. STESD is a safe and effective endoscopic minimally invasive procedure for the esophageal diverticulum that reduces symptoms and improves the quality of life.
8.6.1 Diagnosis of Esophageal Diverticula
An esophageal diverticulum often causes difficulty in swallowing, coughing, anorexia, bad breath, chest pain, heartburn, bucking, or weight loss. Esophageal diverticula are usually diagnosed by endoscopy or an X-ray barium swallow test. Because of its safety, simplicity, and effectiveness, the X-ray barium swallow test is usually superior to endoscopy for diagnosis.
8.6.2 STESD’s Indication and Contraindication
1. Indications: presence of obvious clinical manifestations of esophageal diverticula and difficulty in swallowing as well as endoscopy and X-ray barium swallow test results indicating an esophageal diverticulum.
2. Contraindications: (1) General anesthesia contraindication; (2) Patients with contraindications for gastroscopy examination and treatment; (3) asymptomatic, smaller diverticulum.
8.6.3 Preoperative Preparation
1. Anesthesia and position. All patients undergo general anesthesia with tracheal intubation, lying on their left side; attention should be paid to keep them warm, adjusting the room temperature and the infusion temperature, and avoiding pressure sores.
2. Operating steps
(1) Establishment of the tunnel entrance (figure 8.33): a submucosal injection is conducted at 3 cm above the diverticular septum; the tunnel entrance is established by incising the mucosal layer and exposing the submucosal layer.
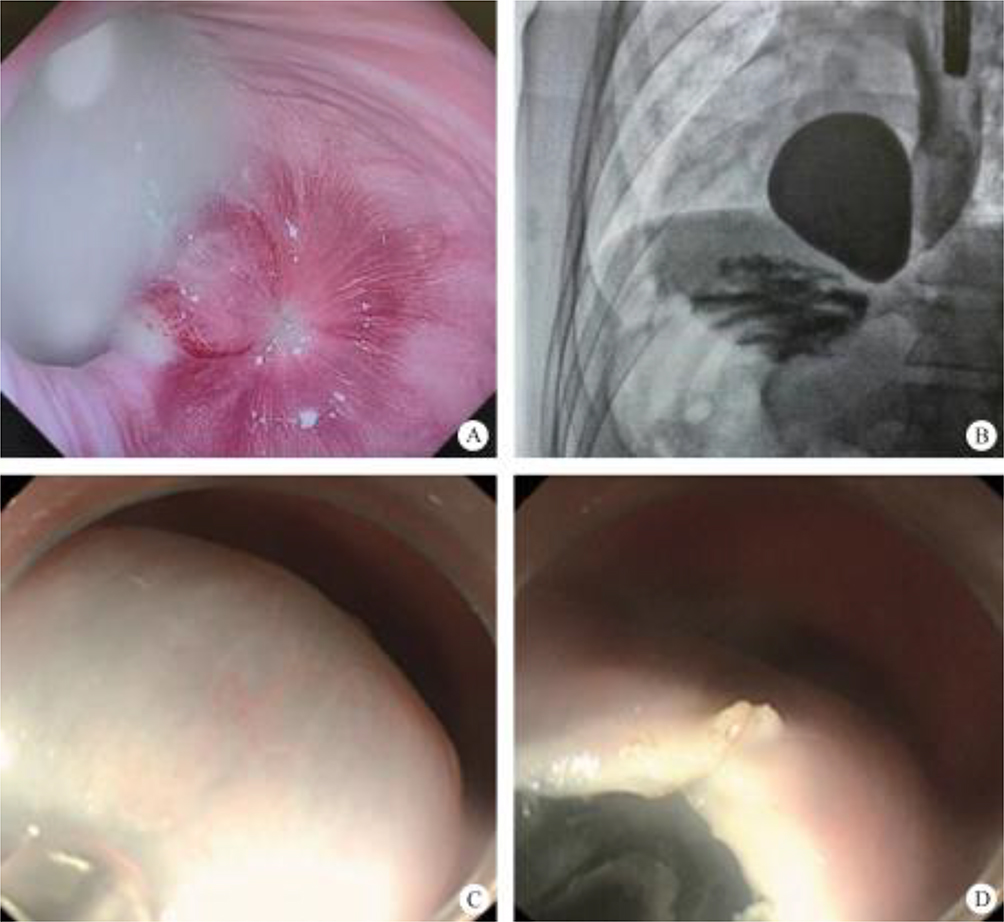

FIG. 8.33 — The steps for establishing the tunnel entrance in peroral super minimally invasive myotomy for esophageal diverticulum. A. Fluid retention observed in a lower esophageal diverticulum. B. Esophageal diverticulum found by contrast. C. Submucosal injection was performed 3cm above the diverticulum; D. Creation of the tunnel entrance by an inverted T-shape incision.
(2) Submucosal tunnel (figure 8.34): by using the endoscopic mucosal dissection technique, a longitudinal tunnel is established by submucosal dissection between the mucous and muscle layers. The tunnel is constructed on both sides of the diaphragm and terminates 1–2 cm from the bottom of the diverticulumand ends at 1–2 cm from the diverticular base, to ensure satisfactory endoscopic observation and enough space for myotomy. Preventive measures must be taken to avoid injuring membranes covered on it during tunnel dissection.
(3) Transverse ridge (figure 8.35): interdiverticular ridges are completed and exposed, the muscle layer is crossed endoscopically to the diverticular base in the middle of the ridges using a knife.
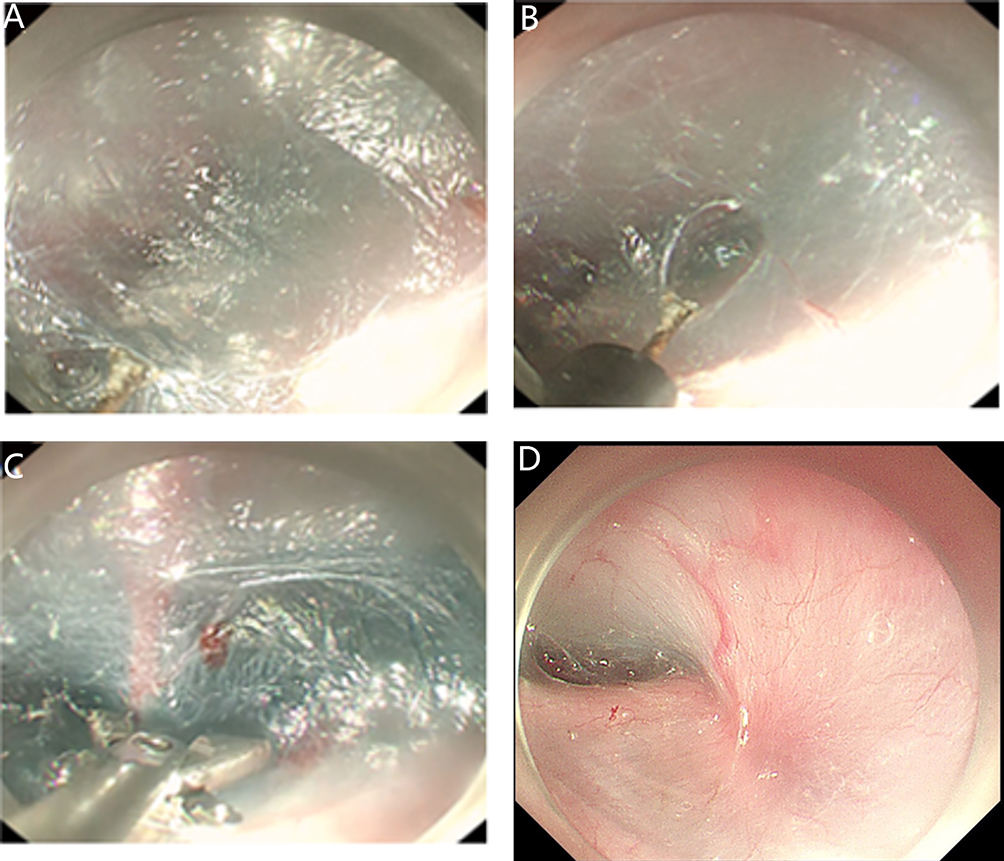

FIG. 8.34 — In-tunnel dissecting steps of peroral super minimally invasive myotomy of an esophageal diverticulum. A. The tunnel is entered by the cap against the mucosal valves along the incision. B. Dissection near the intrinsic muscle layer inside the tunnel. C. Electrocoagulation hemostasis of the thicker vessels. D. Exposed interdiverticular ridges.
(4) The mucosa is incised with metal clamps: once the liquids in the ‘tunnel’ and the esophageal lumen have been completely sucked, the wounds must be rinsed, bleeding spots and small vessels must be electrocoagulated, and the surface mucosa of the ‘tunnel’ must be checked. The endoscope musty be withdrawn through the tunnel’s opening, and the “tunnel entrance” must be occluded by metal clips. After the operation, the dilated esophageal lumen appears without interdiverticular ridges.
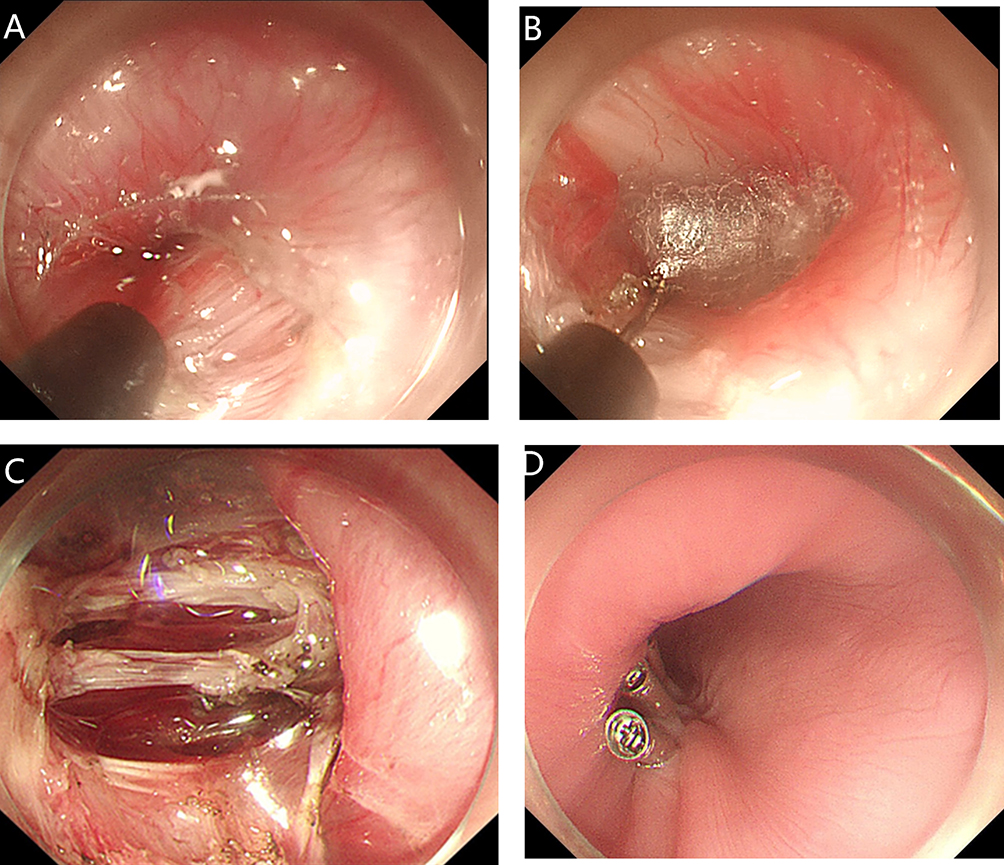

FIG. 8.35 — Steps of muscle layer incision in peroral super minimally invasive myotomy for an esophageal diverticulum. A. The mucosa is separated at both ends, the diverticular ridge is exposed. B. Transverse incision made at the esophageal ridge. C. The incised diverticular ridge. D. Closed tunnel entrance.
(5) Postoperative nursing
After the operation, the patient should fast for food and water, be given treatments like acid suppression, fluid nutritional supplementation, etc., for three to five days, with preventive administration of antibiotics for symptomatic treatment; then he/she must follow a gradual transition diet: liquid diet → semiliquid diet → full diet; postoperative PPIs and a gastric mucosa protective agent will be administered for two months.
(6) A classic case
Case I. Esophageal Zenker’s diverticulum
History: the patient, an elderly man, was admitted into the hospital mainly due to “choking on food for two years, upper full abdominal distension for half a year and discovering esophageal diverticulum for more than 40 days.” Two years before, with no obvious inducing factors, the patient presented with choking feeling after eating solid and dry foods; it could feel better after occasionally vomiting foods, had no discomfort after taking a liquid diet, a semiliquid diet, with no obvious reduction in intake, which he did not pay much attention to. Half a year before, after eating, he presented with upper abdominal full distension after eating foods, with hiccup, without obvious nausea and vomiting, no abdominal pain, venting and passing stool were averagely smooth, with no obvious reduction in eating and weight change. More than 40 days before, the local hospital CT examination showed an esophageal diverticulum; further gastroscopy indicating a giant diverticulum at the upper esophagus, food residue remaining. The patient underwent endoscopic removal of food debris from the esophageal diverticulum, followed by dietary modification to a liquid regimen. While symptomatic improvement was observed in both dysphagia and abdominal distension, persistent postprandial dysphagia, bloating, and hiccups recurred following the consumption of solid foods or large meal volumes. An upper gastrointestinal angiography revealed an upper esophageal diverticulum, gastroesophageal reflux, and gastroptosis. In the past, he suffered from pulmonary emphysema, trachitis, and leukoderma. No abnormalities were found during the physical examination for admission.
Diagnosis: esophageal diverticulum (Zenker’s diverticulum).
After full preoperative evaluation, improving nutrition state and signing the surgery informed consent form, the patient was performed peroral endoscopic super minimally invasive myotomy for Zenker’s diverticulum (Refer to video 8.9, Per-tunnel super minimally invasive myotomy for esophageal Zenker’s diverticulum (See online resources)).
Surgical process: the giant Zenker’s diverticulum is found at the entrance of the esophagus after entering the endoscope, and the diverticular esophageal ridge is at 18 cm from the incisor. After submucosal injection at the esophageal ridge, the submucosal tunnels to the diverticular base and the esophagus are built by a triangle knife. During the tunnel creation process, due to limited operating space at the esophageal entrance, the tunnel is built while myotomy is conducted. The tunnel is built at the diverticular base, vessels are treated with a hemostat, while continuing to incise the muscle layer of the diverticular ridge. After the diverticular ridges are fully cut, the tunnel entrance was occluded with a hemostat. The diverticulum has basically disappeared. The endoscope is entered again; it is withdrawn once it has been confirmed that there were no abnormalities in the esophageal lumen.
Case II. Achalasia with lower esophageal diverticulum
History: the patient, a male adolescent, was admitted into the hospital due to “difficulty in intake and swallowing for three years.” Three years before, due to the repetitive occurrence of vomiting after too fast intake, gastroscopy was performed, showing hiatus hernia and achalasia, considering a giant lower esophageal diverticulum after upper gastrointestinal angiography. In the past, he suffered from congenital autism and epilepsy.
Examination results after admission: gastroscopy showed a diverticulum in the lower esophagus up to the dentate line, inside which was a large amount of liquid, food residue and drug tablets; the cardia was 40 cm from the incisor, there was resistance when the general endoscope passed the cardia; mediastinum CT scan + enhancement indicating dilation of the lower esophagus, consistent with achalasia.
Diagnoses: achalasia; esophageal diverticulum.
After full preoperative evaluation and signing the surgery informed consent form, the patient was performed achalasia POEM surgery and STESD surgical therapy for giant esophageal diverticulum (Refer to video 8.10, Per-tunnel super minimally invasive myotomy for lower esophageal diverticulum (See online resources)).
Surgical process: Upon examination, the esophagus is fully dilated, the cardia is closed, and a large diverticulum can be seen from the lower esophagus to the dentate line with a large amount of food and fluid retention. A submucosal injection was made at 3–5 cm above the diverticulum, the submucosal tunnel was created to the diverticular base and the esophagus with a triangle knife, vessels were treated with a hemostat, the muscle layer of the diverticular ridge was fully incised, the tunnel’s opening was occluded with a hemostatic clip.
After a postoperative fast for food and water, with intravenous acid suppressors and fluid nutritive supplementation for five days, with administration of Supraxen + Ornidazole prophylactically against infection for two days, the patient gradually resumed a normal diet.
(7) Hot-spot issues and progress of the study
Zenker’s diverticulum, also known as pharyngoesophageal diverticulum or pharyngeal pouch, is an acquired pulsion pseudodiverticulum of the hypopharynx, as the nomenclature implies. This protrusion characteristically occurs at Killian’s triangle (the anatomical weak zone between the oblique fibers of the thyropharyngeus muscle and horizontal fibers of the cricopharyngeus muscle), resulting from elevated intraluminal pressure during swallowing against a dysfunctional upper esophageal sphincter. The increased pressure in the lumen causes the esophagus to flow out between two strong pharyngeal muscles—the cricopharyngeal muscle and the hypopharyngeal contractile muscle. Posterior cystic formation occurs when the tension of the hypopharyngeal wall in this area decreases and the pressure in the lumen increases.
The diverticulum in the middle of the thoracic esophagus is classified as Rokitansky’s diverticulum and is formed by the contraction of the mark caused by chronic inflammatory and fibrotic state that pulls the wall of the esophagus outwards. Therefore, they involve the entire esophageal wall (real diverticulum). The apex of the pouch is usually located at a point higher than its entry point, so it rarely produces a noticeable size or produces any symptoms. Rokitansky’s diverticulum has close association with tuberculosis. In contemporary times, the uncontrollable TB cases are very few, and just because of that, Rokitansky’s diverticula with clinical symptoms are rare.
Paradiaphragmatic diverticula are pulsating diverticula, usually located in the distal esophagus. In histology, they are identified as pseudo diverticula lacking a muscle layer. Their pathogenesis is thought to be secondary to esophageal dyskinesia and related to congenital esophageal wall weakness. According to research reports, most para diaphragmatic diverticula are associated with esophageal dyskinesia. Therefore, before deciding on intervention measures, high-resolution manometry (HRM) is recommended to evaluate esophageal motility. A paraprenic diverticulum is more likely to occur on the right side of the esophageal wall, the reason for which being unclear. On the contrary, spontaneous esophageal rupture often occurs through the left wall.
8.7 Super Minimally Myotomy for Achalasia
8.7.1 Introduction
(I) Introduction to achalasia
Achalasia (AC) is a benign esophageal motility disorder, with the characteristics of abnormal esophageal peristalsis; the lower esophageal sphincter (LES) is unable to relax due to the loss of esophageal plexus ganglion cells. The impaired movement push from the esophagus to the stomach can lead to symptoms such as dysphagia, reflux, chest pain, and weight loss. The incidence of achalasia is estimated to be 1.07–2.92 cases/(100 000 persons per year). Autoimmunity, genetic factors and the role of viruses have been regarded as possible causes, but the specific pathogenesis is still unknown.
(II) Diagnosis of achalasia
The initial symptoms of esophageal achalasia are similar to those of other esophageal diseases, such as dysphagia, reflux with weight loss, and require timely endoscopic examination to rule out stenosis and neoplastic esophageal disease. The accuracy and specificity of the diagnosis of achalasia by endoscopy alone need to be improved, and the diagnosis of achalasia requires esophageal manometry. Diagnostic manometry of achalasia is characterized by incomplete relaxation of the LES (integrated relaxation pressure (IRP) greater than the normal upper limit) and the absence of peristalsis at 2/3 of the distal esophagus. For patients with inconclusive esophageal manometry, esophageal barium test is helpful for evaluating esophageal emptiness and EGJ morphology. In addition, measuring EGJ dilatation using a functional lumen imaging probe (FLIP) may also help clarify outcomes in these patients.
According to the Chicago Classification version 4.0 (CC-4) of the esophageal pressurization model demonstrated by high-resolution manometry (HRM), achalasia is classified into the following three subtypes:
Type-I (typical) achalasia: swallowing does not cause significant changes in esophageal pressurization. According to the CC-4 standard, type-I achalasia is a total (100%) absence of peristalsis, and is characterized by an index of distal esophageal contractility (DCI) <100 mmHg.
Type-II achalasia: swallowing causes full esophageal pressurization. According to the CC-4 standard, type-II achalasia is characterized by a 0% absence of peristalsis, and a full esophageal pressure ≥20% after swallowing.
Type-III (spastic) achalasia: swallowing causes premature contractions or spasms that usually lead to closure of the lumen. According to the CC-4 standard, type-III achalasia is characterized by an absence of normal peristalsis, and the presence of ≥20% distal latency <4.5 s and a DCI > 450 mmHg·s·cm of too early (spastic) contraction after swallowing.
(III) Techniques for the implementation of per-tunnel super minimally invasive myotomy for achalasia
The technique applied in per-tunnel super minimally invasive myotomy for achalasia is peroral endoscopic myotomy (POEM), whose methods can be refined. For example, annular myotomy, full-thickness myotomy, progress full-thickness myotomy, glass-type myotomy, peroral endoscopic annular myotomy combined with balloon plastics as well as simultaneous submucosa and muscularis propria myotomy. According to the length of the selected tunnel, it can be divided into short-tunnel POEM and long-tunnel POEM.
8.7.2 Per-Tunnel Super Minimally Myotomy for Achalasia
Per-tunnel super minimally myotomy for achalasia is carried out by peroral endoscopic myotomy (POEM). POEM is a super minimally invasive technique allowing myotomy by a tunnel endoscopic technique for treating achalasia. This surgical method does not require thoracotomy, and the entire surgical procedure is carried out by gastroscopy in a painless state, with a short operation time, small wounds, quick recovery of the patient, who can take food on the day after the operation. The biggest advantage of POEM is that it can basically keep the integral esophageal physiological function, with very low incidence of postoperative complications like reflux esophagitis. With the continuous improvement of endoscopic techniques, POEM has basically replaced the traditional dilation and stent implantation, and has become the first-choice therapy for treating AC. This surgery not only largely overcomes the uncertainty of the efficacy of traditional treatments, but also offers quick recovery and reliable curative effect, fully demonstrating the advantages of super minimally invasive therapy.
(I) Preoperative preparation
The patient must be fully informed of the benefits and risks of POEM in detail, and sign the surgery related informed consent form. Anesthesia evaluation must be refined. A preoperative fast of 24–48 h, a water fast of 6 h and a gastroscopy on the day of the operation are required. A normal saline solution is used to rinse the lumen if necessary to ensure there is no retention of contents in the esophagus. Provide a good field for surgery, meanwhile the occurrence of aspiration should be avoided. Endoscopy should be carried out before the operation to determine its grade in Ling’s classification (table 8.3), assess the difficulty of the surgery, and prepare a personal operation plan. A CO
TAB. 8.3 — Ling’s classification of achalasia.
| Typing | Endoscopic manifestation |
| Type I | Mild dilation of lumen, smooth tube wall without winding |
| II | Dilated lumen, with ring or crescent structure after full inflation |
| II a | A thin ring, without crescent shaped structure |
| II b | With crescent structure, not exceeding 1/3 of the lumen |
| II c | With crescent structure, exceeding 1/3 of the lumen |
| III | Obvious lumen dilation, with diverticular structure formation |
| III 1 | Diverticular structure on the left |
| III r | Diverticular structure on right |
| III lr | Diverticular structure found both on the left and on the right |
(II) Surgical operation and skills
1. Anesthesia and position. All patients undergo general anesthesia with tracheal intubation, lying on their left side; attention should be paid to keep them warm, adjusting the room temperature and the infusion temperature, and avoiding pressure sores. The appropriate position must be chosen. Taking into account the anatomical structure of the esophagus, it is relatively safe to choose the proximal posterior wall of esophagus to establish the submucosal tunnel. The common positions include left-lying position, supine position and supine right shoulder elevated position. The supine position, with the right shoulder raised, has many advantages, and is usually chosen.
2. Operating steps
(1) Routine POEM
1) Specify site of submucosal injection: according to the operation’s grade in Ling’s classification, a standard tunnel or a short tunnel approach must be chosen. We suggest starting the tunnel at the relatively flat area of the posterior wall of the esophagus.
2) Inverted T-shaped opening: after injecting methylene blue and a normal saline solution to form a liquid pad at the selected tunnel opening site, a triangle knife or a hybrid knife must be used to cut the mucosal layer in order to establish the opening (figure 8.36).
3) Creation of the submucosal tunnel: an injection should be made from top to bottom while dissecting the submucosal layer, form a tunnel space, dissect below 2–3 cm of the EGJ. The width of the tunnel must be fully widened to prevent the tunnel from ‘deflecting.’ Vascular bleeding or mucosal defect must be timely treated if any (figure 8.37).
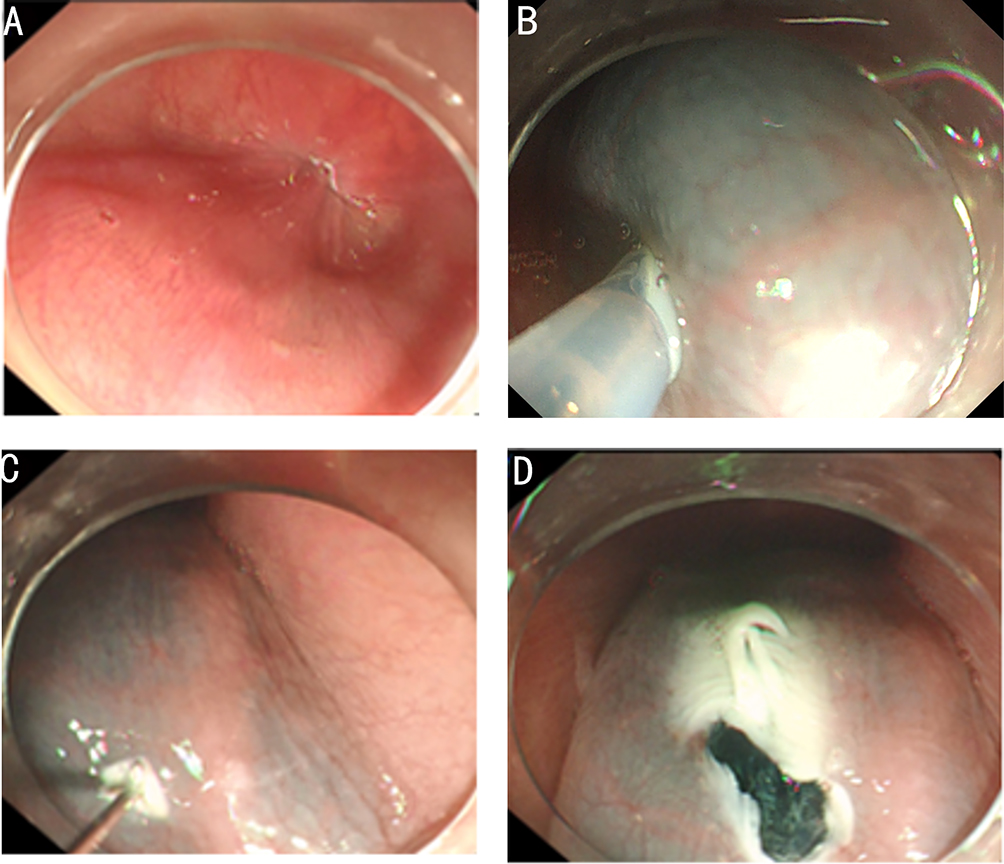
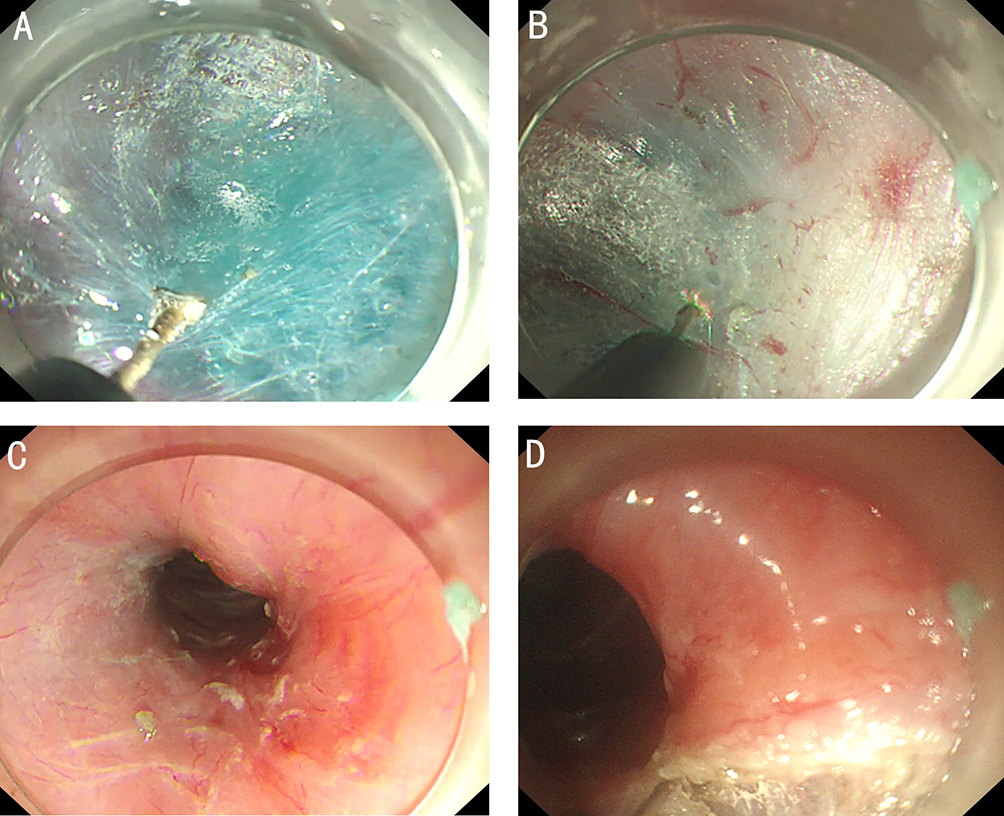

FIG. 8.36 — Steps for building the tunnel entrance in per-tunnel super minimally myotomy for achalasia. A. Endoscopic preventricular stenosis of a patient with achalasia patient. B. A submucosal injection above the stenosis. C. Creation of a tunnel opening with a triangle knife. D. Longitudinal incision to establish a tunnel opening.

FIG. 8.37 — Steps for establishing a submucosal tunnel in per-tunnel super minimally myotomy for achalasia. A. Dissection along the submucosal layer after entering the tunnel. B. Dissection along the submucosal layer near the cardia. C. The establishment of the tunnel is completed. D. Circular muscle layer incision.
4) Progressive myotomy: a circular incision must be made in the muscle, starting 4 cm above the cardiac contraction ring; the incision length is about 2 cm, full-thickness incision must be performed from 2 cm above the cardia to 2 cm below the cardia. Once the progressive myotomy has been completed, the gastroscope can pass again, confirming the degree of cardiac resistance.
5) Closure of the wound: Adequate hemostasis of potential bleeding spots and small vessels must be carried out from the anal to the oral side of the tunnel. After sucking the gas and liquids in the tunnel completely, wounds will be closed from the anal side to the oral side successively by using soft tissue clips (figure 8.38).
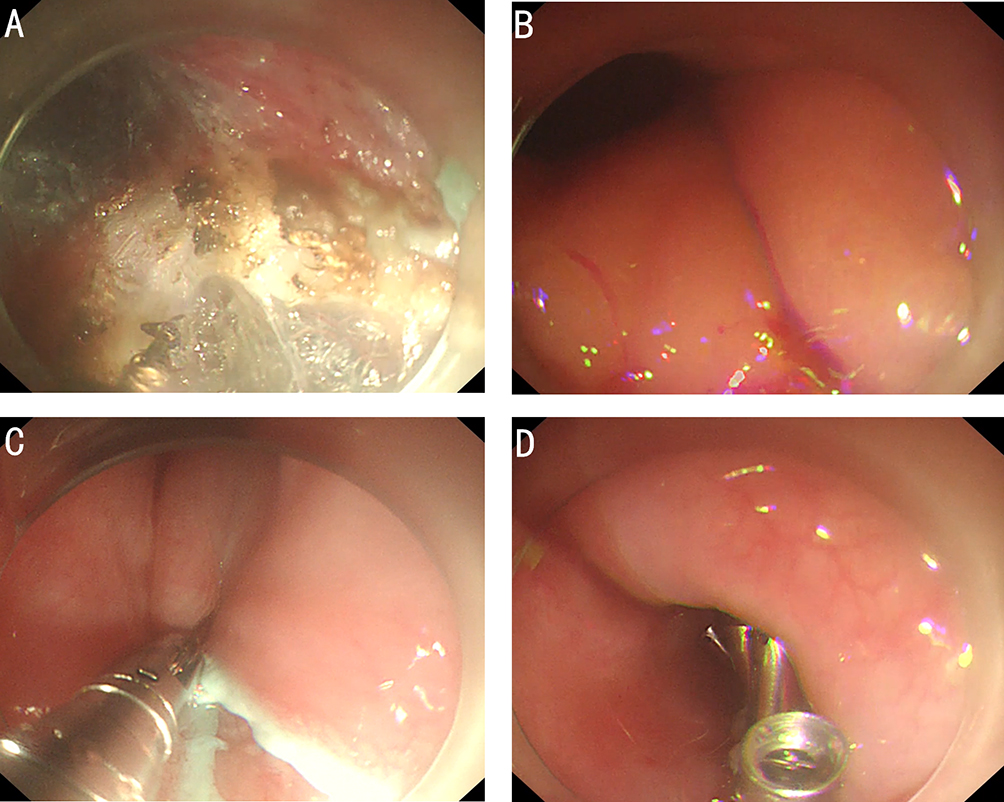

FIG. 8.38 — Steps of myotomy and tunnel opening and closure in per-tunnel super minimally myotomy for achalasia. A. The incision is continued until 2 cm below the cardia. B. Endoscopy exits the tunnel and enters the gastric cavity smoothly through the esophagus. C. The tunnel opening is closed. D. Completion of the closure of the tunnel opening.
(2) Difficult POEM: For patients with achalasia who have a therapeutic history (botox injection, balloon dilation, POEM or laparoscopic Heller myotomy) or a long disease history, the inflammation of the mucous membrane is more severe, and severe submucosal adhesion may exist during the establishment of the tunnel, and the tunnel cannot be completely established below the cardia. In 2014, Professor Linghu Enqiang proposed to combine POEM (percutaneous endoscopic mucosal resection) with SSMD (simultaneous submucosal and muscle dissection) surgical procedures (POEM–SSMD). For patients with submucosal adhesion, this POEM operation plan can be used with success.
1) Creation of the tunnel entrance: a submucosal injection is made, a liquid pad is formed, then an inverted T-shaped incision is made, an IT knife is used to continuously dissect in order to create the tunnel after continuous submucosal.
2) Creation of the tunnel: one should try as much as possible to separate and extend the submucosal tunnel to the site where the serious submucosal layer attaches. At this time the tunnel does not arrive 2 cm below the cardia (figure 8.39).
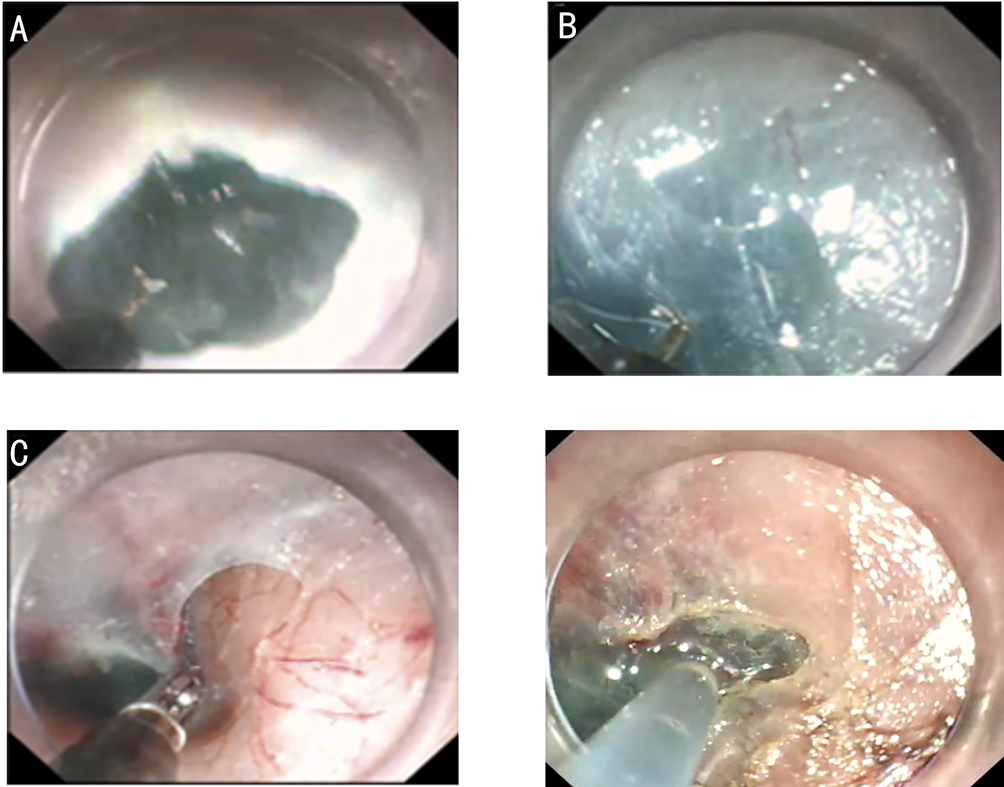

FIG. 8.39 — Steps of creation of a tunnel in per-tunnel super minimally myotomy for achalasia. A. Creation of the opening of an inverted T-shaped tunnel; B. Digging into the submucosal layer to dissect. C. Bare vessels and hemostasis. D. Supplementary injection at the serous adhesion site of the original surgical site.
3) Full-thickness myotomy: a full-thickness myotomy is directly performed on the basis of current tunnel; the submucosal and the muscle layer are simultaneously incised from the end of the adhesion tunnel to 2–3 cm below the cardia. At this time the endoscope is in the mediastinum. The triangle knife cuts the muscle layer laterally first, then cuts the submucosa, and the corresponding surface of the mucosa remains intact (figure 8.40).
4) Hemostasis in the tunnel and closure of the entrance: hemostasis is completed with a hemostat, the endoscope can easily pass the cardia, and then the tunnel’s opening is closed with metal clips (figure 8.40).
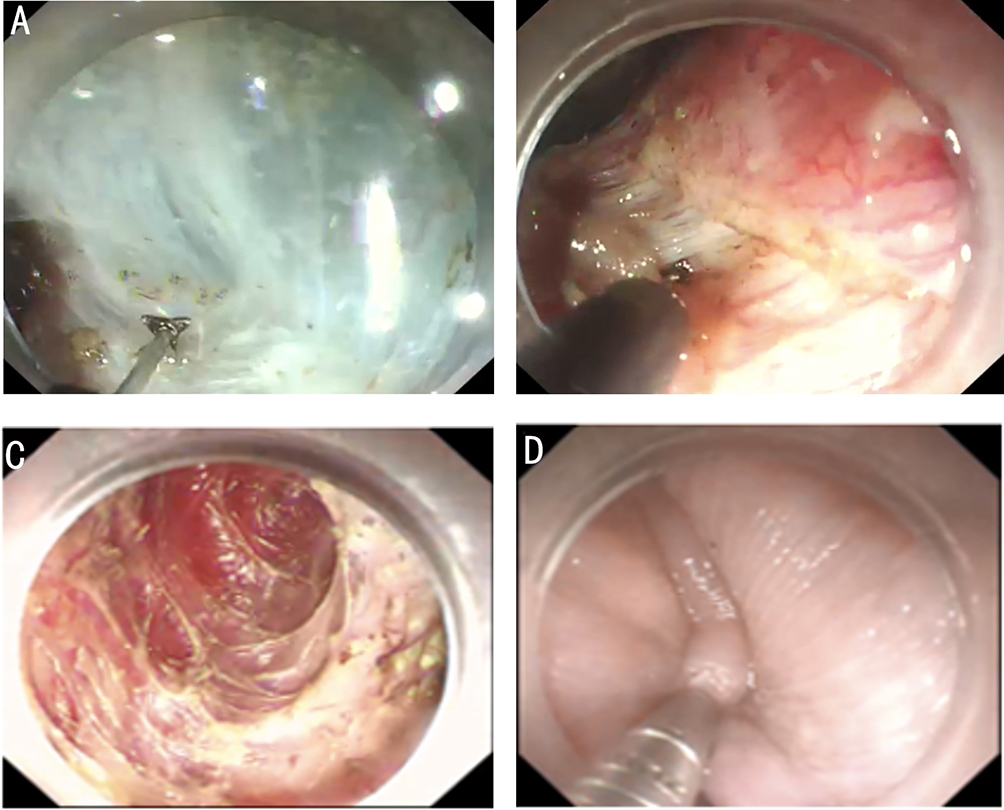

FIG. 8.40 — Steps of muscle layer incision and closure of the tunnel entrance in per-tunnel super minimally myotomy for achalasia. A. Separation along the serous adhesion area and superficial dissection at the level of the serous adhesion area of the original operation. B. Circular incision in the muscle. C. Full-thickness incision until the tunnel’s end. D. Closure of the tunnel entrance.
Refer to video 8.11, Per-tunnel super minimally myotomy for achalasia-1 (See online resources).
(III) Postoperative treatment
The patient must postoperatively fast for food and water for 72 h, switch to a liquid diet at the fourth day, and gradually switch to a normal diet. As he/she fasts for food and water, he must be given therapies like intravenous acid suppression, prophylactic broad-spectrum antibiotics, and nutritive supplementation, etc. He/she should orally take PPIs starting on the fourth day, and continue for eight weeks.
(IV) A classic case
History: the patient, a middle-aged man, was admitted to the hospital mainly due to “poor feeding for more than 30 years, reoccurring for two years.” Thirty years before, with no obvious inducing factors, the patient presented with poor feeding, poor drinking with serious pain behind the sternum. He visited a local hospital, that considered achalasia, and performed Heller surgery in 1994. Symptoms alleviated after surgery. In the preceding two years, poor feeding reoccurred, which was mild, could alleviate after slowly eating or taking meals with water. The external hospital esophagogram considered achalasia. For further diagnosis and treatment, he visited the outpatient department of the hospital where the author is located. Esophageal manometry revealed achalasia (Chicago type II). In the past, he was allergic to penicillin. No abnormalities were found during the physical examination for admission.
Diagnosis: achalasia (Chicago type II) after Heller surgery.
After full preoperative evaluation, improving nutrition condition and signing the surgery informed consent form, the patient was performed achalasia POEM surgery (Refer to video 8.12, Per-tunnel super minimally myotomy for achalasia-2 (See online resources)).
Surgical process: General anesthesia, tracheal intubation, right shoulder elevating position. After the introduction of the endoscope, it can be seen that the esophageal lumen is enlarged and twisted, and that the annular structure exceeds 1/3 of the lumen. Edema of esophageal mucosa and granular change. Tight cardiac ostium, several strips of ulcer erosion can be found in the mucosa above the cardiac ostium, about 39 cm from the incisor, and the endoscope encounters resistance to passing through the cardia. A methylene blue–epinephrine–saline solution is injected submucosally about 32 cm from the incisor. The mucosa is incised with the triangular knife to form an inverted T-shaped opening, and the short tunnel is established 3 cm below the cardia. The adhesion in the tunnel is obvious, a progressive myotomy begins 36 cm from the incisor with the triangle knife, full-thickness myotomy begins at the base of the tunnel 38 cm from the incisor, the bleeding spots in the tunnel cavity and the muscle of the stump are healed with a hot hemostat. The endoscope is withdrawn from the tunnel, and no resistance is observed in passing through the cardia again. The tunnel entrance is closed with several tissue clips. The operation is over. Aspiration and withdrawal. Impression after examination: achalasia POEM surgery (Ling type IIc, mucosal inflammation grading grade E, adhesion grading grade 2).
(V) Hot-spot issues in the consensus and progress of the study
1. The curative effect of POEM depends on the preoperative diagnostic typing and the accurate preparation of the operation plan. In order to improve the therapeutic effect of POEM and reduce the occurrence of complications, one needs to have an accurate diagnostic typing before the operation and to refine the operation plan. The type of opening of the tunnel mucosa, the length of the tunnel and the myotomy method must be determined in accordance with the indications and by avoiding the contraindications. There is sufficient evidence showing that an inverted T-shaped opening can reduce the number of titanium clips and the occurrence of adverse events compared to transverse incision and vertical incision. The tunnel length can be ‘standard’ or ‘short’; the length of the standard tunnel is 10–12 cm (EGJ oral side end 8–10 cm + anal side 2–3 cm), which applies to Ling’s types I and II; the length of the short tunnel is 7–8 cm (EGJ oral side end 5–6 cm + anal side 2 cm), which applies to Ling’s types IIc and III. Compared to tunnels of the same standard, the short tunnel has the same curative effect, but can significantly reduce the operating time, lower the incidence of the intraoperative adverse events. Regarding the myotomy mode, the most widely applied clinically at present are full-thickness myotomy and progressive full-thickness myotomy. The advantage of progressive full-thickness myotomy is that it reduces the occurrence of postoperative gastroesophageal reflux while not affecting the effectiveness of POEM. The end of the tunnel can be determined by a series of methods, such as measuring the distance from the incisor to the cardia before the operation, assessing the morphology of the blood vessels, observing the site of whitening of the anal mucosa and the change in resistance near the LES.
2. The prevention and treatment of postoperative POEM is still the hot-spot issue of study
(1) Mucosal injury: according to the literature, the incidence of mucosal injury is 2.8%–4.8%, and the incidence of perforation is from 0.2%–0.7%, which can easily occur in sites like cardia. After the completion of intraoperative myotomy in the tunnel and adequate hemostasis, the metal clips are used to close the injured wound, meanwhile fibrin glue can be sprayed, and gastrointestinal decompression can be retained if necessary; for patients with postoperative delayed perforation, it can result in mediastinal or abdominal infection. When the infection is limited or the patient’s general condition is good, conservative or endoscopic therapy can be selected and the surgery department should be contacted for consultation if necessary.
(2) Gas-related complications: including subcutaneous emphysema, pneumothorax, mediastinum and pneumoperitoneum, etc., with incidences of 7.5%, 1.2%, 1.1% and 6.8% respectively. Intraoperative use of CO
(3) Delayed hemorrhage: the incidence of serious hemorrhage is 0.2%–0.5%; in this case the endoscopic hemostatic treatment should be immediately performed. The metal clips must be pull out at the tunnel’s entrance; the endoscope enters the tunnel to rinse and clean it, meanwhile bleeding spots are electrocoagulated with a hemostat. The tunnel’s entrance is then occluded again after hemostasis is successful.
(4) Infection: this mainly involves lung infection, infection in the tunnel and mediastinal infection. After infection occurs, the specific site and the severity of infection should be carefully evaluated, antibiotics selected in a reasonable manner, drainage improved if necessary. The more serious risk is mediastinal abscess, and the infection is mostly due to eating food too soon after surgery, or to incomplete closure of the wound, and to the penetration of chyme into the tunnel and into the mediastinum through the muscle incision. Endoscopic therapy should be performed timely, the dirt in the tunnel and mediastinum cleaned, retaining the drainage tube in mediastinum and draining outside from nose. The tunnel entrance does not have to be closed, normal saline solution can be used for repeated rinsing, the period of fasting for food and water should be extended. Antibiotics should be applied intravenously. The selection of antibiotics depends on results of the drug allergy tests, meanwhile a parenteral nutrition treatment will be administered, and the drainage tube will be pulled out after full drainage of the mediastinal abscess.
(5) Fistula of the digestive tract: its incidence is low; maintaining the integrity of the esophageal side mucosa during the operation is key to preventing the fistula of the digestive tract. So injury to the esophageal mucosa should be avoided as much as possible. Once an esophageal fistula occurs, the esophageal metal stent can be used to close the orificium fistulae, meanwhile closed thoracic drainage should be performed and antibiotic treatment upgraded.
3. Whether POEM surgery replaces Heller surgery in the treatment of achalasia. For the treatment of achalasia, POEM or laparoscopic Heller myotomy combined with fundoplication can alleviate the patient’s difficulty in swallowing. But Heller myotomy can have fundus folding at the same time, which effectively reduces the gastroesophageal reflux after the operation. A randomized trial involving 221 adults with achalasia showed no statistically significant difference in clinical success rates between the POEM group and the group undergoing laparoscopic Heller myotomy with fundoplication (83% vs. 82%); clinical success at two years: Eckardt score ≤ 3, with no other treatments. Compared to laparoscopic Heller myotomy, the POEM group had a lower rate of serious adverse events (2.7% vs. 7.3%), but a higher rate of reflux esophagitis at two years (44% vs. 29%).
Super minimally invasive POEM is similar to Heller surgery for the treatment of achalasia, but it has definite advantages, since it avoids thoracotomy, with less trauma for the patient, it allows quick postoperative recovery, a high quality of life, and is easily accepted by patients. The postoperative preventive reflux is also very important. Progressive myotomy effectively reduces the occurrence of reflux, meanwhile patients may also choose C-BLART to treat reflux after surgery.
4. Selection of initial intramuscular dissection. Severe submucosal fibrosis is associated with prolonged surgical duration, increased perioperative complications, and even POEM failure. Recently, Madkour et al. have proposed a novel and successful initial intramuscular interlayer technique, which was used for two serious submucosal fibrosis patients who could not undergo submucosal tunnelling. IIMD (the ‘Madkour technique’), first reported at the mucosal incision level, can be used as a rescue option in patients with long-standing achalasia when a submucosal tunnel cannot be established due to severe submucosal fibrosis.
References
[1] Thrumurthy S. G., Chaudry M. A., Thrumurthy S. S. D., et al. (2019) Oesophageal Carcinoma: risks, prevention, and diagnosis, BMJ 9, 366.
[2] Early Diagnosis and Treatment Group of the Chinese Medical Association Oncology Branch. (2022) Chinese expert consensus on early diagnosis and treatment of esophageal Carcinoma, Zhonghua Zhong Liu Za Zhi 44(10), 1066–1075.
[3] Digestive Endoscopy Terminology Examination Subcommittee. (2021) Terminology of digestive endoscopy. Science Press, Beijing.
[4] Wang H. Y., Zeng X., Bai S. Y., et al. (2020) The safety and efficacy of endoscopic submucosal dissection for treating early oesophageal carcinoma: a meta-analysis, Ann. R. Coll. Surg. Engl. 102(9), 702–711.
[5] Ishihara R., Arima M., Iizuka T., et al. (2020) Japan Gastroenterological Endoscopy Society Guidelines Committee of ESD/EMR for Esophageal Carcinoma. Endoscopic submucosal dissection/endoscopic mucosal resection guidelines for esophageal Carcinoma, Dig. Endosc. 32(4), 452–493.
[6] Su Y. F., Cheng S. W., Chang C. C., et al. (2020) Efficacy and safety of traction-assisted endoscopic submucosal dissection: a meta-regression of randomized clinical trials, Endoscopy 52(5), 338–348.
[7] Libânio D., Pimentel-Nunes P., Bastiaansen B., et al. (2023) Endoscopic submucosal dissection techniques and technology: European Society of Gastrointestinal Endoscopy (ESGE) technical review, Endoscopy 55(4), 361–389.
[8] Wang Y., Ge X. X., Deng X. T., et al. (2022) Current status of gastric traction-assisted endoscopic submucosal dissection, Chin. J. Dig. 42(5), 357–360.
[9] Yu X., Wang G. (2015) Endoscopic treatment of early esophageal cancer, Chin. J. Gastrointest. Surg. 18(09), 860–863.
[10] Kitagawa Y., Ishihara R., Ishikawa H., et al. (2023) Esophageal Carcinoma practice guidelines 2022 edited by the Japan esophageal society: part 1 [published online ahead of print, 2023 Mar 18], Esophagus, 1–30.
[11] Hu W., Yu J., Yao N., et al. (2022) Efficacy and safety of four different endoscopic treatments for early esophageal carcinoma: a network meta-analysis, J. Gastrointest. Surg. 26(5), 1097–1108.
[12] Lu C., Guo S., Li D., et al. (2023) Comparison of the clinical effect of cap-assistted endoscopic full thickness resection and endoscopic submuscosal dissection for gastric small submu-cosal tumors, China Mod. Med. 30(13), 62–66.
[13] Tan B. (2023) The effect and safety of submucosal injection and transparent cap assisted endoscopic mucosal resection in the treatment of large colorectal sessile polyps, Chin. J. Mod. Drug App. 17(14), 41–43.
[14] Liu J., Xiang Y., Yuan Y., et al. (2022) Comparison of endoscopic mucosal resection with endoscopic transparent cap assisted mucosal resection in the treatment of type 1 gastric neuroendocrine neoplasms, Mod. Dig. Interv. 27(09), 1084–1089.
[15] Kim S., Lee B. I. (2024 May) The role of cap-assisted endoscopy and its future implications, Clin. Endosc. 57(3), 293–301. DOI: 10.5946/ce.2023.051. Epub 2024 Feb 7.
[16] Fan X., Wu Q., Li R., et al. (2022) Clinical benefit of tunnel endoscopic submucosal dissection for esophageal squamous Carcinoma: a multicenter, randomized controlled trial, Gastrointest. Endosc. 96(3), 436–444.
[17] Chinese Society of Digestive Endoscopy, Branch of Endoscopic Physicians of Chinese Medical Doctor Association, Beijing Branch of Chinese Society of Digestive Endoscopy, Innovation and Development Branch of Digestive Endoscopy of China Medical Device Industry Association. (2018) Expert consensus on digestive endoscopic tunnel technique (2017, Beijing), Chin. J. Dig. Endosc. 35(1), 1–14.
[18] Tian Y., Du G., Kan J., et al. (2022) A clinical research of modified endoscopic submucosal multi-tunnel dissection for superficial circumferential esophageal cancer over 8 cm in length, Chin. J. Dig. Endosc. 39(12), 983–987.
[19] Li L., Wang W., Yue H., et al. (2019) Endoscopic submucosal multi-tunnel dissection for large early esophageal Carcinoma lesions, Acta Gastroenterol. Belg. 82(3), 355–358.
[20] Zou J., Zhu J., Tang P., et al. (2019) Clinical value of autologous skin-grafting surgery to prevent esophageal stenosis after complete circular endoscopic submucosal tunnel dissection for early esophageal cancer, Chin. J. Dig. Endosc. 36(5), 5. DOI: 10.3760/cma.j.issn.1007-5232.2019.05.003.
[21] Ohki T., Yamato M., Ota M., Takagi R., Murakami D., Kondo M., Sasaki R., Namiki H., Okano T., Yamamoto M. (2012) Prevention of esophageal stricture after endoscopic submucosal dissection using tissue-engineered cell sheets, Gastroenterology 143, 582–588.e582.
[22] Hochberger J., Koehler P., Wedi E., Gluer S., Rothstein R. I., Niemann H., Hilfiker A., Gonzalez S., Kruse E. (2014) Transplantation of mucosa from stomach to esophagus to prevent stricture after cir-cumferential endoscopic submucosal dissection of early squamouscell, Gastroenterology 146, 906–909.
[23] Zou J., Chai N., Linghu E., et al. (2021) Autologous skin-grafting surgery to prevent esophageal stenosis after complete circular endoscopic submucosal tunnel dissection: a case-matched controlled study, Surg. Endosc. 35, 5962–5970.
[24] Ji K., Cai X., Xie Y., et al. (2021) Progress in prevention of esophageal stenosis after endoscopic submuco-sal dissection, Mod. Dig. Interv. 26(12), 1481–1486.
[25] Sakurai T., Miyazaki S., Miyata G., Satomi S., Hori Y. (2007 Jul) Autologous buccal keratinocyte implantation for the prevention of stenosis after EMR of the esophagus, Gastrointest. Endosc. 66(1), 167–173.
[26] Liu Y. (2024) Endoscopic diagnosis and treatment of circumferential superficial esophageal squamous cell neoplasms. Peking Union Medical College Hospital. DOI: 10.27648/d.cnki.gzxhu.2023.000473.
[27] Xiu H., Zhao C. Y., Liu F. G., et al. (2019) Comparing about three types of endoscopic therapy methods for upper gastrointestinal submucosal tumors originating from the muscularis propria layer, Scand. J. Gastroenterol. 54(12), 1481–1486.
[28] Ye L.-P., Zhang Y., Luo D.-H., et al. (2016) Safety of endoscopic resection for upper gastrointestinal subepithelial tumors originating from the muscularis propria layer: an analysis of 733 tumors, Am. J. Gastroenterol. 111(6), 788–796.
[29] Ponte Neto F. L., De Moura D. T. H., Sagae V. M. T., et al. (2021) Endoscopic resection of esophageal and gastric submucosal tumors from the muscularis propria layer: submucosal tunneling endoscopic resection versus endoscopic submucosal excavation: a systematic review and meta-analysis, Surg. Endosc. 35(12), 6413–6426.
[30] Du C., Linghu E. (2017) Submucosal tunneling endoscopic resection for the treatment of gastrointestinal submucosal tumors originating from the muscularis propria layer, J. Gastrointest. Surg. 21(12), 2100–2109.
[31] Jin X. F., Gai W., Du R. L., et al. (2019) Multiband mucosectomy versus endoscopic submucosal dissection and endoscopic submucosal excavation for GI submucosal tumors: short and long term follow-up, BMC Carcinoma 19(1), 893.
[32] Chai N. L., Li H. K., Linghu E. Q., et al. (2019) Consensus on the digestive endoscopic tunnel technique, World J. Gastroenterol. 25(7), 744–776.
[33] Shi Q., Zhong Y., Yao L. (2013) Recent advances in dual-endoscopy combined therapy for gastrointestinal tumors, with a focus on digestive endoscopy, Chin. J. Gen. Surg. 28(10), 3. DOI: 10.3760/cma.j.issn.1007-631X.2013.10.033.
[34] Gonzalez J. M., Debourdeau A., Philouze G., Beyer L., Berdah S., Barthet M. (2018 Feb) Laparoscopic and endoscopic cooperative surgery for difficult resection of posterior esophagogastric junction gastrointestinal stromal tumors, Endoscopy 50(2), 178–179. DOI: 10.1055/s-0043-121136. Epub 2017 Nov 14. PMID: 29136672.
[35] NOTES and Surgical Group of Chinese Society of Digestive Endoscopy, Chinese Medical Association, Digestive Endoscopy Committee of the Endoscopists Branch, Chinese Medical Doctor Association, Gastrointestinal Surgery Group of the Surgical Branch of the Chinese Medical Association. (2023) Chinese consensus on endoscopic diagnosis and managment of gastrointestinal submucosal tumors (version 2023), Chin. J. Pract. Surg. 43(3), 241–251. DOI: 10.19538/j.cjps.issn1005-2208.2023.03.01.
[36] Cui J. X., Gao Y. H., Xi H. Q., et al. (2018) Comparison between laparoscopic and open surgery for large gastrointestinal stromal tumors: a meta-analysis, World J. Gastrointest. Oncol. 10(1), 48–55.
[37] Park S. H., Lee H. J., Kim M. C., et al. (2022) Early experience of laparoscopic resection and comparison with open surgery for gastric gastrointestinal stromal tumor: a ulticenter retrospective study, Sci. Rep. 12(1), 2290.
[38] National Center for Quality Control of Digestive Endoscopy, National Clinical Research Center for Digestive Diseases (Shanghai), Gastrointestinal Early Cancer Alliance (GECA), Branch of Endoscopic Physicians of Chinese Medical Doctor Association, Chinese Society of Digestive Endoscopy (CSDE), Chinese Society of Health Management, Professional Committee of Tumor Endoscopy of Chinese Anti-Cancer Association. (2019) China experts consensus on the protocol of early esophageal cancer and pre-cancerous lesion screening (2019, Xinxiang), Chin. J. Dig. Endosc. 36(11), 793–801. DOI: 10.3760/cma.j.issn.1007-5232.2019.11.001.
[39] Schlemper R. J., Riddell R. H., Kato Y., et al. (2000 Aug) The Vienna classification of gastrointestinal epithelial neoplasia, Gut 47(2), 251–255. DOI: 10.1136/gut.47.2.251.
[40] Linghu E., Feng J., Ma X., et al. (2015) Clinical study of endoscopic radiofrequency ablation for treatment of low-grade gastroesophageal intraepithelial neoplasia, Chin. J. Gastrointest. Endosc. (Electron. Ed.) 2(1), 14–17. DOI: 10.3877/cma.j.issn.2095-7157.2015.01.004.
[41] Feng J. (2017) Experimental and clinical studies on endoscopic radiofrequency ablation for gastric low-grade intraepithelial neoplasia. The General Hospital of the People’s Liberation Army; Chinese PLA Medical School, 1–75. DOI: 10.3877/cma.j.issn.2095-7157.2015.01.004.
[42] Ono S., Fujishiro M., Niimi K., et al. (2009) Predictors of postoperative stricture after esophageal endoscopic submucosal dissection for superficial squamous cell neoplasms, Endoscopy 41, 661–665.
[43] Hanaoka N., Ishihara R., Takeuchi Y., et al. (2012) Intralesional steroid injection to prevent stricture after endoscopic submucosal dissection for esophageal Carcinoma: a controlled prospective study, Endoscopy 44, 1007–1011.
[44] Chinese Society of Digestive Endoscopy, Branch of Endoscopic Physicians of Chinese Medical Doctor Association, Beijing Branch of Chinese Society of Digestive Endoscopy. (2020) Expert consensus on the endoscopic prevention and treatment of benign and malignant esophageal stenosis (2020, Beijing), Chin. J. Gastrointest. Endosc. (Electron. Ed.) 7(4), 165–175.
[45] Adler D. G., Siddiqui A. A. (2017) Endoscopic management of esophageal strictures, Gastrointest. Endosc. 86(1), 35–43.
[46] Xiang J., Linghu E., Li L., Zou J., Wang X., Chai N. (2021) Utility of radial incision and cutting with steroid injection for refractory stricture after endoscopic submucosal dissection for large superficial esophageal squamous cell carcinoma, Surg. Endosc. 35(12), 6930–6937.
[47] Guetner G. C., Werner S., Barrandon Y., et al. (2008) Wound repair and regeneration, Nature 453(7193), 314–321.
[48] Li L., Linghu E., Chai N., et al. (2019) Clinical experience of using a novel self-help inflatable balloon to prevent esophageal stricture after circumferential endoscopic submucosal dissection, Dig. Endosc. 31(4), 453–459.
[49] Li L., Linghu E., Chai N., et al. (2021) Long-term efficacy and safety of a novel self-help inflatable balloon to prevent esophageal stenosis after extensive endoscopic submucosal dissection, Chin. J. Dig. Endosc. 38(9), 712–717.
[50] Vermeulen B. D., de Zwart M., Sijben J., et al. (2020) Risk factors and clinical outcomes of endoscopic dilation in benign esophageal strictures: a long-term follow-up study, Gastrointest. Endosc. 91(5), 1058–1066.
[51] National Center for Quality Control of Digestive Endoscopy, National Clinical Research Center for Digestive Diseases (Shanghai), Gastrointestinal Early Cancer Alliance (GECA), Branch of Endoscopic Physicians of Chinese Medical Doctor Association, Chinese Society of Digestive Endoscopy, Professional Committee of Tumor Endoscopy of Chinese Anti-Cancer Association. (2020) China expert consensus on clinical application of endoscopic radiofrequency ablation for gastrointestinal lesions (2020, Shanghai), Chin. J. Dig. Endosc. 37(2), 77–82.
[52] Hu H., Linghu E. (2017) Current trends of endoscopic treatment for gastroesophageal reflux disease, Chin. J. Gastrointest. Endosc. (Electron. Ed.) 4(1), 36–40.
[53] Chinese Society of Gastroenterology. (2020) Chinese expert consensus of gastroesophageal reflux disease in 2020, Chin. J. Dig. 40(10), 649–663.
[54] Lu D., Bi C. S., Wei X., et al. (2022 Feb 23) A retrospective study of the safety and efficacy of endoscopic radiofrequency therapy under direct vision in 59 patients with gastroesophageal reflux disease from 2 centers in Beijing, China using the gastroesophageal reflux disease questionnaire, Med. Sci. Monit. 28, e933848.
[55] Fass R., Cahn F., Scotti D. J., et al. (2017 Dec) Systematic review and meta-analysis of controlled and prospective cohort efficacy studies of endoscopic radiofrequency for treatment of gastroesophageal reflux disease, Surg. Endosc. 31(12), 4865–4882.
[56] Dughera L., Rotondano G., De Cento M., et al. (2014) Durability of Stretta radiofrequency treatment for GERD: results of an 8-year follow-up, Gastroenterol. Res. Pract. 2014, 531907.
[57] Andreou A., Watson D. I., Mavridis D., et al. (2020) Assessing the efficacy and safety of laparoscopic antireflux procedures for the management of gastroesophageal reflux disease: a systematic review with network meta-analysis, Surg. Endosc. 34(2), 510–520.
[58] Gerson L., Stouch B., Lobonţiu A. (2018 Mar-Apr) Transoral incisionless fundoplication (TIF 2.0): a meta-analysis of three randomized, controlled clinical trials, Chir. (Bucur.) 113(2), 173–184.
[59] Trad K. S., Barnes W. E., Prevou E. R., et al. (2018 Apr) The TEMPO trial at 5 years: transoral fundoplication (TIF 2.0) is safe, durable, and cost-effective, Surg. Innov. 25(2), 149–157.
[60] Ai K., Yu S., Ke L. (2023) Current status of minimally invasive surgery of gastroesophageal reflux disease, J. Surg. Concepts Pract. 28(3), 208–214.
[61] Bell R. C. W., Freeman K., Heidrick R., et al. (2021) Transoral incisionless fundoplication demonstrates durability at up to 9 years, Therap. Adv. Gastroenterol. 14, 1–11.
[62] Sui X., Gao X., Zhang L. et al. (2022) Clinical efficacy of endoscopic antireflux mucosectomy vs. Stretta radiofrequency in the treatment of gastroesophageal reflux disease: a retrospective, single-center cohort study, Ann. Transl. Med. 10, 660.
[63] Inoue H., Ito H., Ikeda H., et al. (2014) Anti-reflux mucosectomy for gastroesophageal reflux disease in the absence of hiatus hernia: a pilot study, Ann. Gastroenterol. 27(4), 346–351.
[64] Garg R., Mohammed A., Singh A., et al. (2022 Jun 10) Anti-reflux mucosectomy for refractory gastroesophageal reflux disease: a systematic review and meta-analysis, Endosc. Int. Open 10(6), E854–E864.
[65] Chou C. K., Chen C. C., Chen C. C., et al. (2023 Feb) Positive and negative impact of anti-reflux mucosal intervention on gastroesophageal reflux disease, Surg. Endosc. 37(2), 1060–1069.
[66] Ren S., Wang Q., Zhu H., et al. (2022) Progress of anti-reflux mucosectomy for refractory gastroesophageal reflux disease, Chin. J. Dig. Endosc. 39(10), 848–851.
[67] Zhong Z., Zhang B., Guo S., et al. (2021) Research progress in anti-reflux mucosectomy for refractory gastroesophageal reflux disease, Int. J. Dig. Dis. 41(06), 409–413.
[68] Katz P. O., Dunbar K. B., Schnoll-Sussman F. H., et al. (2022) ACG clinical guideline for the diagnosis and management of gastroesophageal reflux disease, Am. J. Gastroenterol. 117(1), 27–56.
[69] Nabi Z., Reddy D. N. (2019) Update on endoscopic approaches for the management of gastroesophageal reflux disease, Gastroenterol. Hepatol. 15(7), 369–376.
[70] Liu S., Chai N., Zhai Y., et al. (2020) New treatment method for refractory gastroesophageal reflux disease (GERD): C-BLART (clip band ligation anti-reflux therapy)—a short-term study, Surg. Endosc. 34(10), 4516–4524.
[71] Hu H. Q., Li H. K., Xiong Y., et al. (2018) Peroral endoscopic cardial constriction in gastroesophageal reflux disease, Medicine 97(15), e0169.
[72] Yeh J. H., Lee C. T., Hsu M. H., et al. (2022) Antireflux mucosal intervention (ARMI) procedures for refractory gastroesophageal reflux disease: a systematic review and meta-analysis, Therap. Adv. Gastroenterol. 15, 1–12.
[73] Lv W., Li B., Li Z., et al. (2023) Giant esophageal diverticulum: a case report and literature review, Asian J. Surg. 46, 1817–1818.
[74] Sato H., Takeuchi M., et al. (2019) Esophageal diverticulum: new perspectives in the era of minimally invasive endoscopic treatment, World J. Gastroenterol. 25(12), 1457.
[75] Smith C. D. (2015) Esophageal strictures and diverticula, Surg. Clin. North Am. 95, 669–681.
[76] Parikh M. P., Gupta N. M., Sanaka M. R. (2019) Esophageal third space endoscopy: recent advances, Curr. Treat. Options Gastroenterol. 17, 63–75.
[77] Zhang D., Chen W., Wang Y., et al. (2022) Submucosal tunneling endoscopic septum division for esophageal diverticulum with a median follow-up of 39 months: a multicenter cohort study, Gastrointest. Endosc. 96, 612–619.e1.
[78] Chang C. M., Huang H. H. (2015) Oesophageal diverticulum, Arab J. Gastroenterol. 16, 76–77.
[79] Law R., Katzka D. A., Baron T. H. (2014) Zenker’s diverticulum, Clin. Gastroenterol. Hepatol. 12, 1773–1782; quiz e111-2.
[80] Sturgeon C. T. (1929) Esophageal diverticula, J. Am. Med. Assoc. 92(5), 379–385.
[81] Schick A., Yesner R. (1953) Traction diverticulum of esophagus with exsanguination: report of a case, Ann. Intern. Med. 39, 345–349.
[82] Soares R., Herbella F. A., Prachand V. N., et al. (2010) Epiphrenic diverticulum of the esophagus. From pathophysiology to treatment, J. Gastrointest. Surg. 14, 2009–2015.
[83] D’Journo X. B., Ferraro P., Martin J., et al. (2010) Lower oesophageal sphincter dysfunction is part of the functional abnormality in epiphrenic diverticulum, Br. J. Surg. 96, 892–900.
[84] Sato H., Takahashi K., Mizuno K. I., et al. (2018) Esophageal motility disorders: new perspectives from high-resolution manometry and histopathology, J. Gastroenterol. 53, 484–493.
[85] Tedesco P., Fisichella P. M., Way L. W., et al. (2005) Cause and treatment of epiphrenic diverticula, Am. J. Surg. 190, 891–894.
[86] de Schipper J. P., Pull ter Gunne A. F., Oostvogel H. J., et al. (2009) Spontaneous rupture of the oesophagus: Boerhaave’s syndrome in 2008. Literature review and treatment algorithm, Dig. Surg. 26, 1–6.
[87] Pressman A., Behar J. (2017) Etiology and pathogenesis of idiopathic achalasia, J. Clin. Gastroenterol. 51(3), 195–202.
[88] Jeon H. H., Kim J. H., Youn Y. H., et al. (2017) Clinical characteristics of patients with untreated achalasia, J. Neurogastroenterol. Motil. 23(3), 378–384.
[89] van Hoeij F. B., Ponds F. A., Smout A. J., et al. (2018) Incidence and costs of achalasia in the Netherlands, Neurogastroenterol. Motil. 30(2), e13195.
[90] Harvey P. R., Thomas T., Chandan J. S., et al. (2019) Incidence, morbidity and mortality of patients with achalasia in England: findings from a study of nationwide hospital and primary care data, Gut 68(5), 790–795.
[91] Sadowski D. C., Ackah F., Jiang B., et al. (2010) Achalasia: incidence, prevalence and survival. A population-based study, Neurogastroenterol. Motil. 22(9), e256–e261.
[92] Samo S., Carlson D. A., Gregory D. L., et al. (2017) Incidence and prevalence of achalasia in central Chicago, 2004-2014, Since the widespread use of high-resolution manometry, Clin. Gastroenterol. Hepatol. 15, 366–373.
[93] Li H. K., Linghu E. Q. (2013 Jan 28) New endoscopic classification of achalasia for selection of candidates for peroral endoscopic myotomy, World J. Gastroenterol. 19(4), 556–560.
[94] Li L., Chai N., Linghu E., et al. (2019) Safety and efficacy of using a short tunnel versus a standard tunnel for peroral endoscopic myotomy for Ling type IIc and III achalasia: a retrospective study, Surg. Endosc. 33(5), 1394–1402.
[95] Feng X., Linghu E., Chai N., et al. (2018) Endoscopic submucosal tunnel dissection for large gastric neoplastic lesions: a case-matched controlled study, Gastroenterol. Res. Pract. 2018, 1419369.
[96] Zhang W. G., Linghu E. Q., Chai N. L., et al. (2017) Ling classification describes endoscopic progressive process of achalasia and successful peroral endoscopy myotomy prevents endoscopic progression of achalasia, World J. Gastroenterol. 23(18), 3309–3314.
[97] Chai N., Zhang X., Xiong Y., et al. (2017) Ling classification applied in the preoperative safety and effectiveness assessment of POEM, Surg. Endosc. 31(1), 368–373.
[98] ASGE Technology Committee; Lo S. K., Fujii-Lau L. L., et al. (2016) The use of carbon dioxide in gastrointestinal endoscopy, Gastrointest Endosc. 83(5), 857–865.
[99] Jacobs C., Yang D., Draganov P. V. (2020) Peroral endoscopic myotomy for achalasia: does myotomy position matter? Gastrointest Endosc. 91(2), 298–300.
[100] Inoue H., Tianle K. M., Ikeda H., et al. (2011) Peroral endoscopic myotomy for esophageal achalasia: technique, indication, and outcomes, Thorac. Surg. Clin. 21(4), 519–525.
[101] Du C., Ma L., Chai N., et al. (2018) Factors affecting the effectiveness and safety of submucosal tunneling endoscopic resection for esophageal submucosal tumors originating from the muscularis propria layer, Surg. Endosc. 32(3), 1255–1264.
[102] Wang X. H., Tan Y. Y., Zhu H. Y., et al. (2016 Nov 14) Full-thickness myotomy is associated with higher rate of postoperative gastroesophageal reflux disease, World J. Gastroenterol. 22(42), 9419–9426.
[103] Tanaka S., Kawara F., Toyonaga T., et al. (2018) Two penetrating vessels as a novel indicator of the appropriate distal end of peroral endoscopic myotomy, Dig. Endosc. 30(2), 206–211.
[104] Linghu E. (2017) Interpretation of the expert consensus on digestive endoscopic tunnel technique (2017, Beijing), Chin. J. Gastrointest. Endosc. (Electron. Ed.) 4(04), 159–161.
[105] Haito-Chavez Y., Inoue H., Beard K. W., et al. (2017) Comprehensive analysis of adverse events associated with per oral endoscopic myotomy in 1826 patients: an international multicenter study, Am. J. Gastroenterol. 112(8), 1267–1276.
[106] Weng C. Y., He C. H., Zhuang M. Y., et al. (2022) Peroral endoscopic longer vs shorter esophageal myotomy for achalasia treatment: a systematic review and meta-analysis, World J. Gastrointest. Surg. 14(3), 247–259.
[107] Li H., Linghu E., Wang X. (2012) Fibrin sealant for closure of mucosal penetration at the cardia during peroral endoscopic myotomy (POEM), Endoscopy 44(Suppl 2) UCTN:E215-6.
[108] Zhang W. G., Linghu E. Q., Li H. K. (2017) Fibrin sealant for closure of mucosal penetration at the cardia during peroral endoscopic myotomy: a retrospective study at a single center, World J. Gastroenterol. 23(9), 1637–1644.
[109] Linghu E., Li H., Wang X., et al. (2011) Case report on the use of porcine-derived fibrin sealant for tunnel leak closure during peroral endoscopic myotomy (POEM), Chin. J. Laparosc. Surg. (Electron. Ed.) 4(05), 407–408.
[110] Linghu E. Q. (2019) New classifications of intraoperative bleeding and muscularis propria injury in endoscopic resection, Chin Med. J. (Engl.) 132(15), 1856–1858.
[111] Wang N., Linghu E., Zhai Y., et al. (2013) Incidences of postoperative transient bacteremia underwent peroral endoscopic myotomy: a prospective study, Chin. J. Laparosc. Surg. (Electron. Ed.) 6(06), 430–435.
[112] Anikhindi S. A., Ranjan P., Sachdeva M., et al. (2016 Jul) Self-expanding plastic stent for esophageal leaks and fistulae, Indian J. Gastroenterol. 35(4), 287–293. DOI: 10.1007/s12664-016-0679-3. Epub 2016 Aug 4. PMID: 27488703.
[113] van Halsema E. E., Kappelle W. F. W., Weusten B. L. A. M., Lindeboom R., van Berge Henegouwen M. I., Fockens P., Vleggaar F. P., Spaander M. C. W., van Hooft J. E. (2018) Stent placement for benign esophageal leaks, perforations, and fistulae: a clinical prediction rule for successful leakage control, Endoscopy 50(2), 98–108.
[114] Werner Y. B., Hakanson B., Martinek J., et al. (2019) Endoscopic or surgical myotomy in patients with idiopathic achalasia, N. Engl. J. Med. 381, 2219.
[115] Madkour A., Elfouly A., Sayed H., et al. (2024 Dec) Initial intramuscular dissection as a rescue therapy during peroral endoscopic myotomy for achalasia patients with severe submucosal fibrosis, Endoscopy 56(S 01), E118–E119.
[116] Inoue H., Yamamoto K., Shimamura Y., Azuma D., Ushikubo K., Okada H., Kimoto Y., Nishikawa Y., Tanaka I., Tanabe M., Onimaru M., Navarro M. J. (2024 Jun) Pilot study on anti-reflux mucoplasty: advancing endoscopic anti-reflux therapy for gastroesophageal reflux disease, Dig. Endosc. 36(6), 690–698.
[117] DeWitt J. M., Al-Haddad M., Stainko S., Perkins A., Fatima H., Ceppa D. P., Birdas T. J. (2024 Jan 5) Transoral incisionless fundoplication with or without hiatal hernia repair for gastroesophageal reflux disease after peroral endoscopic myotomy, Endosc. Int. Open 12(1), E43–E49. DOI: 10.1055/a-2215-3415. PMID: 38188922; PMCID: PMC10769579.
Chapter 9 Super Minimally Invasive Surgery of Gastric Diseases
9.1 Super Minimally Invasive Surgery for Early Gastric Cancer
9.1.1 Overview
(1) Overview of gastric cancer
Gastric cancer is one of the most common malignancies worldwide. According to the 2022 Global Cancer Statistics report, gastric cancer ranks fifth in both incidence and mortality among malignant tumors, with approximately 968 400 new cases and 659 900 deaths annually. These figures account for 4.9% and 6.8% of all malignancies, respectively. In China, gastric cancer is particularly prevalent, contributing 37.0% of the global new cases and 39.4% of global deaths, significantly threatening the life and health of the population. Gastric cancer is classified into early gastric cancer (EGC) and advanced gastric cancer (AGC) depending on its progression stage. EGC refers to lesions confined to the mucosal and submucosal layers, with a favorable prognosis and a five-year survival rate exceeding 90%. Conversely, AGC is associated with a low radical resection rate, poor quality of life, and a five-year tumor-related survival rate of less than 30%. Early diagnosis and timely treatment are thus essential for reducing mortality and extending survival in gastric cancer patients.
(2) Diagnosis of early gastric cancer
Patients with early gastric cancer often present with nonspecific clinical symptoms and signs, making gastroscopy and biopsy the primary diagnostic tools and the gold standard. White-light endoscopy (WLE) forms the basis of the examination, where EGC may appear as mucosal discoloration, slight elevations, or depressions. If suspicious lesions are observed under WLE, further assessment using image-enhanced endoscopy (IEE) is recommended. Advanced IEE techniques include chromoscopy, narrow-band imaging (NBI), flexible spectral imaging color enhancement (FICE), linked color imaging (LCI), blue laser imaging (BLI), I-scan high-definition electronic chromoendoscopy, and magnifying endoscopy (ME). Chromoscopy involves spraying dye onto the mucosal surface during conventional endoscopy, enhancing contrast between lesions and normal mucosa to better identify and delineate lesions. Electronic chromoendoscopy allows real-time observation of structural changes and, when combined with ME, effectively utilizes changes in microvascular and surface patterns to improve the accuracy of EGC diagnostic. The integration of artificial intelligence (AI) with IEE shows promise in further enhancing diagnostic accuracy for EGC, offering significant potential in clinical applications.
Endoscopic ultrasonography (EUS) is the most precise method for local staging of gastric cancer, especially in assessing invasion depth and regional lymph node metastasis. However, the accuracy of EUS varies, particularly in differentiating between T1a and T1b stages, due to limitations influenced by lesion morphology and operator expertise. The application of AI may help improve the accuracy of EUS diagnostic.
Computed tomography (CT) assists in evaluating adjacent organs, lymph nodes, and the stage of the disease. Early gastric cancer lesions are challenging to detect with conventional CT. To address this, techniques such as gastric cavity expansion with air or water, multiphase contrast-enhanced scanning, and multiplanar reconstruction are employed. These methods enhance the assessment of the location of the lesion, its depth, its relationships with the surrounding structures and the involvement of the lymph nodes.
(3) Technical methods for implementing super minimally invasive resection for early gastric cancer
Early gastric cancer can be effectively treated with super minimally invasive surgery (SMIS), which aims for curative outcomes through endoscopic approaches. These include natural orifice transluminal, tunnel-based, and multi-cavity techniques: peroral super minimally invasive resection for early gastric cancer is a technique involving the complete local resection of early gastric cancer via an oral endoscopic approach. Per-tunnel super minimally invasive resection for early gastric cancer is a method in which early gastric cancer is resected entirely via a tunnel-based endoscopic approach. Per-multiple-cavity super minimally invasive resection for early gastric cancer is a combined approach involving abdominal skin puncture and oral natural orifice endoscopy, utilizing both digestive endoscopy and laparoscopy either simultaneously or sequentially to achieve complete local resection of early gastric cancer.
The currently applied techniques for super minimally invasive surgery (SMIS) in the treatment of early gastric cancer, along with their previous nomenclature, are summarized in table 9.1.
Comparison of current SMIS techniques with the previous nomenclature.
| Serial no. | Super minimally invasive surgery (SMIS) | Technical method | Previous nomenclature |
| 1 | Peroral SMIS for early gastric cancer | Non-full-thickness resection | Endoscopic submucosal dissection (ESD), traction-assisted ESD, transparent cap-assisted endoscopic mucosal resection (EMR) |
| 2 | Peroral SMIS for early gastric cancer | Full-thickness resection | Endoscopic full-thickness resection (EFTR), traction-assisted EFTR |
| 3 | Per-tunnel SMIS for early gastric cancer | Non-full-thickness resection | Submucosal tunneling endoscopic resection (STER), traction-assisted ESD |
| 4 | Per-multiple-cavity SMIS for early gastric cancer | Multi-cavity minimally invasive resection | Multiscope-assisted therapy |
9.1.2 Peroral Super Minimally Invasive Surgery by Non-Full-Thickness Resection for Early Gastric Cancer
Peroral super minimally invasive surgery by non-full-thickness resection for early gastric cancer involves precise resection of the mucosal layer, muscularis mucosae, and submucosal layer using techniques such as endoscopic submucosal dissection (ESD), traction-assisted ESD, and transparent cap-assisted endoscopic mucosal resection (EMR).
(I) Indications and contraindications
1. Indications: (1) differentiated mucosal cancer (cT1a) without ulceration; (2) differentiated mucosal cancer (cT1a) with ulceration, ≤3 cm in size; (3) high-grade gastric intraepithelial neoplasia (HGIN); (4) undifferentiated mucosal cancer (cT1a) without ulceration, ≤2 cm in size.
2. Contraindications: (1) general anesthesia contraindications, such as anesthetic allergies or severe cardiopulmonary diseases; (2) coagulation disorders or high bleeding risk due to inability to discontinue antiplatelet/anticoagulant therapy; (3) advanced cancer or radiologic evidence of distant metastasis; (iv) pregnancy or other contraindications for endoscopic diagnosis or treatment.
(II) Preoperative preparation
(1) Patients and their families must be fully informed about the risks and benefits of SMIS, and their written informed consent obtained.
(2) Complete preoperative evaluations must be carried out, including blood tests (CBC, biochemistry, coagulation, tumor markers), blood typing, ECG, chest X-ray or CT, abdominal CT, and anesthesia assessment.
(3) Anticoagulant, antiplatelet, or blood-thinning medication use must be verified; they should be discontinued and/or a transition therapy be prescribed as appropriate.
(4) The patient should fast for at least 8 h before the procedure, and oral defoaming and mucolytic agents should be administered preoperatively.
(III) Postoperative management
(1) Strict bed rest for 24 h should be observed, avoiding any strenuous activity. Complications such as fever, palpitations, cold sweats, abdominal pain, hematemesis, or melena, should be monitored.
(2) Fasting should be maintained for 48–72 h, switching to a fresh liquid diet (e.g., rice soup, noodle soup, milk) and then to a semi-liquid diet (e.g., soft noodles, porridge).
Rough or spicy foods and heavy physical activity should be avoided for two weeks. For patients with extensive wounds or bleeding risks, the fasting duration should be extended.
(3) For patients infected with H. pylori, eradication therapy must be recommended postoperatively.
(4) Endoscopic follow-ups must be carried out three, six, and twelve months after the operation, then every year. For non-curative resections, due to the higher risk of recurrence or lymph node metastasis, additional surgical treatment is advised.
I. Endoscopic submucosal dissection (ESD)
Endoscopic submucosal dissection (ESD) is a highly specialized endoscopic technique used for treating early gastric cancer. It involves identifying lesion boundaries, marking the area, injecting the submucosa, adequately lifting the lesions, and gradually dissecting the submucosa from the muscularis propria using an electrosurgical knife. The technique ensures complete en bloc resection of the lesion, followed by wound management. As a super minimally invasive surgery, ESD offers higher en bloc and complete resection rates with lower local recurrence rates compared to endoscopic mucosal resection (EMR).
(A) Surgical procedures and techniques
1. Anesthesia and positioning. All patients undergo tracheal intubation and general anesthesia. The patient is placed in a left lateral decubitus position. Special attention is given to maintaining body warmth, adjusting room and infusion temperatures, and preventing pressure ulcers.
2. Step-by-step procedure
(i) Circumferential marking: the lesion is assessed using magnifying endoscopy and chemical or electronic chromoendoscopy to define the lesion’s boundaries. Circumferential marks are created 3–5 mm away from the lesion edges using an electrosurgical knife or argon plasma coagulation (APC), spacing the marks 2–5 mm apart.
(ii) Submucosal injection: multipoint submucosal injections are performed around the lesion, proceeding distally to proximally. A saline solution/methylene blue/epinephrine mixture is used to separate the submucosal layer from the muscularis propria and adequately lift the lesion.
(iii) Circumferential incision: once the lesion is sufficiently elevated, a circumferential incision is made approximately 3 mm outside the marked boundary using an electrosurgical knife. The incision starts from the distal edge of the lesion. If bleeding occurs, flush to locate the bleeding point and achieve hemostasis using an electrosurgical knife or coagulation forceps (figure 9.1).
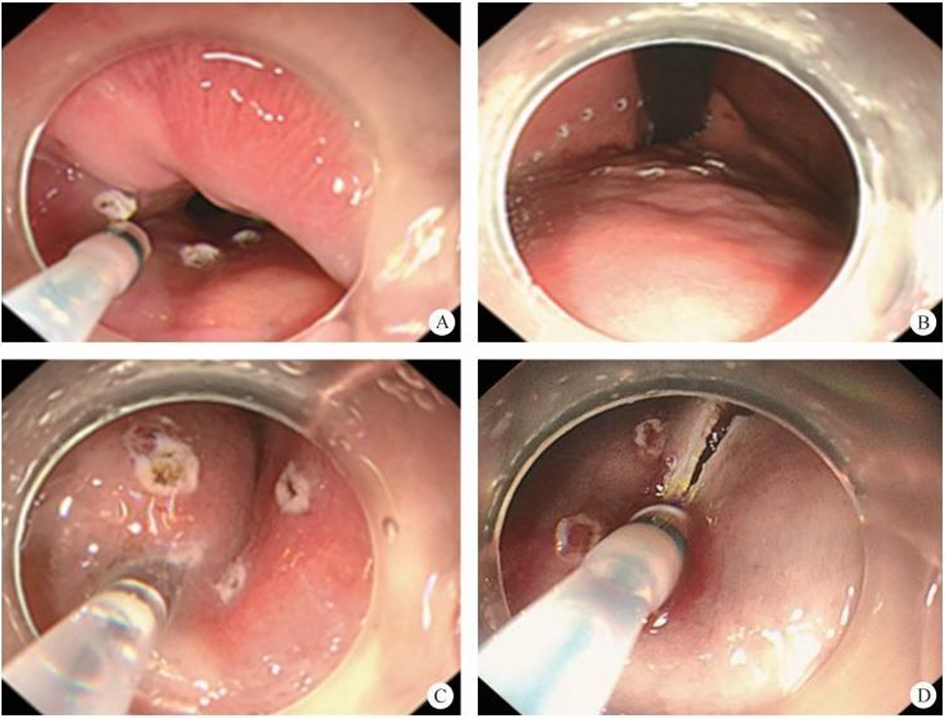

Steps of marking and circumferential incision during peroral super minimally invasive non-full-thickness resection for early gastric cancer. A. Marking the lesion (oral side). B. Completed marking. C. Submucosal injection. D. Incision at the lesion’s margin.
(iv) Submucosal dissection: using an electrosurgical knife, submucosal dissection is performed beneath the lesion until complete resection. During the procedure, submucosal injections must be performed continuously to maintain an adequate elevation of the submucosal layer. Address exposed blood vessels promptly using an electrosurgical knife or a coagulation forceps (figure 9.2).
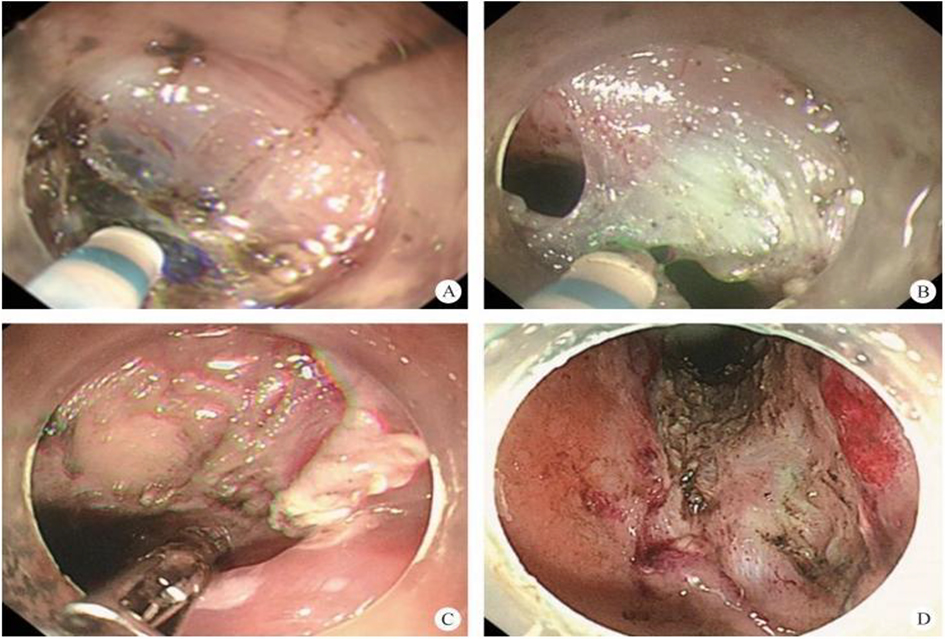

Steps of dissection and wound management in peroral super minimally invasive non-full-thickness resection for early gastric cancer. A. Submucosal dissection along the submucosal layer. B. Resection of the final portion with the assistance of lesion gravity traction. C. Preventive hemostasis on the surface of the wound. D. Observation of the wound to ensure it is completely closed, there are no potential bleeding points, and no injury to the muscularis propria.
(v) Wound management: the blood vessels exposed on the wound surface, especially along the incision margins, are thoroughly coagulated using coagulation forceps or argon plasma coagulation (APC). Biological adhesives or mucosal protectants applied on the surface of the wound if necessary. In case of injury to the muscularis propria, tissue clips can be used to seal the damage and prevent complications.
Refer to video 9.1, Peroral super minimally invasive non-full-thickness resection for early cancer at the gastric cardia and fundus (See online resources).
(B) Classic cases
Case 1: peroral super minimally invasive resection for early cancer of the gastric body
Case History: the patient, an elderly woman, was admitted with a chief complaint of “acid reflux and heartburn for two years, and detection of the gastric body protrusion for over a year.” Imaging and endoscopy: in March 2022, an abdominal CT during a routine health check revealed a 4.0 cm × 2.5 cm mucosal protrusion on the lesser curvature of the gastric body. On February 27, 2023, an enhanced abdominal CT indicated a 2.5 cm × 4.0 cm soft tissue mass protruding into the gastric cavity on the lesser curvature of the gastric body. On April 26, 2023, gastroscopy revealed a mucosal protrusion with a nodular, uneven surface and congestion near the posterior wall of the greater curvature of the gastric body. No clear ulceration was observed. Local biopsy (six specimens) showed chronic inflammation with moderate-to-severe glandular dysplasia. Ultrasonography findings: probe-based endoscopic ultrasonography revealed a heterogeneous internal echotexture with predominantly hyperechoic features. The lesion appeared to originate from the mucosal layer. The gastric wall’s structural layers were partially obscured, but the muscularis propria was intact bilaterally. The lesion’s cross-sectional size was approximately 2.48 cm × 1.33 cm.
Past medical history: hypertension, diabetes, and coronary artery disease. A physical examination revealed no abnormalities.
Diagnosis: early-stage cancer of the gastric body.
Following a comprehensive preoperative evaluation, the patient’s nutritional status was improved, and informed consent for the surgery was obtained. Peroral super minimally invasive non-full-thickness resection was then performed.
Refer to video 9.2, Peroral super minimally invasive non-full-thickness resection for early cancer of the gastric body (See online resources).
Surgical procedure: upon endoscopic insertion, a protrusion was observed near the posterior wall of the greater curvature of the gastric body. The lesion had a nodular, uneven surface with congestion, but no ulceration was detected. The lesion was circumferentially marked using argon plasma coagulation (APC). Submucosal injection of a methylene blue/saline solution was administered, achieving adequate elevation of the lesion. The lesion margins were circumferentially incised with an IT knife. Following additional submucosal injections, the IT knife and the triangle knife were alternately used for layer-by-layer dissection until the lesion was completely detached from the wound bed. Weak areas in the muscularis propria were secured using tissue clips. Hemostasis was meticulously performed on the wound surface using hemostatic forceps, and the area was sprayed with biological glue to ensure adequate protection and promote healing.
Case 2: early cancer of the gastric antrum treated by super minimally invasive resection
Medical history: the patient, an elderly man, was admitted due to a gastric antrum protruding lesion discovered two weeks prior. On April 22, 2023, gastroscopy revealed a localized ulcerative protrusion on the lesser curvature of the gastric antrum, approximately 1.5 × 1.5 cm, with a central depression and elevated periphery. Two biopsies were taken from the soft lesion, and a pathological examination indicated high-grade focal dysplasia of the glandular epithelium. The patient had no significant past medical history except for an early cardia cancer treated with endoscopic mucosal resection in 2011. Physical examination upon admission showed no abnormalities.
Diagnosis: Early cancer of the gastric antrum.
After thorough preoperative evaluation, nutritional optimization, and obtaining informed surgical consent, the patient underwent a super minimally invasive resection via the oral route.
Refer to video 9.3, Super minimally invasive non-full-thickness resection for early cancer of the gastric antrum (See online resources).
Surgical procedure: the lesion, classified as 0-IIa + IIc type, was located on the lesser curvature of the gastric antrum, with surface hyperemia. Using magnifying narrow-band imaging (M-NBI), the lesion’s boundaries were distinct. Irregular microstructures and microvasculature were observed at the lesion’s periphery, with a loss of central microstructure. No white opaque substances were noted. The lesion was lifted with a grasper, revealing a soft texture and no significant adhesions beneath. The lesion’s margins were marked circumferentially with argon plasma coagulation (APC). The mucosal layer was circumferentially incised using a DualKnife, and submucosal dissection was alternated between an IT knife and a Triangle knife. Finally, the lesion was removed using an electrocautery snare. Hemostasis was achieved with hemostatic forceps, and a fibrin sealant was applied to the wound, with no evidence of active bleeding.
Case 3: early gastric angular cancer treated by super minimally invasive resection
Medical history:
The patient, an elderly woman, was admitted due to a high-grade intraepithelial neoplasia in the gastric angle detected over a month prior. On May 5, 2023, gastroscopy revealed a flat, elevated mucosal lesion on the anterior wall of the gastric angle with clear boundaries. A single biopsy from the lesion revealed chronic inflammation with moderate to severe glandular dysplasia. Abdominal CT showed no lymph node metastasis. Physical examination upon admission revealed nothing abnormal.
Diagnosis: early gastric angular cancer.
Following comprehensive preoperative evaluation, nutritional optimization, and obtaining informed consent, the patient underwent a Super Minimally Invasive Resection via the oral route.
Refer to video 9.4, Super minimally invasive non-full-thickness resection for early gastric angular cancer (See online resources).
Surgical Procedure: the lesion, approximately 3 cm in diameter, classified as IIa + IIc, was located on the lesser curvature of the anterior wall of the gastric angle. The lesion’s margins were sprayed with indigo carmine for enhanced visualization. APC was used to mark the lesion circumferentially. Following submucosal injection, the lesion was dissected using a DualKnife and IT knife alternately. Careful hemostasis was achieved, and a fibrin sealant was sprayed onto the wound. No active bleeding was observed.
(III) Key issues and research progress in ESD
1. Prevention of post-ESD bleeding. A critical aspect of safe endoscopic submucosal dissection (ESD) is the prevention of post-ESD bleeding. Postoperative bleeding primarily results from the exposure of mucosal defects after lesion resection. Preventive measures include covering or closing mucosal defects, but the indications for such interventions in early gastric cancer remain unclear. Variations in lesion and defect size contribute to inconsistent findings across studies.
Biodegradable materials, such as polyglycolic acid (PGA) sheets and fibrin glue (FG), have been proposed to cover mucosal defects. Preliminary studies have shown their effectiveness in preventing postoperative bleeding. However, subsequent randomized controlled trials (RCTs) failed to yield robust results, though the intervention group exhibited delayed bleeding onset compared to controls, suggesting a potential benefit in preventing delayed bleeding. Current research on PGA-FG application after gastric ESD is predominantly retrospective or lacking controls, with prospective studies limited to single-center, small-sample designs and follow-up durations of less than one month.
Another approach involves closing mucosal defects, typically using direct clipping. However, closures often fail within a short period due to inadequate adherence of mucosal edges. Advanced endoscopic suturing techniques have recently been introduced for defect closure after gastric ESD. Techniques for closing large defects include purse-string sutures using nylon or dental floss combined with clips, detachable snare-assisted clipping, TTSC, and OTSC, with success rates ranging from 61% to 91.7%. However, these studies primarily focus on lesions smaller than 3 cm in diameter.
2. Establishing follow-up standards after early gastric cancer ESD. For patients undergoing non-curative resection of early gastric cancer through endoscopic submucosal dissection (ESD), deciding whether to perform additional gastrectomy and lymph node dissection is critical. The 5th edition of the Japanese Gastric Cancer Association (JGCA) guidelines, published in 2018, introduced the eCura system to evaluate the curability of ESD for early gastric cancer. This system aids in prognosis assessment and provides a foundation for follow-up and subsequent treatment plans.
For patients with differentiated early gastric cancer who achieve curative resection (eCura A or B), tumor recurrence, rather than lymph node or distant metastasis, is the primary factor affecting prognosis. Conversely, for patients with non-curative resection (primarily eCura C-2), lymph node and distant metastases significantly influence prognosis. Studies show that in patients with intramucosal cancer meeting eCura A and B criteria (excluding SM1-type eCura B), the cumulative incidences of metachronous gastric cancer at five, seven, and ten years were 9.5%, 13.1%, and 22.7%, respectively. These patients require ongoing surveillance.
While ESD indications for early gastric cancer have expanded, patients with non-curative resection (eCura C-2) face higher risks of distant and lymph node metastases. Achieving curative resection necessitates eradicating potentially cancerous lymph nodes. However, the low rate of lymph node metastasis in surgical specimens and the predominance of distant metastases result in limited effectiveness of additional surgical treatment. Therefore, observation without surgery is a feasible alternative.
Determining whether non-curative ESD patients should undergo additional surgery based on the risk of lymph node metastasis remains challenging. Patient factors, such as comorbidities, life expectancy, and surgical tolerance, must also be considered.
The follow-up standards for early gastric cancer ESD based on curability grading need further refinement and clinical validation. Physicians should develop follow-up plans after thorough communication and agreement with patients based on the specific curability grading. This approach not only improves patient adherence but also facilitates practical insights to enhance curability-based follow-up standards, paving the way for more personalized follow-up strategies for different patients with early gastric cancer.
II. Traction-assisted endoscopic submucosal dissection
Traction-assisted endoscopic submucosal dissection (ESD with traction) involves various traction methods during the procedure to fully expose the submucosal dissection field. This technique reduces complications such as bleeding and perforation and shortens operative time. Traction methods can be categorized into intracorporeal and extracorporeal techniques, with most applications reported in case studies.
(I) Surgical techniques
1. Anesthesia and positioning
All patients undergo tracheal intubation and general anesthesia. They are placed in the left lateral decubitus position, with precautions for maintaining body temperature, regulating room and infusion temperatures, and preventing pressure ulcers.
2. Surgical steps
1) Preparation of the traction device: dental floss, silk thread, or nylon thread are attached to one arm of a metal clip extracorporeally.
2) Circumferential marking: magnifying endoscopy, combined with chemical or electronic staining, is used to evaluate the lesion and define its boundaries. Circumferential marking is performed at 3–5 mm from the lesion’s edge using an electrosurgical knife or APC, with intervals of approximately 2 mm between markings.
3) Submucosal injection: injections into the submucosal layer are made at multiple points around the lesion, starting distally and moving proximally, to lift the lesion and separate it from the muscularis propria.
4) Circumferential incision: after sufficient lifting, the mucosa is incised circumferentially along the markings, approximately 3 mm outside the lesion boundary, using an electrosurgical knife. Start from the distal end, and manage bleeding with irrigation to locate the source, followed by electrosurgical or coagulation forceps for hemostasis.
5) Submucosal dissection: after mucosal incision, the submucosal layer is dissected from top to bottom, alternating with submucosal injections. The lesion must be gradually detached while ensuring clear separation between the lesion’s mucosal layer and the muscularis propria. Use a thread-attached metal clip to provide traction, adjusting its direction for optimal visibility and ease of dissection.
6) Wound management: coagulation of exposed blood vessels around the wound edges is performed using coagulation forceps or APC. Fibrin sealants or protective agents are applied as needed. For injuries to the muscularis propria, the defects are closed with clips.
7) Traction methods:
a) Rubber-band traction: For large early gastric cancers, a combination of a rubber band and tissue clips can be employed to achieve multidirectional traction, thereby providing optimal visualization of the surgical field. After partial dissection of the lesion, one end of the rubber band is secured to the partially dissected mucosal layer using a tissue clip, while the other end is anchored to the normal mucosa on the opposite side of the lesion. Multiple traction points can be utilized depending on the dissection progress to achieve the best results. During the procedure, care must be taken to avoid excessive tension on the gastric wall to prevent over-pulling (figure 9.3).
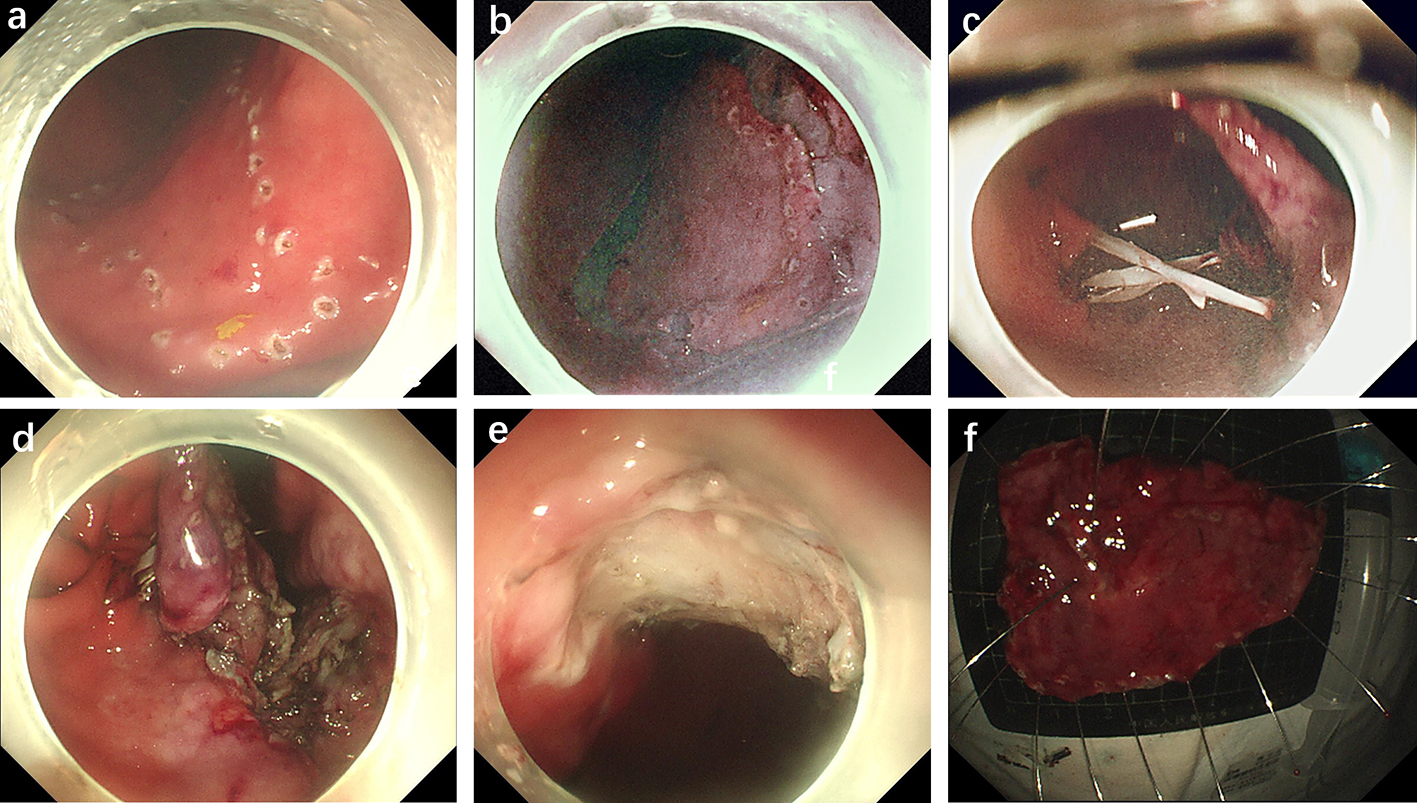

Rubber-band traction method for early gastric cancer in traction-assisted ESD. A. Marked early gastric cancer. B. Circumferential incision around the lesion. C. Multi-directional traction with two sets of rubber bands and tissue clips. D. Modified as rubber band with clip traction. E. Postoperative wound surface. F. the specimen of the lesion.
b) Single-angle traction using tissue clip and dental floss: After partial lesion dissection, one end of the dental floss is attached to a tissue clip, which is then fixed to the partially dissected mucosal layer. The other end of the dental floss is pulled out through the mouth, allowing external traction to provide a controlled pulling force (figure 9.4).
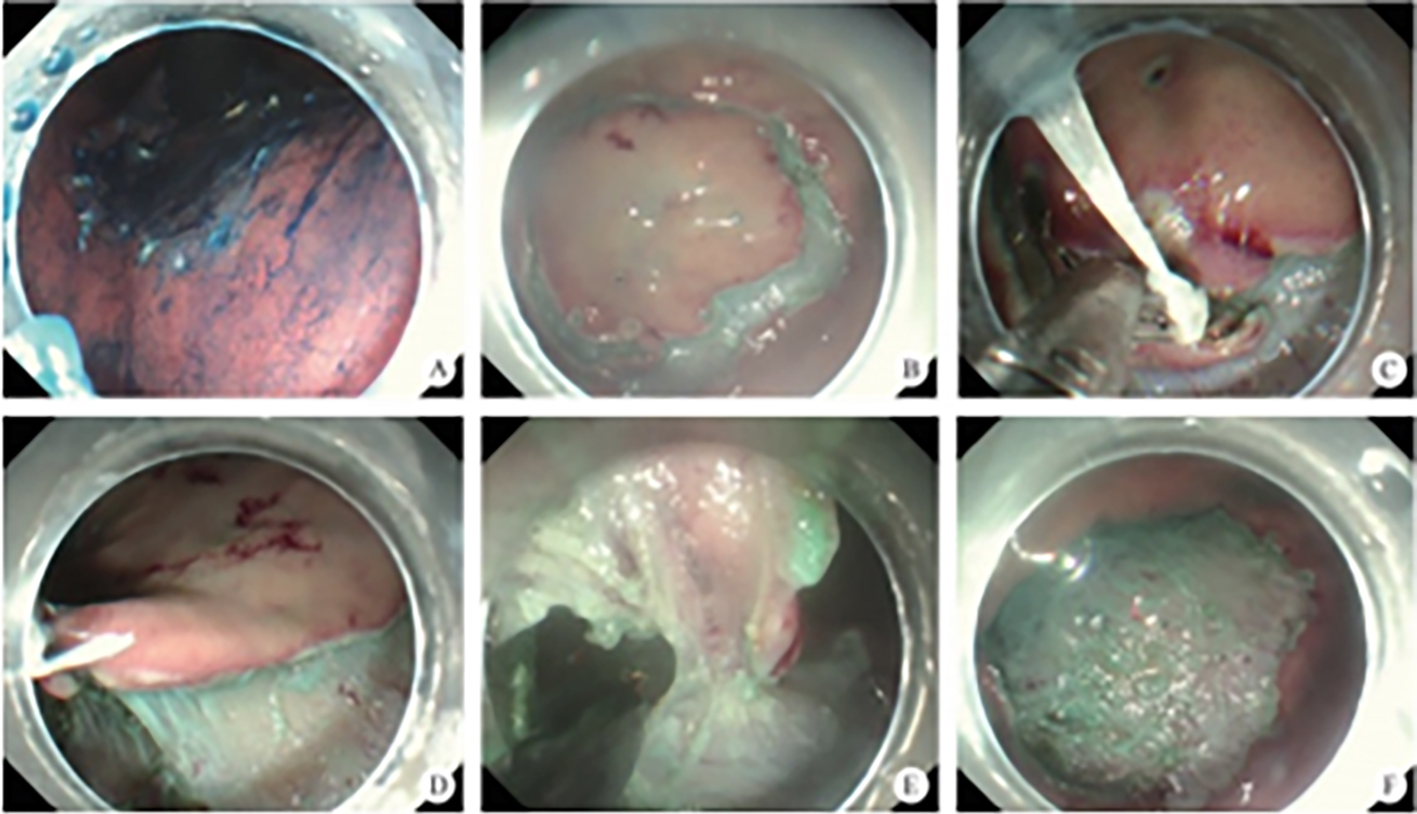

Single-angle traction with tissue clip and dental floss in early gastric cancer traction-assisted ESD. A. Marked early gastric cancer. B. Circumferential incision around the lesion. C. Tissue clip and dental floss fixed to the peeled side of the mucosa. D. Lifting the lesion with dental floss and tissue clip. E. Clear separation of the mucosal and muscular layers after traction. F. Postoperative wound surface.
c) Variable-angle traction using tissue clip and dental floss: if single-angle traction does not provide adequate visualization, an additional tissue clip can be attached to the opposite side of the lesion to create a pulley system. As the dissection progresses, the two fixed tissue clips gradually converge, allowing for continuous adjustment of the traction angle and improved surgical field visualization (figure 9.5).
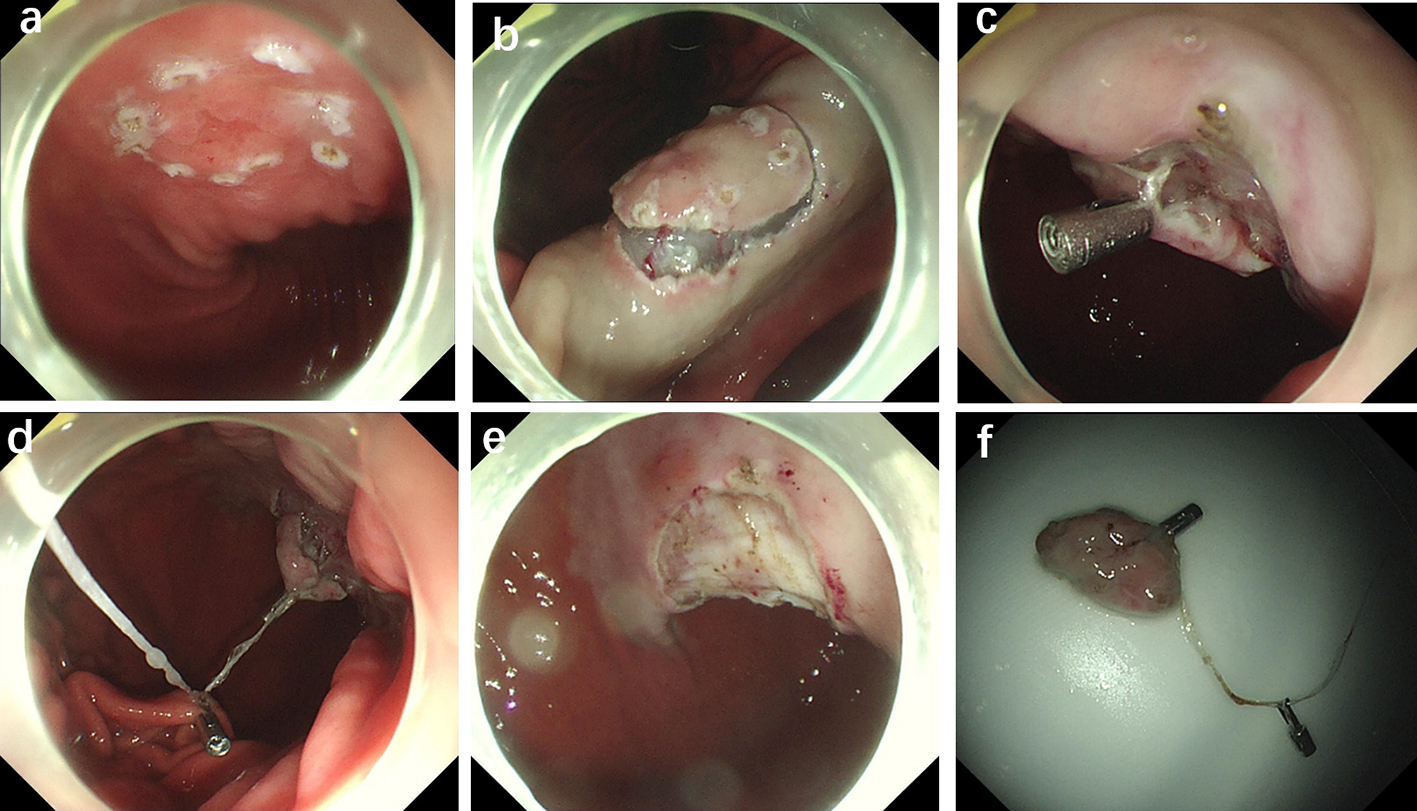

Dual-side traction with tissue clip and dental floss in early gastric cancer traction-assisted ESD. A. Marked early gastric cancer. B. Circumferential incision around the lesion. C. Tissue clip and dental floss fixed to the peeled side of the mucosa. D. Tissue clip secures the dental floss to the contralateral mucosa. E. Postoperative wound surface. F. Tissue clip-dental floss-lesion specimen.
d) Magnetic anchoring and traction-based SMIS surgery for early gastric cancer (figure 9.6): Recent studies have reported the application of magnetic anchoring-assisted super minimally invasive surgery (MAG–SMIS) in the treatment of early gastric cancer (EGC). The principle involves utilizing the flexible angle control enabled by a magnetic force to provide an optimal surgical field. This approach is particularly suitable for extensive lesions or those located in challenging anatomical areas.
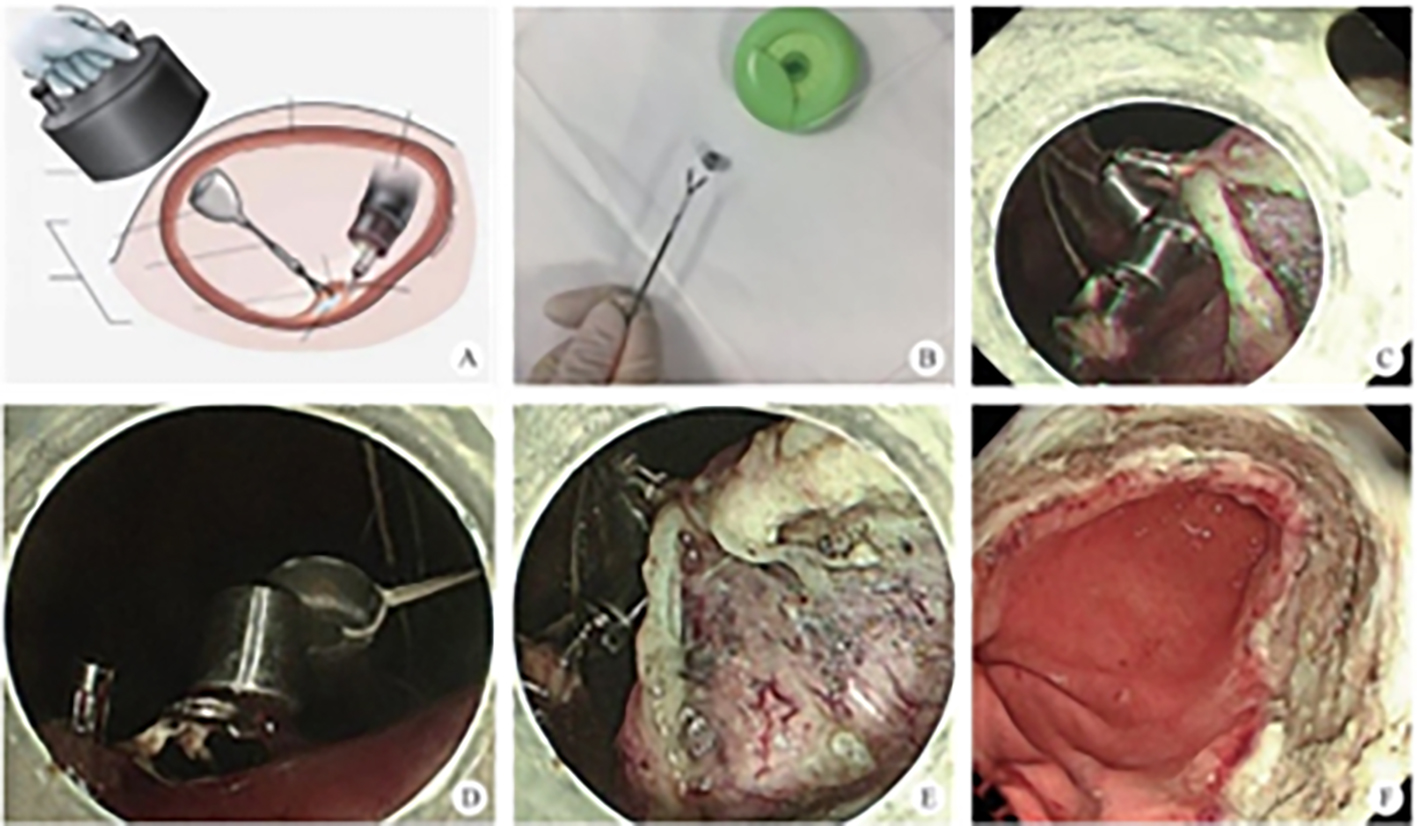

Magnetic anchoring traction method in early gastric cancer SMIS surgery. A. Schematic diagram of magnetic anchoring and traction SMIS surgery. B. Magnetic-assisted instruments used in the procedure. C, D. Internal traction. E. Clear demarcation between the mucosal and muscular layers after traction. F. Postoperative wound surface in SMIS.
Procedural steps for magnetic anchoring and traction-based SMIS surgery for early gastric cancer:
Preoperative preparation and initial procedures: preoperative assessments, marking, injection, and incisions around the lesion margins follow the standard procedures of conventional super minimally invasive SMIS–ESD (submucosal dissection).
Magnetic anchoring implementation: a magnetic hammer is connected to a soft tissue clip using dental floss. The clip is then secured to the lesion and to the slightly mobilized mucosal layer, achieving magnetic anchoring.
Magnetic traction: an external magnet is used to attract the internal magnetic hammer. By adjusting the angle and relative position of the two magnets under endoscopic guidance, a variable-angle magnetic traction system is created, offering an enhanced surgical view for super minimally invasive submucosal dissection of early gastric cancer. While magnetic anchoring technology simplifies endoscopic submucosal dissection (ESD), several areas require optimization in clinical practice.
Miniaturization of external magnets: the size and precision of external magnets need to be further reduced to improve intraoperative control of the internal magnet’s position, meeting clinical demands effectively.
Impact of abdominal wall thickness: the magnetic force is influenced by the abdominal wall thickness of the patient. Excessively obese patients are unsuitable for magnetic anchoring-assisted surgery, underscoring the need for adequate coupling strength of the selected magnets.
Potential biological effects of magnetic fields: magnetic fields may pose potential risks to the human body and interact with ferromagnetic foreign bodies within the patient. Thus magnetic anchoring is contraindicated in patients with cardiac pacemakers, metal stents, or similar devices. Rigorous preoperative screening of patients is essential.
(II) Study of a classic case
Case history: the patient, a middle-aged man, was admitted with a chief complaint of “nausea for over ten years and gastric mucosal lesions discovered during a health examination 23 days ago.” On June 5, 2024, gastroscopy revealed significant congestion and edema of the mucosa at the gastric angle, with surface erosion involving the anterior wall of the antrum, which bled easily upon contact. Histopathological findings indicated: (gastric angle) high-grade intraepithelial neoplasia of the gastric mucosa with scattered small foci and papillary arrangement of cells consistent with adenocarcinoma. Immunohistochemical results showed p53 (+), CK broad-spectrum (+), partial CK7 (+), Ki-67 (70%+), and HER-2 (0). The patient had no previous significant medical history, and physical examination upon admission was unremarkable.
Diagnosis: early-stage gastric antrum cancer.
After thorough preoperative evaluation, nutritional status improvement, and obtaining informed surgical consent, the patient underwent an ESD-assisted dental floss traction procedure. Refer to video 9.5, Super minimally invasive non-full-thickness resection for early cancer of the gastric antrum (See online resources).
Surgical procedure: A protruding lesion was identified on the lesser curvature of the gastric antrum near the gastric angle, involving the anterior and posterior walls of the gastric antrum, with slight central depression and partial surface erosion. Indigo carmine staining was performed, followed by circumferential marking of the lesion with an electrocautery knife. After submucosal injection, the lesion was lifted, and a circumferential mucosal incision was made with an electrocautery knife. A dental floss traction system with clips was placed at the caudal center. Submucosal injection and incision continued along both lateral edges, layer by layer, until reaching the center of the lesion. The traction system was then used to elevate the lesion, enabling further dissection until complete detachment. Visible penetrating vessels between the muscularis and the mucosa were coagulated, and the wound was closed with clips after meticulous hemostasis.
(III) Hot-spot issues and research advances in the consensus
1. Applicability of traction-assisted ESD in early gastric cancer treatment. Due to anatomical factors, most current studies on traction-assisted ESD focus on esophageal and colorectal lesions. Research indicates that traction correlates with shorter operative times, lower adverse event rates, and reduced perforation rates, without significantly impacting R0 resection rates. Subgroup analyses suggest superior efficacy and benefits of traction in esophageal and colorectal ESD. In gastric ESD, the routine advantages of traction are less clear. However, it can be beneficial for lesions in the upper and middle thirds of the stomach, particularly along the greater curvature. Anatomically, these findings are reasonable because vertical traction is challenging unless the lesion is located on the greater curvature.
Reports suggest that the most challenging sites for ESD include the pylorus and duodenal bulb, where complications are more common. For tumors located at the pylorus extending into the duodenum, requiring extensive circumferential resection, complete resection rates are reduced. Traction-assisted ESD can achieve en bloc resection while shortening operative time. The safety, efficacy, and advantages of traction-assisted ESD over conventional ESD for early gastric cancer treatment require further validation.
2. Selection of optimal traction methods and directions. In traction-assisted ESD, limitations in traction methods and directions often reduce efficacy in specific cases. Selecting appropriate traction methods based on lesion location to achieve optimal traction direction is crucial, though such studies are limited. Vertical traction may be the optimal direction for gastric ESD. While other internal traction methods, including pulley systems, dual-scope, and magnetic-anchor techniques, might provide vertical traction, their feasibility in gastric ESD remains unclear and requires evaluation.
A primary limitation of traction systems is the decreasing force as submucosal fibers lose elasticity during ESD. Additional challenges include adjusting traction strength, interference between traction devices and endoscopes, and the potential need for specialized equipment. Common traction methods in gastric ESD include clip-line, internal traction, and sheath traction techniques. The dual-channel endoscope traction method leverages “dual-channel” advantages without requiring endoscope withdrawal for device installation, but the direction of traction is restricted. The clip-line method is widely used due to its simplicity and lack of need for special equipment, though it requires endoscope withdrawal for clip placement and offers limited directional flexibility. Among internal traction methods, the S–O clip-assisted ESD allows multi-directional traction. Studies show that S–O clip-assisted ESD can reduce operative time. Additionally, adaptive traction systems (ATRACT) have been developed to address traction force attenuation. However, these studies often involve high device requirements, single-center designs, and lack external validation.
Currently, the clip-line method is most commonly used in China, with limited application of external snare traction and magnetic traction techniques. In summary, each traction method has its advantages and disadvantages. The ideal traction method should be simple, economical, convenient, effective, and not reliant on overly complex equipment. Future development should focus on devices that provide adjustable and easily controllable traction force and direction, facilitating the safe, efficient, and high-quality execution of ESD, thereby further expanding its clinical application.
9.1.3 Super Minimally Invasive Full-Thickness Resection for Early Gastric Cancer
Super minimally invasive surgery (SMIS) is an effective approach for curing early gastric cancer (EGC). It encompasses non-full-thickness endoscopic resection (e.g., endoscopic submucosal dissection, ESD) and endoscopic full-thickness resection (EFTR). Currently, EFTR is recognized as a safe and effective method for treating gastrointestinal stromal tumors, and its potential for EGC treatment is being increasingly explored. Traditional EFTR techniques often involve perforation followed by closure, which may lead to complications such as pneumoperitoneum or abdominal infection. Here, we report a case of SMIS–EFTR for complete resection of EGC, highlighting its potential to enhance cure rates and reduce recurrence.
(I) Case characteristics
1. Medical history. The patient, a middle-aged man, was admitted for “ulcerative lesion in the upper gastric body discovered 13 days prior.” On July 25, 2023, a gastroscopy performed at an outside hospital revealed an ulcerative lesion approximately 0.6 cm × 0.6 cm on the greater curvature side of the upper gastric body near the posterior wall. The lesion was covered with a thin coating at the center, surrounded by ridge-like mucosal thickening and crowding. Pathological examinations indicated moderately to poorly differentiated adenocarcinoma. The patient reported no symptoms such as abdominal pain, distension, belching, nausea, vomiting, or weight loss. Stool and urinary habits were normal. The patient presented to our gastroenterology department with a preliminary diagnosis of “gastric ulcer: early cancer?” for further evaluation. Relevant medical history included hypertension and previous endoscopic polypectomy for intestinal polyps. The patient had smoked 20 cigarettes daily for 30 years but denied excessive alcohol consumption. He had no significant travel history, surgical history, or other major medical conditions. The family history revealed that the patient’s father had died of rectal cancer.
2. Examinations. Magnifying gastroscopy + NBI: a depressed mucosal lesion (approximately 0.6 cm × 0.6 cm) was detected on the greater curvature side of the upper gastric body near the posterior wall. The lesion surface was covered with mucus, surrounded by crowded nodular ridge-like mucosal elevations with disrupted folds. High-magnification imaging revealed clear boundaries, irregular microvascular architecture, and absent glandular structures (figure 9.7A–C). Pathological examinations confirmed moderately to poorly differentiated adenocarcinoma. Endoscopic ultrasound (EUS): the lesion involved the submucosa, with possible invasion into the muscularis propria (figure 9.7D). PET–CT: no abnormal metabolic activity has been evidenced in the gastric wall. Multiple lymph nodes in the hepato-gastric space were identified without increased metabolic activity. Small reactive lymph nodes with slightly elevated metabolism were noted in the cervical and hilar regions. Abdominal contrast-enhanced CT and ultrasonography: evaluations of bilateral cervical, supraclavicular, axillary, and inguinal regions revealed no evidence of tumor or metastasis. Laboratory tests: blood counts, biochemical profiles, tumor markers, coagulation tests, stool tests (including occult blood), and other routine examinations showed no significant abnormalities.
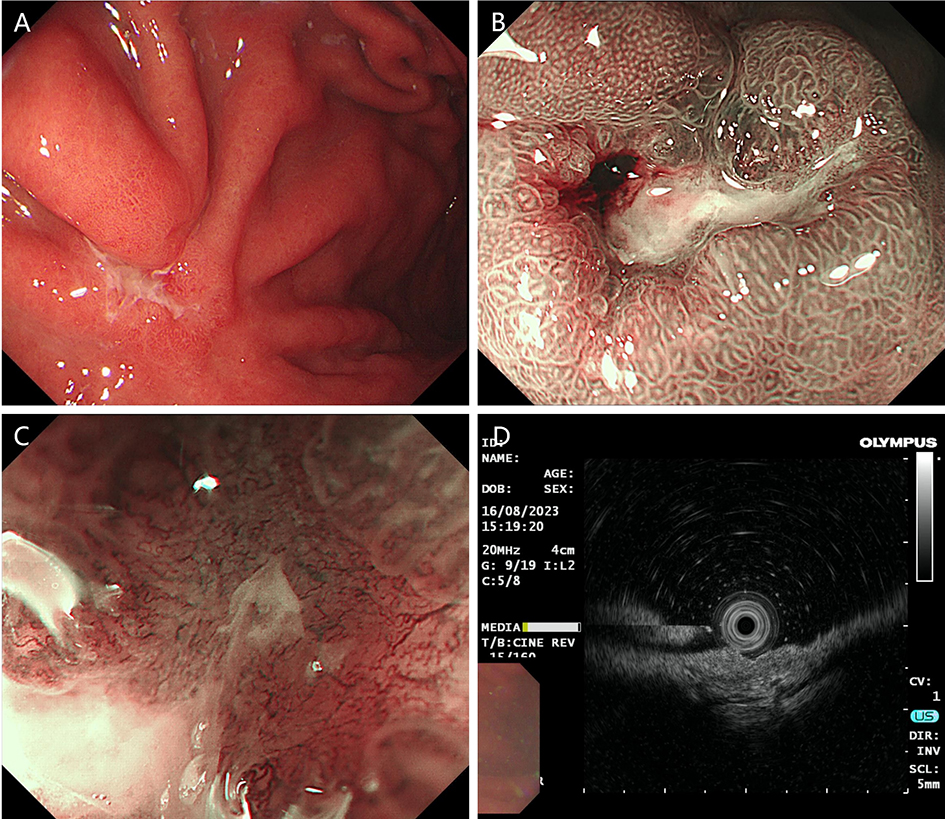

Endoscopic presentation of early gastric cancer. A. Ulcerative lesions observed endoscopically in early gastric cancer. B. Clear boundaries observed with narrow-band imaging (NBI). C. Magnifying endoscopy reveals abnormal microvasculature and an absence of glandular structure. D. Miniprobe endoscopic ultrasonography shows lesion invasion into the submucosal layer.
3. Preoperative preparation. Multidisciplinary consultation involving gastroenterology, general surgery, oncology, and radiology was conducted to formulate a comprehensive treatment plan. Following thorough preoperative discussions and obtaining informed consent for SMIS, the patient underwent super minimally invasive full-thickness resection for early gastric cancer via endoscopy.
(II) Surgical procedures and techniques
1. Anesthesia and positioning. All patients underwent tracheal intubation with general anesthesia and were positioned in the left lateral decubitus position. Care was taken to maintain body warmth, regulate room temperature, and adjust infusion fluid temperature to prevent pressure ulcers.
2. Surgical procedure (figures 9.8 and 9.9)
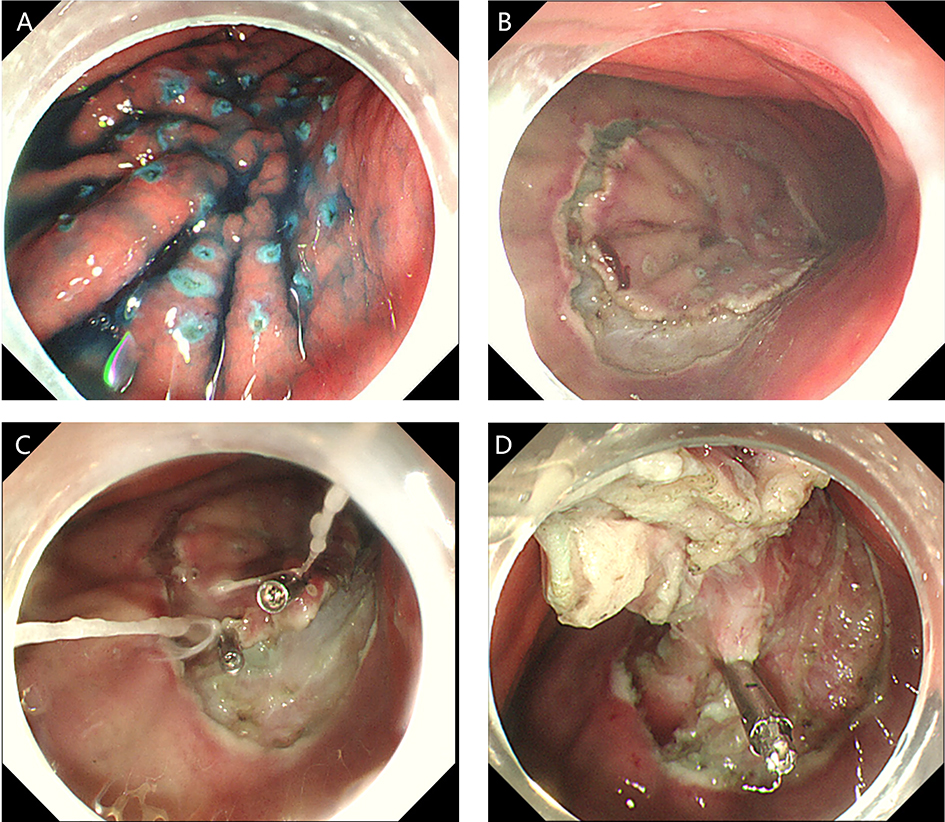
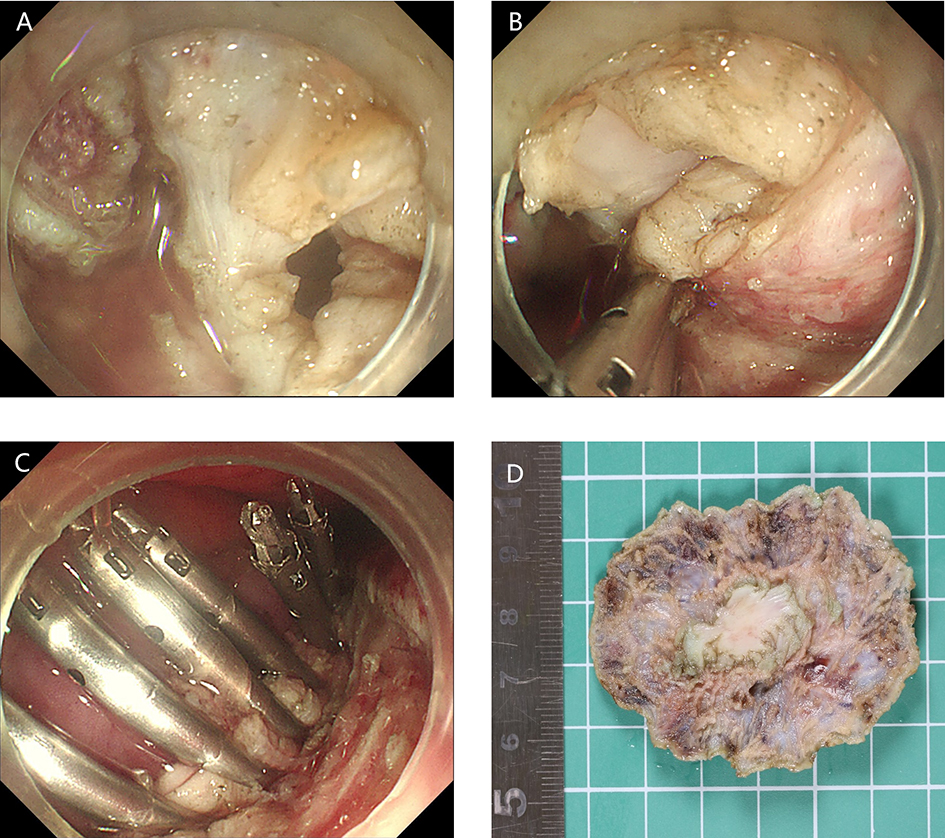

Outer circle steps of super minimally invasive full-thickness resection for early gastric cancer via oral approach. A. Dual-layer marking around the lesion. B. Circumferential incision of the outer mucosal layer. C. Traction of the lesion using tissue clips and dental floss. D. Anchoring the first tissue clip for orientation.

Inner circle steps of super minimally invasive full-thickness resection for early gastric cancer via oral approach. A. Perforation visible after full-thickness incision. B. Simultaneous full-thickness incision and closure of the wound surface with tissue clips. C. Closure of the muscular layer wound surface after full-thickness resection. D. Serosal side of the resected specimen.
(1) Lesion marking: indigo carmine dye was sprayed to delineate the lesion boundaries. The inner circular marking was made circumferentially using argon plasma at a distance of 0.5 cm from the lesion. The outer circular marking was created at a distance of 1 cm from the lesion using argon plasma, with an additional marking on the oral side.
(2) Submucosal injection and dissection: following submucosal injection, the lesion edges were elevated. The outer circular marking of the mucosa was circumferentially incised, followed by submucosal dissection with concurrent injections.
(3) Lesion traction and completion of outer circular ESD: a traction system using two sets of tissue clips and dental floss was applied to achieve a clear delineation between the mucosal and muscularis propria layers. Submucosal injection and circumferential dissection were performed until reaching the first circular marking.
(4) Full-thickness incision after positioning: the traction line, along with the muscularis propria layer, was lifted. A tissue clip was applied to secure and close the inner circular marking of the muscularis propria. Subsequently, a full-thickness incision was performed.
(5) Closure and full-thickness incision: in cases of active perforation, tissue clips were used to close the perforation while performing the full-thickness incision until the lesion was completely detached from the surgical bed.
(6) Wound management: meticulous hemostasis was achieved at the wound site. Additional tissue clips were applied to ensure a secure closure of the wound, followed by the application of biological protein glue for further coverage.
Refer to video 9.6, Super minimally invasive full-thickness resection for early gastric cancer of the gastric body (See online resources).
(III) Postoperative management
(1) Postoperative care: after surgery, patients were treated with continuous gastric tube decompression for two to three days, fasting for three to four days, proton pump inhibitor (PPI) infusion for three to four days, fluid replacement for three to four days, and anti-infective treatment for two days. Gradual dietary transition was initiated, with discharge typically occurring one week postoperatively.
(2) Pathological results of en bloc resection specimen: pathological examination revealed moderately to poorly differentiated adenocarcinoma of the gastric body (located in S9 and S12-16). Microscopic measurements showed a lesion size of 1.7 cm × 1.2 cm, with submucosal invasion to a depth of 1300 μm (SM3). No vascular or lymphatic invasion was detected, and both horizontal and basal resection margins were negative for carcinoma. This case achieved en bloc and curative resection, meeting the standards for expanded indications of endoscopic treatment for early gastric cancer (EGC). Additional surgical or chemotherapeutic intervention was deemed unnecessary. Short-term follow-up, including gastroscopy or magnifying endoscopy, non-contrast chest CT, contrast-enhanced abdominal and pelvic CT, superficial lymph node ultrasound or PET–CT, and tumor marker analysis, was recommended at three months, six months, and one year postoperatively. Two weeks after the surgery, the patient reported no changes in quality of life.
(IV) Hot-spot issues and advances in the consensus
Under the therapeutic goal of “cure the disease with minimal trauma,” surgical techniques have evolved from traditional open surgery and minimally invasive surgery (MIS) to super minimally invasive surgery (SMIS). Traditional surgery achieves disease removal by “resecting part or all of an organ and reconstructing anatomical structures,” but this approach often results in the loss of normal anatomy and unavoidable negative effects on patients. In 2016, Professor Enqiang Linghu recognized the limitations of traditional open and minimally invasive surgery and proposed a novel surgical paradigm that preserves the original anatomical structure while eliminating disease, allowing patients to recover to their pre-illness state. With advancements in endoscopic equipment and surgical techniques, this ideal surgical model is now widely applied in digestive disease treatment.
Surgery remains the most effective curative option for gastric cancer. However, the choice of the surgical approach is a major clinical challenge. While radical gastrectomy with lymph node dissection improves local disease control and survival rates, the extent of necessary lymph node dissection remains controversial. In Western countries, routine D2 lymphadenectomy is now recommended for resectable gastric cancer due to its long-term survival benefits, despite its association with increased postoperative mortality, morbidity, and reoperation rates.
With advances in endoscopic diagnostics, early detection of gastric cancer has become more prevalent, enabling curative treatment through surgery. However, even in cases without pathological lymph node metastasis (LNM), patients undergoing gastrectomy often experience long-term complications such as dumping syndrome and symptoms related to vagotomy. Many resectable EGC cases do not involve LNM, underscoring the need for individualized surgical approaches that balance curability with minimizing postoperative complications, avoiding unnecessary extensive lymphadenectomy.
As early gastric cancers rarely exhibit lymph node metastasis, treatment for certain IMC cases increasingly favors localized resection without lymphadenectomy. This approach effectively cures most patients, preserves the stomach’s anatomical integrity, and avoids complications, maintaining quality of life. For patients with pathologically confirmed IMC following complete tumor resection, lymph node metastasis is presumed absent, eliminating the need for adjuvant therapies such as chemotherapy, additional surgery, or lymphadenectomy. However, potential undetected LNM poses a latent risk, necessitating refined SMIS techniques and treatment strategies.
Preoperative assessment classified this patient as EGC with tumor features of ulcerated, moderately to poorly differentiated adenocarcinoma, diameter ≤ 20mm, submucosal invasion on EUS evaluation, and no imaging evidence of LNM. Although the case exceeded current standard ESD indications, the patient and family strongly requested stomach-preserving treatment. After thorough expert evaluation and informed consent, the patient underwent ‘super minimally invasive surgery–endoscopic submucosal dissection–full-thickness resection (SMIS–ESD–FTR).’ This procedure established a standardized operational protocol for this technique.
According to the 5th edition of the JGCA guidelines, endoscopic curative resection for early gastric cancer (EGC) is classified into eCura A to C-2. Radical resection is classified as eCura A, expanded radical resection as eCura B, and the outcomes of various differentiated mucosal carcinomas under expanded radical resection are also categorized as eCura A. Non-curative resection is divided into eCura C-1 and eCura C-2, with eCura C-1 corresponding to partially ineffective cases and eCura C-2 to other cases. A risk scoring system is established based on five indicators: tumor diameter, vertical margin, lymphatic vessel invasion, vascular invasion, and depth of invasion. The system stratifies patients into low-risk, intermediate-risk, and high-risk groups, with lymph node metastasis (LNM) risks of 2.5%, 6.7%, and 22.7%, respectively. At the First Medical Center, Department of General Surgery, a retrospective cohort study collected clinical data from 1633 EGC patients who underwent radical gastrectomy and had postoperative pathological staging as early gastric cancer between December 2005 and December 2020. This dataset was used to develop a nomogram prediction model for lymph node metastasis in EGC. Multivariate analysis identified the following independent risk factors for lymph node metastasis in EGC: female sex, ulcerative tumor morphology, vascular and lymphatic emboli, tumor long axis ≥3 cm, greater invasion depth, and poor differentiation.
Based on the risk factors for lymph node metastasis in EGC and the eCura risk stratification system, treatment strategies after endoscopic therapy are guided accordingly. For this patient, postoperative pathology indicated complete and curative resection. Pathological features included moderately to poorly differentiated adenocarcinoma of the gastric body, submucosal invasion depth of 1300 μm (SM3), lesion size of 1.7 × 1.2 cm, and no vascular or lymphatic invasion. The horizontal and basal resection margins were clear of cancer. Therefore, treatment options for this patient include adjuvant chemotherapy, surgery, or surveillance. After comprehensive evaluation, the patient opted for postoperative adjuvant chemotherapy with S-1 (tegafur) administered at 60 mg for two weeks followed by a one-week rest, for a total of six cycles. This regimen aligns with the NCCN Clinical Practice Guidelines in Oncology for Gastric Cancer and the Chinese Society of Clinical Oncology (CSCO) guidelines for single-agent S-1 use in neoadjuvant chemotherapy for resectable gastric cancer.
This case holds significant importance as it establishes a standard surgical protocol for super minimally invasive full-thickness resection (SMIS–ESD–FTR) for EGC. SMIS–ESD–FTR involves three key steps: marking two circles around the lesion to define the surgical area, performing standard ESD on the outer circle, and conducting full-thickness resection within the inner circle. The procedure addresses four major challenges:
Difficulty in closing full-thickness defects: this is resolved through a muscle-to-muscle closure technique. Specifically, dual-circle marking is employed, with the inner circle marked similarly to classic submucosal dissection and the outer circle involving expanded marking with submucosal preservation. Full-thickness resection is performed on the inner circle, and the preserved muscular layer from the outer circle is closed using clips.
Complications such as pneumoperitoneum and peritoneal infection from perforation: a stepwise closure-resection approach is used to prevent gas and fluid leakage into the abdominal cavity, with decompression as needed.
Hemostasis of bleeding from extramural gastric vessels: before cutting the muscular layer, tissue clips and dental floss traction are used to elevate the lesion. The first clip seals the inner circle, followed by full-thickness cutting with electrocautery while applying additional clips to block bleeding. This technique effectively prevents extramural bleeding, which is difficult to control endoscopically.
Selection of surgical instruments: quick muscular incision requires the use of a triangular knife. During closure-resection procedures, non-conductive tissue clips are selected to avoid severe burns caused by electrical conductivity.
In conclusion, with advancements in gastrointestinal endoscopic techniques, local resection without lymphadenectomy is increasingly accepted. This case represents an exploratory and standardized approach to super minimally invasive full-thickness resection for gastric cancer, expanding its indications. Postoperative pathology should be carefully monitored to determine the necessity of additional surgery or chemotherapy.
9.1.4 Early Gastric Cancer: Super Minimally Invasive Resection via the Tunnel Technique
The super minimally invasive resection of early gastric cancer can be achieved using the endoscopic submucosal tunnel dissection (ESTD) technique. ESTD, a branch of the Digestive endoscopic tunnel technique (DETT), is an innovative method for the complete resection of early gastrointestinal cancers through the creation of a submucosal tunnel. Compared to conventional endoscopic submucosal dissection (ESD), ESTD offers several advantages: reduced frequency of submucosal injections during dissection, improved exposure of the operative field due to mutual traction of tissues within the tunnel, and blunt dissection facilitated by the transparent cap on the endoscope tip. These features enhance dissection efficiency and lower the complication rate. Preliminary studies have shown that ESTD is associated with shorter operative time, faster dissection speed, higher en bloc resection rates, and fewer complications than traditional ESD.
(I) Indications and contraindications of ESTD
1. Indications. ESTD is primarily indicated for the resection of large early gastric cancers with a transverse diameter ≥3 cm, as well as lesions with ulceration or severe fibrosis. Suitable anatomical locations include the cardia, the lesser curvature of the gastric body, and the greater curvature of the gastric antrum.
2. Contraindications. Lesions with a depth of invasion exceeding the submucosal layer as detected by endoscopic ultrasound (EUS). Presence of lymph node or distant metastases confirmed by EUS or other imaging techniques. Advanced or undifferentiated carcinoma. Contraindications for endoscopy or anesthesia, such as coagulation disorders or cardiopulmonary dysfunction.
(II) Surgical procedure and techniques
1. Anesthesia and positioning. All patients undergo tracheal intubation under general anesthesia and are positioned in the left lateral decubitus position. Care must be taken to maintain warmth, adjust room and infusion temperatures, and prevent pressure ulcers.
2. Standard steps of ESTD (figures 9.10 and 9.11)

Steps for determining the lesion and performing bilateral incisions during super minimally invasive tunnel resection for early gastric cancer. A. White-light endoscopy used to identify the lesion. B. Lesion marking following border delineation using indigo carmine staining. C. Submucosal injection followed by incision of the distal (anal) mucosa. D. Submucosal injection followed by incision of the proximal (oral) mucosa.

Steps of submucosal tunnel creation and mucosal resection in super minimally invasive surgery for early gastric cancer. A. Submucosal injection and dissection are performed simultaneously to establish a tunnel from the oral to the anal side. B. Dissection continues until the anal-side tunnel incision is reached. C. Mucosal incisions are made on both sides. D. The lesion is completely dissected, and the wound surface is treated with coagulation forceps.
(1) Circumferential marking: after evaluating the lesion with magnifying endoscopy and confirming its boundaries using chemical or electronic chromoendoscopy, circumferential markings are made 3–5 mm from the lesion margin. An electrocautery knife or argon plasma coagulation (APC) is used to create marks spaced approximately 2 mm apart.
(2) Submucosal injection: multiple injections are administered around the lesion, starting from the distal side and proceeding to the proximal side. This raises the mucosal layer from the muscularis propria to facilitate separation.
(3) Mucosal incision: using an electrocautery knife, incisions are made on the distal (anal) and proximal (oral) sides of the lesion, following the circumferential markings. The incision length is approximately 1.5–2.0 cm.
(4) Tunnel creation: submucosal dissection begins from the proximal incision, creating a submucosal tunnel toward the distal incision. During tunnel creation, the markings on both sides should be clearly observed, and the tunnel direction should align with the lesion’s morphology and trajectory to avoid excessive mucosal dissection.
(5) Lesion resection: the lesion is completely resected by sequentially incising the mucosa along the boundaries on both sides with an electrocautery knife.
(6) Wound management: the exposed wound surface, especially around the resection margins, is treated with sufficient coagulation using coagulation forceps or APC. Biological adhesives or mucosal protectants may be applied as needed. In cases of muscularis propria damage, tissue clips should be used to seal the injury.
(III) Key issues and advances in consensus
1. Indications for ESTD in early gastric cancer. Current consensus on endoscopic treatment for early gastric cancer (EGC) indicates that endoscopic submucosal tunnel dissection (ESTD) is primarily suited for the removal of large EGC lesions with a horizontal diameter ≥3 cm, as well as lesions accompanied by ulceration or severe fibrosis. However, no case reports or relevant studies are currently available specifically targeting ulcerated and fibrotic lesions. Additionally, in cases of recurrence following ESD for early gastric cancer, the original resection site often exhibits significant scar formation and adhesion, which diminishes the effectiveness of submucosal injection. This makes repeated ESD relatively challenging and increases the risk of bleeding and intrinsic muscular layer injury. The efficacy of ESTD for treating in situ recurrent early gastric cancer warrants further exploration.
2. Challenges in submucosal tunnel creation and balancing the benefits of ESTD. Due to the unique anatomical and physiological characteristics of the stomach, such as its large and non-linear lumen, non-fixed position, and high flexibility, creating a submucosal tunnel is technically more challenging compared to the esophagus. Lesions located at the pyloric ring or in areas of significant curvature, such as the gastric fundus, are particularly difficult for submucosal tunnel formation.
As ESTD and submucosal tunneling endoscopic resection (STER) share the same third-space endoscopic principles, optimal locations for creating submucosal tunnels include the cardia, lesser curvature of the gastric body, and greater curvature of the antrum. ESTD can be attempted for lesions with a horizontal diameter >3 cm. Previous studies have demonstrated that lesions located on the lesser curvature of the distal gastric body are suitable for ESTD. However, large-scale studies evaluating ESTD treatment for early gastric cancer in various locations are still lacking. Further research is needed to determine the optimal cut-off points for lesion length and width when choosing between ESD and ESTD. The consensus also suggests that ESTD and traction-assisted techniques are both applicable for lesions on the greater curvature of the stomach, potentially reducing operative time. However, no comparative studies have yet been conducted on ESTD and traction-assisted ESD for treating early gastric cancer in specific locations (e.g., the greater curvature). Identifying the best treatment approach for early gastric cancer in such special locations remains an area for further investigation.
9.1.5 Early Gastric Cancer Treated with Multi-Channel Super Minimally Invasive Resection
The treatment of early gastric cancer with multi-channel super minimally invasive resection is achieved through laparoscopic and endoscopic cooperative surgery (LECS). LECS combines the advantages of endoscopy and laparoscopy, allowing local gastric resection and regional lymph node dissection. This technique expands the indications for endoscopic treatment while ensuring curative outcomes, preserving the normal anatomical structure and function of the stomach to the greatest extent possible. Initially, LECS involved endoscopic identification of lesions followed by en bloc full-thickness gastric wall resection and laparoscopic closure of the gastric wall defect. This process, however, required creating communication between the gastric cavity and the abdominal cavity, increasing the risk of tumor dissemination and implantation. Subsequent modifications to LECS have reduced these risks to varying degrees.
(A) Indications and contraindications for laparoscopic and endoscopic cooperative surgery
1. Indications. ① Early gastric cancer patients meeting the absolute indications for endoscopic submucosal dissection (ESD) but facing technical challenges during endoscopic procedures, such as tumors with a diameter >3 cm, tumors located on the greater curvature of the gastric body or the anterior gastric wall, or tumors exhibiting severe ulcerative changes. ② Elderly patients unable to tolerate radical surgery or those with low requirements for tumor radicality.
2. Contraindications ① Contraindications to general anesthesia, such as allergies to anesthetic agents or severe cardiopulmonary diseases. ② Coagulation disorders or inability to discontinue antiplatelet or anticoagulant medications, leading to a high risk of intraoperative bleeding. ③ Advanced-stage cancer or imaging evidence of distant metastasis. ④ Pregnancy or other contraindications to endoscopic diagnosis and treatment.
(B) Preoperative preparation
1) Patients and their families should be provided with detailed information about the risks and benefits of super minimally invasive resection and obtain written informed consent.
2) Preoperative assessments, including routine blood tests, blood biochemistry, coagulation function, eight-item serum tests, blood typing, electrocardiography, chest X-ray or CT, abdominal CT, and anesthesia evaluation, along with other endoscopy-related examinations, must be completed.
3) Use of anticoagulants, antiplatelet agents, or other blood-thinning medications must be verified, and their use adjusted or discontinued based on the patient’s condition.
4) At least 8 h of fasting and water restriction must be observed on the day of surgery. Oral defoaming and mucolytic agents must be administered preoperatively.
5) Surgery must be prepared according to standard surgical protocols.
(C) Surgical procedures and techniques (figure 9.12)
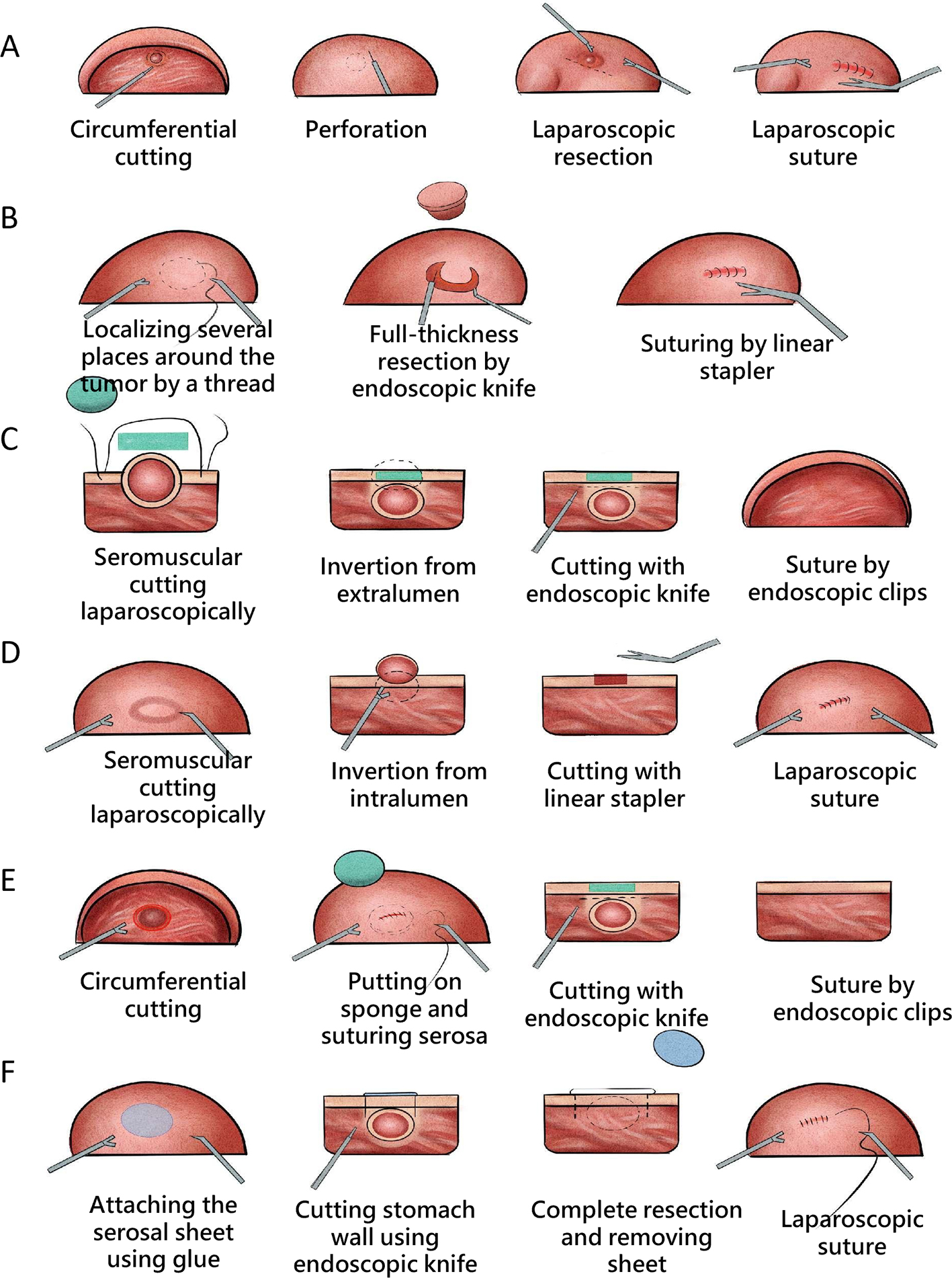

Six types of LECS procedures.
1. Classic exposure LECS. This approach combines laparoscopic gastric wall resection and ESD. Lesions are identified endoscopically, followed by en bloc full-thickness resection using endoscopy and/or laparoscopy. Gastric wall defects are closed laparoscopically using staplers or sutures. However, due to the risk of tumor spillage or implantation, its application in early gastric cancer is limited.
2. Inverted LECS. In this method, mucosal incision lines are pre-marked endoscopically, and submucosal dissection is completed endoscopically. During full-thickness resection via endoscopy and laparoscopy, the tumor remains protruding into the gastric cavity, with the surrounding gastric wall tissue maintained in an elevated state. Gastric wall defects are closed using staplers, and the tumor specimen is extracted orally through endoscopy. However, this technique also involves full-thickness gastric wall incision, which establishes communication between the gastric and abdominal cavities, posing risks of gastric content contamination and tumor cell dissemination into the peritoneal cavity.
3. Non-exposure endolaparoscopic full-thickness resection with the simple-suturing technique (NESS-EFTR). This laparoscopy-assisted endoscopic full-thickness resection involves a combination of seromuscular layer suturing via laparoscopy to achieve gastric wall inversion, endoscopic full-thickness resection (EFTR), and endoscopic mucosal suturing using loops and clips.
4. Non-exposed endoscopic wall-inversion surgery (NEWS). This technique begins with endoscopic mucosal marking and incision line determination around the lesion. Under endoscopic guidance, serosal marking is performed laparoscopically. A liquid cushion is created through endoscopic submucosal injection, followed by seromuscular layer incision and suturing via laparoscopy. The lesion is inverted into the gastric cavity, after which ESD is performed. Due to its multi-step nature, high technical demands, and extended operation time, NEWS is generally not recommended for lesions larger than 3 cm. The tumor specimen is retrieved orally via endoscopy.
5. Combined laparoscopic and endoscopic approach for neoplasia with a non-exposure technique (CLEAN-NET). This approach begins with endoscopic mucosal marking, supplemented by laparoscopic serosal marking under endoscopic guidance. Four full-thickness sutures are placed around the predetermined serosal incision line via laparoscopy to secure and elevate the gastric wall. The seromuscular layer is incised outside the sutures, lifting the full-thickness gastric wall containing the lesion and the adjacent mucosa. The gastric wall is then resected and closed simultaneously using a laparoscopic stapler, with the tumor specimen extracted laparoscopically. Throughout the procedure, the continuity of the mucosal layer is preserved, preventing communication between the gastric and abdominal cavities, effectively mitigating the risks of gastric content leakage and tumor cell dissemination into the peritoneal cavity. However, precise delineation of the tumor margin remains challenging on the serosal side, and special care is required to prevent positive surgical margins.
6. ESD combined with laparoscopic lymph node dissection. This technique involves performing ESD first, followed by laparoscopic regional lymph node dissection based on the primary tumor’s location and gastric lymphatic drainage patterns.
(IV) Hot-spot issues and research progress in the consensus
1. Requirements for the location and size of lesions in LECS for early gastric cancer (EGC). Currently, existing guidelines recommend laparoscopic radical gastrectomy (D1 or D1+ lymph node dissection) for EGC cases with cT1a and cT1b staging outside the absolute indications for ESD. However, some Japanese and Korean scholars are actively exploring the use of LECS for EGC patients who are challenging to treat with ESD. These include tumors larger than 3 cm, located on the greater curvature of the gastric body or the anterior gastric wall, or exhibiting severe ulcerative changes. Both classic LECS and inverted LECS require opening the gastric wall during the procedure, which poses risks of tumor dissemination into the abdominal cavity or leakage of gastric contents. Kikuchi et al. proposed a closed LECS technique that avoids gastric wall opening; however, its application for EGC has not yet been reported. CLEAN-NET requires the tumor to protrude completely outside the gastric wall to ensure adequate margins, limiting its applicability for EGC located on the posterior wall of the upper third of the stomach. Although large-scale clinical studies confirming the efficacy and safety of LECS techniques for EGC are lacking, laparoscopy-assisted endoscopic full-thickness resection (LAEFTR) and NEWS have been reported to be applicable for EGCs of any location or size, ensuring sufficient vertical and horizontal margins. If intraoperative findings suggest altered gastric morphology or postoperative complications arise, standard gastrectomy can be performed as an additional procedure without significantly affecting the patient’s postoperative quality of life or residual gastric function. For patients with preoperative indications of lymph node metastasis, D1 or D1+ lymph node dissection can be performed laparoscopically, preserving the gastric function while achieving curative outcomes. Each LECS-based treatment method has its advantages and limitations, and currently, no standardized criteria exist for lesion location and size requirements for different LECS techniques. Further research with large-scale databases is necessary to establish these criteria.
2. Requirements for lymph node dissection in LECS. The potential risk of lymph node metastasis is a crucial factor in selecting treatment methods for EGC. LECS, by combining ESD for lesion removal with laparoscopic lymph node dissection, embodies the concept of super minimally invasive surgery, preserving the anatomical integrity and function of the stomach. Li et al. demonstrated that combining ESD with laparoscopic sentinel lymph node dissection minimizes unnecessary gastrectomies and excessive lymph node dissection. They identified submucosal infiltration, tumor diameter >2 cm, and lymphovascular invasion as potential risk factors for postoperative pathological positive lymph node metastasis.
The Korean SENTINEL Node Navigation Oriented Tailored Approach (SENORITA) research group has pioneered the feasibility of sentinel node navigation surgery (SNNS) in treating EGC. Short-term results confirm that local resection combined with sentinel node drainage area dissection is non-inferior in surgical safety compared to traditional laparoscopic gastrectomy. Theoretically, local resection is advantageous for preserving the gastric function, thereby improving nutritional status and quality of life postoperatively. However, evaluations remain based on retrospective big data analyses of surgical specimens. Current limitations of SNNS include cumbersome intraoperative lymph node tracing, a certain failure rate, and a proportion of false-negative intraoperative lymph node biopsies. These issues hinder its clinical application for EGC surgical treatment. Further research is required to determine the optimal indications and tracing methods for supplementary lymph node dissection in combined therapies.
Future exploration of lymph node metastasis patterns in EGC through big data analysis is expected to guide individualized lymph node dissection strategies, offering greater hope for EGC patients.
9.2 Super Minimally Invasive Surgery for Gastric Low-Grade Intraepithelial Neoplasia
9.2.1 Overview
(1) Overview of gastric low-grade intraepithelial neoplasia
Gastric low-grade intraepithelial neoplasia (LGIN) refers to atypical cellular and structural characteristics that differ to varying degrees from the normal gastric mucosa from which it originates. These include mild to moderate dysplasia and are classified as precancerous lesions of gastric cancer. Histologically, LGIN is characterized by mild crowding of gastric glandular cells that are similar in size and shape, with orderly basal alignment and mild to moderate nuclear hyperchromasia. Endoscopically, LGIN typically presents as flat, elevated lesions with smooth surfaces and pale mucosa. Treatment options include super minimally invasive radiofrequency ablation (RFA) under endoscopy. According to a clinical study involving 253 cases, this technique demonstrated a short-term (one-year) cure rate of 91.5% and a long-term (three-year) cure rate of 88.5%. Postoperative complications were minimal, with only mild upper abdominal pain lasting ≤14 days, and no reported cases of bleeding, perforation, or infection. These findings suggest that RFA has the potential to become a mainstream treatment modality for gastric LGIN in the future.
(2) Diagnosis of gastric low-grade intraepithelial neoplasia
The clinical diagnosis of gastric LGIN primarily relies on histopathological examination of biopsy specimens obtained through endoscopy, evaluated based on the WHO/Vienna classification criteria. However, the biopsy method itself has inherent randomness and limitations, leading to varying degrees of discrepancy between the biopsy findings and the true nature of the lesion. Even with advanced endoscopic techniques such as magnifying endoscopy (ME) combined with narrow-band imaging (NBI), these limitations cannot be entirely eliminated. Therefore, where feasible, timely diagnostic resection of suspected lesions to obtain macroscopic specimens for a comprehensive pathological assessment of the lesion is recommended. For all biopsy-diagnosed LGIN lesions, standardized, detailed endoscopic evaluation should be performed, which includes assessment of lesion size, surface morphology, phenotype, and coloration. Special attention should be paid to ME + NBI techniques for observing lesion boundaries and surface microstructures. Where necessary, chromoendoscopy with dyes such as indigo carmine or acetic acid may be employed. Lesions larger than 2 cm, lesions with clearly defined boundaries, or those exhibiting abnormal surface microstructures suggest a higher likelihood of pathological progression. These should be considered high-risk factors within LGIN and warrant endoscopic intervention.
(3) Technical methods and previous nomenclature in the implementation of super minimally invasive surgery for gastric low-grade intraepithelial neoplasia
Gastric low-grade intraepithelial neoplasia (LIN) can achieve curative outcomes through super minimally invasive surgery (SMIS), utilizing endoscopic treatments via natural orifice transluminal approaches. These techniques include: endoscopic radiofrequency ablation (RFA); argon plasma coagulation (APC); Holmium laser therapy (HLT). Each method represents a minimally invasive approach designed to optimize therapeutic efficacy while reducing patient burden.
9.2.2 Low-Grade Intraepithelial Neoplasia of the Stomach: Peroral Super Minimally Invasive Radiofrequency Ablation
The peroral super minimally invasive radiofrequency ablation (RFA) procedure, previously referred to as endoscopic radiofrequency ablation (ERFA), involves the application of radiofrequency electrodes to the target lesion under endoscopic guidance. The technique utilizes radiofrequency current to induce coagulative necrosis of local tissues, aiming to eradicate pathological changes. ERFA demonstrates an efficacy rate of 91.3% with no reported severe complications. Its short-term therapeutic efficacy in treating low-grade intraepithelial neoplasia (LGIN) is comparable to endoscopic submucosal dissection (ESD).
(A) Indications and contraindications
1. Indications. (1) Patients diagnosed with gastric LGIN based on strict and standardized magnifying endoscopy screening and biopsy in accordance with the WHO/Vienna classification criteria. (2) Lesion morphology conforms to the Paris classification type 0-II as observed during endoscopy.
2. Contraindications. (1) Confirmed or suspected high-grade intraepithelial neoplasia (HGIN) or early gastric cancer based on standardized endoscopic screening and biopsy following WHO/Vienna classification. (2) Presence of contraindications to endoscopic treatment or examination, such as severe cardiopulmonary dysfunction or uncorrectable coagulopathy. (3) Concurrent malignancies in other organs or systems. (4) Pregnancy. (5) Long-term use of anticoagulant medications without the possibility of discontinuation.
(B) Preoperative preparation
1. The patient and the family should be informed in detail of the potential outcomes of gastric LGIN, including progression to high-grade intraepithelial neoplasia (HGIN) or early gastric cancer, long-term stability, or natural regression. Discuss the potential clinical benefits, risks, adverse events, postoperative recovery management, follow-up, and other relevant matters associated with the super minimally invasive radiofrequency ablation procedure.
2. Routine preoperative examinations, including blood routine, blood biochemistry, coagulation function, and serological tests must be completed and verified. Additional assessments such as electrocardiogram (ECG), chest X-ray (or chest CT), anesthesia evaluation, and any other necessary diagnostic procedures must be carried out. Antiplatelet or anticoagulant medications should be discontinued at least one week before surgery.
2. The patient must fast for at least 8 h, refraining from food and water. Fifteen minutes before the procedure, 50–60 ml of an anti-foaming and anti-mucolytic agent containing streptokinase granules and simethicone must be administered, followed by 10 min of positional rotation on the bed. Five minutes prior to the procedure, the patient should hold 5–10 ml of tetracaine hydrochloride gel solution in his/her mouth, swallowing it after 1–2 min.
(C) Surgical procedures and techniques
1. Anesthesia and positioning. All patients undergo intravenous general anesthesia and are positioned in the left lateral decubitus position. Vital signs are monitored continuously, and oxygen supplementation is provided. Alternatively, sedation with intravenous midazolam and pethidine may be administered.
2. Surgical steps (figure 9.13)
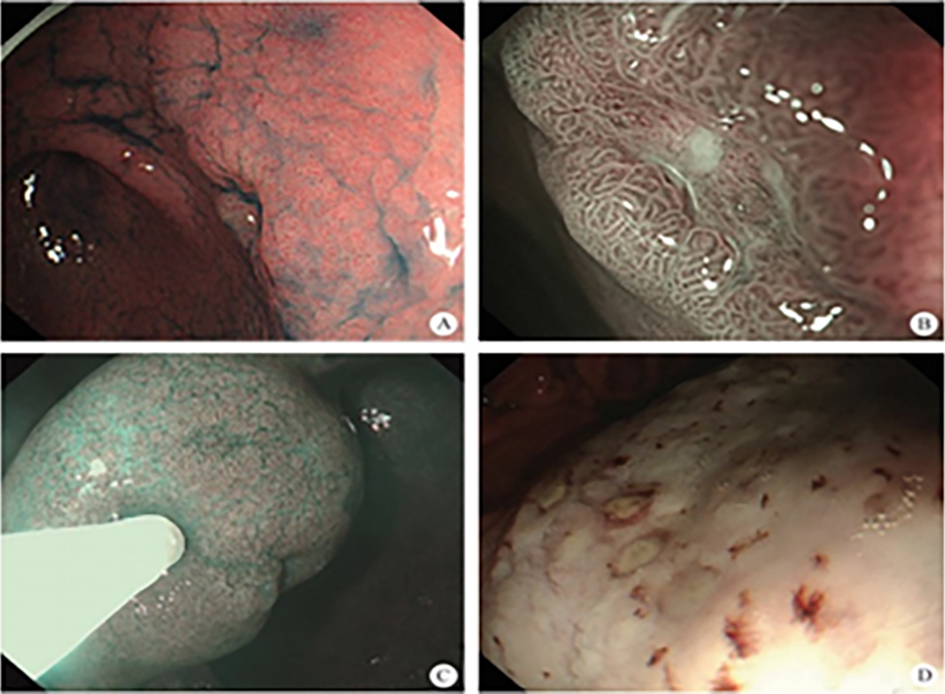

Steps of endoscopic radiofrequency ablation (RFA). A. Indigo carmine staining: enhances visualization of lesion boundaries and surface details. B. Re-evaluation with magnifying endoscopy after staining: confirms the lesion’s location and extent. C. Submucosal injection: elevates the lesion by injecting a saline solution beneath the mucosa. D. RFA coverage: the ablation area extends 0.5–1 cm beyond the lesion boundaries to ensure comprehensive treatment.
(1) Observation and staining: before treatment, magnifying endoscopy (ME) combined with narrow-band imaging (NBI) should be conducted to reconfirm the lesion location, define its boundaries, and assess surface microstructures and microvasculature. This process also helps exclude high-grade intraepithelial neoplasia (HGIN) or early gastric cancer. If these methods do not sufficiently visualize the lesion, indigo carmine staining may be employed as an auxiliary technique. Following the observation, a biopsy should be performed within the lesion area to provide further diagnostic evidence.
(2) Boundary marking and submucosal injection: for relatively large lesions observed during endoscopy, marking the lesion boundaries under ME is crucial to ensure complete coverage during subsequent RFA. Additionally, submucosal injection must be made at the edges of the lesion using a multi-point approach, injecting 2–3 ml of saline solution at each point to sufficiently elevate the lesion mucosa.
(3) Endoscopic radiofrequency ablation (RFA): the magnifying endoscope must be replaced with a therapeutic endoscope (e.g., GIF-Q260J/HQ290) and the radiofrequency ablation generator must be connected, with an output power of 57 W and an energy density of 15 J/cm
Refer to video 9.7, Super minimally invasive radiofrequency ablation for gastric low-grade intraepithelial neoplasia (See online resources).
(D) Postoperative management
1. The patient should fast for 4–6 h postoperatively, with appropriate fluid replacement provided.
2. Feeding should begin with cool liquid foods, gradually switching to semi-liquid and regular diets as tolerated.
3. Proton pump inhibitors (PPIs) and gastric mucosal protective agents must be administered for one month postoperatively. Close monitoring of adverse events is recommended through telephone and outpatient follow-ups.
4. Patients are advised to undergo endoscopic follow-ups at three months, six months, one year, two years, three years, four years, and five years after the procedure. Each follow-up should include magnifying endoscopy (ME) combined with narrow-band imaging (NBI) and biopsies of the treated area for histopathological examination.
(E) Key issues and research progress in consensus
1. Refining indications for RFA in gastric LGIN. While RFA is appropriate for treating isolated gastric LGIN, its inability to provide post-procedural histopathology underscores the critical importance of precise preoperative diagnosis. Preoperative magnifying endoscopy (ME) combined with narrow-band imaging (NBI) is essential for accurate lesion evaluation. Additionally, multi-site targeted biopsies of identified lesions and suspicious areas are crucial. If findings suggest highly suspicious or inescapable high-grade intraepithelial neoplasia (HGIN) or early gastric cancer, RFA treatment should be approached with caution, considering endoscopic resection techniques such as endoscopic mucosal resection (EMR) or endoscopic submucosal dissection (ESD), or even surgical intervention.
2. Post-RFA management and follow-up for LGIN. Gastric LGIN carries a potential for malignancy, necessitating standardized, meticulous endoscopic assessment. Lesions > 2 cm, lesions with well-defined margins, and those exhibiting abnormal surface microstructures are high-risk factors for pathological progression. A large-scale chinese study with up to 10 years of clinical follow-up reported that 51.0%–78.7% of LGIN cases could regress, while 0.45%–14.3% progressed to cancer. These divergent outcomes complicate clinical decision-making, requiring further discussion on monitoring versus active intervention criteria.
Expert consensus suggests: LGIN with well-defined boundaries observed under high-definition endoscopy should undergo immediate endoscopic minimally invasive treatment, with a one-year follow-up post-treatment. LGIN with poorly defined boundaries should be managed with medication alongside endoscopic monitoring, with follow-up endoscopy within one year. Untreated LGIN with clear boundaries should be re-evaluated every six months using high-definition chromoendoscopy. High-risk LGIN for pathological progression should undergo meticulous re-evaluation and targeted biopsy three months after diagnosis. If pathology examinations confirm repeatedly LGIN, endoscopic diagnostic resection is recommended. For LGIN without high-risk features, re-evaluation and biopsy are advised three months after the initial diagnosis. If LGIN is reconfirmed, endoscopic ablation treatments (e.g., RFA, argon plasma coagulation [APC]) or close endoscopic surveillance every three months are recommended.
3. Role of submucosal injection in RFA. The primary complications of RFA include abdominal pain, bleeding, and perforation, with low incidence rates. Previous research indicates comparable rates of total scar formation and complete healing between groups with and without submucosal injection after RFA at one month of follow-up. However, patients in the submucosal injection group experienced less pain and predominantly developed white scars, while those without injection showed more red scars.
Submucosal fluid cushion injection during RFA for gastric LGIN protects the submucosa and the muscularis propria, alleviates postoperative pain, promotes tissue healing, reduces scar formation and fibrous adhesions, and minimizes potential complications for future ESD procedures. Additionally, for lesions in specific locations such as the gastric angle or lesser curvature of the gastric body, submucosal injection enhances lesion elevation, improves electrode contact, and facilitates the procedure.
9.2.3 Low-Grade Gastric Intraepithelial Neoplasia: Super Minimally Invasive Argon Plasma Coagulation
Super minimally invasive argon plasma coagulation (APC), previously referred to as argon plasma coagulation, is a non-contact ablative technique. Its mechanism involves ionized argon gas sprayed onto the target mucosa, transferring high-frequency electrical energy to tissues, which coagulate under high temperatures. International studies report short-term local recurrence rates of 4%–10% following APC treatment of early gastric cancer and gastric dysplasia. Chinese studies indicate an efficacy rate of 81.8% based on a follow-up one month after APC.
(I) Surgical procedure and techniques
1. Preoperative Assessment: magnifying endoscopy with narrow-band imaging (ME + NBI) is performed to confirm the location of the lesion, define its boundaries, and assess the surface microstructure and microvascular patterns.
2. Procedure: the APC catheter is inserted through the endoscopic channel, extending it approximately 1 cm beyond the scope. Maintain a distance of 0.3–0.5 cm from the lesion and mark the lesion circumferentially.
3. Coagulation: the APC device is positioned close to the surface of the lesion and the coagulation treatment is applied.
(II) Current consensus and research progress
1. Comparison of APC, RFA, and endoscopic resection (EMR, ESD). Clinical studies highlight the promising efficacy and safety of radiofrequency ablation (RFA) in the treatment of gastric LGIN. Comparative studies show that APC is slightly less effective than RFA and is better suited for lesions ≤ 2 cm. Single-center data (pending publication) reveal comparable short-term outcomes for RFA and ESD in treating gastric LGIN, though ESD exhibits slightly superior long-term results without significant statistical differences. Retrospective studies indicate higher recurrence rates with APC compared to ESD. However, recurrence after APC was managed successfully with additional treatment and follow-up. While ESD demonstrates higher efficacy, it poses a greater risk of perforation and bleeding and requires longer procedure times than APC. In contrast, APC and RFA offer advantages such as simplicity, safety, minimal trauma, rapid recovery, and suitability for outpatient settings. These benefits support their potential for broader clinical adoption, provided indications are carefully defined and long-term efficacy is further investigated.
2. Indications for gastric LGIN selection for APC therapy. Retrospective studies suggest that APC is an effective alternative to endoscopic resection for gastric LGIN <10 mm without scarring, with some studies extending this indication to lesions <20 mm. For larger lesions, careful consideration and close monitoring are necessary. APC is particularly suitable for elderly patients with comorbidities or those unable to tolerate anesthesia or surgery. Research focusing on this demographic may further validate its efficacy.
3. Risk factors and follow-up for recurrence after APC. Lesions > 2 cm are an independent risk factor for local recurrence due to uneven coagulation depth and intensity with APC. Studies on the effects of submucosal injection and power settings during APC for gastric adenomas suggest that low power (40 W) and the absence of mucosal elevation may contribute to recurrence. While submucosal injection does not significantly improve APC efficacy in elderly patients, follow-up periods remain limited. Further research on intraoperative APC parameters and their relationship to recurrence is needed.
9.2.4 Low-Grade Gastric Intraepithelial Neoplasia: Super Minimally Invasive Holmium Laser Therapy
(I) Overview
Super minimally invasive Holmium laser therapy for low-grade gastric intraepithelial neoplasia (LGIN) utilizes a Holmium laser (Ho: YAG laser), a pulsed near-infrared laser. Its light is highly absorbable by water, with a thermal effect depth typically less than 500 μm, minimizing damage to surrounding normal tissues. Holmium laser therapy has been widely applied in digestive tract interventions, particularly for early-stage tumors, Barrett’s esophagus, and atypical hyperplasia.
(II) Consensus and research progress
1. Efficacy of Holmium laser in treating LGIN. Compared to traditional electrocautery, Holmium laser therapy causes less thermal injury to surrounding tissues. Delivered via flexible optical fibers, it reduces blind spots, making it suitable for use across all regions of the gastrointestinal tract. Studies report a high success rate in treating early gastrointestinal cancers with Holmium laser, with no postoperative bleeding, perforation, or residual lesions observed in follow-ups within one to three months. However, clinical studies on Holmium laser therapy for early cancers and intraepithelial neoplasias remain limited, and its efficacy has yet to be confirmed through comparative trials with other first-line treatments. Previous research indicates that Holmium laser is effective in managing cancerous and scar-induced esophageal strictures, as well as restenosis after stent placement. For gastric LGIN combined with esophageal strictures or lesions located in specific regions such as the cardia, Holmium laser may represent a feasible dual-benefit treatment approach.
2. Power settings for Holmium laser. Ideally, Holmium laser energy should be adjusted to limit ablation to superficial layers, such as the mucosa and submucosa, avoiding damage to the muscularis propria to reduce complications like perforation. Experimental studies have explored the thermal damage to the gastric wall in rats under various laser power and duration settings, as well as the effects of Holmium laser-assisted ESD on porcine gastric walls, including postoperative perforation rates. However, these findings have not yet been translated into clinical applications.
3. Indications for endoscopic Holmium laser therapy. Current Chinese and international consensus guidelines on gastric LGIN do not address endoscopic microwave coagulation therapy or Holmium laser therapy, and their applicability for treating gastric LGIN requires further exploration and validation.
9.3 Super Minimally Invasive Resection of Gastric Subepithelial Tumors
9.3.1 Overview
(1) Introduction to gastric subepithelial tumors
Subepithelial lesion (SELs) are protuberant lesions originating from the muscularis mucosa, submucosa, or muscularis propria of the stomach, although some may arise from extraluminal structures. Typically, SETs smaller than 2 cm in diameter are asymptomatic and are often discovered incidentally during endoscopic examinations. However, certain types or locations of SETs may present with symptoms such as abdominal pain, bleeding, or obstruction as the lesion enlarges or the disease progresses. Gastric SETs include both non-neoplastic lesions, such as ectopic pancreatic tissue, and neoplastic lesions. Among the latter, gastrointestinal stromal tumors, lipomas, Brunner’s gland adenomas, granular cell tumors, schwannomas, and glomus tumors are generally benign, with less than 15% demonstrating histological malignancy.
(2) Diagnosis of gastric subepithelial tumors
1. Conventional white-light endoscopy is the first-line modality for detecting gastric SETs. It provides information about lesion location, size, mobility, mucosal color, morphology, and the presence of erosion or bleeding. However, it cannot determine the nature or origin of the lesion, especially for SETs exhibiting an extraluminal growth pattern. Therefore, conventional endoscopy alone is insufficient for definitive diagnosis.
2. Endoscopic ultrasonography (EUS) is the primary diagnostic tool for gastric SETs, offering detailed insights into the lesion’s origin, intramural or extramural location, size, shape, echogenicity, vascularity, and associated lymphadenopathy. These features help assess histological characteristics and malignancy potential. EUS has a sensitivity of 92% in distinguishing extraluminal compression from intraluminal lesions. The overall sensitivity and specificity of EUS for predicting malignancy potential are 64% and 80%, respectively. Advanced techniques such as contrast-enhanced EUS and EUS elastography further facilitate differentiation between stromal tumors and other lesions, with stromal tumors showing high enhancement and lipomas or leiomyomas demonstrating low enhancement.
3. Computed tomography (CT) and magnetic resonance imaging (MRI) are recommended for staging, treatment planning, and prognostic evaluation, particularly for suspected malignant lesions or tumors larger than 2 cm in diameter.
4. Necessity and efficacy of biopsy for histopathological examination
1) Lesions diagnosed as benign by routine endoscopy and EUS: tissue sampling may be conducted for lesions such as lipomas, cysts, or ectopic pancreatic tissue.
2) Suspicious or indeterminate lesions: for lesions with potential malignancy or when endoscopy and EUS cannot conclusively assess malignancy, techniques such as EUS-guided fine-needle aspiration (EUS–FNA), EUS-guided fine-needle biopsy (EUS–FNB), or mucosal incision-assisted biopsy (MIAB) may be employed for preoperative pathological evaluation.
3) Endoscopic resection without preoperative pathology: considering the limitations of EUS–FNA and its impact on subsequent endoscopic procedures, direct endoscopic resection may be performed for lesions meeting the criteria for endoscopic surgery. In such cases, complete resection can be achieved by experienced endoscopists at centers with advanced endoscopic capabilities, obviating the need for preoperative pathological diagnosis.
(3) Technical approaches for super minimally invasive resection of gastric subepithelial tumors
Super minimally invasive surgery (SMIS) offers a curative approach for gastric subepithelial tumors through innovative techniques utilizing natural orifice transluminal access, tunnel-based pathways, or multi-cavity access. The primary methods include:
Peroral super minimally invasive resection of subepithelial lesions: this technique involves the local and complete resection of gastric subepithelial tumors via a peroral endoscopic approach.
Per-tunnel super minimally invasive resection of subepithelial lesions: this method achieves local and complete resection of gastric subepithelial tumors through a tunnel created endoscopically within the gastric wall.
Per-multiple-cavity super minimally invasive resection of subepithelial lesions: this approach combines transabdominal percutaneous puncture and natural orifice transluminal endoscopic access. It employs a hybrid technique involving both digestive endoscopy and laparoscopy, performed simultaneously or sequentially, to achieve local and complete tumor resection.
The currently implemented SMIS techniques for gastric subepithelial tumors and their historical nomenclature are summarized in table 9.2.
Technical methods and historical nomenclature for super minimally invasive surgery (SMIS) of gastric subepithelial tumors.
| No. | SMIS technique | Technical method | Historical nomenclature |
| 1 | Traction-assisted endoscopic submucosal dissection | Peroral super minimally invasive resection | Endoscopic submucosal dissection (ESD)Traction-assisted endoscopic submucosal dissectionCap-assisted endoscopic mucosal resection (EMR)Endoscopic mucosal resection (EMR)Endoscopic submucosal tumor excavation |
| 2 | Peroral super minimally invasive resection of gastric subepithelial tumors | Super minimally invasive full-thickness resection | Endoscopic full-thickness resection (EFTR)Traction-assisted endoscopic full-thickness resection |
| 3 | Per-tunnel super minimally invasive resection of gastric subepithelial tumors | Super minimally invasive non-full-thickness resection | Endoscopic submucosal tunneling dissectionTraction-assisted endoscopic submucosal dissection |
| 4 | Per-multiple-cavity super minimally invasive resection of gastric subepithelial tumors | Super minimally invasive multi-cavity resection | Combined multiscope therapy |
9.3.2 Super Minimally Invasive Resection of Gastric Subepithelial Tumors
The super minimally invasive resection of gastric subepithelial tumors can be performed using the endoscopic submucosal excavation (ESE) technique. ESE incorporates technical principles from endoscopic submucosal dissection (ESD) and endoscopic mucosal resection (EMR). It typically involves a circular ‘cap-lifting’ incision around the tumor to remove the overlying mucosa, fully exposing the subepithelial tumor. This approach aims to preserve tumor integrity, enhance surgical radicality, and reduce intraoperative complications. The complete resection rate for ESE reaches 92%, achieving a 100% resection rate for tumors with a diameter ≤1.5 cm and 77.8% for tumors >1.5 cm in diameter.
(I) Indications and contraindications of ESE
1. Indications
(a) Tumors with malignant potential suspected in preoperative examinations or confirmed through biopsy pathology, particularly when preoperative assessment indicates a tumor diameter ≤2 cm, with low risk of recurrence or metastasis, and deemed resectable by endoscopy. For tumors >2 cm, endoscopic resection can be considered in units with mature endoscopic techniques and experienced endoscopists, provided no lymph node or distant metastasis is evident and complete resection is feasible.
(b) Symptomatic gastric subepithelial tumors presenting with bleeding or obstruction.
(c) Benign tumors confirmed by preoperative examination or pathology but in patients unable to adhere to regular follow-up, or those with significant tumor growth within a short follow-up period, or strong preference for endoscopic treatment.
(d) Subepithelial lesions (SELs) ≥2 cm or tumors confirmed by preoperative imaging (EUS, CT) to protrude into the gastric lumen, where endoscopic snaring is challenging.
2. Contraindications
(a) Lesions with confirmed lymph node or distant metastasis.
(b) For some SELs with confirmed lymph node or distant metastasis requiring large biopsy specimens for histopathological diagnosis, ESE may be considered a relative contraindication.
(c) Patients with poor general health, as determined through comprehensive preoperative evaluation, who are unable to tolerate endoscopic surgery.
(II) Preoperative preparation
(a) Thorough anesthesia evaluation and completion of imaging studies such as endoscopic ultrasonography (EUS) and abdominal CT are essential preoperative steps.
(b) Preoperative fasting: patients should abstain from food and water for at least 8 h prior to the procedure.
(III) Surgical procedure and techniques
1. Marking, circumferential incision, and tumor dissection (figure 9.14)
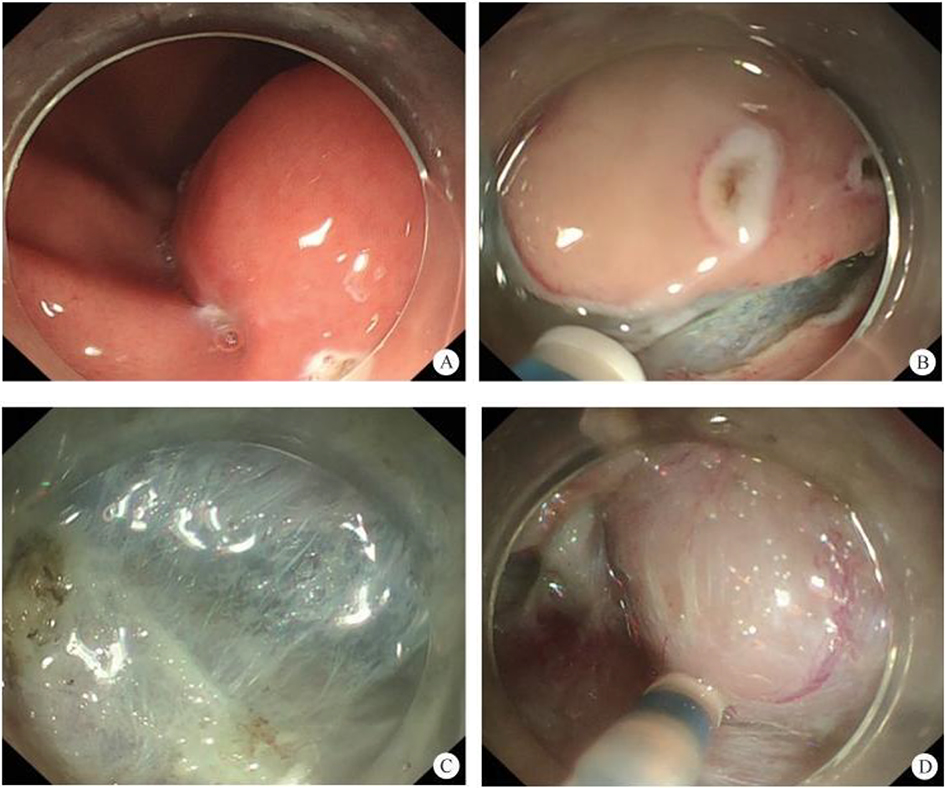

Steps of marking, circumferential incision, and tumor dissection in super minimally invasive resection of gastric subepithelial tumors. A. Identification of the lesion and circumferential marking. B. Circumferential incision around the lesion. C. Submucosal dissection to locate the lesion. D. Dissection along the tumor’s margin.
(a) Marking: the lesion is identified and marked circumferentially.
(b) Submucosal injection: an injection needle is used to introduce a mixed solution into the submucosal layer surrounding the lesion.
(c) Circumferential incision: a DualKnife is used to incise the mucosa along the marked points at the lesion’s edge.
(d) Dissection: the submucosal tissue is gradually separated from the muscular fibers around the tumor capsule using instruments such as the DualKnife, IT2 Knife, or Triangle Knife.
2. Tumor resection and wound management (figure 9.15)
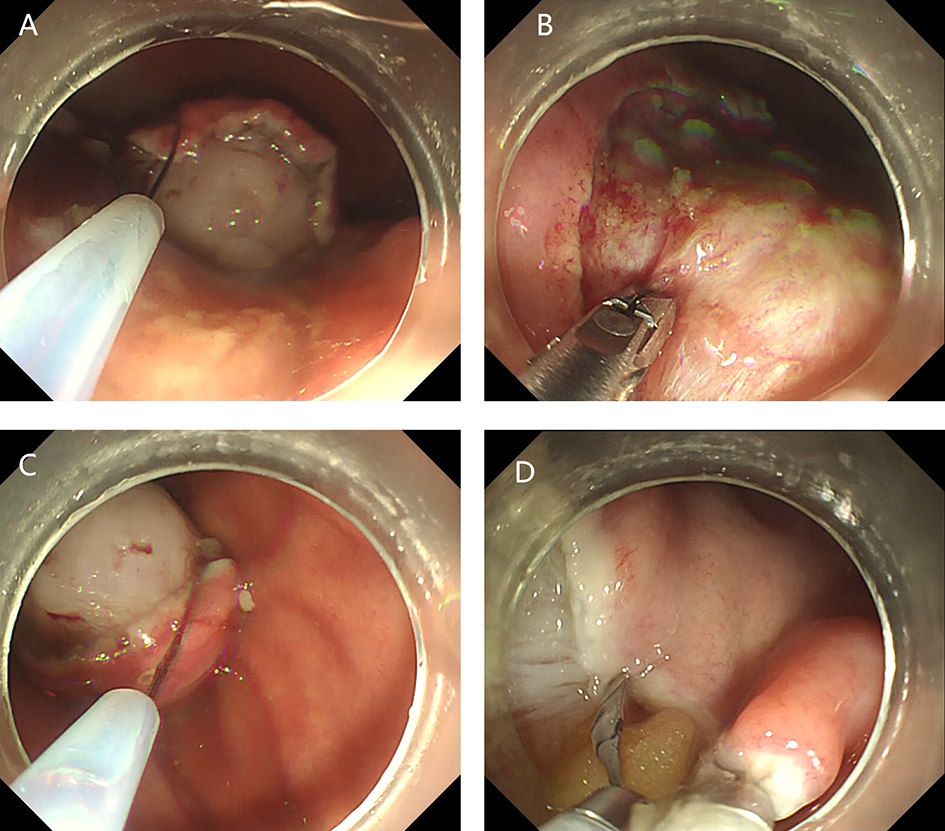

Steps of tumor resection and wound management in super minimally invasive resection of gastric subepithelial tumors. A. Tumor dissection until most of the lesion is separated from the wound surface, followed by resection using a snare. B. Hemostasis of residual blood vessels using hemostatic forceps. C. Removal of large lesions with the assistance of a snare. D. Closure of small perforations using tissue clips.
(a) Tumor resection: the tumor is resected using an electrosurgical knife or a snare.
(b) Hemostasis: after resection, carefully manage the wound surface with electrocoagulation to prevent delayed bleeding.
(c) Ulcer closure: in many cases, the mucosa covering the SEL is also removed, making it difficult to close the artificial ulcer with clips. If feasible, close the ulcer to reduce the risks of perforation, infection, and delayed bleeding.
Refer to video 9.8, Super minimally invasive resection of a stromal tumor of the gastric fundus (See online resources).
(IV) Postoperative management
(1) Strict bed rest
Patients should remain strictly on bed rest for 24 h postoperatively to avoid vigorous physical activities. Monitor closely for signs of complications, such as fever, palpitations, cold sweats, abdominal pain, hematemesis, or melena, which may indicate infection or bleeding.
(2) Dietary modifications
Postoperatively, fasting is required for 48–72 h. Subsequently, patients may switch to a cool liquid diet (e.g., rice soup, noodle soup, milk), followed by a gradual progression to a semi-liquid diet (e.g., soft noodles, porridge). They must avoid coarse, spicy, or irritating foods, and refrain from heavy physical exertion for two weeks. For patients with significant bleeding or extensive wound surfaces, the fasting period should be extended. For those undergoing endoscopic treatment for gastric subepithelial lesions (SELs), routine administration of proton pump inhibitors (PPIs) is recommended. After resuming oral intake, patients should continue taking PPIs and mucosal protective agents for six to eight weeks postoperatively to promote wound healing.
(3) Use of antibiotics
The decision to use antibiotics should be based on a comprehensive assessment of factors such as wound condition, procedure duration, surgical techniques employed, the presence of complications, and other infection-related risks.
(V) A classic case
Case history
The patient, a young man, was admitted with the chief complaint of “intermittent melena for four months and a submucosal protrusion at the cardia detected over three months.” On February 20, 2023, gastroscopy revealed, 42 cm from the incisors, a protruding lesion on the posterior wall of the cardia with a central depression covered with white exudate. The lesion occupied approximately 2/5 of the circumferential lumen. Retroflexion of the gastroscope did not reveal additional lesions. Endoscopic ultrasonography (EUS) indicated an anechoic mass at the lesion site, oval in shape, protruding both inward and outward from the lumen. The boundaries were distinct, with the largest cross-sectional diameter measuring 2.8 cm × 2.5 cm. The internal echoes were heterogeneous, displaying medium-grade irregular echoes with septations, originating from the submucosa or the muscularis propria. Abdominal enhanced CT reconstruction and MRI suggested a submucosal lesion at the gastric fundus below the cardia, with a probable diagnosis of gastrointestinal stromal tumor (GIST). The patient had no significant medical history. Physical examination upon admission showed no abnormalities.
Diagnosis: subepithelial tumor of the cardia.
After thorough preoperative evaluation, nutritional status optimization, and obtaining informed consent for surgery, the patient underwent a super minimally invasive peroral resection procedure. Refer to video 9.9, Super minimally invasive peroral resection of subepithelial tumor of the cardia) (See online resources).
Surgical procedure: circumferential scanning with EUS at 43 cm from the incisors revealed a round, hypoechoic lesion with mixed echoes, originating from the muscularis propria and enclosed within an intact capsule. Color Doppler Flow Imaging (CDFI) indicated scattered blood flow signals at the lesion margins and within the lesion. The cross-sectional dimensions were approximately 27.4 mm × 33.1 mm. Gastroscopy showed a spherical submucosal protrusion at the cardia and gastric fundus, with edematous mucosa on its surface. A depressed ulcer with thin exudate was observed at the lesion apex. A mixture of methylene blue, epinephrine, and normal saline solution was injected at the lesion’s oral side, resulting in good lifting. The mucosal layer at the lesion’s oral side was incised using a DualKnife, and the submucosa and muscularis propria were progressively separated, exposing the white tumor using an IT Knife. Alternating use of IT Knife, triangle knife, and snare was employed to dissect the tumor. Bleeding vessels were treated with hemostatic forceps. Following complete tumor resection, hemostatic forceps and argon plasma coagulation (APC) were used to manage the surgical bed, and tissue clips secured weak points in the muscularis propria. Three biological glue applications were used to cover the surgical area. A gastrointestinal decompression tube was inserted under endoscopic guidance. The procedure concluded with exhalation and withdrawal of the endoscope.
(VI) Hot-spot issues and research advances in the consensus
1. Feasibility of non-submucosal injection ESE. The standard procedure for endoscopic submucosal excavation (ESE) involves submucosal injection to separate the mucosa from the muscularis propria, facilitating tumor removal. However, a clinical issue arises when submucosal injection displaces smaller subepithelial tumors (SETs) (<2 cm) due to mucosal elevation, potentially prolonging the time required to locate the tumor and increasing the risks of bleeding and perforation, especially for tumors <1 cm. Studies have demonstrated that non-submucosal injection ESE is a safe and effective approach for treating gastric stromal tumors, particularly for tumors <2 cm. This method is associated with shorter tumor exposure time, reduced tumor excavation time, lower surgical costs, and shorter hospital stays compared to the submucosal injection group. For small, luminally protruding SETs, the consensus recommends endoscopic snare resection. Non-submucosal injection ESE offers an alternative treatment strategy, enabling complete tumor removal. However, further comparative studies are needed to confirm the efficacy of this approach.
2. ESE combined with snare resection to simplify surgery. A Chinese study reported that endoscopic ligation device-assisted ESE for SETs simplifies the complexity of endoscopic operations and shortens surgical time. However, the transparent cap diameter of the ligation device limits its use to lesions with diameters ≤2 cm. Recent case reports have described a mucosal snare resection-endoscopic submucosal excavation (MSR–ESE) technique that avoids electrocautery marking and submucosal injection, thereby preventing iatrogenic perforation. MSR–ESE is expected to emerge as a safe, time-saving, and effective option for treating submucosal tumors. Guidelines suggest that for SELs ≥2 cm in diameter or those with luminal protrusion confirmed by preoperative EUS or CT, ESE may be performed when endoscopic snare resection is challenging. However, the indications for MSR–ESE still require further exploration due to the diameter limitations of the snare device.
3. Indications for ESE. Consensus on the choice of endoscopic resection methods for gastric SETs has been established, with recommendations based on factors such as tumor size and luminal or extraluminal protrusion. Techniques such as endoscopic snare resection, ESE, submucosal tunneling endoscopic resection (STER), and endoscopic full-thickness resection (EFTR) are gradually being recommended. Given the variability in SET location, origin, and size, an international team developed a scoring system to predict the difficulty of minimally invasive surgery for gastric SETs. The scoring system considers three clinical factors: tumor location, size, and growth pattern. Patients scoring above 3 points experienced greater intraoperative blood loss and longer hospital stays, providing a basis for optimal treatment selection.
The latest expert consensus suggests that for SETs with a shallow surface, preoperative EUS and CT confirmation of luminal protrusion, and the feasibility of complete one-time snare resection, endoscopic snare resection is appropriate. For SETs ≥ 2 cm in diameter or those with luminal protrusion confirmed by preoperative imaging, ESE is recommended when snare resection is difficult. However, no preferred treatment method has been identified for smaller SETs with inconspicuous luminal protrusion. Current studies support the safety and efficacy of ESE for smaller SETs (<2 cm), though comparative studies with snare resection are lacking. The application of ESE continues to expand, often in combination with snare resection to reduce adverse events and shorten surgical time. Multicenter prospective studies are needed to guide optimal treatment strategies.
9.3.3 Peroral Super Minimally Invasive Full-Thickness Resection of Gastric Subepithelial Tumors
Peroral super minimally invasive full-thickness resection of gastric subepithelial tumors can be achieved through endoscopic full-thickness resection (EFTR) combined with endoscopic suturing. EFTR is one of the endoscopic treatment methods for gastric subepithelial tumors, often requiring endoscopic suturing to close the defect. The success rate of EFTR can reach 100%, with a very low complication rate. Proper closure of the perforation site post-EFTR is critical to its success. The most commonly used technique is closure with metallic clips. For larger defects that cannot be closed directly, negative pressure suction may be applied to draw the omentum into the gastrointestinal lumen, and metallic clips are used to anchor the omentum and mucosa at the defect margins, a technique known as the ‘omentum patch suturing technique.’ Other methods include ‘purse-string suturing’ using nylon threads combined with metallic clips. In recent years, new techniques such as over-the-scope clip (OTSC) systems and OverStitch suturing have also been reported.
(I) Indications and contraindications
1. Indications: ① SETs in locations where tunneling is difficult to establish or with maximum transverse diameters >3.5 cm that are unsuitable for STER; ② SETs with subserosal or partial extraluminal growth, with diameters <5 cm; ③ intraoperative findings of tumor adherence to the serosal layer that precludes separation.
2. Contraindications: ① General contraindications for general anesthesia, such as allergic reactions to anesthetic agents or pregnancy; ② contraindications for gastroscopic examination and treatment, such as severe cardiopulmonary disease, hematological disorders, coagulation abnormalities, or the inability to discontinue anticoagulant/antiplatelet therapy; ③ preoperative imaging or histological characteristics indicating malignant potential in gastric SETs; ④ high risk of lymph node metastasis or confirmed lymph node or distant metastases.
(II) Surgical procedure and techniques
1. Lesion identification and circumferential incision (figure 9.16). A transparent cap must be attached to the tip of the endoscope to ensure better visualization, tissue traction, and positioning, as well as to facilitate hemostasis in case of bleeding. The gastroscope is advanced to the lesion site, and argon plasma coagulation (APC) is used to mark the lesion circumferentially. A mixture of methylene blue and saline solution is injected into the submucosal layer, and the mucosa is circumferentially incised.
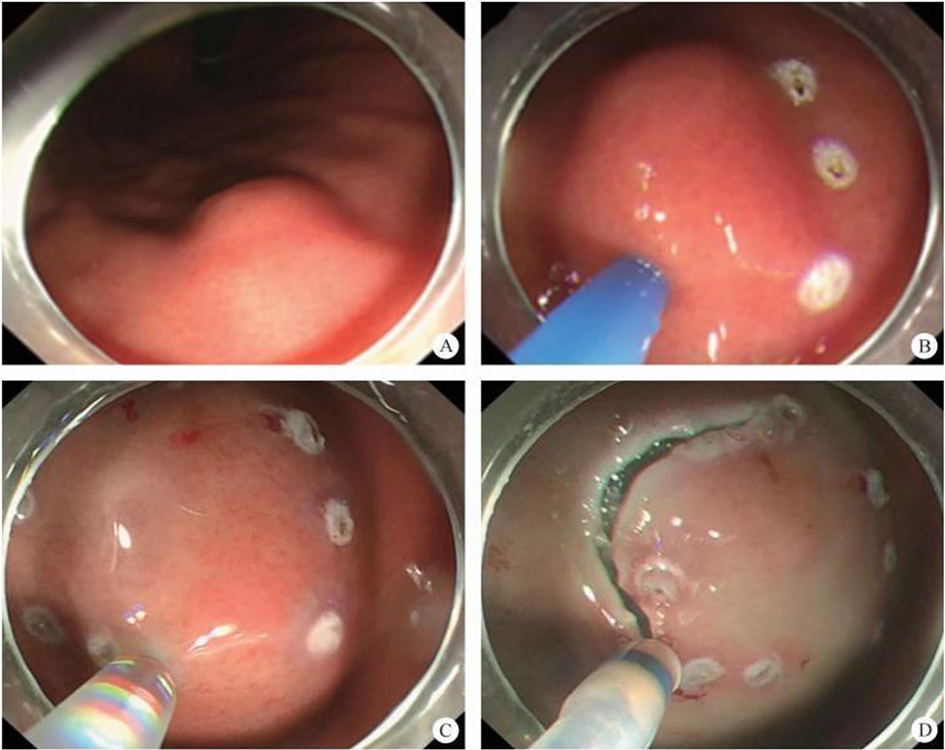

Steps of circumferential mucosal incision in peroral super minimally invasive full-thickness resection of gastric subepithelial tumors. A. Submucosal lesion in the gastric body. B. Circumferential marking with argon plasma coagulation (APC). C. Submucosal injection. D. Circumferential mucosal incision edges.
2. Layer-by-layer dissection (figure 9.17). The dissection is performed progressively along the submucosal layer from the top downward until a semicircular incision is achieved. Subsequently, the dissection continues around the lesion, separating the muscularis propria. Traction is applied to pull the lesion into the lumen.
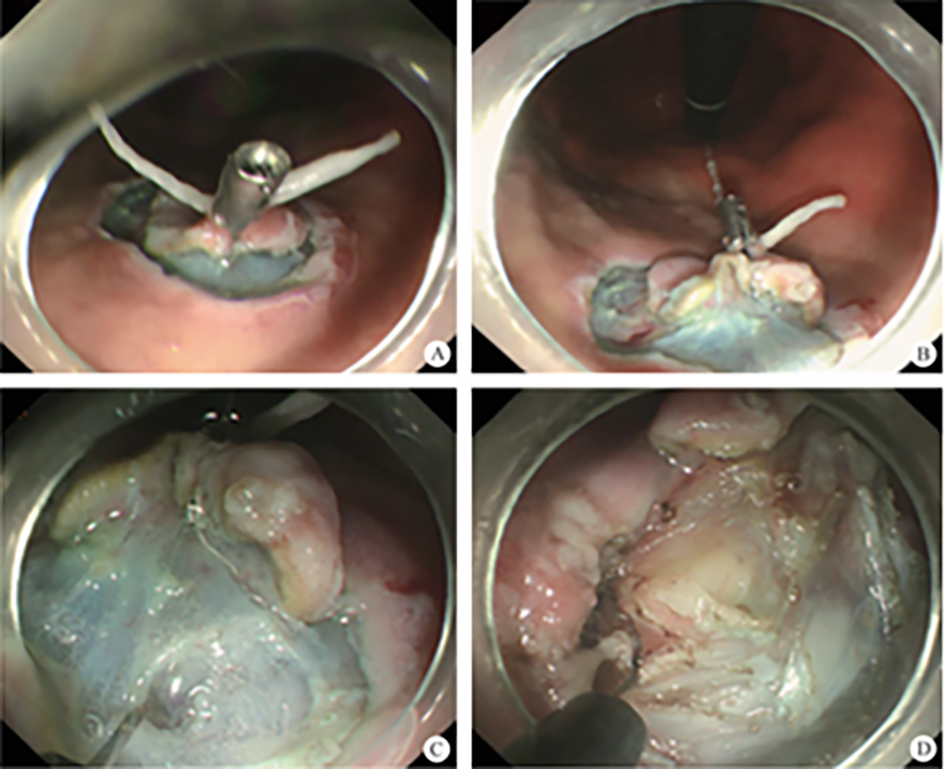

Dissection steps of peroral super minimally invasive full-thickness resection of gastric subepithelial tumors. A. Full-thickness resection performed under dental floss traction. B. Partial closure of the incised muscularis layer using tissue clips. C. Continued resection following partial closure of the incision. D. Closure of the incision with multiple tissue clips.
3. Full-thickness resection. After the full-thickness incision has been made over a certain length, a transparent cap is used to assist in pushing the tumor into the lumen, fully exposing the dorsal side within the field of view. Under direct visualization, the tumor is gradually excised along the outer margin from the serosal side. This ensures the integrity of the tumor capsule and allows precise handling of serosal blood vessels to prevent the tumor from falling into the abdominal cavity upon complete release (figure 9.18).
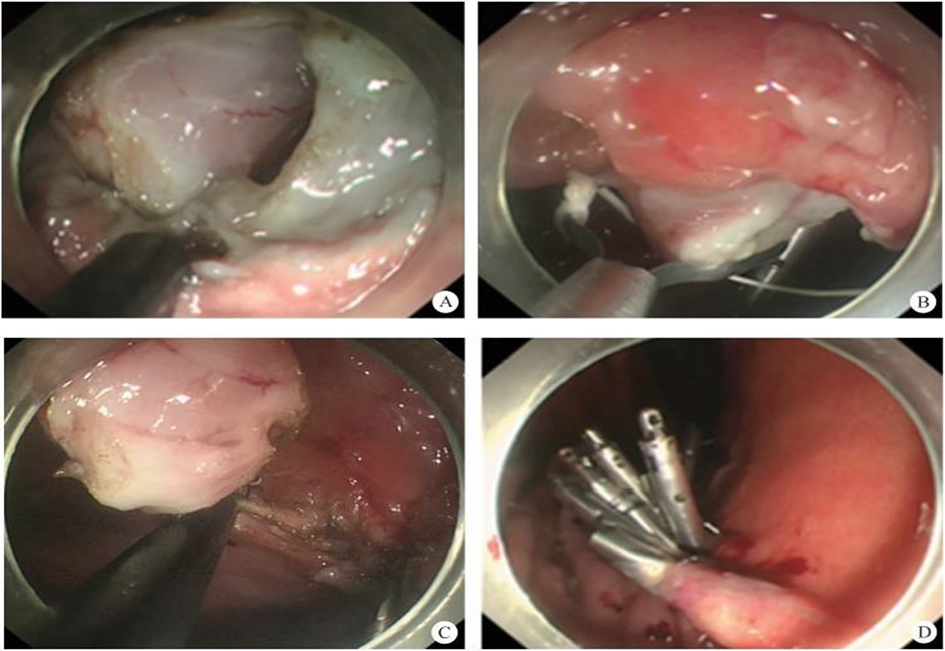

Tumor resection and wound management steps of peroral super minimally invasive full-thickness resection of gastric subepithelial tumors. A. Full-thickness resection performed under dental floss traction. B. Partial closure of the incised muscularis layer using tissue clips. C. Continued resection following partial closure of the incision. D. Closure of the incision with multiple tissue clips.
4. Endoscopic suturing (figure 9.18)
(1) Clip closure: since the mucosa covering the SEL is also excised, clips often cannot be used to close the artificial ulcer. Closure of the ulcer is recommended, if possible, to reduce the risk of perforation, infection, and delayed bleeding. For lesions located deeply in the muscularis propria (MP) layer or with thin ulcer walls, clips are used to prevent perforation. In some cases, fibrin sealants are applied to close the defect.
(2) Nylon rope and clip method: a nylon loop is introduced through one working channel of the endoscope and positioned to fit the defect. Titanium clips are then inserted through another working channel. The first clip anchors the nylon loop to the defect edge, followed by additional clips evenly distributed along the entire edge of the defect. The clips are symmetrically placed with equal spacing on both sides. Once all clips are secured, the nylon loop is tightened, fully closing the defect. Under endoscopic visualization, the tightened clips appear as a cluster over the closed defect.
(3) Stepwise closure: a method involving simultaneous closure and resection can also be applied. This includes muscularis-to-muscularis closure, supplemented with interrupted mucosal-to-mucosal suturing in areas where gaps remain.
(III) Classic cases
Case 1: Peroral super minimally invasive resection of a tumor in the gastric body’s muscularis propria
Case history: the patient, a middle-aged woman, was admitted due to a “gastric body protruding lesion detected for 10 months.” Gastroscopy revealed a submucosal protrusion approximately 1.0 cm in diameter on the lesser curvature of the gastric body, with a smooth surface. Endoscopic ultrasonography (EUS) indicated the lesion originated from the muscularis propria, presenting as homogenous hypoechoic, with clear boundaries. The measured dimensions were 7.6 mm × 4.0 mm, with intraluminal protrusion. The patient had no significant medical history.
Diagnosis: suspected stromal tumor of the gastric body.
After thorough preoperative evaluation, nutritional optimization, and informed consent, the patient underwent peroral Super minimally invasive resection. Refer to video 9.10, Peroral super minimally invasive full-thickness resection of a subepithelial tumor in the gastric body (See online resources).
Surgical process: a protruding lesion approximately 1 cm in diameter with normal mucosal coverage was identified on the anterior wall near the lesser curvature of the gastric body midsection. Circumferential marking with APC was followed by a semicircular incision using a DualKnife. Dental floss traction was employed to assist in tumor handling. The lesion was resected in full thickness using an IT Knife and a triangle knife, with meticulous hemostasis achieved via hemostatic clips. A fibrin sealant was applied to cover the wound.
Case 2: Peroral super minimally invasive resection of a tumor in the lesser sac of the gastric fundus
Case history: the patient, an elderly woman, was admitted due to a “submucosal protruding lesion in the gastric fundus detected for over four years.” Gastroscopy revealed a subepithelial tumor in the gastric body of uncertain nature, with a high probability of being a stromal tumor. Endoscopic ultrasonography (EUS) indicated that the lesion originated from the muscularis propria near the lesser curvature of the gastric fundus, close to the cardia, measuring 1.5 cm × 1.5 cm. The patient had a history of hypertension, hypothyroidism, and hyperlipidemia but showed no abnormalities upon physical examination for admission.
Diagnosis considerations: suspected gastrointestinal bleeding and gastric stromal tumor.
After comprehensive preoperative evaluation, nutritional optimization, and obtaining informed consent, the patient underwent peroral super minimally invasive resection.
Refer to video 9.11, Peroral super minimally invasive full-thickness resection of a tumor in the lesser sac of the gastric fundus (See online resources).
Surgical process: Intraoperative findings revealed a submucosal protrusion measuring approximately 1.5 cm × 1.5 cm near the cardia on the lesser curvature of the gastric fundus. The lesion had a smooth surface with minimal secretions. Circumferential marking was performed using argon plasma coagulation (APC) at the lesion margin. A needle was used for submucosal injection, achieving satisfactory lifting of the lesion. Dental floss was employed to assist in traction, and the lesion was dissected along its margin using an IT Knife. Partial defects in the muscularis layer were closed completely with metallic clips, and meticulous hemostasis was achieved using hemostatic forceps. The wound was sealed with a biological protein glue spray, and the lesion was successfully excised.
(IV) Hot-spot issues and research advances in the consensus
1. The key to EFTR success lies in proper closure of perforation sites. A critical step in EFTR (endoscopic full-thickness resection) remains the full-thickness suturing technique. In addition to traditional methods such as metallic clip suturing and nylon-titanium ‘purse-string suturing,’ new closure systems have emerged, including over-the-scope clip (OTSC) systems, the Apollo OverStitch Sx system, Over-the-Scope Suturing (OSS) systems, TTSS systems, and the ZeoSuture M endoscopic suturing device. Despite advancements, EFTR has not yet become a routine part of endoscopic practice. According to an international survey, 32% of respondents reported technical difficulties with endoscopic suturing. Further clinical research is needed to establish the safety, efficacy, and feasibility of EFTR and to incorporate emerging suturing devices into everyday endoscopic practice. Additionally, new challenges need to be addressed, such as developing advanced devices to improve areas where EFTR falls short compared to surgical techniques, including robust hemostatic instruments and staplers.
2. EFTR requirements and limitations due to the size of the lesion. Advances in resection techniques and suturing devices have facilitated EFTR, expanding its indications for treating subepithelial lesions (SELs) in the gastrointestinal tract. Consensus guidelines recommend EFTR for tumors with a maximum transverse diameter >3.5 cm unsuitable for STER (submucosal tunneling endoscopic resection). No clear upper limit on lesion size has been defined, but current devices impose practical limitations. For SELs >5 cm, individualized treatment decisions are necessary, and clinical reports on such cases are limited. Additional clinical data are required to further explore and expand EFTR indications.
3. Choosing the EFTR approach. EFTR can be categorized into exposed and non-exposed techniques based on whether the thoracic or abdominal cavity is exposed during the procedure.
Exposed EFTR: this involves resection followed by suturing and can be further divided into tunnel and non-tunnel techniques.
Non-tunnel exposed EFTR refers to conventional EFTR, while tunnel-exposed EFTR resembles STER but also involves dissection of the muscularis propria around the lesion.
The existing literature does not extensively distinguish between tunnel-exposed EFTR and STER. Non-exposed EFTR: OTSC-based non-exposed EFTR and other devices, such as the full-thickness resection device (FTRD), are gaining clinical attention. The FTRD integrates resection and suturing in one device by using a transparent cap with a snare and OTSC. The lesion is pulled into the cap for resection and simultaneous closure. This approach prevents contamination or dissemination of gastrointestinal contents into the abdominal cavity, significantly reducing postoperative complications. However, limitations include a restricted resection range, reduced flexibility, and challenges in gastric applications due to the thick and less mobile gastric wall. Most studies on FTRD focus on the lower gastrointestinal tract, and the technology remains in the exploratory phase due to immature device development.
4. Future integration of EFTR with lymph node dissection. With EFTR technology maturing, complete full-thickness resection is no longer a technical challenge. Current EFTR applications primarily target benign or low-grade malignant lesions originating from the muscularis propria in the gastrointestinal tract. However, its potential extends further. Future research may focus on the combination of EFTR with endoscopic lymph node dissection in malignant gastrointestinal tumors. As tumor infiltration depth increases, the risk of lymph node metastasis also rises. Combining EFTR with effective sentinel lymph node detection technology could enable simultaneous full-thickness tumor resection and endoscopic lymphadenectomy, marking a new direction for EFTR technology.
9.3.4 Peroral Super Minimally Invasive Non-Full-Thickness Resection of Gastric Subepithelial Tumors
Peroral Super minimally invasive non-full-thickness resection of gastric subepithelial tumors can be performed using the endoscopic submucosal resection with ligation (ESMR-L) technique. ESMR-L involves resecting the lesion using a snare in conjunction with a cap-assisted or ligation device. The lesion is suctioned into the cap and resected using a preloaded snare. This technique carries a bleeding risk of 4%–13% and a perforation risk of 5%.
(I) Indications and contraindications for ESMR-L
1. Indications: for superficial SELs (subepithelial lesions) with preoperative EUS and CT confirming intraluminal protrusion, ESMR-L can be used when the lesion can be completely resected in a single session with a snare.
2. Contraindications: ① severe cardiopulmonary disease, shock, coma, acute perforation of the upper gastrointestinal tract, altered consciousness, severe or acute pharyngeal disease, severe acute inflammation of the esophagus or stomach, aortic aneurysm, or severe cervical/thoracic spinal deformities; ② coagulation disorders or bleeding tendencies; ③ lesions with obvious surface ulcers or scars; ④ submucosal tumors originating from the muscularis propria; ⑤ lesions that are too large (>2 cm).
(II) Surgical procedure and techniques (figures 9.19 and 9.20)
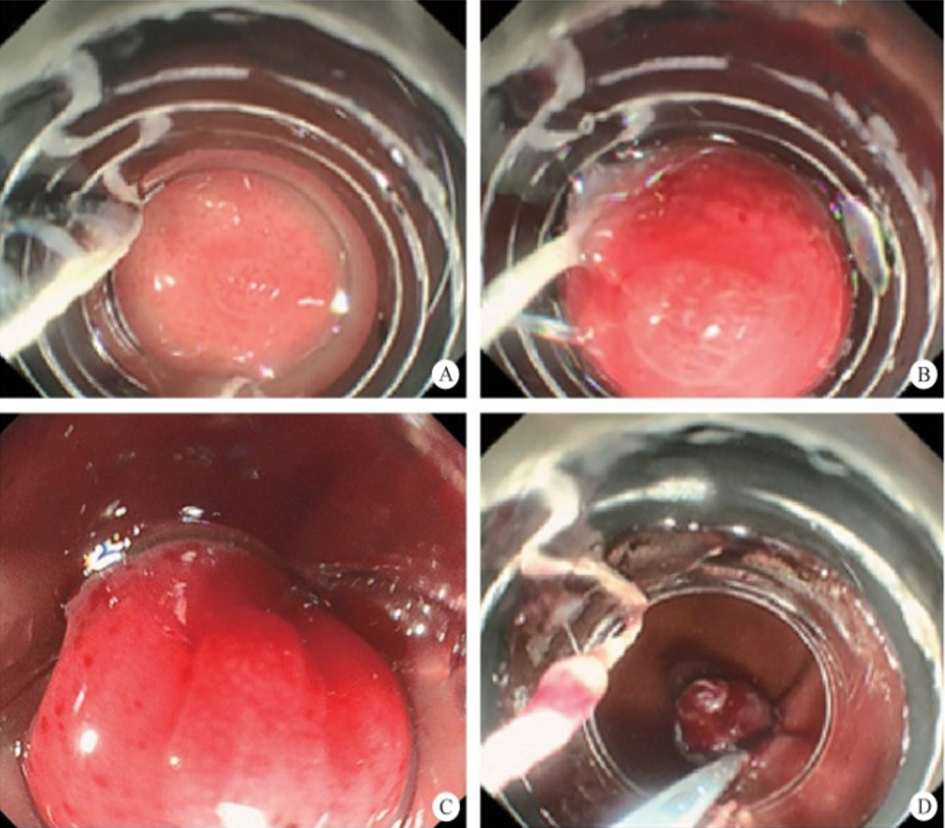
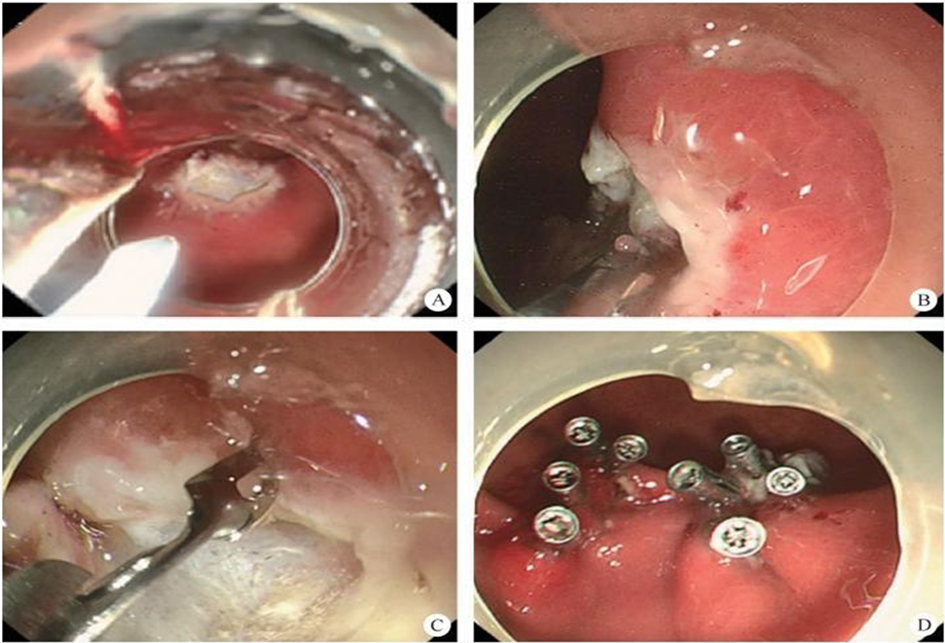

Super minimally invasive mucosal resection with ligation of gastric subepithelial tumors. A. Multiple gastric neuroendocrine tumors, planned for ligation combined with snare resection. B. Ligation device positioned close to the lesion, centering it within the field. C. Suction applied to draw the lesion along with surrounding normal mucosa into the transparent cap. D. Snare positioned below the rubber band to perform electrocautery resection.

Tumor resection and wound closure steps of super minimally invasive mucosal resection with ligation of gastric subepithelial tumors. A. Complete resection of the lesion. B. Prophylactic hemostasis of exposed vessels at the wound site. C. Wound closure using tissue clips. D. Sequential resection of multiple lesions with wound closure using tissue clips.
1. Lesion identification and circumferential marking. A transparent cap must be attached to the endoscope tip to ensure better visualization, tissue traction, and precise positioning, as well as to facilitate hemostasis in case of bleeding. The endoscope is advanced to the lesion site, and argon plasma coagulation (APC) is used to circumferentially mark the lesion.
2. Ligation and snaring. A ligation device is mounted on the front end of the endoscope. The endoscope is inserted, and negative pressure suction is applied to draw the lesion into the transparent cap of the ligation device. A rubber band is then released, ligating the base of the suctioned lesion tightly and creating a polyp-like structure. The endoscope is withdrawn, and the ligation device is removed. The endoscope is reinserted with a snare, which is positioned 1–2 mm below the ligated rubber band and used to resect the lesion.
3. Wound management. After the lesion is resected, meticulous hemostasis is performed using electrocautery to prevent delayed bleeding. Tissue clips can be applied to close the wound site as needed.
(III) Postoperative management
1. Patients should strictly rest in bed for 24 h postoperatively, avoiding significant physical activity. Monitor for symptoms such as fever, palpitations, cold sweats, abdominal pain, hematemesis, or melena, which could indicate infection or bleeding complications.
2. Patients should fast for 48–72 h, followed by a transition to cool liquid foods (e.g., rice soup, noodle soup, milk), then gradually shift to semi-liquid foods (e.g., soft noodles, porridge). Avoid rough or spicy foods, and refrain from heavy physical activity for two weeks. Patients with large wound areas or bleeding may require prolonged fasting. Routine administration of proton pump inhibitors (PPIs) is recommended for gastric SEL patients after endoscopic treatment, with continued oral PPIs and mucosal protectants for six to eight weeks postoperatively to promote wound healing.
3. The decision to use antibiotics should be based on a comprehensive assessment of the condition of the wound, the duration of the procedure, the type of operation, the presence of complications, or of other related infection risks.
(IV) A classic case
Case history: the patient, a young man, presented with a six-year history of acid reflux and belching. Gastroscopy revealed more than 10 polyps in the gastric fundus and body, each measuring between 0.2 and 0.4 cm in diameter, with smooth surfaces, which were removed using biopsy forceps. Pathological examinations indicated chronic inflammation with acute inflammation in the mucosa of the gastric body and fundus, partial glandular hyperplasia, focal lymphoid hyperplasia in the lamina propria, and multiple nests of small round cells within the lamina propria. Immunohistochemical diagnosis confirmed neuroendocrine tumors with CK20 (+ in individual cells). The patient had no significant medical history, and physical examination upon admission was unremarkable.
Diagnosis: multiple gastric neuroendocrine tumors.
After a thorough preoperative evaluation, nutritional optimization, and obtaining informed consent, the patient underwent peroral super minimally invasive non-full-thickness resection.
Refer to video 9.12, Peroral super minimally invasive non-full-thickness resection of multiple gastric neuroendocrine tumors (See online resources).
Surgical process: scattered, multiple flat, elevated lesions were observed in the gastric body. Several lesions were resected using a multi-band mucosal resection device. Hemostatic forceps were used to manage the wound sites, and tissue clips were applied to close each resection site. The remaining lesions were ligated using the multi-band ligation device, resulting in dark purple discoloration of the mucosa at the lesion sites.
(V) Hot-spot issues and research advances
1. Timing of ESMR-L application. Guidelines from the European Society of Gastrointestinal Endoscopy (ESGE) and the American Society for Gastrointestinal Endoscopy (ASGE) recommend endoscopic submucosal dissection (ESD) as the preferred treatment for most superficial gastric neoplastic lesions. However, for deeper lesions originating from the muscularis propria, ESD has limited efficacy, increasing procedural complexity and the risk of complications.
Endoscopic submucosal resection with ligation (EMR-L) involves suctioning the lesion into a ligation device, releasing a band to ligate it, and resecting the lesion below the band using a snare. Studies have shown that EMR-L achieves complete resection in patients with esophageal submucosal tumor without perforation. However, due to the limited band diameter (1 cm), it is challenging to completely resect submucosal tumors larger than 1 cm. Transparent caps and ligation devices are inadequate for tumors >2 cm or those originating from the muscularis propria. Before resection, it is crucial to determine whether the tumor is mobile and not fixed. Although EMR-L presents a low risk of bleeding, it has a higher perforation rate and does not guarantee a cure. Consequently, current guidelines rarely recommend this method.
2. Improvements in ESMR-L for precise lesion resection. A common issue with ESMR-L is the failure to ligate the lesion at the first attempt, resulting in ligation of only the mucosa or submucosa. To address this, research teams have developed a method combining pre-incision of the mucosa with ESMR-L. This approach involves visualizing and suctioning the lesion for more accurate resection. While preliminary results from small-sample studies are promising, ongoing randomized controlled trials are necessary to further investigate the feasibility and safety of this technique.
9.3.5 Trans-Multi-Cavitary Super Minimally Invasive Resection of Gastric Subepithelial Tumors
The trans-multi-cavitary super minimally invasive resection technique, previously referred to as laparoscopic and endoscopic cooperative surgery (LECS), combines endoscopy and laparoscopy to precisely locate, resect, and safely close lesions. LECS synergizes the advantages of laparoscopy and endoscopy while mitigating their respective limitations. Variants include laparoscopy-assisted endoscopic resection (LAER), endoscopy-assisted laparoscopic resection (EALR), and combined laparoscopic-endoscopic resection (CLER).
(I) Indications and contraindications
1. Indications
1) Endoscopy-assisted laparoscopic resection (EALR): ① tumors originating from the muscularis propria with extraluminal growth; ② Gastric subepithelial tumors in special locations such as the gastric fundus, cardia, and pyloric canal; ③ lesions with unclear invasion depth under endoscopic visualization; ④ large SELs unsuitable for endoscopic resection.
2) Laparoscopy-assisted endoscopic resection (LAER): ① tumors < 5 cm in diameter, suitable for endoscopic resection, with intraluminal growth and invasion limited to the submucosal layer; ② gastric subepithelial tumors in special locations such as the gastric fundus, cardia, and pyloric canal.
2. Contraindications
① Lesions with confirmed lymph node or distant metastases.
② SELs with suspected lymph node or distant metastases requiring large biopsies for pathology evaluation (relative contraindication).
③ Patients with poor general condition deemed unable to tolerate endoscopic surgery based on preoperative evaluation.
(II) Hot-spot issues and research advances
1. Indications for LECS in the treatment of SELs. While indications for endoscopic SEL resection are well-established, those for LECS remain undefined. LECS has theoretical advantages, such as enhanced precision and safety, but its clinical adoption faces challenges, including the need for experienced practitioners, technical complexity in deep resection and hemostasis, and insufficient long-term clinical benefit data. Extensive clinical research is required to define LECS indications.
Gastrointestinal stromal tumors (GISTs), a subtype of SELs, have malignant potential, necessitating complete resection without damaging the pseudocapsule to reduce recurrence risks. Asian guidelines do not recommend endoscopic resection of small GISTs due to potential pseudocapsule damage. The NCCN guidelines suggest laparoscopic surgery for GISTs <5 cm in favorable locations. LECS, combining laparoscopic and endoscopic strengths, may provide a more effective treatment option for SELs, particularly GISTs, in the future.
2. Selection of LECS approach. LECS (laparoscopic and endoscopic cooperative surgery) is primarily divided into exposed and non-exposed procedures, where ‘exposure’ refers to whether there is communication between the gastrointestinal lumen and the peritoneal cavity. Exposed LECS includes classic LECS, inverted LECS, and laparoscopy-assisted endoscopic full-thickness resection (LAEFR). Inverted LECS builds upon classic LECS by pushing the tumor into the gastrointestinal lumen to minimize the risk of tumor cell dissemination. LAEFR involves manual suturing of the gastric wall defect under laparoscopic guidance during the final closure process, allowing for the precise resection of lesions. It is considered a more reasonable alternative to other laparoscopic procedures for SELs (subepithelial lesions). For patients with lesions extending into the submucosa and a risk of lymph node metastasis, particularly those unsuitable for gastrectomy, LAEFR combined with lymphadenectomy can be performed for staging or treatment. Non-exposed LECS includes the clean non-exposure technique (CLEAN–NET) and non-exposed endoscopic wall-inversion surgery (NEWS), both effectively addressing intraoperative tumor dissemination issues. CLEAN–NET involves identifying lesion boundaries endoscopically, followed by full-thickness gastric wall resection under laparoscopy. Compared to CLEAN–NET, NEWS is limited by the size of the lesion, necessitating accurate tumor size assessment via ultrasound or CT before surgery. Currently, non-exposed LECS is considered the optimal choice for small GISTs (gastrointestinal stromal tumors) with gastric mucosal ulceration. This approach effectively prevents the risk of tumor dissemination caused by gastric wall perforation following full-thickness resection. For SEL patients with a high risk of lymph node metastasis, CLEAN–NET combined with sentinel lymph node navigation surgery enables intraoperative diagnosis of lymph node metastasis. By mapping lymphatic flow, this approach minimizes the extent of lymph node and gastric resection, though its efficacy requires further validation through multicenter studies with large sample sizes.
3. Application of other specialized endoscopic and laparoscopic techniques for the treatment of SELs. In addition to conventional endoscopy, advanced tools such as endoscopic ultrasonography (EUS) and capsule endoscopy can also be utilized in laparoscopic-endoscopic cooperative treatment. EUS provides accurate assessment of SEL origin and localization, offering significant prospects for combined treatment approaches in SEL management. Capsule endoscopy, when combined with laparoscopy, can be applied to the treatment of GISTs, though it is more commonly used for lesions in the lower gastrointestinal tract. Capsule retention is a potential complication of this approach. By integrating laparoscopy, it is not only possible to recover the capsule, but also to establish a definitive diagnosis and to carry out a complete resection of small colorectal diseases.
9.4 Super Minimally Invasive Surgery for Gastroparesis
9.4.1 Overview
Gastroparesis (GP) is a syndrome characterized by delayed gastric emptying and dysfunction of the pyloric sphincter, resulting from altered gastric motility. Clinically, it manifests as symptoms such as bloating, nausea, vomiting, early satiety, and loss of appetite. Although the global epidemiology of GP remains unclear, a large-scale study from a national claims database in the United States reported a standardized prevalence rate of 267.7 per 100 000 individuals. Common etiologies of GP include diabetes mellitus, followed by upper abdominal surgeries, neurological disorders (e.g., Parkinson’s disease), medication-induced cases, and idiopathic causes.
(A) Diagnosis of gastroparesis
The diagnosis of gastroparesis requires meeting three criteria: delayed gastric emptying, exclusion of mechanical obstruction, and a disease duration of more than three months. Clinical symptoms alone are insufficient to determine delayed gastric emptying, necessitating objective testing methods. Currently effective assessments include breath testing, gastric emptying scintigraphy, and wireless motility capsule evaluation. Gastric emptying scintigraphy is considered the ‘gold standard’ for diagnosing delayed gastric emptying in clinical practice.
(B) Technical methods and former nomenclature in super minimally invasive surgery for gastroparesis
Super minimally invasive surgery for gastroparesis encompasses three primary techniques: peroral endoscopic myotomy (POEM) of the pylorus; endoscopic botulinum toxin injection into the pylorus; endoscopic pyloric stent placement.
9.4.2 Super Minimally Invasive Tunnel Myotomy for Gastroparesis
Super minimally invasive tunnel myotomy for gastroparesis, formerly known as gastric peroral endoscopic pyloromyotomy (G-POEM), is one of the endoscopic treatment methods for refractory gastroparesis. As a super minimally invasive pyloric sphincteroplasty, G-POEM demonstrates a short-term (one-year) success rate of 50%–80%, with an adverse event rate of approximately 10%. Current evidence suggests that G-POEM is superior to gastric electrical stimulation and surgical pyloroplasty for treating gastroparesis.
1. Indications and contraindications
(1) Indications
(a) Refractory gastroparesis, defined as persistent symptoms despite lifestyle modifications and failed pharmacological therapy (e.g., proton pump inhibitors, metoclopramide, domperidone, mosapride citrate, erythromycin, tricyclic antidepressants, or selective serotonin reuptake inhibitors), severe adverse drug reactions (e.g., rapid tachyphylaxis, extrapyramidal symptoms, or arrhythmias), or requiring repeated hospitalization for nutritional support.
(b) All patients undergoing preoperative evaluation for gastroparesis, including gastroscopy, gastric emptying scintigraphy (GES), contrast-enhanced ultrasonography of gastric emptying, and gastroparesis cardinal symptom index (GCSI), demonstrating consistent findings of gastroparesis.
(c) Etiologies such as diabetic gastroparesis, gastroparesis following proximal gastrectomy, or esophageal resection.
(2) Contraindications
(a) Luminal gastric outlet obstruction caused by mass lesions.
(b) Contraindications to general anesthesia.
(c) Contraindications to gastroscopic evaluation or treatment.
(d) Coexistence of gastrointestinal malignancies.
2. Preoperative preparation
(1) Intravenous fluids must be administered to correct metabolic imbalances (e.g., ketoacidosis, uremia, hypoglycemia, or hyperglycemia). Enteral nutrition and glycemic control are needed for diabetic patients. Iatrogenic gastroparesis, particularly opioid-induced cases, must be addressed.
(2) Based on the severity of gastroparesis, a liquid diet must be observed for two days before surgery, fasting one day prior, or fasting for two days. On the day of surgery, patients should fast from food and water for at least 8 h.
(3) For patients with gastric retention identified via gastroscopy, preoperative gastric lavage under standard endoscopy is performed to prevent complications such as tracheal aspiration during general anesthesia.
3. Surgical procedure and techniques (continued)
(1) Anesthesia and positioning. All patients undergo general anesthesia with endotracheal intubation. The patient is positioned in the left lateral decubitus position. Precautions include maintaining body temperature, regulating room temperature and infusion fluid temperature, and preventing pressure ulcers.
(2) Surgical steps
(a) Creating the tunnel entrance: the gastroscope is advanced to approximately 5 cm proximal to the pylorus along the greater curvature of the gastric antrum. The tunnel’s entrance is marked with four argon plasma coagulation points (optional). A submucosal injection of 6–8 ml of a methylene blue/saline solution/epinephrine mixture is administered. A reverse T-shaped (or transverse) incision is made to create the tunnel entrance (figure 9.21).
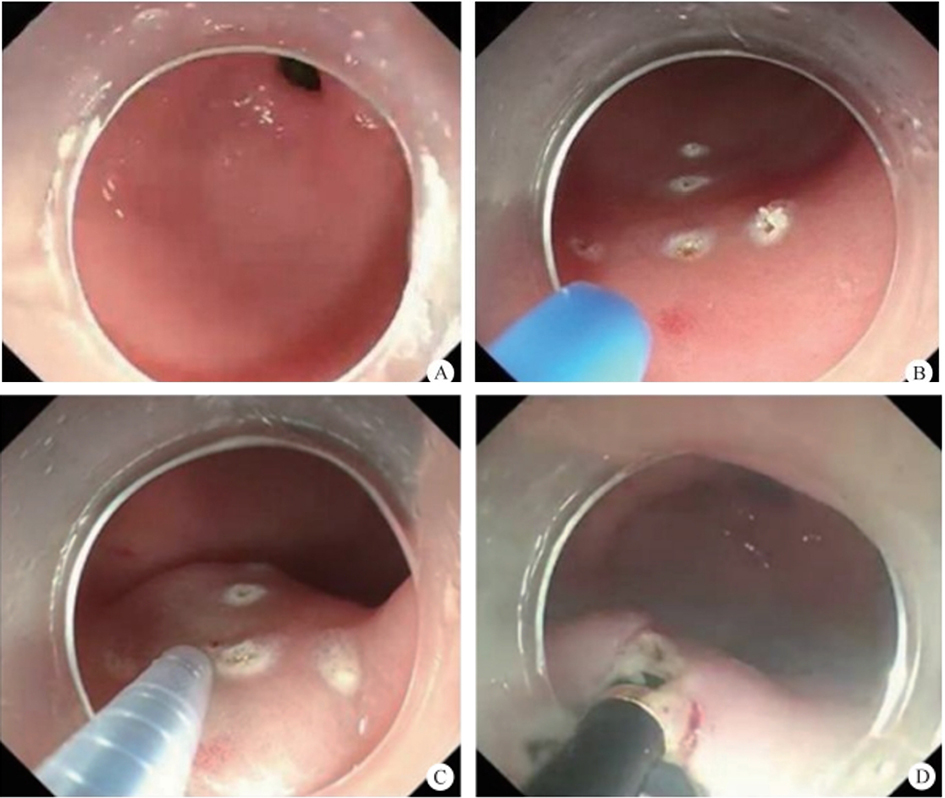

Steps for creating the tunnel entrance in super minimally invasive tunnel myotomy for gastroparesis. A. The gastroscope is positioned with the tip 5 cm proximal to the pyloric ring. B. The tunnel entrance is marked using argon plasma coagulation (APC). C. Submucosal injection is performed. D. The tunnel’s entrance is created.
(b) Establishing the submucosal tunnel: the submucosal layer is dissected from proximal to distal along the greater curvature of the gastric antrum to establish a submucosal ‘tunnel’ extending to the pyloric ring (crescent-shaped landmark). Care is taken to remain close to the muscle layer during dissection. Repeated submucosal injections are performed during the process to minimize the risk of injury to the mucosal layer (figure 9.22).
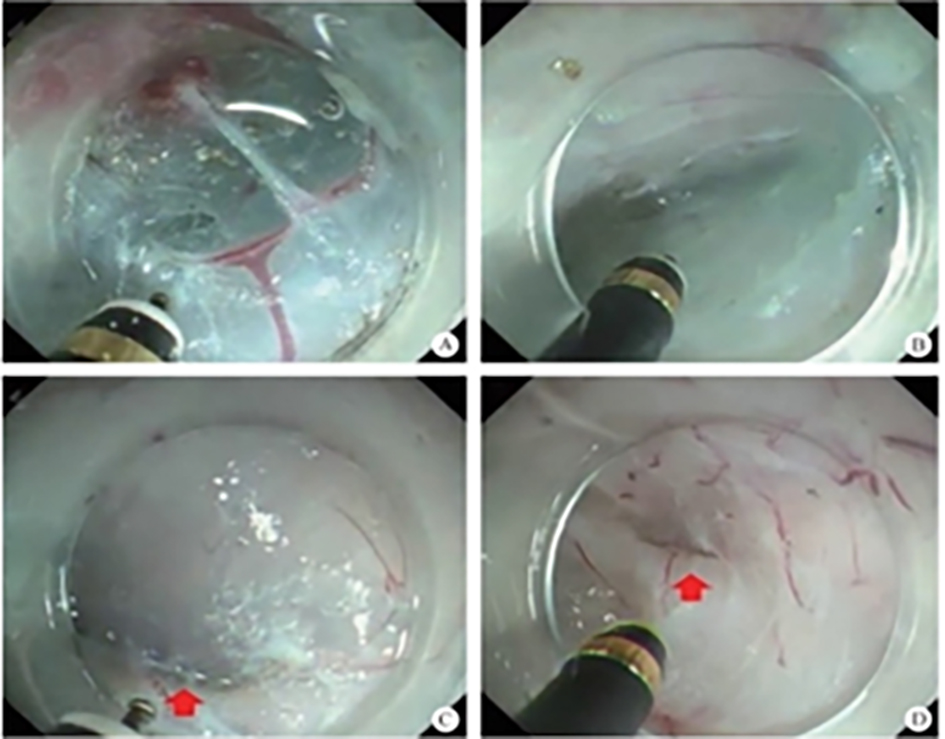

Steps for establishing the submucosal tunnel in super minimally invasive tunnel myotomy for gastroparesis. A. The gastroscope enters the submucosal tunnel. B. Dissection is performed close to the muscle layer. C. Dissection continues after the structure of the pyloric ring has been identified. D. The pyloric ring structure is fully exposed (indicated by the red arrow pointing to the pyloric ring).
(c) Pyloromyotomy: under direct endoscopic visualization, the pyloric sphincter is incised progressively from distal to proximal (anus-to-oral direction). The incision begins at the pyloric ring and is performed in a gradual, superficial-to-deep manner. A full-thickness incision is made at the pyloric ring over a length of 1–2 cm. The circular muscle of the gastric antrum is then incised longitudinally while preserving the longitudinal muscle layer. The incision extends to 1–2 cm below the tunnel’s entrance to minimize the risk of extensive perforation (figure 9.23).
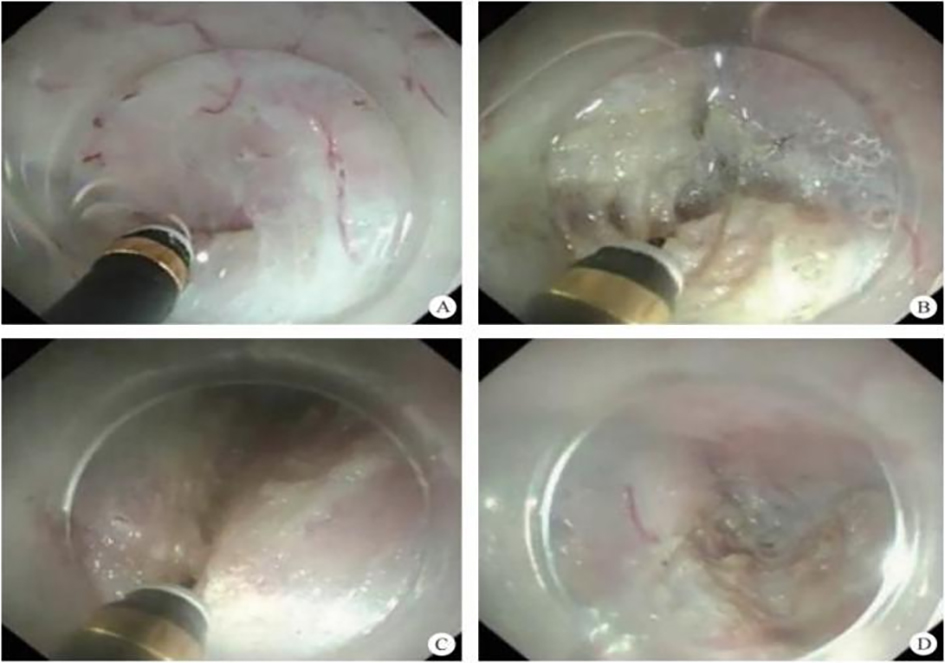

Steps of pyloromyotomy in super minimally invasive tunnel myotomy for gastroparesis. A. The muscle layer is incised starting at the pyloric ring. B. Full-thickness incision of the muscle layer is performed. C. The circular muscle of the gastric antrum is incised. D. Gradual progressive myotomy is completed.
4. Inspection and closure of the tunnel
After completing the myotomy, one has to make sure that the gastroscope passes through the pylorus without resistance. All gas and fluid from the submucosal ‘tunnel’ must be sucked and the surgical field must be rinsed. Any bleeding point or small vessel must be coagulated using an electrocautery device. The mucosal incision is closed using multiple metallic clips to achieve complete closure of the tunnel’s entrance (figure 9.24).
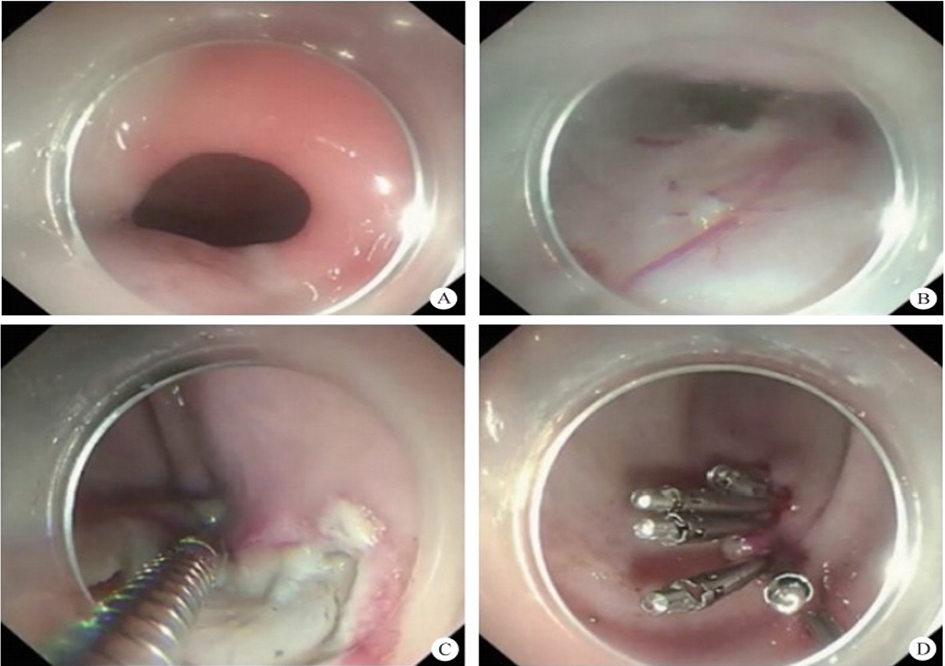

Steps for inspecting and closing the tunnel in super minimally invasive tunnel myotomy for gastroparesis. A. Enlargement of the pyloric opening. B. Inspection of the tunnel for bleeding and hemostasis where necessary. C. Closure of the tunnel entrance using tissue clips. D. Completion of the closure of the tunnel entrance.
5. Postoperative management
(1) Patients should abstain from oral intake, receive acid suppression therapy, and intravenous fluid replacement for three to five days.
(2) Prophylactic anti-infective treatment with third-generation cephalosporins combined with ornidazole is recommended for 48 h, alongside other symptomatic management measures.
(3) Gradual dietary progression is advised: from liquid diet to semi-liquid diet, and eventually to a regular diet.
(4) Postoperative treatment with proton pump inhibitors (PPIs) and gastric mucosal protective agents should continue for one month. Blood glucose, arterial blood gases, and biochemical parameters must be closely monitored. Any metabolic disorder should be corrected promptly.
6. Study of a classic case
Case history: the patient, a young woman, was admitted with a chief complaint of “recurrent nausea and vomiting for two years.” Two years prior, due to persistent nausea and vomiting that made oral intake impossible, she had undergone jejunal feeding tube placement for enteral nutrition. She had a medical history of type-1 diabetes mellitus for 18 years.
Upon admission, diagnostic evaluations revealed the following.
Gastroscopy indicated impaired gastric motility without evidence of neoplastic or space-occupying lesions. Abdominal CT scan (plain and enhanced) showed mild dilation of the gastric antrum, ruling out organic obstructive gastrocolorectal diseases. Gastroparesis symptom score (GCSI): 35 points. Gastric emptying scintigraphy findings: solid gastric emptying half-time: 158.44 min. Gastric emptying rate within two hours: 9% (figure 9.25).
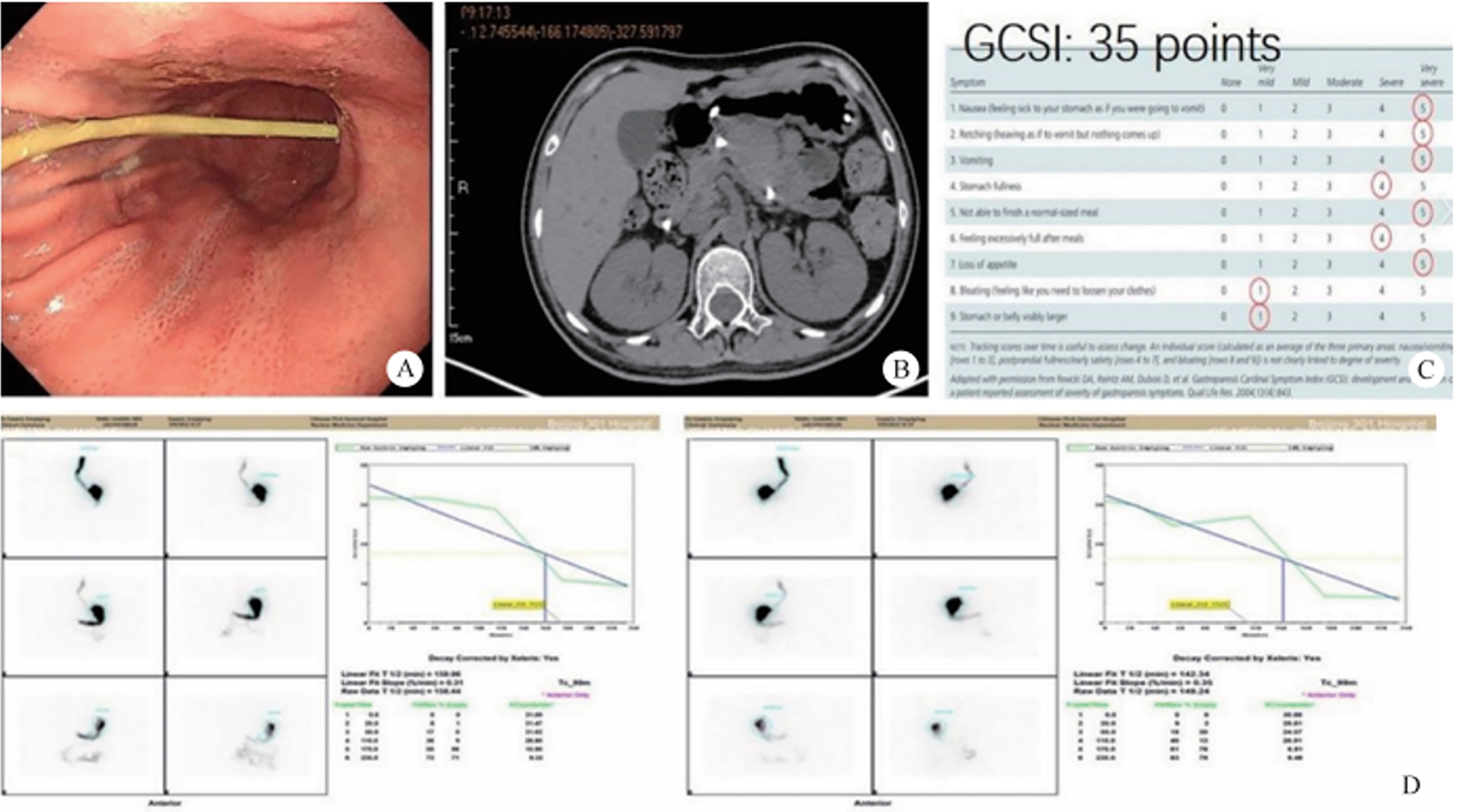

Examination results of a patient with gastroparesis. A. Gastroscopy showing a jejunal feeding tube within the stomach and impaired gastric motility. B. Contrast-enhanced abdominal CT revealing mild dilation of the gastric antrum without evidence of tumors or obstructive lesions. C. Gastroparesis cardinal symptom index (GCSI) score reaching as high as 35 points. D. Gastric emptying scintigraphy indicating delayed gastric emptying consistent with gastroparesis.
Diagnosis: the patient was diagnosed with diabetic gastroparesis.
After comprehensive preoperative evaluation, nutritional status optimization, and obtaining informed surgical consent, the patient underwent G-POEM (gastric peroral endoscopic myotomy).
Refer to video 9.13, Super minimally invasive peroral endoscopic myotomy for gastroparesis (See online resources).
Postoperative outcome: the jejunal feeding tube was removed postoperatively. The patient was kept nil by mouth and received intravenous acid suppression and fluid replacement for five days. Prophylactic anti-infective therapy with Shupushen (esomeprazole) and ornidazole was administered for two days. Gradual dietary progression was implemented, and the patient returned to a normal diet. One month postoperatively, the GCSI score improved to 12 points, and blood glucose levels were more stable compared to preoperative levels.
7. Hot-spot issues and research progress in the consensus
(1) Research on the pathogenesis of gastroparesis remains limited to animal studies. Current studies on the pathogenesis of gastroparesis primarily rely on animal experiments, revealing mechanisms involving neural, immune, and inflammatory factors. 1) Exogenous denervation leading to delayed gastric emptying. 2) Loss of interstitial cells of Cajal (ICCs), causing reduced smooth muscle contraction and disrupted rhythm. 3) Functional alterations in M2 immune cells, resulting in ICC and smooth muscle damage. 4) Absence of neuronal nitric oxide synthase (nNOS), impairing inhibitory signaling pathways and hindering gastric emptying. 5) Uncoordinated gastric antral contractions. 6) Pylorospasm affecting gastric emptying. Due to the difficulty in obtaining gastric muscularis propria samples from gastroparesis patients, most research on the disease’s pathogenesis is limited to animal models.
(2) Selection of candidates for G-POEM surgery. Ideal candidates for G-POEM (gastric peroral endoscopic myotomy) include:
(a) Adults with refractory gastroparesis confirmed by gastroscopy to have no pyloric obstruction.
(b) Patients with delayed gastric emptying verified by gastric emptying scintigraphy (GES), particularly with a 4-h retention rate >20%.
(c) Patients exhibiting moderate to severe symptoms (mean GCSI score > 2), particularly those with predominant nausea and vomiting.
The decision to perform G-POEM should be based on a comprehensive clinical evaluation, rather than solely on GES findings or GCSI scores. Clinical experience suggests that patients with predominant nausea and vomiting respond better to G-POEM, while those with predominant abdominal pain have poorer outcomes, though supporting data is lacking. Patients unresponsive to gastric electrical stimulation, pyloric stenting, or botulinum toxin injection may also be considered for G-POEM. Current studies do not definitively determine whether symptom relief following botulinum toxin injection or pyloric stenting predicts successful G-POEM outcomes.
(3) Optimization and follow-up of G-POEM surgery. Experts recommend the use of an IT knife to enhance the safety margin of myotomy, particularly during the early learning curve or in challenging cases. A meta-analysis of 10 studies reported a one-year success rate of 61% for G-POEM. Some patients with short-term symptom relief experience symptom recurrence over time. Research on repeated G-POEM procedures is currently lacking. A single-center study abroad proposed a ‘dual myotomy’ technique during G-POEM, involving a second pyloromyotomy near the first during the procedure. Six-month follow-ups showed that this approach was technically feasible, did not significantly increase surgical time, and demonstrated better outcomes than single myotomy. However, its long-term efficacy requires further follow-up and comparative studies. Current guidelines recommend follow-up GES at four to eight weeks postoperatively and outpatient visits at one to three months. GCSI scoring should be conducted during every pre- and post-operative follow-up visit to quantify gastroparesis symptoms. However, due to the limited availability of GES in many hospitals, long-term follow-up studies on G-POEM outcomes remain scarce in China.
(4) G-POEM as a new avenue for research into gastroparesis pathogenesis and treatment. Studies indicate that interstitial cells of Cajal (ICCs) play a key role in transmitting neural signals to gastrointestinal smooth muscle and are central to the pathogenesis of gastroparesis. Apoptosis and oxidative stress contribute to ICC loss. Conventional endoscopy allows only mucosal biopsy, restricting research on gastroparesis pathogenesis. The advent of G-POEM enables muscularis propria biopsy during myotomy, providing new opportunities for foundational research and etiology-based treatments for gastroparesis.
9.4.3 Gastric Peroral Super Minimally Invasive Botulinum Toxin Injection (BoNT)
1. Overview
Gastric peroral super minimally invasive botulinum toxin injection involves administering botulinum toxin (BoNT) into the pylorus. This intervention, despite ongoing debate, remains widely used in clinical practice due to its relative safety. BoNT inhibits neuromuscular transmission through the following mechanism: it binds to receptor sites at motor or sympathetic nerve terminals, enters the nerve endings, and suppresses acetylcholine release, leading to muscle relaxation and paralysis. When injected into muscles at therapeutic doses, BoNT induces partial chemical denervation, causing localized paralysis. Paralysis of the pyloric muscle reduces gastric outlet pressure, thereby promoting gastric emptying. Endoscopic botulinum toxin injection (BTI) is performed under endoscopic visualization, where the appropriate amount of BoNT is injected into four quadrants of the pyloric sphincter region using an injection needle (figure 9.26). In most cases, patients can be discharged on the same day of the procedure. Dietary progression is initiated based on the patient’s tolerance and gradually advanced.

Endoscopic injection of botulinum toxin.
2. Hot-spot issues and research progress in the consensus
(1) Indications for BTI in treating gastroparesis. Manometric studies in some patients with gastroparesis (GP) have shown increased pyloric tone and prolonged phasic contractions, a phenomenon referred to as ‘pylorospasm.’ Botulinum toxin is a potent neurotransmitter inhibitor. While early studies demonstrated that intrapyloric botulinum toxin injections improved gastroparesis symptoms in diabetic patients, two larger double-blind, placebo-controlled studies revealed only modest improvements in gastric emptying without significant symptom relief compared to placebo. Consequently, the consensus does not recommend intrapyloric botulinum toxin injections as a treatment for gastroparesis. However, a study indicated potential benefits for gastroparesis patients with reduced pyloric distensibility, as measured preoperatively using the EndoFLIP system (Crospon, Galway, Ireland), who underwent intrapyloric botulinum toxin injection. Additionally, type A botulinum toxin is widely used for pediatric spastic disorders. A meta-analysis suggested that intrapyloric injection of type-A botulinum toxin is safe for children and can provide temporary relief to patients with refractory upper gastrointestinal symptoms, whether or not they have gastroparesis. Therefore, subgroup analyses are necessary to identify appropriate candidates among patients with ‘pylorospasm’ to maximize treatment benefits.
(2) Impact of BTI on subsequent treatments. For patients who have not previously undergone botulinum toxin injections, submucosal injection during G-POEM (gastric peroral endoscopic myotomy) can facilitate the separation of the mucosal and muscular layers. However, prior botulinum toxin injections may lead to the fusion of the mucosal and muscular layers, resulting in dense scarring and an opaque submucosal layer, which poses challenges for establishing a submucosal tunnel. This consideration is one reason why recent consensus and studies do not recommend endoscopic botulinum toxin injection for gastroparesis treatment. Some studies have shown that clinical symptom improvement after BTI is a predictor of G-POEM effectiveness in gastroparesis patients. However, further research is needed to evaluate the advantages and disadvantages of performing G-POEM after BTI.
(3) Choosing between BTI and G-POEM for gastroparesis treatment. Recent interest has focused on the mechanical (G-POEM) and chemical (BTI) approaches to treating refractory gastroparesis. G-POEM is emerging as a promising technique for refractory gastroparesis. A recent double-blind randomized study compared the clinical efficacy of G-POEM and BTI. G-POEM demonstrated a higher short-term (three-month) clinical success rate than BTI, though the difference was not statistically significant. However, the follow-up duration of this study was relatively short. Large-scale, long-term follow-up, high-quality clinical studies are needed to further validate these findings.
9.4.4 Peroral Super Minimally Invasive Pyloric Stenting for Gastroparesis
1. Overview
Peroral super minimally invasive pyloric stenting for gastroparesis involves the placement of a self-expanding metal stent (SEMS) at the pylorus. The proximal flange of the stent is positioned in the prepyloric antrum and secured to the gastric wall using endoscopic sutures or clips to maintain the pylorus in an open state, thereby facilitating normal gastric emptying. Complications associated with stent placement include stent migration, perforation, and bleeding. Proximally migrated stents can be retrieved endoscopically using forceps through the stomach and esophagus. Distally migrated stents are typically monitored via radiographic imaging until their safe passage through the intestinal tract is confirmed. Since pyloric stenting is not considered a long-term treatment option, its current application is limited to temporary treatment for refractory gastroparesis patients hospitalized due to severe symptoms or as a predictive tool to identify patients who may respond to subsequent treatments, such as G-POEM.
2. Surgical procedure and techniques
A self-expanding metal stent is deployed across the pylorus using a delivery system. Under direct endoscopic visualization, the stent is released while observing the yellow marker at the proximal end of the stent, eliminating the need for X-ray guidance. The stent fully expands in the trans-pyloric position, with its proximal end located in the gastric antrum (figure 9.27).

Steps of peroral super minimally invasive pyloric stenting for gastroparesis. A. Placement of the delivery system across the pylorus. B. Deployment of the stent under direct endoscopic visualization. C. Full expansion of the stent, with the proximal end located in the gastric antrum.
3. Hot-spot issues and research progress in the consensus
Pyloric stent placement, while effective in alleviating symptoms, also increases the risk of complications such as stent migration, reflux, perforation, bleeding, and new stenosis caused by tissue hyperplasia induced by the stent. Consequently, pyloric stenosis due to benign diseases is considered a contraindication for stent placement. Reports suggest that pyloric stent placement can significantly improve symptoms in patients with gastroparesis. Endoscopic placement of self-expanding metal pyloric stents (SEMCs), fixed to the gastric side through suturing, has been tested in small, open-label studies, particularly for refractory gastroparesis. Temporary placement of SEMCs can substantially reduce the risk of complications. However, reports on the long-term efficacy of temporary transpyloric stent treatment for refractory gastroparesis after stent removal remain limited. Case studies have indicated that temporary trans-pyloric SEMC placement is a safe, effective, and minimally invasive option. Despite case reports documenting symptom improvement, the safety and efficacy of transpyloric stent placement for refractory gastroparesis require further investigation due to the lack of data from prospective, controlled trials and concerns about stent migration. Current guidelines do not recommend routine use of pyloric stents for gastroparesis patients.
References
[1] Bray F., Laversanne M., Sung H., et al. (2024) Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries, CA Cancer J. Clin. 74(3), 229–263.
[2] Li P., Li Z., Linghu E., et al. (2024) Chinese national clinical practice guidelines on the prevention, diagnosis, and treatment of early gastric cancer, Chin. Med. J. (Engl.) 137(8), 887–908.
[3] Expert Group of the Beijing Municipal Science and Technology Commission Major Project Study on Standardized Treatment of Early Gastric Cancer. (2019) Expert consensus on standardized endoscopic resection of early gastric cancer (2018, Beijing), Chin. J. Dig. Endosc. 36(6), 381–392.
[4] Fujishiro M. (2024) Advanced diagnostic and therapeutic endoscopy for early gastric cancer, Cancers (Basel) 16(5).
[5] Liu S., Zhang M., Yang Y., et al. (2024) Establishment and validation of a risk score model based on EUS: assessment of lymph node metastasis in early gastric cancer, Gastrointest. Endosc.
[6] Uema R., Hayashi Y., Kizu T., et al. (2024) A novel artificial intelligence-based endoscopic ultrasonography diagnostic system for diagnosing the invasion depth of early gastric cancer, J. Gastroenterol. 59(7), 543–555.
[7] Gao Z., Yu Z., Zhang X., et al. (2023) Development of a deep learning model for early gastric cancer diagnosis using preoperative computed tomography images, Front. Oncol. 13, 1265366.
[8] Digestive Endoscopy Terminology Examination Subcommittee. (2021) Terminology of digestive endoscopy. Science Press, Beijing.
[9] ASGE Standards of Practice Committee; Forbes N., Elhanafi S. E., Al-Haddad M. A., et al. (2023 Sep) American Society for Gastrointestinal Endoscopy guideline on endoscopic submucosal dissection for the management of early esophageal and gastric cancers: summary and recommendations, Gastrointest. Endosc. 98(3), 271–284.
[10] Tsuji Y., Fujishiro M., Kodashima S., et al. (2015 Apr) Polyglycolic acid sheets and fibrin glue decrease the risk of bleeding after endoscopic submucosal dissection of gastric neoplasms (with video), Gastrointest. Endosc. 81(4), 906–912.
[11] Kataoka Y., Tsuji Y., Hirasawa K., et al. (2019 Jul) Endoscopic tissue shielding to prevent bleeding after endoscopic submucosal dissection: a prospective multicenter randomized controlled trial, Endoscopy 51(7), 619–627.
[12] Lee B. I., Kim B. W., Kim H. K., et al. (2011) Routine mucosal closure with a detachable snare and clips after endoscopic submucosal dissection for gastric epithelial neoplasms: a randomized controlled trial, Gut Liver 5(4), 454–459.
[13] Maekawa S., Nomura R., Murase T., et al. (2015) Complete closure of artificial gastric ulcer after endoscopic submucosal dissection by combined use of a single over-the-scope clip and through-the-scope clips (with videos), Surg. Endosc. 29(2), 500–504.
[14] Japanese Gastric Cancer Association. (2021) Japanese gastric cancer treatment guidelines 2018 (5th edition), Gastric Cancer 24(1), 1–21.
[15] Abe S., Oda I., Suzuki H., et al. (2015) Long-term surveillance and treatment outcomes of metachronous gastric cancer occurring after curative endoscopic submucosal dissection, Endoscopy 47(12), 1113–1118.
[16] Libânio D., Pimentel-Nunes P., Bastiaansen B., et al. (2023 Apr) Endoscopic submucosal dissection techniques and technology: European Society of Gastrointestinal Endoscopy (ESGE) technical review, Endoscopy 55(4), 361–389.
[17] Su Y. F., Cheng S. W., Chang C. C., et al. (2020) Efficacy and safety of traction-assisted endoscopic submucosal dissection: a meta-regression of randomized clinical trials, Endoscopy 52, 338–348.
[18] Nagata M., Namiki M., Fujikawa T., et al. (2023 Jun) Impact of traction direction in traction-assisted gastric endoscopic submucosal dissection (with videos), Dig. Dis. Sci. 68(6), 2531–2544.
[19] Grimaldi J., Louis-Jean M., Lafeuille P., et al. (2024 Mar 6) Endoscopic submucosal dissection with adaptive traction strategy: first prospective multicenter study, Gastrointest. Endosc. S0016-5107(24)00148-2.
[20] Zhao X., Yao F. (2019) Assisted traction techniques in endoscopic submucosal dissection (ESD), Chin. J. Dig. Endosc. 36(8), 541–547.
[21] Linghu E. (2022) A new stage of surgical treatment: super minimally invasive surgery, Chin. Med. J. 135(1), 1–3.
[22] Li X., Zhang W., Gao F., et al. (2023) A modified endoscopic full-thickness resection for gastrointestinal stromal tumors: a new closure technique based on the instruction of super minimally invasive surgery, Endoscopy 55(S 01), E561–E562.
[23] Aslanian H. R., Sethi A., Bhutani M. S., et al. (2019) ASGE guideline for endoscopic full-thickness resection and submucosal tunnel endoscopic resection, VideoGIE 4(8), 343–350.
[24] Gu Y., Li G., Wang J., et al. (2020) Comparing the impact of laparoscopic sleeve gastrectomy and gastric cancer surgery on resting-state brain activity and functional connectivity, Front. Neurosci. 14, 614092.
[25] Linghu E. (2016) Super minimally invasive surgery: a new stage in the history of surgery, Chin. J. Gastrointest. Endosc. (Electron. Ed.) 3(3), 97–98.
[26] Li G. Z., Doherty G. M., Wang J. (2022) Surgical management of gastric cancer: a review, JAMA Surg. 157(5), 446–454.
[27] Watanabe A., Hamilton T. D. (2022) Lymph node mapping in gastric cancer: a pilot study in Western patients, Can. J. Surg. 65(5), E630–E634.
[28] Sakamoto E., Safatle-Ribeiro A. V., Ribeiro Jr U. (2022) Advances in surgical techniques for gastric cancer: indocyanine green and near-infrared fluorescence imaging. Is it ready for prime time? Chin. J. Cancer Res. 34(6), 587–591.
[29] Wei J., Bu Z. (2020) Sentinel lymph node detection for gastric cancer: promise or pitfall? Surg. Oncol. 33, 1–6.
[30] Kim S., Bae W. J., Ahn J. M., et al. (2021) MicroRNA signatures associated with lymph node metastasis in intramucosal gastric cancer, Mod. Pathol. 34(3), 672–683.
[31] Ono H., Yao K., Fujishiro M., et al. (2021) Guidelines for endoscopic submucosal dissection and endoscopic mucosal resection for early gastric cancer (second edition), Dig. Endosc. 33, 4–20.
[32] Ajani J. A., D’Amico T. A., Bentrem D. J., et al. (2022) Version 2.2022, NCCN clinical practice guidelines in oncology, J. Natl. Compr. Cancer Netw. 20(2), 167–192.
[33] Wang F. H., Zhang X. T., Li Y. F., et al. (2021) The Chinese Society of Clinical Oncology (CSCO): clinical guidelines for the diagnosis and treatment of gastric cancer, 2021, Cancer Commun. (Lond.) 41(8), 747–795.
[34] Feng X., Linghu E., Chai N., et al. (2018 Mar 6) Endoscopic submucosal tunnel dissection for large gastric neoplastic lesions: a case-matched controlled study, Gastroenterol. Res. Pract. 2018, 1419369, 1–9.
[35] Lin C., Zhang Z., Wang L. (2023) Progress in the treatment of early gastric cancer with laparoscopic and endoscopic cooperative surgery, Chin. J. Gastrointest. Surg. 26, 798–802.
[36] Zhao P. Y., Ma Z. F., Jiao Y. N., et al. (2022) Laparoscopic and endoscopic cooperative surgery for early gastric cancer: perspective for actual practice, Front. Oncol. 12, 969628.
[37] Japanese Gastric Cancer Association. (2023 Jan) Japanese gastric cancer treatment guidelines 2021 (6th edition), Gastric Cancer 26(1), 1–25.
[38] Nunobe S., Hiki N., Gotoda T., et al. (2012) Successful application of laparoscopic and endoscopic cooperative surgery (LECS) for a lateral-spreading mucosal gastric cancer, Gastric Cancer 15(3), 338–342.
[39] Kikuchi S., Nishizaki M., Kuroda S., et al. (2017) Nonexposure laparoscopic and endoscopic cooperative surgery (closed laparoscopic and endoscopic cooperative surgery) for gastric submucosal tumor, Gastric Cancer 20(3), 553–557.
[40] Zang L., Zuo B. (2022) Application value of laparoscopy and endoscopy combined surgery in the treatment of early gastric cancer, Chin. J. Pract. Surg. 42(10), 1119–1122.
[41] Li H., Zhao L. L., Zhang X. C., et al. (2020) Combination of endoscopic submucosal dissection and laparoscopic sentinel lymph node dissection in early mucinous gastric cancer: role of lymph node metastasis, World J. Clin. Cases 8, 3474–3482.
[42] An J. Y., Min J. S., Hur H., et al. (2020) Laparoscopic sentinel node navigation surgery versus laparoscopic gastrectomy with lymph node dissection for early gastric cancer: short-term outcomes of a multicentre randomized controlled trial (SENORITA), Br. J. Surg.
[43] Schlemper R. J., Riddell R. H., Kato Y., et al. (2000 Aug) The Vienna classification of gastrointestinal epithelial neoplasia, Gut 47(2), 251–255.
[44] Wang N. J., Chai N. L., Tang X. W., et al. (2022 Mar 15) Clinical efficacy and prognostic risk factors of endoscopic radiofrequency ablation for gastric low-grade intraepithelial neoplasia, World J. Gastrointest. Oncol. 14(3), 724–733.
[45] Beijing Branch of Chinese Society of Digestive Endoscopy. (2019) Expert consensus on standardized diagnosis and treatment of gastric low-grade intraepithelial neoplasia (2019, Beijing), Chin. J. Gastrointest. Endosc. (Electron. Ed.) 6(2), 49–56.
[46] The Paris endoscopic classification of superficial neoplastic lesions: esophagus, stomach, and colon: November 30 to December 1, 2002, Gastrointest. Endosc. (2003 Dec) 58(6 Suppl), S3–S43.
[47] Wang N., Chai N., Linghu E., et al. (2020) The clinical study of endoscopic radiofrequency ablation in the treatment of gastric low-grade intraepithelial neoplasia, Chin. J. Gastrointest. Endosc. (Electron. Ed.) 7(03), 117–120.
[48] Wu B., Linghu E., Yang J., et al. (2011) Clinical pathologic features of low-grade intraepithelial neoplasia and its turnover, J. Chin. PLA Postgrad. Med. Sch. 32(06), 598–600.
[49] Niu X. (2016) Protective role of submucosal fluid padding in the ablation of gastric low-grade intraepithelial neoplasia (LGIN). Nankai University, pp. 1–50.
[50] Wang N., Chai N., Li L., et al. (2022 Jun 22) Comparison of endoscopic radiofrequency ablation and argon plasma coagulation in patients with gastric low-grade intraepithelial neoplasia: a large-scale retrospective study, Can. J. Gastroenterol. Hepatol. 2022, 2349940.
[51] Jung S. J., Cho S. J., Choi I. J., et al. (2013) Argon plasma coagulation is safe and effective for treating smaller gastric lesions with low-grade dysplasia: a comparison with endoscopic submucosal dissection, Surg. Endosc. 27(4), 1211–1218.
[52] Wang H., Ahn J. Y., Noh J. H., et al. (2024 Jan 23) Clinical outcomes of argon plasma coagulation for the treatment of gastric low grade dysplasia, Gastrointest. Endosc. S0016-5107(24)00045-2.
[53] Li B., Chai N., Tang X., et al. (2021) Risk factors of local recurrence of gastric low-grade intraepithelial neoplasia after endoscopic argon plasma coagulation, Chin. J. Laparosc. Surg. (Electron. Ed.) 14(02), 104–108.
[54] Liu J., Wang Z., Zhang Z., et al. (2016) Endoscopic argon plasma coagulation treated low-grade gastric intraepithelial neoplasia in 62 elderly patients, Chin. J. Health Care Med. 18(06), 445–447.
[55] Mao Y., Qiu H., Liu Q., et al. (2013 Nov) Endoscopic holmium: YAG laser ablation of early gastrointestinal intramucosal cancer, Lasers Med. Sci. 28(6), 1505–1509.
[56] Lu Z., Mao Y., Liu Q., et al. (2001) Observation of Ho: YAG laser via endoscopy for 99 cases with esophagostenosis caused by carcinoma and scar, Chin. J. Phys. Ther. (03), 16–17.
[57] Lu Z., Liu Q., Mao Y., et al. (2002) Ho: YAG laser treatment of tumor and hyperplastic tissue ingrowth in expandable esophageal stents, Chin. J. Laser Med. Surg. (02), 18–20.
[58] Saccomandi P., Quero G., Costamagna G., et al. (2017 Jul) Effects of Nd: YAG laser for the controlled and localized treatment of early gastrointestinal tumors: preliminary in vivo study, Annu. Int. Conf. IEEE Eng. Med. Biol. Soc. 2017, 4533–4536.
[59] Pyo H., Kim H., Kang H. W. (2021 Sep) Evaluations on laser ablation of ex vivo porcine stomach tissue for development of Ho: YAG-assisted endoscopic submucosal dissection (ESD), Lasers Med. Sci. 36(7), 1437–1444.
[60] Wiech T., Walch A., Werner M. (2005) Histopathological classification of nonneoplastic and neoplastic gastrointestinal submucosal lesions, Endoscopy 37, 630–634.
[61] Aghdassi A., Christoph A., Dombrowski F., et al. (2018) Gastrointestinal stromal tumors: clinical symptoms, location, metastasis formation, and associated malignancies in a single center retrospective study, Dig. Dis. 36, 337–345.
[62] Polkowski M. (2005) Endoscopic ultrasound and endoscopic ultrasound-guided fine-needle biopsy for the diagnosis of malignant submucosal tumors, Endoscopy 37, 635–645.
[63] Rösch T., Kapfer B., Will U., et al. (2002) Accuracy of endoscopic ultrasonography in upper gastrointestinal submucosal lesions: a prospective multicenter study, Scand. J. Gastroenterol. 37, 856–862.
[64] Jacobson B. C., Bhatt A., Greer K. B., et al. (2023 Jan 1) ACG clinical guideline: diagnosis and management of gastrointestinal subepithelial lesions, Am. J. Gastroenterol. 118(1), 46–58.
[65] Surgical Group of the Chinese Society of Digestive Endoscopy, Chinese Medical Association, Natural Orifice Transluminal Endoscopic Surgery (NOTES) Group of the Chinese Society of Digestive Endoscopy, Chinese Medical Association, Digestive Endoscopy Committee of the Endoscopists Branch, Chinese Medical Doctor Association, et al. (2023) Chinese consensus on endoscopic diagnosis and management of gastrointestinal submucosal tumors (version 2023), Chin. J. Dig. Endosc. 40(04), 253–263.
[66] Cao B., Lu J., Tan Y., et al. (2021) Efficacy and safety of submucosal tunneling endoscopic resection for gastric submucosal tumors: a systematic review and meta-analysis, Rev. Esp. Enferm. Dig. 113(1), 52–59.
[67] Huang L., Jia Y. X., Lyu B., et al. (2022 Apr 4) Effects of endoscopic submucosal excavation with non-submucosal injection on stromal tumors in stomach, Front. Oncol. 12, 792445.
[68] Wang Z., Liu D., Liu X., et al. (2020) Application of endoscopic submucosal excavation assisted with ligation device for treatment of gastrointestinal submucosal tumors, Chin. J. Gastroenterol. 25(06), 358–362.
[69] Ye Y., Hong Y., Wei W., et al. (2024 Dec) Mucosal snare resection-endoscopic submucosal excavation: a novel technology, Endoscopy 56(S 01), E192–E193.
[70] Akashi Y., Ogawa K., Narasaka T., et al. (2022 Apr) A scoring system to predict surgical difficulty in minimally invasive surgery for gastric submucosal tumors, Am. J. Surg. 223(4), 715–721.
[71] Dalal I., Andalib I. (2022) Advances in endoscopic resection: a review of endoscopic submucosal dissection (ESD), endoscopic full thickness resection (EFTR) and submucosal tunneling endoscopic resection (STER), Transl. Gastroenterol. Hepatol. 7, 19.
[72] Granata A., Martino A., Ligresti D., et al. (2021 Jul 27) Closure techniques in exposed endoscopic full-thickness resection: overview and future perspectives in the endoscopic suturing era, World J. Gastrointest. Surg. 13(7), 645–654.
[73] Kaan H. L., Ho K. Y. (2020 Sep) Endoscopic full thickness resection for gastrointestinal tumors - challenges and solutions, Clin. Endosc. 53(5), 541–549. DOI: 10.5946/ce.2019.161. Epub 2020 Feb 17.
[74] ASGE Technology Committee; Aslanian H. R., Sethi A., Bhutani M. S., et al. (2019 Jun 29) ASGE guideline for endoscopic full-thickness resection and submucosal tunnel endoscopic resection, VideoGIE 4(8), 343–350.
[75] Sharzehi K., Sethi A., Savides T. (2022 Nov) AGA clinical practice update on management of subepithelial lesions encountered during routine endoscopy: expert review, Clin. Gastroenterol. Hepatol. 20(11), 2435–2443.e4.
[76] Ang K., Gao P., Cai M., et al. (2023) Endoscopic full-thickness resection, indication, methods and perspectives, Dig. Endosc. 35(2), 195–205.
[77] Kim S. Y., Kim K. O. (2018) Endoscopic treatment of subepithelial tumors, Clin. Endosc. 51, 19–27.
[78] Zhang D., Lin Q., Shi R., et al. (2018 Dec) Ligation-assisted endoscopic submucosal resection with apical mucosal incision to treat gastric subepithelial tumors originating from the muscularis propria, Endoscopy 50(12), 1180–1185.
[79] Li S., Li Q., Liu M., et al. (2024 May) Precutting endoscopic band ligation-assisted resection is safe and effective for treating gastric submucosal tumors from the muscularis propria, Dig. Dis. Sci. 69(5), 1762–1769.
[80] Ntourakis D., Mavrogenis G. (2015 Nov 21) Cooperative laparoscopic endoscopic and hybrid laparoscopic surgery for upper gastrointestinal tumors: current status, World J. Gastroenterol. 21(43), 12482–12497.
[81] Demetri G. D., von Mehren M., Antonescu C. R., et al. (2010 Apr) NCCN Task Force report: update on the management of patients with gastrointestinal stromal tumors, J. Natl. Compr. Cancer Netw. 8(Suppl. 2), S1–S41; quiz S42-4.
[82] Nishida T., Hirota S., Yanagisawa A., et al. (2008 Oct) GIST Guideline Subcommittee. Clinical practice guidelines for gastrointestinal stromal tumor (GIST) in Japan: English version, Int. J. Clin. Oncol. 13(5), 416–430.
[83] Lu A., Zong Y., Zheng H. (2010) Expert consensus on laparoscopic and endoscopic cooperative surgery for gastrointestinal diseases, Chin. J. Pract. Surg. 30(08), 667–668.
[84] Nishida T. (2018 Feb 8) Asian consensus guidelines for gastrointestinal stromal tumor: what is the same and what is different from global guidelines, Transl. Gastroenterol. Hepatol. 3, 11.
[85] Cao Y., Qi Z., Zhong Y. (2023) Progress in the application of laparoscopic and endoscopic cooperative surgery in gastrointestinal stromal tumors, Chin. J. Gastrointest. Surg. 26(10), 997–1000.
[86] Ye Y. Z., Yin Y., Huh S. Y., et al. (2022) Epidemiology, etiology, and treatment of gastroparesis: real-world evidence from a large US national claims database, Gastroenterology 162, 109–121.e5.
[87] Shen S., Xu J., Lamm V., et al. (2019 Jan) Diabetic gastroparesis and nondiabetic gastroparesis, Gastrointest. Endosc. Clin. N. Am. 29(1), 15–25.
[88] Schol J., Wauters L., Dickman R., et al. (2021) United European Gastroenterology (UEG) and European Society for Neurogastroenterology and Motility (ESNM) consensus on gastroparesis, United Eur. Gastroenterol. J. 9, 287–306.
[89] DeSantis E. R., Huang S. (2007) Botulinum toxin type A for treatment of refractory gastroparesis, Am. J. Health Syst. Pharm. 64, 2237–2240.
[90] Hasler W. L. (2005) Nausea, gastroparesis, and aerophagia, J. Clin. Gastroenterol. 39, S223–S229.
[91] Ukleja A., Tandon K., Shah K., et al. (2015) Endoscopic botox injections in therapy of refractory gastroparesis, World J. Gastrointest. Endosc. 7, 790–798.
[92] Arts J., Holvoet L., Caenepeel P., et al. (2007) Clinical trial: a randomized controlled crossover study of intrapyloric injection of botulinum toxin in gastroparesis, Aliment. Pharmacol. Ther. 26, 1251–1258.
[93] Friedenberg F. K., Palit A., Parkman H. P., et al. (2008) Botulinum toxin A for the treatment of delayed gastric emptying, Am. J. Gastroenterol. 103, 416–423.
[94] Camilleri M., Parkman H. P., Shafi M. A., et al. (2013) Clinical guideline: management of gastroparesis, Am. J. Gastroenterol. 108, 1837.
[95] Desprez C., Melchior C., Wuestenberghs F., et al. (2019) Pyloric distensibility measurement predicts symptomatic response to intrapyloric botulinum toxin injection, Gastrointest. Endosc. 90, 754–760 e1.
[96] Ezaizi Y., Hasan B., Manini M. L., et al. (2022 Sep) Intrapyloric botulinum toxin A injection for gastroparesis and functional upper gastrointestinal symptoms in children: mayo clinic experience, review of the literature, and meta-analysis, Paediatr. Drugs 24(5), 539–545.
[97] Saumoy M., Nassani N., Ortiz J., et al. (2017 Oct) Gastric peroral endoscopic myotomy for gastroparesis, after botulinum toxin injection, Endoscopy 49(10), E256–E257.
[98] Wadhwa V., Gonzalez A., Azar F., et al. (2023 Jun) Response to botulinum toxin may predict response to peroral pyloromyotomy in patients with gastroparesis, Endoscopy 55(6), 508–514.
[99] Gonzalez J. M., Mion F., Pioche M., et al. (2024 Feb 19) Gastric peroral endoscopic myotomy versus botulinum toxin injection for the treatment of refractory gastroparesis: results of a double-blind randomized controlled study, Endoscopy.
[100] Liu K., Enke T., Aadam A. (2020) Endoscopic approaches to gastroparesis, Gastroenterol. Hepatol. (NY) 16, 458–463.
[101] Camilleri M. (2016 Aug) Novel diet, drugs, and gastric interventions for gastroparesis, Clin. Gastroenterol. Hepatol. 14(8), 1072–1080.
[102] Liang G. G., Zhang Q. K., Zhang G. X., et al. (2019 Feb 27) Therapeutic effect of a temporary transpyloric stent in refractory post-surgical gastroparesis: a case report, BMC Surg. 19(1), 27.
[103] Lacy B. E., Tack J., Gyawali C. P. (2022 Mar) AGA clinical practice update on management of medically refractory gastroparesis: expert review, Clin. Gastroenterol. Hepatol. 20(3), 491–500.
[104] McCurdy G. A., Gooden T., Weis F., et al. (2023) Gastric peroral endoscopic pyloromyotomy (G-POEM) in patients with refractory gastroparesis: a review, Therap. Adv. Gastroenterol. 16, 17562848231151289.
[105] Aziz M., Gangwani M. K., Haghbin H., et al. (2023) Gastric peroral endoscopic myotomy versus surgical pyloromyotomy/pyloroplasty for refractory gastroparesis: systematic review and meta-analysis, Endosc. Int. Open 11, E322–E329.
[106] Camilleri M., Sanders K. M. (2022) Gastroparesis, Gastroenterology 162, 68–87.e1.
[107] Vosoughi K., Ichkhanian Y., Benias P., et al. (2022) Gastric peroral endoscopic myotomy (G-POEM) for refractory gastroparesis: results from an international prospective trial, Gut 71, 25–33.
[108] Khashab M. A., Wang A. Y., Cai Q. (2023 Jun) AGA clinical practice update on gastric peroral endoscopic myotomy for gastroparesis: commentary, Gastroenterology 164(7), 1329–1335.e1.
[109] Kamal F., Khan M. A., Lee-Smith W., et al. (2022) Systematic review with meta-analysis: one-year outcomes of gastric peroral endoscopic myotomy for refractory gastroparesis, Aliment. Pharmacol. Ther. 55, 168–177.
[110] Abdelfatah M. M., Li B., Kapil N., et al. (2020) Short-term outcomes of double versus single pyloromyotomy at peroral endoscopic pyloromyotomy in the treatment of gastroparesis (with video), Gastrointest. Endosc. 92, 603–609.
[111] Shah R., Calderon L. F., Sanders B. E., et al. (2022) Quantification of interstitial cells of Cajal in the gastric muscle of patients with gastroparesis at per-oral endoscopic pyloromyotomy: a novel approach for future research in pathogenesis of gastroparesis, Dig. Dis. Sci. 67, 4492–4499.
Chapter 10 Super Minimally Invasive Therapies for Duodenal Diseases
10.1 Peroral Super Minimally Invasive Resection for Early Duodenal Cancer
1. Overview
Early duodenal carcinoma refers to malignant epithelial tumors of the duodenum confined to the mucosa or submucosa, regardless of lymph node metastases. Clinical manifestations are nonspecific and may include abdominal pain, nausea, vomiting, fatigue, and weight loss. Peroral super minimally invasive resection for early duodenal cancer can be performed using techniques such as endoscopic submucosal dissection (ESD), endoscopic mucosal resection (EMR), or traction-assisted endoscopic submucosal dissection (TA-ESD). Traction methods utilize external forces to achieve better surgical fields and have demonstrated an R0 resection rate of 96.9% for duodenal lesions, with an adverse event rate of 4.9%. Compared to conventional ESD, TA-ESD offers shorter operative times, higher en bloc resection rates, and improved R0 resection rates.
2. Diagnosis of early duodenal cancer
The primary diagnostic basis for early duodenal cancer includes preoperative endoscopy and biopsy pathology. Endoscopic findings typically show polypoid or superficial lesions, while endoscopic ultrasonography reveals hypoechoic lesions confined to the mucosal or submucosal layer. Pathological types include high-grade adenoma (HGA) and superficial adenocarcinoma (SAC) limited to the submucosa. Studies indicate that biopsy accuracy for duodenal lesions is relatively lower than that for conventional endoscopic examinations and may induce fibrosis. Typical endoscopic features of early duodenal cancer include: tumor diameter >5 mm; rough or nodular surface; depressed or mixed-type morphology (type IIa + IIc or type IIc).
3. Indications and contraindications for traction-assisted endoscopic submucosal dissection
(1) Indications
① High-grade adenoma (HGA) with a diameter ≥10 mm or with evidence of growth, as well as adenocarcinoma confined to the submucosa (in situ carcinoma and T1-stage adenocarcinoma). ② Early duodenal carcinoma located on the lateral or posterior wall of the duodenum. ③ Early duodenal carcinoma involving the submucosa or showing positive non-elevation signs due to fibrosis. ④ Lesions pathologically diagnosed as low-grade adenoma but exhibiting macroscopic features such as redness and roughness, raising suspicion of adenoma with malignant transformation.
(2) Contraindications
① Duodenal carcinoma invading the muscularis propria. ② Early duodenal carcinoma with confirmed lymph node metastasis. ③ Patients with coagulation disorders. ④ Anesthetic contraindications due to severe cardiopulmonary diseases.
4. Preoperative preparation
① Patient evaluation: assess the general condition of the patient. Discontinue antiplatelet, anticoagulant, or other blood-invigorating traditional Chinese medicines at least one week before surgery. ② Fasting: an 8-h preoperative fasting period, including food and water must be observed. ③ Imaging and localization: a high-frequency probe endoscopic ultrasound must be performed in order to determine the lesion’s location, depth, size, layers involved, and blood supply. An enhanced abdominal CT must be conducted to assess the involvement of surrounding tissues, lymph nodes, and proximity of adjacent vessels and organs.
5. Techniques and procedures for traction-assisted endoscopic submucosal dissection
(a) Lesion marking and submucosal injection: a gastroscope must be advanced to the duodenal lesion. Circumferential markings must be created using argon plasma coagulation or a mucosal incision knife approximately 5 mm from the lesion margin. 6–8 ML of a mixture of methylene blue, saline solution, and sodium hyaluronate must be injected into the submucosal layer. The mucosa should be incised on the oral side of the lesion to expose the submucosal layer adequately.
(b) Submucosal dissection: After circumferential mucosal incision, dissect along the submucosal layer from top to bottom. Submucosal injections must be repeated during the dissection to progressively peel the lesion away from the tissue until partial detachment is achieved.
(c) Traction device placement: One side of an ‘8-shaped’ traction ring must be anchored to the lesion and the other side to the opposite intestinal wall. This separates the submucosal layer from the muscularis propria, providing a clear surgical field. Dissection must be continued until the lesion is completely detached (figure 10.1).

FIG. 10.1 — Steps of circumferential incision and traction placement in peroral super minimally invasive resection of early duodenal cancer. A. Early laterally spreading carcinoma under white-light endoscopy. B. Circumferential mucosal incision and partial submucosal dissection, with one end of the ‘8-shaped’ traction ring fixed to the lesion side. C. The opposite end of the ‘8-shaped’ ring secured to the anal-side opposite edge of the lesion using a tissue clip. D. Continued dissection of the lesion until complete detachment from the tissue surface.
(d) Hemostasis. After achieving hemostasis, the wound must be secured using the ‘8-shaped’ ring and tissue clips. First, one end of the ring is fixed to one side of the wound margin and then the other end is secured to the opposite side using tissue clips. Once the wound is reduced in size, it is closed routinely with additional tissue clips. Finally, the wound is sealed with hemostatic clips and a gastrointestinal decompression tube is positioned (figure 10.2).
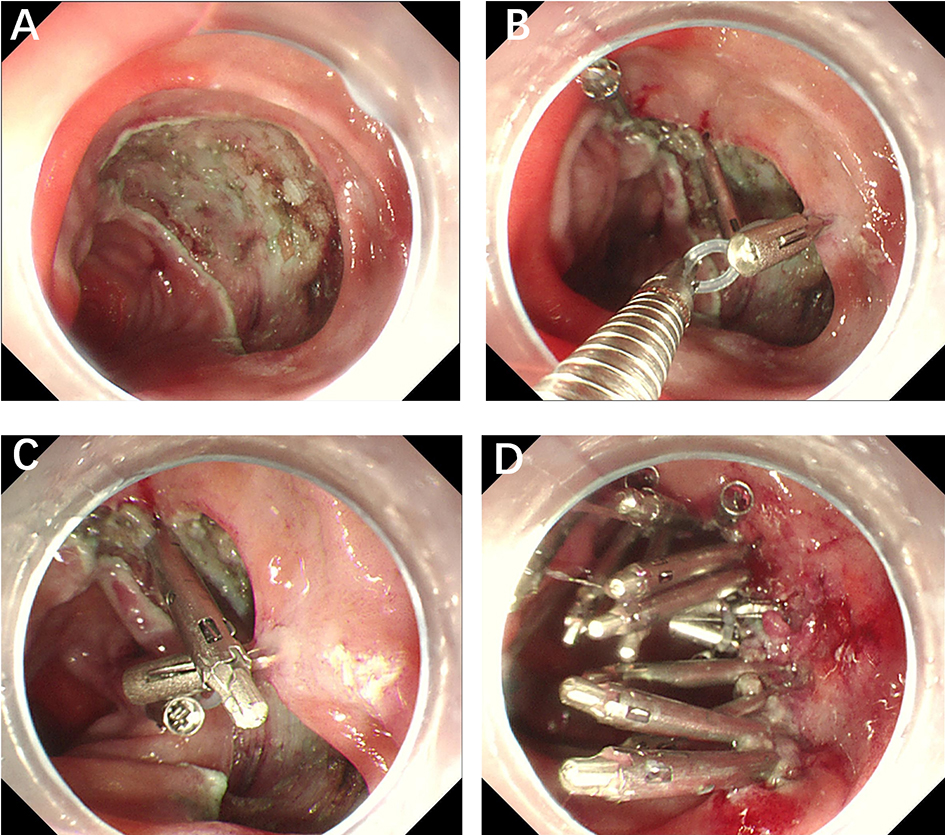

FIG. 10.2 — Steps of wound management in peroral super minimally invasive resection of early duodenal cancer. A. Post-resection wound. B, C. Placement of ‘8-shaped’ rings to reduce the size of the wound. D. Closure of the wound with tissue clips.
6. Postoperative management
Fasting and water restriction must be prolonged for three to five days. Acid-suppressing and enzyme-inhibiting medications must be administered as intravenous fluids. Prophylactic anti-infection therapy with third-generation cephalosporins and ornidazole must be provided for 48 h. The patient should switch gradually to oral intake: liquid diet → semi-liquid diet → regular diet. Proton pump inhibitors (PPIs) and gastric mucosal protective agents should be administered for one month postoperatively.
7. A classic case
Medical history: the patient, an elderly man, was admitted with a chief complaint of “duodenal and colonic polyps identified eight months ago.” Eight months before, the patient experienced intermittent abdominal bloating and discomfort. Gastroscopy revealed a laterally spreading tumor (LST) of 2 cm × 2.5 cm located on the opposite side of the major papilla in the descending duodenum. The lesion appeared whitish, granular, uneven, and well-demarcated, occupying approximately one-third of the lumen. The patient had a history of coronary artery disease, hypertension, and lung cancer. Physical examination upon admission revealed no abnormalities.
Diagnosis: laterally spreading polyp in the descending duodenum.
After comprehensive preoperative evaluation, nutritional optimization, and informed consent, the patient underwent a peroral super minimally invasive resection of the duodenal LST (Refer to video 10.1, Peroral super minimally invasive non-full-thickness resection of duodenal LST (See online resources)).
Surgical process: During the procedure, a laterally spreading lesion was observed on the opposite side of the major papilla, occupying approximately one-third of the lumen. A saline solution of methylene blue, epinephrine, and sodium hyaluronate was injected submucosally, achieving adequate elevation. The DualKnife was used to circumferentially incise the mucosal layer. With the aid of dental floss and an ‘8-shaped’ traction ring, a Triangle Knife and an IT Knife were used to separate the submucosal layer from the muscularis propria step by step. After complete lesion dissection, the wound was closed using ‘8-shaped’-ring-assisted tissue clips.
Hot-spot issues and research progress
The unique anatomical features of the duodenum—such as a narrow lumen, rich blood supply, limited visualization, Brunner’s gland hyperplasia, and biopsy-induced fibrosis—pose significant technical challenges for procedures like conventional endoscopic mucosal resection (cEMR) and endoscopic submucosal dissection (ESD). These factors often lead to poor submucosal lifting signs. Consequently, various gastrointestinal endoscopy societies recommend limiting duodenal ESD to endoscopists with extensive experience in ESD, and the indications for this procedure remain controversial.
Compared to EMR, ESD has higher rates of intraoperative and delayed perforation. The safety and local recurrence rates of duodenal early cancer treated with either technique remain uncertain, necessitating further prospective studies. As a minimally invasive procedure, ESD offers curative potential through en bloc resection of early gastrointestinal tumors, making it an attractive option for tumor removal.
Challenges in duodenal ESD
Anatomical challenges: the transition from the stomach to the duodenum presents a sharp angle, which impairs endoscopic control and leads to vertical entry into the muscularis propria during ESD.
Histological challenges: the presence of Brunner’s glands and a thin muscularis propria results in suboptimal submucosal lifting during injection and increases the risk of full-thickness injury to the muscular layer.
These challenges contribute to a higher incidence of adverse events in duodenal ESD, including intraoperative complications, delayed bleeding, and perforation. Consequently, duodenal ESD should currently be limited to endoscopists with substantial experience in ESD at other sites.
ESD is the only endoscopic therapy capable of reliably achieving R0 resection of mucosal cancers and precancerous lesions of various shapes and sizes throughout the gastrointestinal tract. In contrast, EMR is a safe and effective technique for most non-ampullary duodenal adenomas. Current evidence suggests that ESD has a higher overall resection rate than EMR, facilitating accurate histological evaluation of resected specimens. Additionally, ESD achieves higher R0 resection rates and lower local recurrence rates compared to EMR. However, the rates of intraoperative and delayed perforation are also higher with ESD than with EMR.
As advancements in instrumentation and endoscopic techniques continue, the R0 resection rate and safety of ESD for early duodenal cancer are improving. Nevertheless, prospective studies are needed to further evaluate the safety, efficacy, and indications of duodenal ESD compared to EMR.
Innovative techniques to enhance duodenal ESD
The fundamental techniques of duodenal ESD are similar to those used for other gastrointestinal sites. However, several specialized methods have been developed to address the unique challenges of duodenal ESD:
Water pressure method: filling the duodenal lumen with a saline solution improves submucosal visualization by opening the mucosal flap and leveraging water jet functions. A prospective study demonstrated that this approach, combined with ESD knives equipped with water jet capabilities, significantly reduces operative time.
Pocket creation method (PCM): PCM is an alternative technique for duodenal ESD that stabilizes the endoscope, even in difficult locations such as the duodenal angle. Studies evaluating P CM for duodenal ESD have shown that it is associated with higher en bloc resection rates, faster dissection speeds, and lower perforation rates.
Double-balloon endoscopy: this method addresses anatomical challenges such as excessive gastric and duodenal angles and the vertical entry of the muscularis propria. By stabilizing the endoscope’s tip, double-balloon endoscopy enables precise and safe duodenal ESD.
Endoscopists should employ techniques to minimize adverse events during procedures. These may include piecemeal resection, defect closure methods, non-contact hemostasis, and other emerging technologies, with specific approaches tailored to individual cases. Evidence supporting the routine prophylactic closure of defects with clips following duodenal EMR is limited. Non-contact hemostatic agents have been successfully used to minimize post-EMR bleeding in the duodenum, but the supporting data remain insufficient.
ESD has become the first-line treatment for superficial esophageal and gastric tumors. This technique allows for en bloc resection of lesions of almost any size, accompanied by low rates of adverse events and recurrence. Traction strategies are increasingly utilized to enhance ESD, demonstrating significant benefits in esophageal, colorectal, and selected gastric ESD procedures. After completing mucosal incision and submucosal layer trimming, various traction techniques have been developed to elevate the mucosal flap and provide better exposure of the submucosal space. This improves visualization and enables precise identification of the dissection plane and vascular structures. While gravity, positional changes, and distal attachment caps may suffice in some cases, these methods may be inadequate in certain anatomical locations.
Traction techniques
Non-device-assisted traction: gravity-assisted ESD; tunneling techniques; tunnel-bridge methods; pocket creation ESD; tissue traction.
Device-assisted external traction: clip-with-line techniques; clip-snare methods; external forceps; dual endoscope techniques; dual-channel endoscopes; device-assisted internal traction: dual-clip traction; intraluminal traction wire; magnetic bead traction.
10.2 Peroral Super Minimally Invasive Resection of Duodenal Papillary Adenomas
10.2.1 Overview
Most duodenal papillary adenomas are asymptomatic and are often identified during routine upper gastrointestinal endoscopy. When symptoms do occur, they may include jaundice (16.6%), pain (14.4%), pancreatitis (4.1%), and cholangitis (1%). Furthermore, up to 38% of cases are associated with common bile duct stones. Tumors of the duodenal papilla are rare in the general population (0.04%–0.12%), but with the increasing use of upper gastrointestinal endoscopy and endoscopic retrograde cholangiopancreatography (ERCP), these tumors are now more frequently detected during asymptomatic stages. Most papillary tumors are adenomatous and are believed to follow an adenoma-carcinoma sequence, necessitating their removal. Traditionally, the primary treatment for duodenal papillary adenomas has been pancreaticoduodenectomy, which is associated with significant trauma and high mortality rates. Endoscopic papillectomy was first reported in Japan in 1983, but early iterations of the procedure had a complication rate exceeding 60%, particularly for postoperative bleeding. Professor Linghu Enqiang significantly improved this technique by introducing methods to seal the surgical wound and by placing pancreatic or bile duct stents to separate bile and pancreatic secretions in the papillary region. These advancements markedly reduced the incidence of complications. In 2020, Professor Linghu reported that the overall adverse event rate (including bleeding and postoperative pancreatitis) in the group treated with the modified technique was significantly lower than in the conventional group (13.0% vs. 41.5%, P = 0.018). Endoscopic papillectomy has since become the preferred treatment for duodenal papillary adenomas. Compared to open surgery, it is associated with higher success rates, lower recurrence rates, and fewer adverse events, making it widely accepted as a safe and effective therapeutic approach.
1. Diagnosis of duodenal papillary adenomas
Lateral spreading variants are defined when the extramural portion of the duodenal wall exceeds the size of the adenoma or when an ampullary tumor demonstrates lateral spread greater than 10 mm beyond the ampullary crest. Endoscopic features suggestive of benign ampullary tumors typically include regular surface or margins, a soft appearance, and mobility. Endoscopic biopsy and histological examination can further enhance the diagnostic accuracy for duodenal adenomas.
Endoscopic ultrasound (EUS), magnetic resonance cholangiopancreatography (MRCP), contrast-enhanced multidetector computed tomography (MDCT), and detailed endoscopic retrograde cholangiopancreatography (ERCP) are commonly employed prior to ampullary adenoma resection. These imaging modalities are used to evaluate the presence of submucosal invasion (SMI), lymph node metastasis, intraductal involvement, and other pancreatic anatomical variations.
2. Indications and contraindications
(1) Indications. (i) Low-grade intraepithelial neoplasia. (ii) High-grade intraepithelial neoplasia without lymphovascular invasion on biopsy. (iii) Intraductal extension <10 mm. (iv) Lateral spreading tumors of the papilla, regardless of size. (v) Small papillary adenomas.
(2) Contraindications (i) Lymphovascular invasion detected on biopsy. (ii) Malignant lesions confirmed as cancer on pathological diagnosis. (iii) Intraductal extension >10 mm. (iv) Evidence of invasive disease during evaluation, such as involvement of the bile or pancreatic ducts.
3. Preoperative preparation
Patients should undergo thorough medical evaluation and provide fully informed consent regarding the procedure and its alternatives, including surgical and observational approaches. Observation may be suitable for elderly patients or those with significant comorbidities, as these lesions typically progress to overtly symptomatic cancer at a slow rate and may not be ideal candidates for endoscopic papillary resection (EPR).
4. Surgical techniques and procedures
(1) Anesthesia and positioning. General intravenous anesthesia was administered, and the patient was placed in the prone position.
(2) Surgical steps
(a) Submucosal injection for lesion elevation or non-injection.
(b) The widely adopted snaring technique for en bloc resection was employed, consisting of the following steps:
(c) Fully or nearly fully open the snare, with the proximal end of the snare partially within the working channel.
(d) Anchor the snare tip at the proximal end of the lesion, slightly aligning it to the right of the long axis of the funnel. This allows for better control of the snare and minimizes slippage.
(e) Gradually advance the snare from the working channel and position it to completely capture the ampullary adenoma.
(f) Gently push the duodenoscope distally and slowly open the elevator, applying slight pressure to maintain the snare tip in a compressed position above the duodenal wall.
(g) Slowly close the snare while keeping it parallel to the duodenal wall. When fully closed, the ampullary lesion should move independently from the duodenal wall. To confirm complete capture of the lesion, move the snare back and forth with the elevator open.
(h) Once these steps are completed, and invasive disease or deeper tissue involvement is excluded, perform electrosurgical resection by tightening the snare with the elevator open.
(i) Super-selective cannulation of the bile duct or pancreatic duct was performed using a guidewire, which was left in place within the duct.
(j) Electrocautery forceps and argon plasma coagulation were employed for hemostasis. Metal clips were used to close the wound.
(k) Stents were implanted in the bile duct or pancreatic duct as necessary.
(l) Fibrin glue was sprayed over the closed wound surface for additional reinforcement.
(m) Radiographic imaging was conducted to confirm gas evacuation from the stomach and proper stent placement.
(n) The excised specimen was sent for pathological analysis. Refer to video 10.2, Peroral super minimally invasive resection of a duodenal ampullary adenoma (See online resources).
5. Hot-spot issues and research advances in the consensus
Ampullary tumors are rare, with an annual incidence of less than 1/100 000, accounting for only 0.6%–0.8% of gastrointestinal cancers. The male-to-female ratio is approximately 1.5. Although the incidence of this disease in younger individuals (<45) has increased over the past 20 years, the trend remains more stable in the elderly population. Most benign or malignant ampullary tumors are sporadic; however, if diagnosed at a younger age, a genetic predisposition must be considered. Precursor lesions may originate from pancreatic-type mucosa or pancreatobiliary-type ampullary mucosa, constituting the two primary histological subgroups. The intestinal type evolves through the well-documented adenoma-carcinoma sequence, whereas the pancreatobiliary type arises from pancreatic ductal intraepithelial neoplasia precursors.
Papillary tumors may be confined to the ampullary mound, but in 6.9%–43.8% of cases, they exhibit extra-ampullary components or intraductal manifestations. Lateral spreading lesions (LST) of the papilla may also have extra-ampullary components and share features with superficial non-ampullary duodenal tumors. Currently, no ideal endoscopic classification system exists for ampullary tumors.
Endoscopic biopsy combined with routine hematoxylin and eosin staining is considered essential for diagnosing ampullary tumors. However, the reported diagnostic accuracy of preoperative biopsies ranges from 38.3% to 85%, with histological underestimation rates reaching up to 30% and overestimation rates approximately 15%. Thus, endoscopic biopsy alone is insufficient to determine the most appropriate treatment, potentially leading to undertreatment and increased risks.
Regarding the treatment of ampullary tumors, well-designed prospective studies comparing the efficacy of endoscopic papillectomy (EP) and surgical treatment (transduodenal ampullectomy or pancreatoduodenectomy) are still lacking. With the growing popularity of EP, surgical treatments have become less common. Nevertheless, transduodenal ampullectomy remains an acceptable choice in the following scenarios where EP may not be suitable: intraductal involvement; technical inability to perform EP (e.g., G. diverticulum, tumor size > 4 cm); incomplete resection with positive margins after EP; local recurrence not amenable to endoscopic treatment. It is crucial to emphasize that the experience of the operator is pivotal in ensuring favorable outcomes and minimizing morbidity for both EP and transduodenal ampullectomy. Generally, ampullary adenomas smaller than 3 cm can be treated with en bloc endoscopic resection, while tumors larger than 4 cm are better suited for surgical intervention. For ampullary tumors measuring 3–4 cm, a case-by-case evaluation is recommended. Lateral spreading tumors involving the duodenal papilla (LST-p), which extend ≥10 mm beyond the ampullary mound or exhibit extra-ampullary components involving the duodenal wall, are typically larger than papillary adenomas. The European Society of Gastrointestinal Endoscopy (ESGE) recommends treating such cases with endoscopic resection, albeit with consideration of higher risks for intraoperative and delayed bleeding.
Post-endoscopic papillectomy (EP) is most commonly associated with post-procedural pancreatitis, with an incidence rate of 11.9% (95% CI, 10.4–13.6; IIII
Some researchers advocate for submucosal injection prior to the resection of ampullary tumors to better delineate the lateral extent of the lesion. If the ‘non-lifting sign’ is observed, indicating deep infiltration, endoscopic resection is deemed unsuitable. However, other researchers advise against submucosal injection for the following reasons:
The central location of ampullary tumors, constrained by the bile and pancreatic ducts, may prevent adequate lifting;
The injection could create a ‘dome effect’, complicating effective snare placement for en bloc resection.
Reports suggest that injection may increase the risk of post-resection pancreatitis. Currently, there is little clinical data to confirm or refute these claims, leaving this aspect of the procedure under debate.
Despite the lack of clear evidence from controlled trials, many researchers recommend performing cholangiography and pancreatography before EP to rule out intraductal extension exceeding 10 mm. However, limited data exists on whether dual-duct sphincterotomy affects cannulation rates, pancreatic duct stent placement, or outcomes of subsequent ampullary tumor resections.
There is also no consensus regarding the optimal electrosurgical current and power settings for EP. Some authors advocate the use of pure cutting current to avoid edema associated with coagulation modes. However, pure cutting current may elevate the risk of bleeding. Others prefer mixed current or alternating cut/coagulation modes for better procedural control and hemostasis.
10.3 Peroral Super Minimally Invasive Resection of Subepithelial Duodenal Tumors
1. Overview
With the increasing availability of digestive endoscopy and the development of endoscopic ultrasonography (EUS), the detection rate of subepithelial duodenal tumors has significantly risen. Most of these lesions are benign and cannot be effectively biopsied, leading to the conventional strategy of regular follow-up. This approach not only imposes an economic burden on patients but also adds psychological stress. With advancements in endoscopic techniques, peroral super minimally invasive resection has emerged as a novel treatment option for subepithelial duodenal tumors.
2. Diagnosis of subepithelial duodenal tumors
(1) Conventional white-light endoscopy serves as the initial step in detecting subepithelial duodenal tumors. It provides information on the lesion’s location, size, mobility, mucosal coloration, morphology, and the presence of erosion or bleeding. However, it cannot determine the nature or origin of the lesion, particularly for subepithelial tumors with exophytic growth patterns, which cannot be diagnosed solely through conventional endoscopy.
(2) EUS is the frontline diagnostic method for subepithelial duodenal tumors. It provides detailed information about the lesion’s origin, intramural or extramural location, size, shape, echogenicity, vascularity, and associated lymphadenopathy. These characteristics enable the evaluation of histological features and even malignant potential. EUS has a sensitivity of 92% in differentiating extraluminal compression from true intraluminal lesions.
(3) CT and MRI are recommended for staging, treatment planning, and prognosis evaluation, particularly in cases of suspected malignancy or larger tumors. These imaging modalities help assess the tumor’s characteristics and its potential for malignancy.
(4) The necessity and utility of histopathological examination for subepithelial duodenal tumors depend on the clinical context. Pathological evaluation aids in confirming the diagnosis, guiding treatment strategies, and assessing malignancy risk, although the effectiveness varies based on tumor type and size.
3. Indications and contraindications
(1) Indications: ① Tumors originating from the submucosa and muscularis propria of the duodenum. ② Patients who refuse or are unsuitable for surgical treatment.
(2) Contraindications
(a) Lesions with confirmed lymph node or distant metastases.
(b) For certain lesions with confirmed lymph node or distant metastases (subepithelial lesions, SELs), large biopsy samples required for pathological examination may be considered a relative contraindication.
(c) Patients who, following detailed preoperative evaluation, are determined to be in poor general condition and unable to tolerate endoscopic surgery.
Surgical procedures and techniques
(1) Anesthesia methods and considerations. All patients undergo endotracheal intubation for general anesthesia in the left lateral position. Care should be taken to maintain body temperature, adjust room temperature, and regulate infusion fluid temperature to prevent pressure sores. Patients are instructed to swallow defoaming agents, and 4% lidocaine is used for topical anesthesia of the pharynx. Oxygen is administered via a nasal cannula at 1 to 2 l/min, followed by intravenous anesthesia. For patients with active intestinal peristalsis, antispasmodic agents may be used.
(2) Surgical procedure (figures 10.3–10.5)
(a) Super minimally invasive snare resection:
The lesion is identified under white light, and its submucosal location is confirmed via endoscopic ultrasonography. A snare is used to encircle the lesion, followed by electrocautery and resection. The surgical site is inspected for bleeding or perforation. Residual blood vessels at the site are coagulated using electrocautery, and tissue clips are applied to seal the wound.
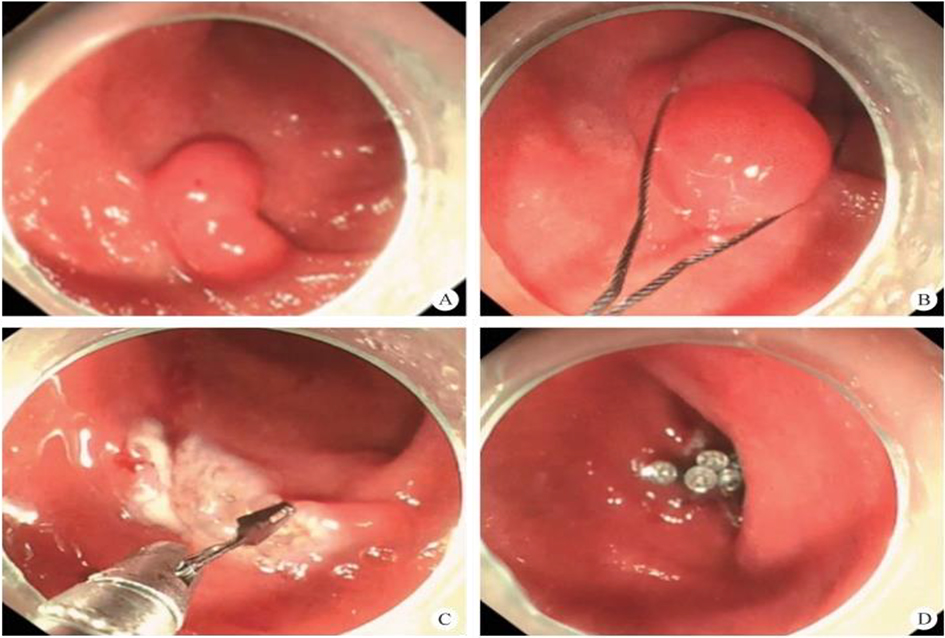

FIG. 10.3 — A. Submucosal lesion of the duodenum. B. Snaring the lesion with a snare device. C. Clean surgical site after complete resection. D. Closure of the surgical site with multiple tissue clips.
(b) Super minimally invasive tumor enucleation
1) Lesion identification and marking: duodenal subepithelial tumors are identified endoscopically. Endoscopic ultrasonography is used to assess the depth of the lesion and determine the presence or absence of lymph node metastases. The lesion is marked circumferentially using a DualKnife or argon plasma coagulation (APC) around its periphery.
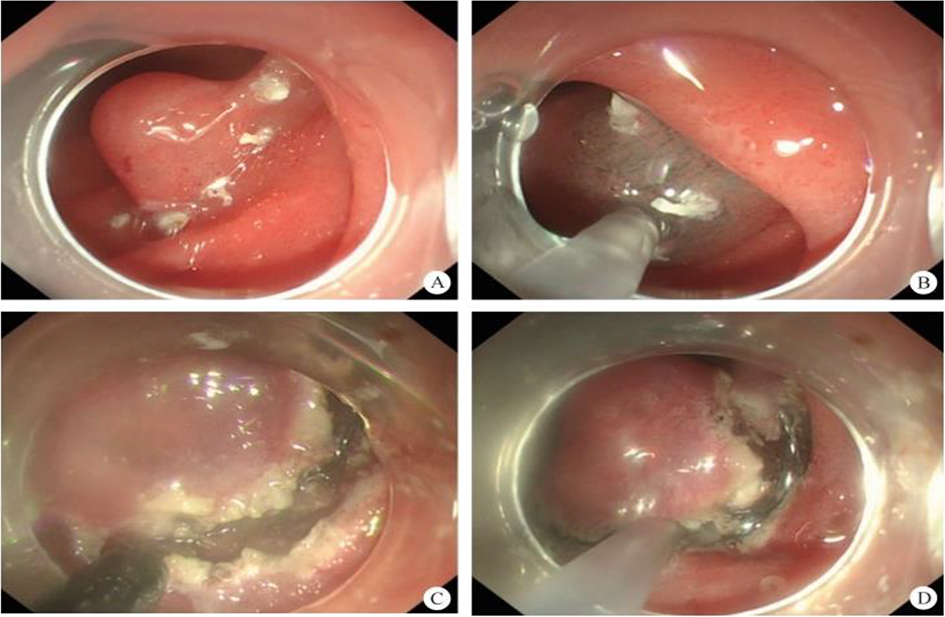
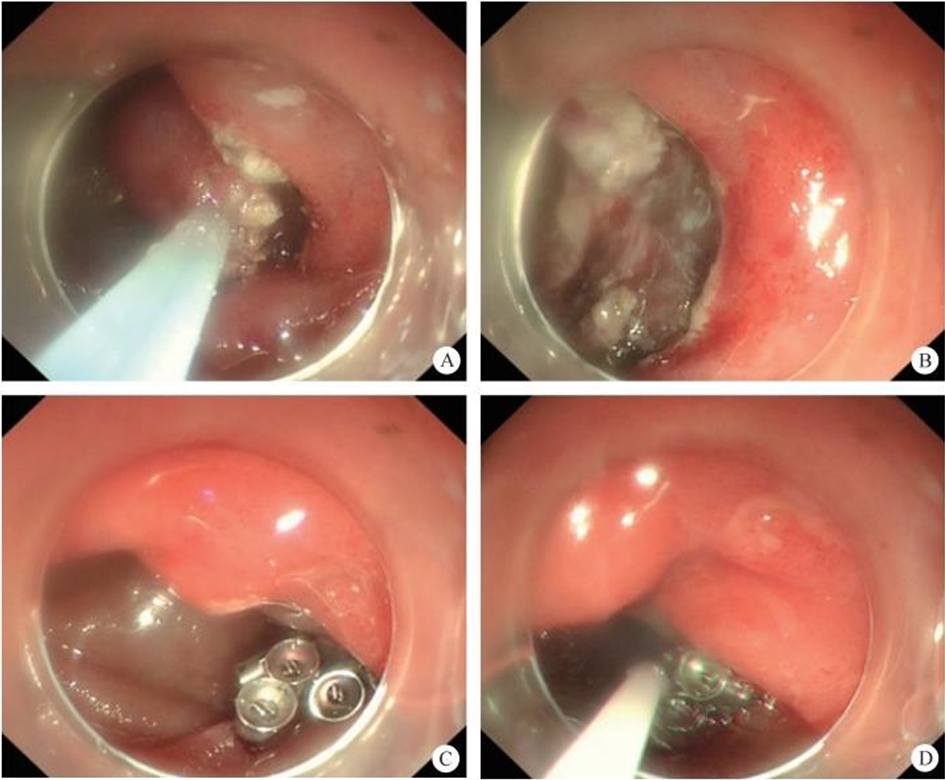

FIG. 10.4 — Steps of circumferential incision during the peroral super minimally invasive resection of a duodenal subepithelial tumor. A. Marking the lesion. B. Submucosal injection. C. Circumferential incision of the mucosa. D. Assistance with a snare device.

FIG. 10.5 — Steps of tumor resection and wound management during peroral super minimally invasive resection of a duodenal subepithelial tumor. A. Snaring and resecting the lesion. B. Confirmation of a tumor-free wound surface. C. Closure of the wound with tissue clips. D. Application of biological glue to seal the wound.
2) Submucosal injection and lesion dissection: a mixture of epinephrine, fructose solution, and methylene blue is injected into the submucosa to create an elevated mucosal state. The mucosal and submucosal layers are incised at the marked points to fully expose the muscularis propria and the tumor. The tumor is dissected using an IT knife, with the area flushed with epinephrine saline solution to maintain a clear field. Hemostasis is achieved using electrocautery forceps as needed.
3) Optional snare resection and wound management: once the tumor is completely dissected, the lesion base is treated by electrocautery using a snare device if necessary. Postoperatively, the wound is sealed entirely with metallic stents.
4. Classic cases
Case 1: subepithelial tumor of the descending duodenum treated with peroral super minimally invasive resection
Medical history: the patient, a middle-aged man, presented with “upper abdominal discomfort and melena for two weeks”. Gastroscopy revealed a submucosal protruding lesion in the descending duodenum. The patient had a history of hepatitis A, hypertension, and colon cancer. Physical examination upon admission showed no abnormalities.
Diagnosis: subepithelial tumor of the duodenum.
Following thorough preoperative evaluation, nutritional improvement, and signing of the informed surgical consent form, the patient underwent a peroral super minimally invasive resection of a duodenal subepithelial tumor (Refer to video 10.3, Peroral super minimally invasive resection of a subepithelial tumor in the descending duodenum-1 (See online resources)).
Surgical procedure: a dumbbell-shaped submucosal protruding lesion covered with normal, slightly translucent mucosa was observed in the descending duodenum. Mini-probe endoscopic ultrasonography (EUS) revealed a hypoechoic lesion originating from the submucosal layer, with an intact muscularis propria. The lesion’s diameter was approximately 4.6 mm in cross-section. The lesion was resected using a snare device with electrocautery. Multiple tissue clips were used to close the wound, and the site was covered with a layer of biological glue.
Case 2: subepithelial tumor of the duodenum treated with peroral super minimally invasive resection
Medical history: the patient, a middle-aged man, presented with “submucosal protrusion in the duodenum and multiple colonic polyps detected 25 days ago”. Gastroscopy revealed a submucosal protrusion approximately 1.5 cm in size in the descending duodenum. The patient had a history of papillary thyroid carcinoma. Physical examination upon admission showed no abnormalities.
Diagnosis: submucosal protrusion of the duodenum.
Following thorough preoperative evaluation, nutritional improvement, and signing of informed surgical consent form, the patient underwent a peroral super minimally invasive resection of the duodenal subepithelial tumor (Refer to video 10.4, Peroral super minimally invasive resection of a subepithelial tumor in the descending duodenum-2 (See online resources)).
Surgical procedure: a submucosal protrusion approximately 1.5 cm in size, covered with normal mucosa, was observed in the descending duodenum. Mini-probe EUS showed a dumbbell-shaped, hypoechoic lesion located in the submucosal layer, with an intact muscularis propria. The lesion’s cross-sectional dimensions were approximately 0.9 cm × 0.5 cm. The lesion area was marked circumferentially with APC. A methylene blue/epinephrine/normal saline solution was injected into the submucosa, achieving good elevation. The mucosal layer was incised circumferentially using a DualKnife, and the lesion was completely resected with a snare device using electrocautery. Hemostasis was achieved with hemostatic forceps. Multiple tissue clips and hemostatic clips were used to close the wound, and a layer of biological glue was applied to cover the site.
5. Key issues and research progress in consensus
The diagnosis of duodenal subepithelial tumors (SETs) primarily relies on direct endoscopic visualization, endoscopic ultrasonography (EUS), EUS-guided fine-needle biopsy (EUS–FNB), and endoscopic resection for evaluation and treatment guidance. Complete resection of duodenal SETs is recommended and should be performed in an inpatient hospital setting. Considering the high risk of residual or recurrent tumors, close endoscopic follow-up is advised after endoscopic resection. Additionally, due to an increased risk of colorectal neoplasms compared to the general population, all patients with duodenal SETs are advised to undergo colonoscopy screening.
Duodenal SETs are protruding lesions originating from the muscularis mucosae, submucosa, or muscularis propria, or they may represent extraluminal lesions. These types of lesions are often detected during routine endoscopic examinations, most commonly as incidental findings. Submucosal tumors can appear as protrusions of the organ wall with intact mucosa, comparable to the adjacent mucosa. Most patients with these lesions are asymptomatic, although in rare cases, they may experience pain, obstruction, or gastrointestinal bleeding. In terms of coloration, the overlying mucosa of most lesions appears normal; however, some lesions may present a yellowish, whitish, or reddish appearance. The consistency of duodenal SETs can be assessed using closed biopsy forceps as a probing tool, with the ‘pillow sign’ being 98% specific for diagnosing lipomas. Larger lesion size, growth during follow-up, or the presence of (hemorrhagic) ulcers may indicate a risk of malignant transformation.
There are no comparative studies on the use of magnifying endoscopy or chromoendoscopy in the endoscopic diagnosis of duodenal SETs, likely due to the limited application of these techniques in lesions typically covered by normal mucosa.
EUS is the first-line diagnostic tool for SETs, providing information on lesion origin, intramural or extramural location, size and shape, echogenicity, vascular involvement, and associated lymph node abnormalities. These characteristics allow for histological evaluation and assessment of malignancy potential. EUS has a 92% sensitivity in distinguishing extraluminal compression from intraluminal lesions. Artificial intelligence (AI) has shown remarkable performance in medical imaging diagnostics and holds potential value in aiding the diagnosis of SETs. AI demonstrates higher diagnostic accuracy than endoscopy experts, but its clinical application and associated ethical issues require further investigation. Lesion size and vascular involvement are two additional features predictive of the malignancy potential of SETs. The overall sensitivity and specificity of EUS in predicting SET malignancy potential are 64% and 80%, respectively. Interpretation of EUS images is operator-dependent, emphasizing the need for advanced imaging or tissue diagnostics to enhance EUS accuracy.
When the diagnosis is established, benign lesions such as leiomyomas, lipomas, ectopic pancreas, granular cell tumors, schwannomas, and glomus tumors do not require monitoring, as the risk of malignancy or complications is anecdotal. There is no evidence supporting the benefit of surveillance in these patients. Conversely, confirmed malignant conditions, such as lymphoma and metastatic lesions, require individualized treatment. For neuroendocrine neoplasms (NENs), most patients benefit from treatment (endoscopic or surgical) rather than monitoring. The only exception is type-1 gastric NENs smaller than 10 mm, where the risk of progression is very low, making surveillance a viable option.
The primary indication for super minimally invasive resection of duodenal subepithelial tumors is non-ampullary, non-functional duodenal neuroendocrine neoplasms (d-NENs) measuring <20 mm. Ampullary d-NENs and functional d-NENs exhibit more aggressive pathophysiology, with a higher incidence of synchronous lymph node and liver metastases, and are therefore considered suitable for tumor surgery. Non-functional, non-ampullary d-NENs in stage G1 and measuring <20 mm have a much lower risk of metastasis, particularly those <10 mm, making them appropriate candidates for local resection. However, for lesions >20 mm, the risk of metastasis increases, and tumor surgery becomes the recommended treatment.
Significant differences exist in the size and depth of invasion of lesions treated with various techniques. Considerable heterogeneity is also noted across studies, with various methods employed, including endoscopic mucosal resection (EMR) (commonly with band ligation, circumferential incision, or traction), endoscopic submucosal dissection (ESD), or endoscopic full-thickness resection (EFTR). ESD generally demonstrates a higher R0 resection rate compared to EMR but is associated with greater complexity and a higher risk of perforation. Due to the higher risks of malignant transformation and metastasis, current guidelines do not recommend EMR for duodenal gastrointestinal stromal tumors (GISTs). However, EFTR has shown favorable outcomes in a study involving 32 patients, with en bloc and R0 resection rates of 100%. Further research is needed to determine the role of extended full-thickness resection devices and whether local resection could be expanded to specific subgroups of duodenal GISTs.
Endoscopic submucosal excavation (ESE) is a minimally invasive alternative to surgical resection. The standard steps of ESE typically include thermal marking to outline the target lesion, submucosal injection, lesion resection using tools such as a hook knife, insulated-tip knife, or DualKnife, and closure of the mucosal incision with clips.
While submucosal tunneling endoscopic resection (STER) and ESE are considered safe and feasible techniques for removing submucosal tumors originating from the muscularis propria, the lack of robust data and inconsistencies in published reports make it difficult to determine the superior approach. Both techniques appear to be effective and safe, with no differences in complete resection rates, en bloc resection rates, recurrence rates, or overall adverse event rates (including bleeding and perforation). Hospital stays are also similar, with endoscopic treatment shortening hospitalization compared to traditional surgical resection.
However, ESE has the advantage of shorter procedural time compared to STER. STER requires a longer execution time, likely due to the need for tunneling through anatomical layers until the lesion is exposed. In contrast, ESE involves dissection at the corresponding organ wall layer directly over the lesion. Despite these findings, all studies to date are retrospective, and further high-quality data are needed to validate these conclusions.
10.4 Peroral Super Minimally Invasive Resection of Duodenal Polyps
10.4.1 Overview
Duodenal polyps constitute a small proportion of gastrointestinal tumors and have a relatively low incidence. Approximately 40% of cases may be associated with hereditary conditions (e.g., familial adenomatous polyposis [FAP]), while the remaining cases are sporadic. In recent years, the incidence of duodenal polyps has been increasing annually due to the rising frequency of endoscopic examinations in the general population and improvements in endoscopic technology. As duodenal polyps follow an adenoma-carcinoma sequence similar to that of colorectal adenocarcinoma and serve as precursors to duodenal cancer, active treatment via super minimally invasive resection is necessary.
10.4.2 Diagnosis of Duodenal Polyps
Most duodenal polyps are asymptomatic and lack a history of gastrocolorectal diseases. They are primarily discovered through endoscopic or imaging examinations. Common diagnostic methods include the following: routine white-light endoscopy provides information about the location, size, mobility, mucosal coloration, morphology, and signs of erosion or bleeding of the lesion. Endoscopic ultrasonography (EUS) further evaluates the characteristics of duodenal polyps by offering insights into their origin, intramural or extramural positioning, size and shape, echogenicity, vascular features, and associated lymph node abnormalities. These features facilitate the assessment of histological properties and even malignant potential. For polyps suspected of having malignant potential or those of larger size, computed tomography (CT) and magnetic resonance imaging (MRI) are recommended for staging, treatment planning, and prognostic evaluation. A histopathological biopsy is the gold standard for determining the nature of duodenal polyps and serves as a guide for subsequent treatment.
10.4.3 Indications and Contraindications
1. Indications
(1) Tumors suspected to have malignant potential based on preoperative evaluations or confirmed by histopathological biopsy, especially polyps with a preoperative assessment indicating a diameter ≤2 cm, a low risk of recurrence or metastasis, and a likelihood of complete resection.
(2) Polyps with a diameter >2 cm that are suspected to be low-risk duodenal polyps, provided that preoperative evaluations rule out lymph node or distant metastases. In such cases, endoscopic resection can be considered in specialized centers with mature endoscopic treatment techniques, performed by experienced endoscopists, ensuring the feasibility of complete tumor removal.
2. Contraindications
(1) Identify lesions with lymph nodes or distant metastases.
(2) After a detailed preoperative evaluation, it was determined that the patient was in poor general condition and could not tolerate endoscopic surgery.
10.4.4 Preoperative Preparation
(1) Gastrointestinal preparation: a liquid diet should be started one to two days before surgery, and fasting and water restriction observed for at least 6 h prior to anesthesia.
(2) Preoperative examinations: all necessary tests, including routine bloodwork, biochemical analysis, coagulation profile, electrocardiography (ECG), and echocardiography, among other relevant evaluations, must be completed.
(3) Medication discontinuation: anticoagulant medications must be discontinued as follows: aspirin (seven days prior), low-molecular-weight heparin (three days prior), heparin (6–12 h prior), and warfarin (three days prior).
10.4.5 Surgical Procedure and Techniques
(I) Endoscopic mucosal resection (EMR)
The procedure is performed with the patient in the left lateral decubitus position. Indigo carmine staining can be applied to delineate the boundaries of the lesion, and endoscopic ultrasonography is used to assess the depth of the lesion and the presence of lymph node metastases. The most commonly employed treatment methods are hot snare polypectomy (HSP) and conventional endoscopic mucosal resection (EMR) (figure 10.6).
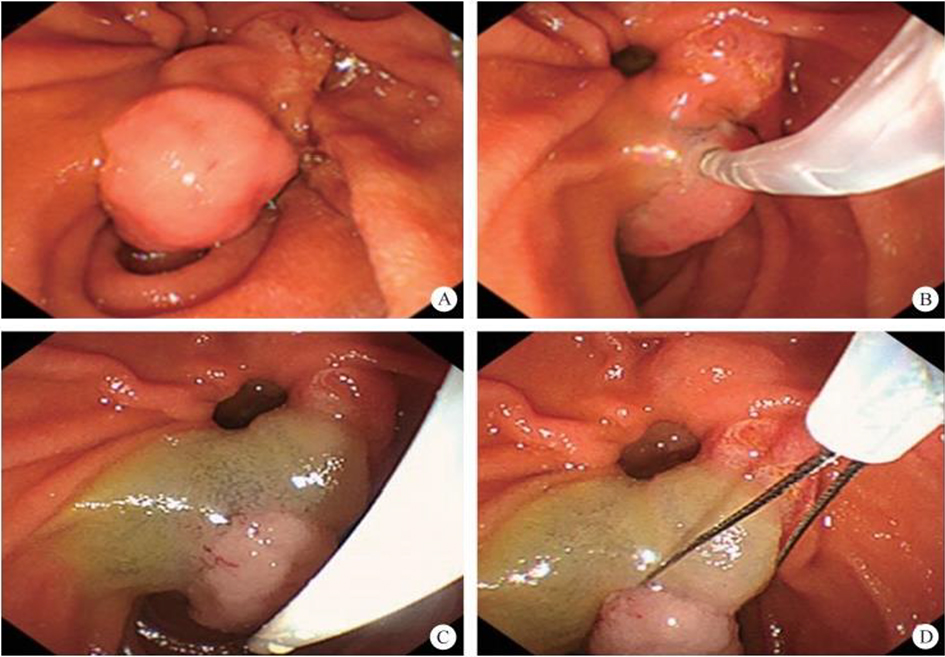

FIG. 10.6 — Steps of mucosal injection and snaring in peroral super minimally invasive resection of duodenal polyps. A. Periampullary lesion in the duodenum, adjacent to a diverticulum. B. Submucosal injection performed between the lesion and the ampulla. C. Adequate injection creates a safe separation distance between the lesion and the ampulla. D. The snare is repeatedly maneuvered to securely enclose and capture the lesion for complete resection.
Hot snare polypectomy (HSP): after the snare wire is looped around the duodenal polyp and positioned slightly above its base, the polyp is lifted and resected. For larger polyps, piecemeal resection is performed in stages using electrocautery. Conventional endoscopic mucosal resection (EMR): following submucosal injection, the snare is used to enclose and excise the lesion. Hemostasis is achieved at the resection site, and the defect is closed with endoclips (figure 10.7).
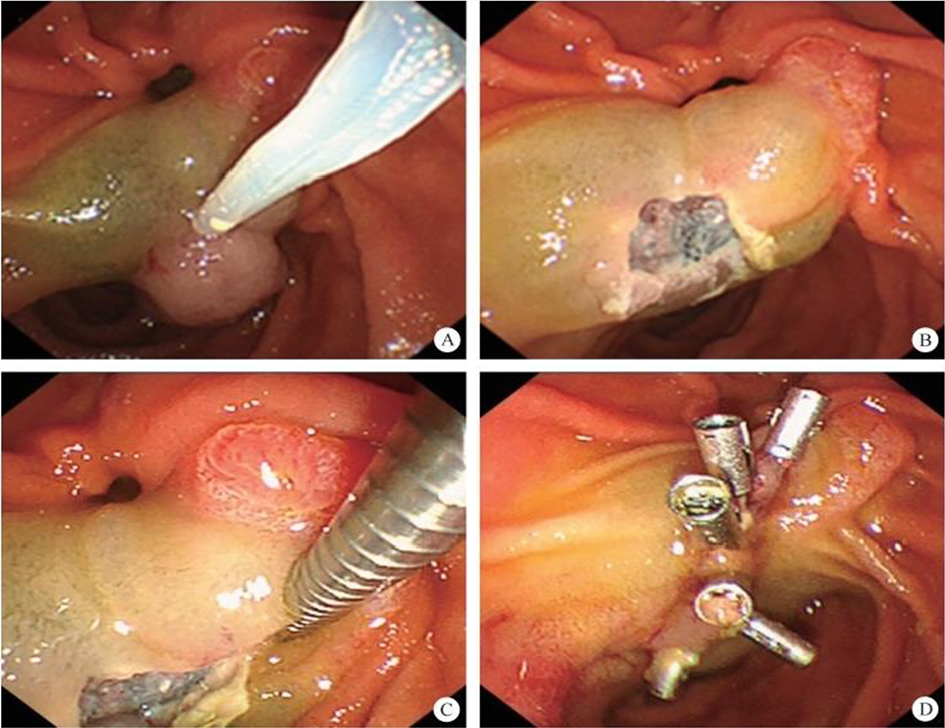

FIG. 10.7 — Steps of snare resection and defect management in peroral super minimally invasive resection of duodenal polyps. A. The lesion is completely encircled and the snare tightened. B. Post-resection, the surgical site is clean, with the ampulla unaffected. C. The defect is closed with endoclips, maintaining a safe distance from the ampulla. D. The defect is completely sealed.
Refer to video 10.5, Peroral super minimally invasive non-full-thickness resection of duodenal adenomas-1 (See online resources).
(II) Endoscopic nylon rope-assisted resection (figures 10.8–10.10)
(1) The procedure begins with the patient in the left lateral decubitus position. After identifying the polyp under white light, a nylon rope is looped around the base of the lesion via a dental floss attachment, and the lesion is dragged through the pylorus into the gastric antrum.
(2) Using dental floss for traction, the nylon rope’s tail is used to secure the lesion at the gastric antrum. Another nylon rope, also attached via dental floss, is looped around the base of the lesion, and two hemostatic clips are applied to close the stalk.
(3) The stalk is severed using an IT knife, and the blood vessels in the stalk are managed with hemostatic forceps. The lesion is completely excised.
(4) The residual wound is inspected for complications such as bleeding or perforation, and hemostasis is achieved. The wound is then sealed with endoclips.
10.4.6 Hot-Spot Issues and Research Progress in the Consensus
Duodenal polyps are the most common lesions of the duodenum, often incidentally discovered during upper gastrointestinal endoscopy, with a prevalence of 0.1%–0.8% among all endoscopic examinations. Duodenal polyps account for 10%–20% of these lesions and can occur sporadically or in association with familial adenomatous polyposis (FAP).
Endoscopy serves as the cornerstone for the management of duodenal polyps, primarily through the diagnostic and therapeutic approach of peroral super minimally invasive resection.
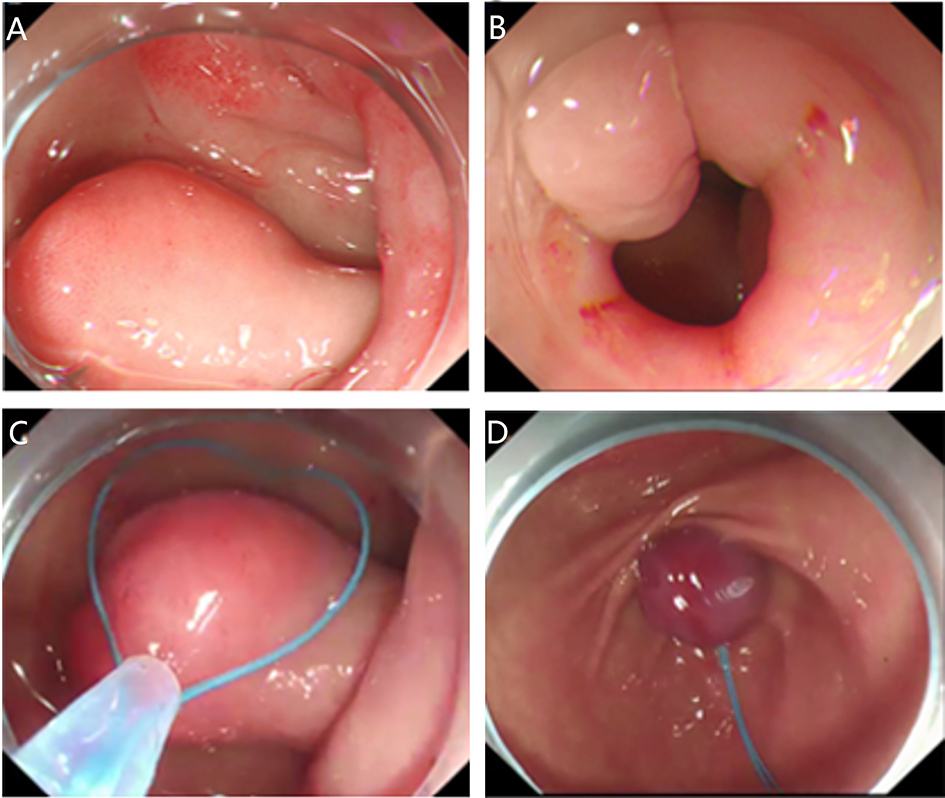
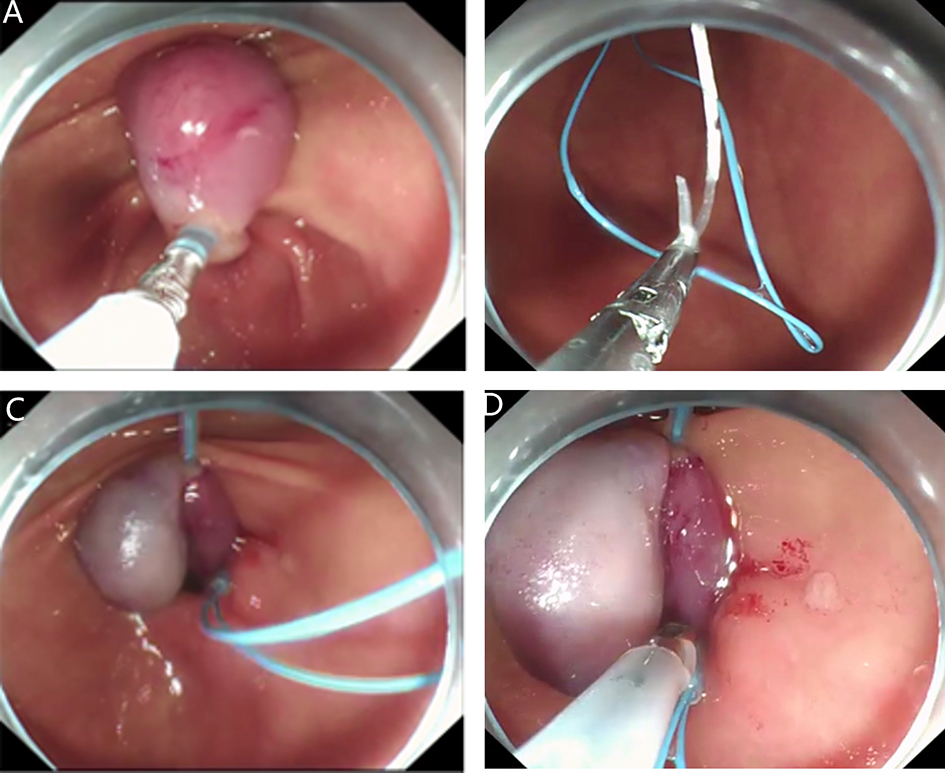
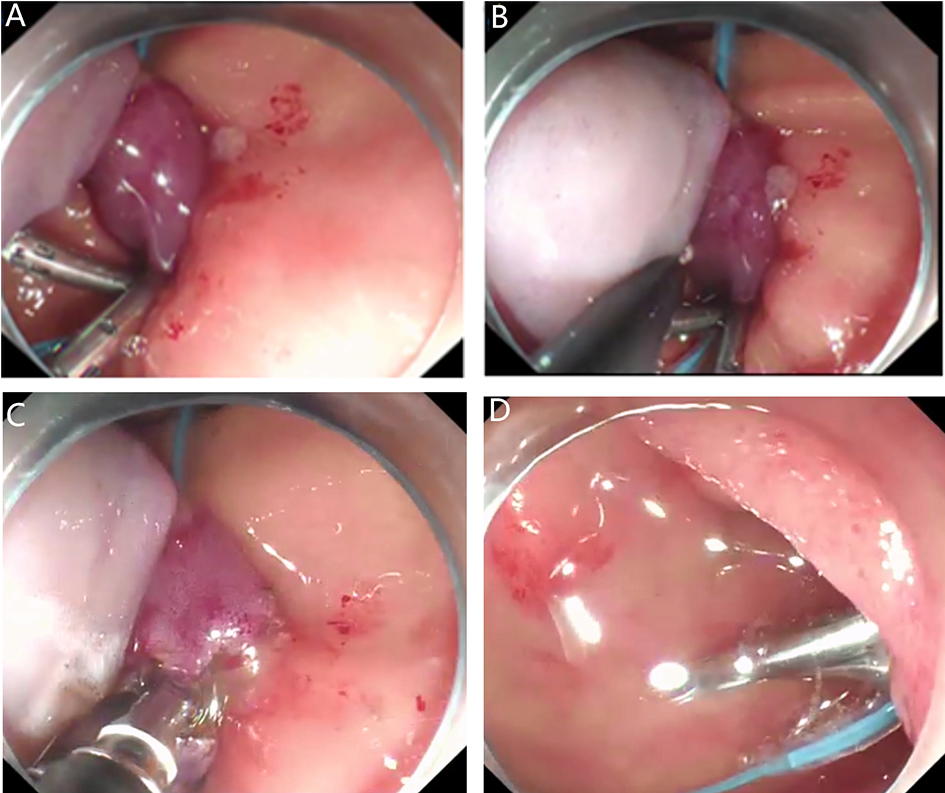

FIG. 10.8 — Steps of nylon rope-assisted ligation in endoscopic resection of duodenal polyps. A. Polypoid lesion with a broad stalk in the duodenum. B. The lesion exhibits a thick and elongated stalk. C. Nylon rope is prepared for ligation. D. The base of the lesion is secured with nylon rope ligation.

FIG. 10.9 — Steps of resection in nylon rope-assisted endoscopic removal of duodenal polyps. A. The head of the lesion is further ligated with a nylon rope. B. Tissue clips are applied in combination with tensioning the nylon rope for traction. C. The stalk of the lesion is exposed through traction. D. Additional tissue clips are applied to the base of the lesion for reinforced closure.

FIG. 10.10 — Steps of nylon rope-assisted endoscopic resection of duodenal polyps. A. Complete closure of the lesion’s base. B. The stalk is severed using an IT knife. C. Hemostasis is achieved via electrocautery. D. Post-resection view of the wound site.
Refer to video 10.6, Peroral super minimally invasive non-full-thickness resection of duodenal polyps-2 (See online resources).
Duodenal adenomas may occur in patients with hereditary predisposition syndromes to gastrointestinal tumors, predominantly familial adenomatous polyposis (FAP). These adenomas can be diagnosed through endoscopic screening. Sporadic non-ampullary duodenal adenomas (SNDA) comprise less than 10% of all duodenal polyps, while ampullary adenomas are even rarer. Villous polyps or duodenal polyps larger than 10 mm in diameter have a higher likelihood of malignant transformation and, therefore, require resection. Symptoms associated with duodenal polyps include iron-deficiency anemia, gastrointestinal bleeding, abdominal pain, and dyspeptic symptoms caused by duodenal obstruction. The three main types of duodenal adenomas are: sporadic non-ampullary duodenal adenomas (SNDA); ampullary adenomas; adenomas associated with familial adenomatous polyposis (FAP). SNDA has an incidence rate of 0.03%–6.9% among all duodenal polyps, and its prevalence has been increasing due to the rising frequency of endoscopic examinations. It is most commonly found in the second portion of the duodenum and carries up to an 85% risk of malignancy, necessitating active surveillance.
FAP is an autosomal dominant hereditary syndrome caused by germline pathogenic mutations in the APC tumor suppressor gene. Non-ampullary duodenal adenomas are commonly observed in FAP. A 20-year retrospective study found that FAP-associated adenomas accounted for 60% of duodenal adenomas. Patients with FAP-associated adenomas were diagnosed at a younger age (39.5 vs. 66.5 for sporadic adenomas), had longer follow-up periods (100 months vs. 43 months), and showed higher local recurrence rates (72.5% vs. 52%). The management of duodenal adenomas in FAP patients is based on regular endoscopic monitoring every one to five years, guided by the Spigelman scoring system and the presence of ampullary lesions. Endoscopic treatment aims at resecting the most prominent lesions, thereby gradually reducing the Spigelman score.
Non-ampullary duodenal adenomas are typically unique, sessile polyps located in the second portion of the duodenum. They most commonly exhibit Paris 0-Is morphology (observed in 17.5%–57% of cases) or 0-IIa morphology (30%–65.3% of cases). A thorough endoscopic evaluation requires not only conventional upper endoscopy but also side-viewing duodenoscopy to comprehensively inspect the left side of the descending duodenum and rule out adenoma extension to the major papilla. Cap-assisted endoscopy is particularly useful for detecting polyps hidden between duodenal folds. Endoscopic forceps biopsy may induce submucosal scarring and fibrosis, complicating subsequent endoscopic resection and increasing the risk of complications. Therefore, optical diagnostic techniques such as virtual chromoendoscopy and magnifying endoscopy are recommended for lesion assessment.
Several studies have demonstrated that endoscopic characteristics can help distinguish between low-grade and high-grade duodenal adenomas. Features indicative of high-grade dysplasia include: irregular, coarse, nodular mucosal patterns or a lack of identifiable patterns, reticular microvascular patterns, marginal distribution of white opaque substances (caused by lipid accumulation in the mucosa), lesion diameter >5 mm, pale reddish color and presence of depressed areas. These findings are strongly associated with duodenal adenomas exhibiting high-grade dysplasia, highlighting the importance of careful evaluation and tailored management.
References
[1] Tashima T., Jinushi R., Ishii N., et al. (2022) Effectiveness of clip-and-thread traction-assisted duodenal endoscopic submucosal dissection: a propensity score-matched study (with video), Gastrointestinal. Endosc. 95(5), 918–928.e3. 10.1016/j.gie.2021.12.024.
[2] Li A., Shen J. (2022) Current endoscopic diagnosis treatment strategy for superficial nonampullary duodenal tumours, Eur. J. Med. Res. 27(1), 290. 10.1186/s40001-022-00940-4.
[3] Kakushima N., Kanemoto H., Sasaki K., et al. (2015) Endoscopic and biopsy diagnoses of superficial, nonampullary, duodenal adenocarcinomas, World J. Gastroenterol. 21(18), 5560–5567. 10.3748/wjg.v21.i18.5560.
[4] Suwa T., Yoshida M., Ono H., et al. (2022) Issues and prospects of current endoscopic treatment strategy for superficial non-ampullary duodenal epithelial tumors, Curr. Oncol. (Toronto, Ont.) 29(10), 6816–6825. 10.3390/curroncol29100537.
[5] Vanbiervliet G., et al. (2021) Endoscopic management of ampullary tumors: European Society of Gastrointestinal Endoscopy (ESGE) guideline, Endoscopy 53, 429–448.
[6] Deprez P. H., et al. (2022) Endoscopic management of subepithelial lesions including neuroendocrine neoplasms: European Society of Gastrointestinal Endoscopy (ESGE) guideline, Endoscopy 54, 412–429.
[7] Pimentel-Nunes P., et al. (2022) Endoscopic submucosal dissection for superficial gastrointestinal lesions: European Society of Gastrointestinal Endoscopy (ESGE) Guideline – Update 2022, Endoscopy 54, 591–622.
[8] Muramoto T., et al. (2022) Superficial nonampullary duodenal epithelial tumor: a rare case with signet-ring cell carcinoma component diagnosed by magnifying endoscopy, Endoscopy 55, E90–E91.
[9] Akahoshi K., et al. (2020) Current challenge: endoscopic submucosal dissection of superficial non-ampullary duodenal epithelial tumors, Curr. Treat. Options Oncol. 21.
[10] Kobara H., et al. (2022) Clinical and technical outcomes of endoscopic closure of postendoscopic submucosal dissection defects: literature review over one decade, Dig. Endosc. 35, 216–231.
[11] Al Ghamdi S. S., Ngamruengphong S. (2023) Endoscopic submucosal dissection in the stomach and duodenum, Gastrointest. Endosc. Clin. North Am. 33, 67–81.
[12] Nunes F. G., et al. (2024) Conventional versus traction-assisted endoscopic submucosal dissection for esophageal, gastric, and colorectal neoplasms: a systematic review and meta-analysis of randomized controlled trials.
[13] Khan S., et al. (2022) Advancing endoscopic traction techniques in endoscopic submucosal dissection, Front. Oncol. 12.
[14] Nagata M. (2023) Device-assisted traction methods in colorectal endoscopic submucosal dissection and options for difficult cases, 15, 265–272.
[15] Libânio D., et al. (2023) Endoscopic submucosal dissection techniques and technology: European Society of Gastrointestinal Endoscopy (ESGE) technical review, Endoscopy 55, 361–389.
[16] Klein A., et al. (2015) Endoscopic resection of advanced and laterally spreading duodenal papillary tumors, Dig. Endosc. 28, 121–130.
[17] de Campos S. T., Bruno M. J. (2022) Endoscopic papillectomy, Gastrointest. Endosc. Clin. North Am. 32, 545–562.
[18] Li D., et al. (2023) Clip and dental floss traction-assisted endoscopic mucosal resection for early carcinoma of the duodenal papilla, Endoscopy 55, E440–E441.
[19] Deprez P. (2011) Endoscopic diagnosis and treatment of upper gastrointestinal tumors, Endoscopy 43, 966–970.
[20] Goda K. (2022) Duodenum: endoscopic diagnosis of superfcial non-ampullary duodenal epithelial tumors, Dig. Endosc. 34(Suppl 2), 68–72.
[21] Fleischmann C., Probst A., Ebigbo A., et al. (2021) Endoscopic submucosal dissection in Europe: results of 1000 neoplastic lesions from the German endoscopic submucosal dissection registry, Gastroenterology.
[22] Toya Y., Endo M., Oizumi T., Akasaka R., Yanai S., Kawasaki K., et al. (2020) Diagnostic algorithm of magnifying endoscopy with crystal violet staining for nonampullary duodenal epithelial tumors, Dig. Endosc. 32, 1066–1073.
[23] Vanbiervliet G., Moss A., Arvanitakis M., Arnelo U., Beyna T., Busch O., Deprez P. H., Kunovsky L., Larghi A., Manes G., Napoleon B., Nalankilli K., Nayar M., Pérez-Cuadrado-Robles E., Seewald S., Strijker M., Barthet M., van Hooft J. E. (2021) Endoscopic management of superficial nonampullary duodenal tumors: European Society of Gastrointestinal Endoscopy (ESGE) guideline, Endoscopy 53, 522–534.
[24] Amoyel M., Belle A., Dhooge M., et al. (2022) Endoscopic management of non-ampullary duodenal adenomas, Endosc. Int. Open 10, E96–E108.
[25] Kim S. Y., Kim K.-O. (2018) Endoscopic treatment of subepithelial tumors, 51, 19–27.
[26] Wong V. W. Y., et al. (2017) Endoscopic treatment of subepithelial lesions of the gastrointestinal tract, 15, 603–617.
[27] Cai M.‐Y., et al. (2018) Endoscopic full‐thickness resection for gastrointestinal submucosal tumors, Dig. Endosc. 30, 17–24.
[28] Ye L., et al. (2023) Calcified fibrous tumor in the stomach: a rare case of gastric submucosal tumor treated with endoscopic submucosal excavation, Endoscopy 55, E1201–E1202.
[29] Neto F. L. P., et al. (2021) Endoscopic resection of esophageal and gastric submucosal tumors from the muscularis propria layer: submucosal tunneling endoscopic resection versus endoscopic submucosal excavation: a systematic review and meta-analysis, Surg. Endosc. 35, 6413–6426.
[30] Tan Y., et al. (2017) Endoscopic resection of gastric gastrointestinal stromal tumors, 2, 115–115.
[31] Almario J. A., Zhang L. Y., et al. (2023) Through-the-scope suture closure of nonampullary duodenal endoscopic mucosal resection defects: a retrospective multicenter cohort study, Endoscopy, undefined.
[32] Valerii G., Tringali A., Landi R., et al. (2018) Endoscopic mucosal resection of non-ampullary sporadic duodenal adenomas: a retrospective analysis with long-term follow-up, Scand. J. Gastroenterol. 53, 490–494.
[33] Alkhatib A. A. (2019) Sporadic nonampullary tubular adenoma of the duodenum: prevalence and patients’ characteristics, Turk. J. Gastroenterol. 30, 112–113.
[34] Yamasaki Y., Takeuchi Y., Kanesaka T., et al. (2020) Differentiation between duodenal neoplasms and non-neoplasms using magnifying narrow-band imaging – do we still need biopsies for duodenal lesions? Dig. Endosc. 32, 84–95.
[35] Coriat R., et al. (2022) Endoscopic management of non-ampullary duodenal adenomas, 10, E96–E108.
[36] Kim G. E., Siddiqui U. D. (2023) Endoscopic resection techniques for duodenal and ampullary adenomas, VideoGIE 8, 330–335.
[37] Funayama Y., et al. (2023) Advancements in endoscopic management of small-bowel polyps in Peutz–Jeghers syndrome and familial adenomatous polyposis, Ther. Adv. Gastroenterol. 17.
[38] Gaspar J. P. (2016) Approach to the endoscopic resection of duodenal lesions, World J. Gastroenterol. 22, 600.
[39] Chen W.-C., Wallace M. B. (2015) Endoscopic management of mucosal lesions in the gastrointestinal tract, Exp. Rev. Gastroenterol. Hepatol. 10, 481–495.
[40] Lei J. E. Q., Ling-Hu N. L., et al. (2020) Novel endoscopic papillectomy for reducing postoperative adverse events (with videos), World J. Gastroenterol. 26(40), 144–153. 10.3748/wjg.v26.i40.6250.
Chapter 11 Super Minimally Invasive Surgery for Biliary Diseases
11.1 Choledocholithiasis and Cholangitis – Super Minimally Invasive Surgery
11.1.1 Overview
Choledocholithiasis is generally asymptomatic or presents with mild abdominal discomfort. When a stone becomes impacted and causes biliary obstruction, patients may experience abdominal pain or jaundice. In cases of secondary cholangitis, the classic Charcot’s triad may appear. If the condition worsens, the Reynolds’ pentad of acute obstructive suppurative cholangitis may develop.
(I) Diagnosis
When cholangitis is present, serum total bilirubin and direct bilirubin levels may be elevated, along with an increase in white blood cell count or neutrophil count.
Endoscopic ultrasound (EUS) is not affected by intestinal gas, which allows for a very precise diagnosis of choledocholithiasis. If the diameter of the common bile duct exceeds 1 cm, choledocholithiasis is highly suspected. EUS is particularly valuable for diagnosing stones in the distal common bile duct.
Magnetic resonance cholangiopancreatography (MRCP) can clearly demonstrate the degree and extent of bile duct dilation, the location, size, and distribution of stones, and the level of biliary obstruction.
Computed tomography (CT) plays an important role in diagnosing choledocholithiasis.
(II) Application of a duodenal papillary supporter
Super minimally invasive surgery involves performing endoscopic treatment within the bile duct and the pancreas through the duodenal papilla without damaging the papilla itself. This technique is made possible by the use of a single mushroom-head duodenal papillary supporter (12 mm in diameter, 25–30 mm in length) developed by Professor Enqiang Linghu’s team. The preoperative and postoperative papillary pressure test results are consistent. Notably, compared to self-expandable metallic stents (SEMS) used in previous studies, this supporter offers the following advantages: (1) the new papillary supporter is shorter than other stents, making the procedure more convenient, (2) the papillary supporter features a 70-degree angle design on the bile duct side, (3) the papillary supporter adopts a single mushroom-head design.
For distal common bile duct (CBD) stones, after stent placement, stones may become lodged between the longer SEMS and the bile duct wall. Jun et al. suggest that before placing the stent, the patient’s position should be slightly tilted to move the stone from the distal CBD to the mid-CBD. However, this method is not always effective. Therefore, the design of the 70° angle on the bile duct side of the papillary supporter ensures that during stent release, the stone is pushed into the upper or middle CBD. Additionally, the length of the papillary supporter is minimized to avoid stone impaction between the supporter and the bile duct mucosa. On the other hand, the mushroom-head design of the papillary side of the supporter prevents the supporter from entering the CBD due to continuous friction with the bile duct scope.
Furthermore, the papillary supporter has the following two additional advantages: (1) the supporter helps straighten the ampullary lumen of the common bile duct, facilitating smoother operations under direct visualization with the oral bile duct scope, (2) after the papillary supporter is inserted, there is enough space between the supporter and the bile duct scope to allow for the injection of physiological saline solution for biliary drainage, preventing excessive bile duct pressure and ultimately reducing the incidence of cholangitis.
11.1.2 Endoscopic Super Minimally Invasive Retrieval of Choledocholithiasis
(I) Overview
Currently, endoscopic retrograde cholangiopancreatography (ERCP) is the standard treatment for choledocholithiasis. However, the ERCP stone retrieval process involves radiation exposure and the need for endoscopic papillary sphincterotomy (EST). EST may lead to the loss of Oddi’s sphincter function and cause complications such as bleeding, perforation, cholangitis, biliary tract malignancy, and recurrent choledocholithiasis, both in the short and long term. More importantly, EST is not suitable for patients who cannot discontinue dual antiplatelet (anti-coagulant or anti-platelet) therapy.
The introduction of oral bile duct endoscopy has made it possible to retrieve CBD stones under direct visualization with minimal radiation exposure. Using a single mushroom-head duodenal papillary supporter facilitates stone retrieval under oral bile duct endoscopy and avoids the need for EST. This method, referred to as choledocholithotripsy with endoscopic papillary supporter under direct visualization (CETPS), is particularly effective for treating small CBD stones and sludge-like CBD stones. This technique has the dual advantages of preserving Oddi’s sphincter function while allowing for stone retrieval under direct visualization, with minimal radiation damage.
(II) Indications and contraindications
1. Indications: (1) sludge-like choledocholithiasis; (2) small choledocholithiasis with a transverse diameter ≤ 1 cm; (3) single or multiple choledocholithiasis; (4) pregnant women or other populations who cannot tolerate radiation exposure during ERCP; (5) choledocholithiasis in patients who cannot discontinue dual antiplatelet therapy.
2. Contraindications: (1) acute obstructive suppurative cholangitis; (2) poor general condition of the patient, age ≥ 65 or those with a bleeding tendency; (3) large or multiple complex choledocholith.
(III) Preoperative preparation
1. Gastrointestinal preparation: a liquid diet must be started one to two days before surgery. Fasting is required for 6 h prior to anesthesia, and water intake should be withheld for 2 h. For colorectal surgeries, a laxative or enema may be used to cleanse the intestines.
2. Preoperative examinations: complete blood count, blood biochemistry, coagulation function tests, electrocardiogram (ECG), echocardiography, and other relevant investigations should be performed.
(IV) Surgical steps and techniques
1. Anesthesia and positioning: depending on the patient’s condition and the specifics of the procedure, the surgery can be performed under deep sedation or intravenous general anesthesia. The patient is typically placed in a prone position, though supine or left lateral decubitus positioning may be used in special circumstances. Care should be taken to maintain body temperature, adjust the room temperature, and ensure the infusion fluid temperature is regulated to avoid pressure sores.
2. Surgical steps (figures 11.1 and 11.2):
(1) The duodenoscope is inserted into the duodenal papilla.
(2) A bow-shaped knife along with a guidewire is used for bile duct cannulation.
(3) After successful bile duct cannulation, a single mushroom-head papillary supporter is placed between the distal common bile duct and the papilla.
(4) For sandy-like CBD stones, the removal of negative pressure suction stones is performed under endoscopic guidance. For single large CBD stones, an oral bile duct endoscope is inserted into the common bile duct. Under direct vision, a stone retrieval basket is introduced through the oral bile duct endoscope channel for stone removal. For multiple large CBD stones, an oral bile duct endoscope is inserted into the common bile duct. Under direct vision, a stone retrieval balloon is introduced through the oral bile duct endoscope channel for stone removal.
(5) The oral bile duct endoscope is reinserted into the CBD to confirm the absence of residual stones.
(6) Finally, the papillary supporter is removed.
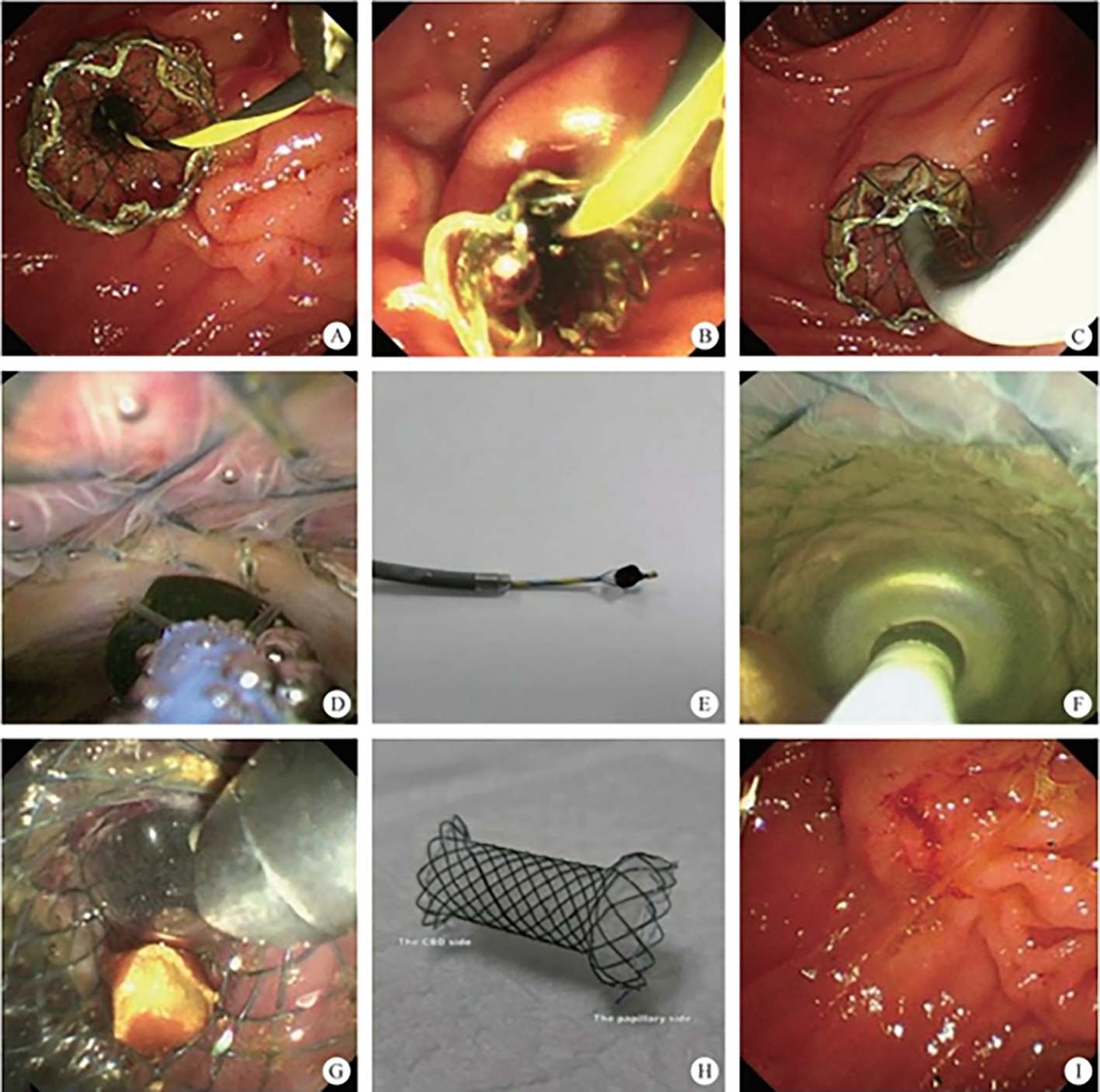
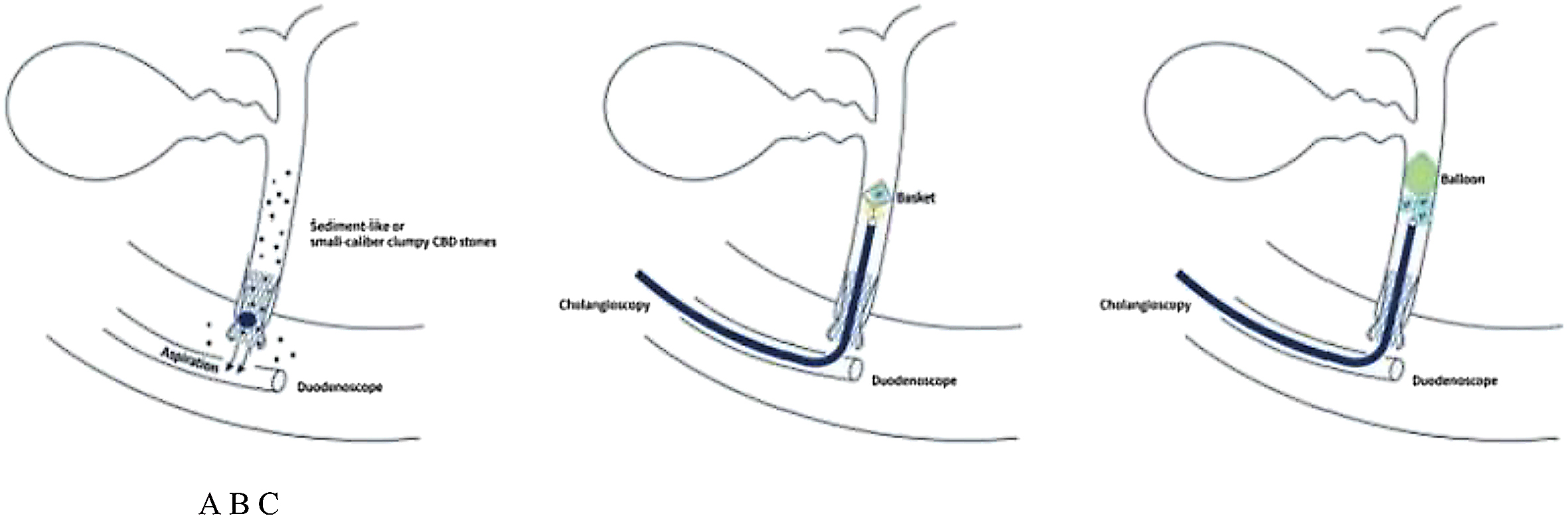

FIG. 11.1 — Process of biliary stone extraction under direct visualization with duodenal papillary supporter. A. After successful bile duct cannulation, a single mushroom-head duodenal papillary supporter is placed between the distal common bile duct and the duodenal papilla. B. For sludge-like CBD stones, negative pressure suction is used for stone retrieval under endoscopic guidance. C. The oral bile duct endoscope is inserted into the common bile duct. D. For a single solid CBD stone, an extraction basket is inserted through the oral bile duct endoscope channel under direct visualization to retrieve the stone. The basket can be seen grasping the stone. E. The stone is extracted from the body. F, G. For multiple solid CBD stones, the oral bile duct endoscope is inserted into the common bile duct, and an extraction balloon is inserted through the oral bile duct endoscope channel under direct visualization to remove the stones. H. The duodenal papillary supporter is removed from the body. I. After the papillary supporter is removed, the papilla appears normal.

FIG. 11.2 — Schematic diagram of the operative process for bile duct stone removal under direct vision using papillary supporter and bile duct endoscopy. For sandy-like CBD stones, negative pressure suction stone removal is performed under endoscopic guidance. B. For single large CBD stones, an oral bile duct endoscope is inserted into the common bile duct. Under direct vision, a stone retrieval basket is introduced through the oral bile duct endoscope channel for stone removal. C. For multiple large CBD stones, an oral bile duct endoscope is inserted into the common bile duct. Under direct vision, a stone retrieval balloon is introduced through the oral bile duct endoscope channel for stone removal.
(V) Postoperative management
(1) Postoperatively, the patient should fast from food and water for one day. If no abnormal abdominal signs are observed and postoperative laboratory parameters show no significant abnormalities, a gradual transition from liquid to semi-liquid diet can be initiated.
(2) Postoperative PPI and antibiotic therapy should be administered for three days. If postoperative pancreatitis or gastrointestinal bleeding occurs, the duration of treatment should be extended accordingly.
(3) In the case of postoperative pancreatitis, somatostatin therapy should be initiated promptly. For gastrointestinal bleeding, if conservative treatment is ineffective, endoscopic, surgical, or interventional methods should be promptly considered for hemostasis.
(VI) Postoperative follow-up
It is recommended to conduct close follow-up through phone calls and outpatient visits to record any adverse events. Additionally, patients will be advised and encouraged to return for endoscopic reexamination at three months, six months, one year, two years, three years, four years, and five years after surgery.
(VII) A classic case
History: the patient, an elderly man, was admitted with a chief complaint of “abdominal pain for three days.” EUS and CT examination revealed multiple stones in the common bile duct and gallstones in the gallbladder.
Diagnosis: common bile duct stones, gallbladder stones.
After thorough preoperative assessment and obtaining informed consent for surgery, the patient underwent bile duct stone removal via endoscopic retrograde cholangiopancreatography (ERCP) under direct vision (Refer to video 11.1, Super minimally invasive bile duct stone extraction via duodenal papillary bile duct endoscopy (See online resources)).
Using the papillary supporter technique for direct endoscopic visualization of the bile ducts sandy-like stones were suctioned out.
Stone removal procedure: A covered stent with a diameter of 12 mm and a length of 30 mm was placed in the distal common bile duct and papilla. The bile duct endoscope was inserted into the common bile duct. Multiple sandy-like stones were observed in the bile duct. These stones were removed through the suction function of the bile duct endoscope.
Postoperatively, the patient was fasted for one day. PPIs and antibiotics were administered for three days. The patient developed hyperamylasemia postoperatively, but no abdominal pain was noted. A follow-up two weeks after surgery revealed no residual stones or cholangitis.
(VIII) Hot-spot issues and research progress in the consensus
The cholangioscopy-assisted extraction through novel papillary support (CETPS) method is a safe and feasible treatment for smaller CBD stones (diameter ≤ 1.0 cm) or sandy-like CBD stones. It has a high technical success rate, with a low incidence of adverse events such as perforation, bleeding, and cholangitis.
Compared to ERCP combined with EST for CBD stone treatment, the primary advantage of CETPS is the preservation of the function of the sphincter of Oddi, avoiding associated adverse events such as bleeding, perforation, cholangiocarcinoma, and recurrent CBD stones. Jun et al. confirmed that the use of a transpapillary SEMS effectively protects the function of the sphincter of Oddi. Additionally, a study by Cho et al. demonstrated that in patients undergoing dual-antiplatelet therapy, SEMS could be used for CBD stone extraction without causing bleeding or thromboembolism.
In terms of postoperative pancreatitis (PEP) incidence, theoretically, using a papillary supporter can avoid unnecessary pancreatic duct cannulation during stone removal, thereby reducing the occurrence of pancreatitis. However, on the other hand, excessive compression from the papillary supporter may also induce PEP. Therefore, finding an optimal balance between sufficient support and a reasonable PEP rate will be a key direction for further improvement of this papillary supporter.
Another significant advantage of CETPS over traditional ERCP is the treatment under direct vision. First, it minimizes radiation exposure to both the patient and the surgical team, which is particularly beneficial for special patient groups such as pregnant women and children. Second, the technique is equipped with specialized stone retrieval baskets and balloons for the oral bile duct endoscope, which can be deployed under direct vision in the most appropriate position to effectively grasp or drag stones. Moreover, the operator can promptly detect related adverse events such as CBD perforation, bleeding, and damage.
In summary, CETPS combines the benefits of preserving the function of the sphincter of Oddi and stone removal under direct vision. The clinical application of a single mushroom-head papillary supporter provides a smooth access route for the oral bile duct endoscope, reducing the technical difficulty of stone removal under direct vision. However, research on the super minimally invasive technique of bile duct stone removal using cholangioscopy-assisted extraction with a papillary supporter is still limited. Further studies are needed to assess its safety, efficacy, and broader applicability.
11.1.3 Peroral Super Minimally Invasive Papillotomy
(I) Overview
Peroral super minimally invasive papillotomy, previously known as endoscopic clipping papillotomy, is a technique that involves endoscopic retrograde cholangiopancreatography (ERCP) and associated procedures such as endoscopic sphincterotomy (EST), endoscopic papillary balloon dilation (EPBD), and endoscopic sphincterotomy plus balloon dilation (ESBD). These procedures are widely used for the diagnosis and treatment of various diseases, including bile duct stones, bile duct tumors, and pancreatic tumors, with high diagnostic value and definitive therapeutic effects.
The sphincter of Oddi, also known as the papillary sphincter, is composed of the distal bile duct sphincter, the distal pancreatic duct sphincter, and the sphincter of the duodenal papilla. It functions as a one-way valve, precisely regulating the secretion and storage of bile and pancreatic juices, while preventing the retrograde reflux of duodenal contents, bacteria, and other substances. EST can disrupt the structure of the sphincter of Oddi, potentially causing partial or permanent loss of its function. This can lead to duodenal content reflux, recurrent bile duct stones, bacterial colonization of the bile ducts, liver abscesses, and other complications. Endoscopic clipping papillotomy (endoclip papilloplasty) refers to the procedure where, after endoscopic papillotomy of the duodenal papilla, metal clips are applied to continuously occlude the papillary opening while maintaining patency for bile duct drainage. This technique aims to repair the papillary structure and restore the function of the sphincter of Oddi, aligning with the principles of super minimally invasive surgery.
(II) Indications and contraindications
1. Indications. ① Confirmed common bile duct stones through imaging such as MRCP. ② Common bile duct stones with a diameter of less than 2.5 cm. ③ Preoperative evaluation predicts that the entire common bile duct stone can be removed in a single procedure.
2. Contraindications. ① Previous sphincterotomy or papillary balloon dilation. ② Benign or malignant bile duct strictures.
(III) Preoperative preparation
1. Preoperative counseling. Preoperatively, a comprehensive evaluation of treatment effectiveness and risks should be conducted, considering factors such as the diameter of the common bile duct, the size of the bile duct stones, and the length of sphincterotomy. The surgeon, senior physician, or primary assistant should fully communicate the details with the patient or the family members, providing all necessary information. A written informed consent form should be signed by the patient or his/her authorized representative.
2. Preparation of the patient. (1) Correction of abnormal coagulation function: preoperative coagulation tests should be conducted, and any abnormalities in platelet count, International Normalized Ratio (INR), etc., should be corrected as much as possible. Patients on antiplatelet therapy should discontinue the medication five to seven days prior to surgery.
(2) Prevention of pancreatitis: indomethacin suppositories should be administered preoperatively to reduce the incidence of postoperative pancreatitis.
(3) Prophylactic antibiotics: for patients with bile duct infection or those in an immunosuppressive state, broad-spectrum antibiotics should be used prophylactically, covering Gram-negative bacteria, enterococci, and anaerobes.
(4) Fasting: the patient should fast from food and water for 8 h prior to the surgery.
(IV) Surgical steps and techniques
1. Anesthesia and positioning. Depending on the patient and clinical circumstances, surgery can be performed under deep sedation or intravenous general anesthesia. The standard surgical position is prone, but in special cases, supine or left lateral decubitus positioning may be used. Appropriate measures should be taken to keep the patient warm, regulate the ambient temperature and control the temperature of intravenous fluids to prevent pressure ulcers.
2. Surgical procedure (figure 11.3). (1) Stent insertion into the bile duct: after the bile duct stones are removed, a plastic bile duct stent should be placed in the common bile duct using a stent introducer along the guidewire to prevent inadvertent closure of the common bile duct during papillotomy.
(2) Pancreatic duct stent insertion: if the guidewire has entered the pancreatic duct three times or more during the bile duct cannulation process, or if a pre-cut pancreatic sphincterotomy is performed, a pancreatic duct stent should be placed. A guidewire should be left in place, and a plastic stent should be inserted into the pancreatic duct using a stent introducer along the guidewire.
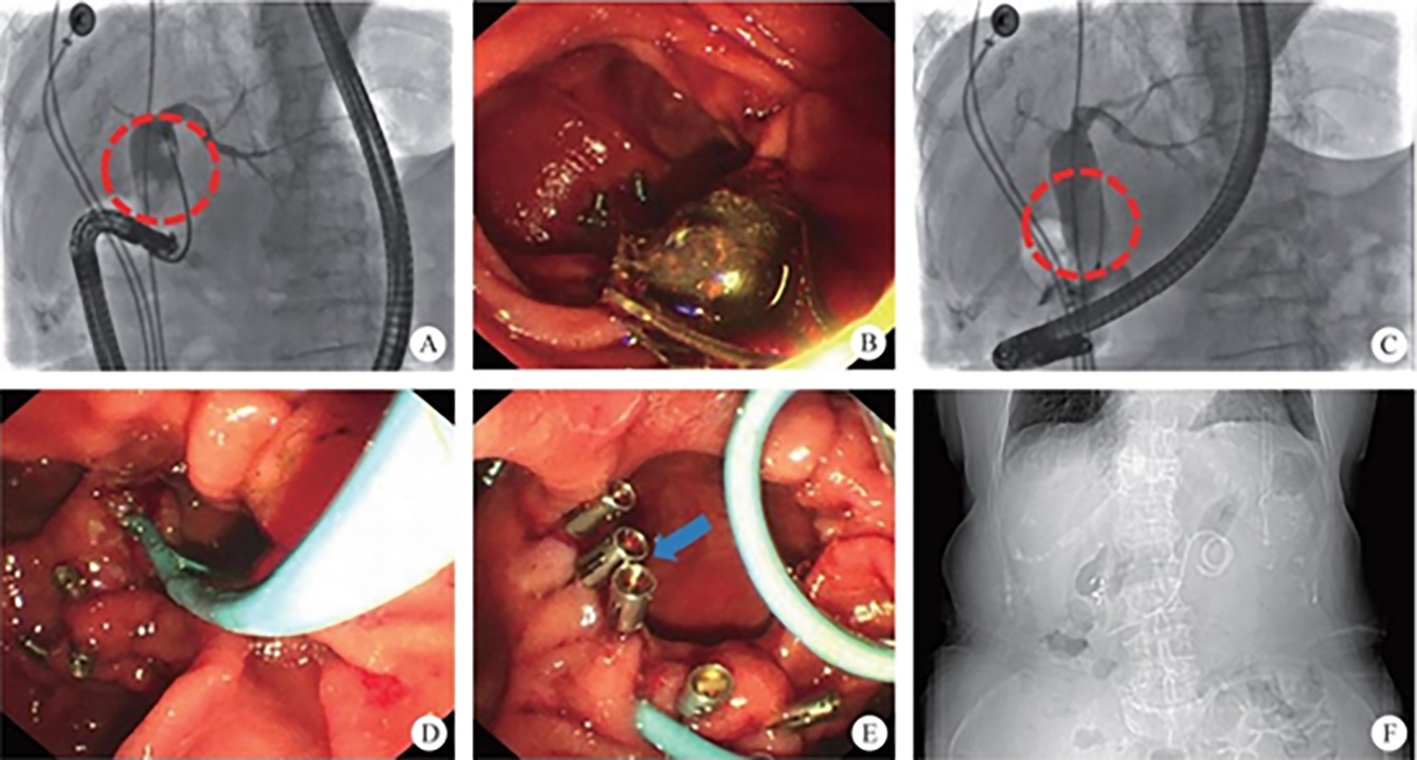

FIG. 11.3 — A. ERCP imaging shows common bile duct stones. B. Bile duct stones are removed after sphincterotomy. C. Post-removal, imaging shows no remaining stones in the bile duct. D. A self-expanding bile duct stent (custom-made) is inserted. E. Tissue clips are used to reshape the sphincter of Oddi. F. Abdominal X-ray confirms that the stent is in place.
(3) Clipping and closing the papilla
A rotatable tissue clip should be used to gradually close the papillotomy from proximal to distal in a ‘zipper’ fashion. The position and direction of the tissue clip should be adjusted according to the bile duct opening and its axis. Care must be taken to avoid inadvertently clipping the bile and pancreatic duct stent during the procedure. The number of tissue clips used should correspond to the size of the papillotomy, with four to five clips typically being used.
(4) Stent removal
If there are no adverse reactions upon close postoperative monitoring, the bile and pancreatic duct stents should be removed via endoscopy after three weeks.
(V) Hot-spot issues in the consensus
The sphincter of Oddi is composed of the bile duct sphincter, the pancreatic duct sphincter, and the duodenal sphincter. It performs three main functions: regulating the amount of bile entering the duodenum, preventing the retrograde flow of intestinal contents into the bile and pancreatic ducts, and controlling the degree of gallbladder filling. As a ‘valve’ for the bile-pancreatic duct channel, the sphincter of Oddi effectively prevents the reflux of duodenal contents, prevents retrograde infections such as bacterial contamination, and indirectly regulates the secretion and storage of bile and pancreatic juice. It plays an irreplaceable role in maintaining the normal physiological function of the bile and pancreatic ducts. Once damaged, it leads to a reflux of duodenal contents, bacterial retrograde infections, and increases the risk of biliary complications such as cholangitis, stone formation, and even bile duct cancer. Therefore, protecting the structure and function of the sphincter of Oddi is crucial in clinical practice.
Endoscopic sphincterotomy (EST) can impair the normal physiological function of the sphincter of Oddi. The bile duct pressure gradient and basal pressure of the sphincter of Oddi are completely lost 15–17 years after EST, resulting in bile duct reflux. Reduced frequency and amplitude of sphincter activity are associated with the formation of bile duct stones. For large CBD stones (diameter > 1 cm), large EST (>1 cm) is usually performed. To reduce the long-term risk of recurrence of bile duct stones, the European Society of Gastrointestinal Endoscopy (ESGE) recommends endoscopic papillary balloon dilation (EPBD) or endoscopic papillary large balloon dilation (EPLBD) as alternatives to large EST. However, the basal pressure of the Oddi sphincter also significantly decreased after EPBD, dropping from 13.6 mmHg to 6.3 mmHg and 9 mmHg to 3.3 mmHg after one week, respectively. After one month, the basal pressure of the sphincter of Oddi can return to 9.3 mmHg, and after one year, it can return to 4.2 mmHg, which is still significantly lower than preoperative values. For EPLBD, the basal pressure of the sphincter of Oddi drops from 30.4 mmHg to 6.4 mmHg after the procedure, and similar results are observed one year after EPLBD. However, compared to EST, EPBD or EPLBD shows similar recurrence rates for CBD stones.
In 2018, Huang Yonghui et al. reported on endoscopic clip-assisted papilloplasty, a new technique for repairing the structure and function of the sphincter of Oddi. This technique uses a harmonious clip to close the papillary tissue after EST injury, promoting the healing of the duodenal papilla incision and restoring the function of the sphincter of Oddi. In experiments involving three patients and four animals, the method not only promoted healing but also reduced the occurrence of post-EST cholangitis and stone formation. However, due to the small sample size and lack of long-term clinical observations on cholangitis and stone recurrence, this method is still at the clinical observation and research stage. It is currently believed that the success of endoscopic clip-assisted papilloplasty is related to the morphology of the papilla. Generally, patients with long papillae are more suitable for papilloplasty, with a higher success rate. In contrast, papillae with a single or constricted opening are less suitable for this procedure, and challenges may arise in cases of diverticular papillae or severely edematous papillae, where clipping may even fail.
Endoscopic clip-assisted papilloplasty is an innovative technique for restoring the function of the sphincter of Oddi and preventing post-EPLBD cholangitis and recurrence of CBD stones. However, further exploration and research are still needed. The formation of scarring at the lower segment of the bile duct papilla post-papilloplasty, its impact on long-term bile drainage, and its potential in reducing recurrence of cholangitis and stone formation still require further validation through multi-center, large-scale, long-term follow-up randomized controlled trials.
11.1.4 Acute Suppurative Cholangitis and Peroral Super Minimally Invasive Drainage
(I) Overview
Acute suppurative cholangitis is an acute purulent inflammation of the biliary system caused by bacterial infection. The underlying cause is biliary obstruction combined with bacterial infection, with Escherichia coli being the most common pathogen. During the onset of acute suppurative cholangitis, the Reynolds pentad may appear, which includes abdominal pain, chills and high fever, jaundice, shock, and central nervous system symptoms. If not treated in a timely manner, it can progress to multiple organ dysfunction syndrome (MODS) and even lead to death. Peroral Super Minimally invasive drainage for acute suppurative cholangitis refers to a procedure in which drainage of the biliary system is performed endoscopically through the mouth. This technique preserves the function of the sphincter of Oddi while achieving biliary drainage and biliary decompression.
(II) Diagnosis
Clinically, the Reynolds pentad may be present, and in severe cases, multiple organ dysfunction syndrome (MODS) may occur. Laboratory tests typically show elevated white blood cell counts, with neutrophils predominating, and elevated serum C-reactive protein levels. Biochemical markers indicative of cholestasis, such as total bilirubin with an increase in direct bilirubin, alkaline phosphatase, and γ-glutamyl transpeptidase (GGT), are also elevated. Abdominal ultrasound, CT, and MRCP can show varying degrees of biliary dilation and biliary obstruction. Endoscopic ultrasound (EUS) provides a more comprehensive view of the intrahepatic and extrahepatic bile ducts, offering significant diagnostic value for identifying the underlying cause.
(III) Indications and contraindications
1. Indications: diagnosis of acute suppurative cholangitis confirmed through symptoms, laboratory tests, and imaging.
2. Contraindications: 1) unstable vital signs, with a poor prognosis for tolerating surgery. 2) severe coagulopathy. 3) upper gastrointestinal obstruction, where endoscopic access to the duodenal papilla is not possible.
(IV) Preoperative preparation
1. Preoperative consultation. Preoperatively, a comprehensive assessment of the patient’s condition, the potential outcomes and risks of the surgery, and alternative treatment options is conducted through the patient’s medical history, symptoms, and auxiliary examinations. This information is communicated clearly to the patient or the family by the surgeon, senior physician, or primary assistant, and the patient (or his/her representative) is required to sign a written informed consent form.
2. Patient preparation
(1) Stabilization of Vital Signs: If the patient has unstable vital signs and cannot tolerate surgery, symptomatic treatments, including oxygen therapy, anti-infection treatment, fluid resuscitation, and maintenance of water-electrolyte and acid-base balance, are provided to stabilize vital signs. In severe cases, the patient may be admitted to the ICU.
(2) Active prevention of pancreatitis: preoperative administration of indomethacin suppositories is used to reduce the incidence of postoperative pancreatitis.
(3) Antibiotic therapy: Before blood culture results are available, third-generation cephalosporins in combination with nitroimidazole antibiotics are used. For patients with severe infections or high risk of antibiotic resistance, carbapenem antibiotics are recommended. Once blood culture results are returned, antibiotics should be adjusted based on the findings.
(4) Preoperative fasting: the patient should fast from food and water for at least 8 h before the procedure.
(E) Surgical procedure and techniques
1. Anesthesia and positioning. Depending on the patient’s condition and clinical factors, the surgery can be performed under deep sedation or intravenous general anesthesia. The standard surgical position is prone; however, in special cases, the supine or left lateral position may be used. It is essential to ensure the patient’s warmth, regulate room temperature, and maintain the temperature of fluids to prevent pressure sores.
2. Surgical steps
(1) Biliary cannulation: the duodenoscope is inserted into the descending part of the duodenum to locate the duodenal papilla. Under DSA fluoroscopy, a papillary incision knife or contrast catheter is used to guide the insertion of a guidewire, which is then advanced into the common bile duct, where the guidewire is left in place.
(2) Biliary imaging: a contrast agent is injected into the papillary incision knife or contrast catheter, and endoscopic retrograde cholangiopancreatography (ERCP) is performed to identify any stenosis, stones, or other lesions in the bile ducts. The guidewire is advanced beyond the lesion site to ensure adequate positioning.
(3) Choice of the drainage method: depending on factors such as the location of the lesion, the cause of the disease, the diameter of the bile ducts, the expected prognosis, and financial considerations, either endoscopic nasobiliary drainage (ENBD), which provides external drainage, or endoscopic biliary stent placement (EBS), which offers internal drainage, may be selected. For ENBD, nasobiliary tubes of various shapes and sizes may be used. For EBS, plastic or metal stents of different lengths and diameters can be chosen.
(4) External drainage procedure: after locking the guidewire, the papillary incision knife or contrast catheter is removed. A nasobiliary tube, selected based on the appropriate size and shape, is inserted into the hepatic hilum or higher, ensuring it passes beyond the lesion. The tube is carefully advanced under fluoroscopic guidance while the endoscope is slowly withdrawn to ensure that the distal end of the nasobiliary tube remains stationary and curves appropriately in the stomach. The proximal end of the tube is passed through the nasal passage and fixed in place. Final confirmation of correct positioning is done under fluoroscopy.
(5) Internal drainage procedure: after locking the guidewire, the papillary incision knife or contrast catheter is removed. The selected biliary stent, along with its delivery system, is introduced into the bile duct, and the stent is deployed in the predetermined position. The delivery system and guidewire are then removed. After stent placement, air is aspirated, and bile should flow out of the stent, confirming successful drainage.
(F) Hot-spot issues in the consensus
1. Choice between ENBD and ERBD. In the comparison of the efficacy between emergency endoscopic and laparoscopic treatment of acute obstructive suppurative cholangitis caused by extrahepatic biliary stones, endoscopic treatment is preferred for patients with an established diagnosis of AOSC. For patients with septic shock, biliary obstruction should be relieved within six hours of admission. For elderly AOSC patients with grade-III disease, early ENBD drainage can be performed, with subsequent treatment carried out after the patient’s condition improves. In emergency treatment of acute obstructive suppurative cholangitis, both biliary plastic stent placement and endoscopic nasobiliary drainage (ENBD) can achieve good drainage outcomes. There are no significant statistical differences in laboratory indices, liver function, symptom recovery time, or complication rates following both procedures, allowing for a rational choice based on the patient’s condition.
Currently, endoscopic biliary drainage (EBD) has become the gold standard for the safety and practicality of acute cholangitis treatment. Whether the pathology is benign or malignant, endoscopic biliary drainage can be applied for various forms of acute cholangitis. EBD includes endoscopic sphincterotomy (EST) alone, EST combined with ENBD, or endoscopic retrograde biliary drainage (ERBD). EST involves using endoscopic instruments to incise the duodenal papilla and extract stones with the aid of various endoscopic tools. After years of clinical experience, over 90% of common bile duct stones can be treated using this method. With favorable long-term outcomes, EST has become the preferred treatment for common bile duct stones. Experienced endoscopists have a success rate of up to 98%, without the need for a T-tube and with minimal intra-abdominal interference, unaffected by previous surgery, adhesions, age, or comorbidities. However, for large common bile duct stones, endoscopic stone removal can be challenging. With the development of laser lithotripsy, both Chinese and international studies have successfully treated and gained extensive experience in stone extraction. For elderly patients or those with specific diseases who are not candidates for surgery, EST presents an excellent alternative. However, the efficacy and safety of EST in acute cholangitis remain controversial due to complications such as bleeding.
ENBD is an external drainage technique that has the advantage of being able to irrigate blocked ducts and perform bile culture. However, if the patient is uncomfortable with the nasobiliary tube position, it may lead to dislodgement, especially in elderly patients. Furthermore, there is a risk of electrolyte and fluid loss, as well as of tube collapse due to kinking. ERBD is an internal drainage technique, with two types of biliary stents available: plastic and metal. Plastic stents are easier to insert and remove, more cost-effective than metal stents, but are prone to biofilm and sludge obstruction. The choice of the stent depends on factors such as practicality, cost, and the preference of the ERCP operator. The advantages of ERBD include no discomfort for the patient, with no risk of electrolyte or fluid loss, but there are risks of stent migration or blockage. Randomized controlled trials have found no significant difference in the prognosis of AOSC drainage between ENBD and ERBD without prior EST. According to the Tokyo Guidelines, AOSC patients with coagulopathy or those receiving anticoagulant therapy should avoid percutaneous transhepatic cholangiography (PTCD). ENBD or ERBD should be used for such patients until coagulopathy and cholangitis are improved, after which biliary stone treatment can be performed.
2. Endoscopic ultrasound-guided biliary drainage as a useful alternative following ERCP failure. Endoscopic retrograde cholangiopancreatography (ERCP) has long been the standard technique for preoperative biliary drainage (PBD). Recently, endoscopic ultrasound-guided biliary drainage (EUS–BD) has emerged as a useful alternative after ERCP failure. However, the optimal strategy for EUS–BD remains controversial. There is no consensus on the best EUS–BD technique for PBD, and the choice of a method depends on the patient’s condition, the site of biliary obstruction, postoperative anastomosis, and the preference of the endoscopist. Both ERCP and EUS–BD are comparable in terms of efficacy and safety in randomized controlled trials comparing ERCP and EUS–BD as primary drainage for obstructive jaundice. Although further studies are required, EUS–BD may be a viable and safe alternative to ERCP for PBD in cases of malignant biliary obstruction after ERCP failure.
11.2 Super Minimally Invasive Surgery of Biliary Strictures
11.2.1 Overview
Biliary stricture can be caused by a variety of benign and malignant diseases. Early identification of the nature of the stricture and timely intervention are crucial for guiding subsequent treatments and improving patient prognosis. At the time of initial diagnosis, it is usually easy to determine whether biliary obstruction is present, but the specific nature of the obstruction can sometimes be difficult to identify. Even moderate obstruction causing jaundice, if untreated for several months, can progress to secondary biliary cirrhosis. Some clinical studies have shown that biliary strictures following cholecystectomy, if not treated promptly, may progress to secondary biliary cirrhosis 15–62 months later. Key elements in the assessment and treatment of biliary strictures include clarifying the pathological nature of the stricture, relieving biliary obstruction, and considering treatment modalities such as endoscopy, percutaneous methods, or surgical approaches. These measures help alleviate symptoms, prevent disease progression, and ultimately improve the patient’s quality of life.
(I) Diagnosis of biliary strictures
Differentiating between benign and malignant biliary strictures is of utmost importance in clinical practice, as it directly affects the patient’s treatment and prognosis. Clarifying the nature of the stricture and relieving biliary obstruction are often not independent processes and can often be achieved in a single intervention. The nature of the stricture is commonly determined through a combination of history review, laboratory tests, invasive and non-invasive imaging, and various histological sampling techniques.
Benign biliary strictures (BBS) are most commonly iatrogenic, typically resulting from surgical trauma, such as after cholecystectomy or liver transplant. Other causes include primary sclerosing cholangitis, chronic pancreatitis, and IgG4-related cholangiopathy. Malignant biliary strictures account for 54%–95% of cases, with the most common causes being cholangiocarcinoma and pancreatic cancer located at the head or uncinate process of the pancreas. However, malignant biliary strictures often do not present with obvious symptoms or signs in the early stages, making diagnosis challenging. The majority of cases are diagnosed via imaging, with approximately 70% of patients diagnosed at an advanced stage, leading to a five-year survival rate of less than 5%. Early and accurate diagnosis of malignant biliary strictures remains a significant challenge.
In recent years, the rapid development of endoscopic techniques, such as cholangioscopy, endoscopic ultrasound, and laser confocal microscopy, has greatly enhanced the clinician’s toolkit. These advanced techniques not only allow for direct visualization of the size and location of the lesions, but they also enable the direct acquisition of tissue samples for pathological examination. This has significantly improved the ability to diagnose biliary strictures early and differentiate between benign and malignant conditions.
In the treatment of biliary strictures, the concept of super minimally invasive techniques has gradually emerged. Endoscopic biliary dilation and endoscopic biliary stent placement have now replaced traditional minimally invasive surgical procedures and have become the first-line treatments for biliary strictures. With the advent of peroral cholangioscopy, it is now possible to visualize the biliary system dynamically through natural orifices, facilitating biliary exploration, precise biopsy of suspicious lesions, and investigation of indeterminate biliary strictures (IBS). This has greatly enhanced diagnostic and therapeutic capabilities.
(II) Technical methods and previous names in super minimally invasive surgery of biliary strictures
The super minimally invasive surgery of biliary strictures can be achieved through previously established methods, such as endoscopic biliary dilation and endoscopic biliary stent placement.
11.2.2 Super Minimally Invasive Dilatation for Biliary Stricture at the Duodenal Papilla
(I) Overview
Super minimally invasive dilatation for biliary stricture can be achieved through endoscopic biliary dilation. This technique involves guiding a dilatation catheter or balloon to the stricture site via a guide wire, gradually expanding the narrowed segment of the bile duct to restore its normal diameter. The key to endoscopic treatment of biliary stricture lies in passing through the stricture segment, dilating the stenosis, and preventing and managing restenosis.
(II) Indications and contraindications
Endoscopic treatment for biliary stricture first requires passage through the stricture segment, which necessitates continuity of the common bile duct. For patients with complete transection or ligation of the common bile duct, where the guide wire cannot pass through the lesion, endoscopic treatment is not feasible.
1. Indications. MRCP or ERCP imaging shows biliary stricture, and the guide wire can pass through the narrowed segment.
2. Contraindications. ① Complete transection or ligation of the common bile duct, where the guide wire cannot pass through the stricture segment; ② severe cardiopulmonary insufficiency, rendering the patient unable to undergo endoscopic surgery; ③ significant coagulopathy.
(III) Key points of endoscopic biliary dilation procedure
Endoscopic treatment for biliary stricture involves two steps: passing through the stricture segment and dilating the stricture. For patients with complete transection or ligation of the common bile duct, where the guide wire cannot pass through the lesion, endoscopic treatment is not feasible. Once the instruments have passed through the stricture and entered the deeper part of the bile duct, cholangiography should be performed to determine the type and characteristics of the biliary stricture (figure 11.4). Benign biliary strictures are generally shorter, asymmetric, and exhibit significant fibrosis, making them more difficult to traverse compared to those caused by tumors. When the hilum is involved, these strictures are more complex, and passage is even more challenging. Therefore, it is usually necessary to use a small-diameter hydrophilic guide wire (diameter 0.021 in or 0.018 in) with a straight or curved tip to traverse the stricture. Guide wire manipulation requires good fluoroscopic imaging and a high level of technical skill. Forceful manipulation should be avoided as it may lead to false passages. After inflating a balloon at the distal end of the stricture, traction should be applied in a downward direction to straighten the bile duct, which facilitates alignment of the guidewire and the stricture segment. A controllable directional catheter or sphincterotome may also be used to attempt passage through the stricture. Once the hydrophilic guide wire has passed through the stricture, it can be replaced with a stiffer guide wire to facilitate dilation.
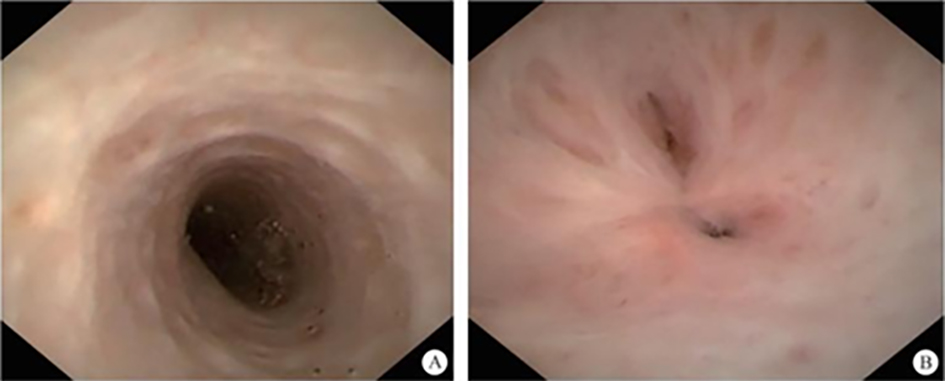

FIG. 11.4 — Endoscopic images of the biliary duct. A. Scar stricture visible within the bile duct. B. Near-complete occlusion of the bile duct wall.
Dilation of the stricture has two objectives: reopening the biliary stricture to restore bile drainage and maintaining the stricture open to prevent restenosis. After the guidewire passes through the stricture segment, a 5Fr or 6Fr catheter is advanced along the guidewire. Mechanical dilation or balloon dilation can then be performed. Mechanical dilation typically uses a dilator (e.g., a 6–9.5Fr Cunningham–Cotton dilator; Cook Endoscopy, Winston-Salem, NC) to assess the degree of stenosis, which guides the placement of a stent. If mechanical dilation is ineffective, small-diameter water-filled balloons (typically 4 mm, 6 mm, or 8 mm) are used for dilation. The balloon is usually inflated to a diameter of 1–2 mm larger than that of the downstream bile duct.
(IV) Hot-spot issues and research progress in the consensus
1. Clinical value of endoscopic biliary dilation. A prospective study demonstrated that endoscopic biliary dilation and biliary stent placement for the treatment of biliary anastomotic stricture after liver transplantation have similar clinical success rates (71% vs. 73%), with a lower complication rate for endoscopic dilation (4.3% vs. 13.6%).
2. Restenosis after endoscopic biliary dilation. A retrospective study involving 165 patients with biliary stricture, with a median follow-up of eight years, found that, for patients with anastomotic stricture after liver transplantation who underwent regular endoscopic biliary dilation, the clinical success rate was 79.1%, proving the safety and effectiveness of the procedure. While dilation can provide immediate relief, whether for single or multiple strictures, a single dilation session via endoscopy or percutaneous puncture cannot fully resolve the issue, with a high restenosis rate (47%). Preventing and managing restenosis can be addressed through repeated dilation and placement of multiple plastic stents or covered metal stents.
11.2.3 Biliary Stricture: Super Minimally Invasive Endoscopic Stent Placement at the Duodenal Papilla
(I) Overview
Endoscopic biliary stent placement for biliary stricture at the duodenal papilla has demonstrated significant therapeutic efficacy and high safety. The goal is to alleviate biliary obstruction, maintain long-term bile duct patency, and preserve liver function. Endoscopic treatment has become the first-line therapy for most biliary strictures. ERCP dilation of the biliary stricture combined with biliary stent placement can achieve a favorable biliary drainage effect. The effectiveness, success rate, and prognosis of endoscopic intervention depend on the specific etiology of the biliary stricture, whether appropriate endoscopic techniques and stents are selected, and the adequacy of the procedure.
For benign biliary strictures, such as anastomotic stricture after liver transplantation, multiple plastic stents may be used to provide greater biliary support. Another option is the fully covered self-expandable metallic stent (FCSEMS), which, compared to multiple plastic stents, avoids the need for repeated ERCP procedures, the continual addition and replacement of plastic stents, and offers a viable choice for benign biliary strictures. For malignant biliary strictures, endoscopic biliary stent placement effectively relieves jaundice, protects liver function, and, compared to surgical palliative procedures, results in shorter postoperative hospital stays with no significant difference in the incidence of early complications.
(II) Key points of endoscopic multiple biliary stent placement (figure 11.5)
For benign biliary stricture, simple endoscopic biliary dilation has a high restenosis rate. In addition to biliary dilation, the placement of biliary stents can help maintain patency of the stricture segment for a longer period, allowing scar remodeling and consolidation. If mechanical or balloon dilation fails, placement of a 5Fr or 6Fr nasobiliary drain for 24–48 h followed by endoscopic stent placement may increase the chance of success. Alternatively, a spiral stent retrieval device (Soehendra stent retriever; Cook Endoscopy) and a 3Fr angioplasty balloon catheter can be used for dilation. Following this, a 5Fr balloon dilator can be employed, and a stent can be further placed.
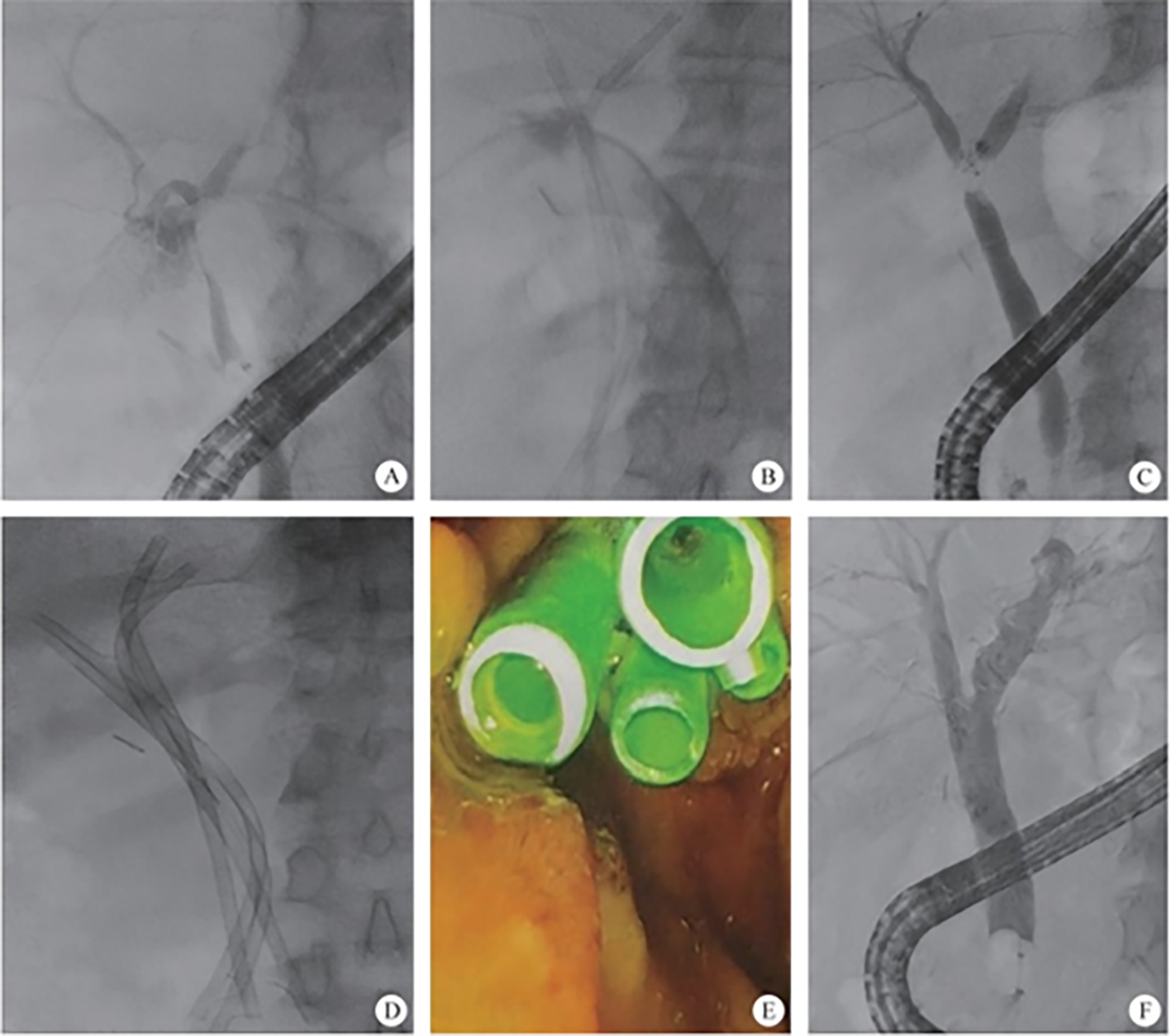

FIG. 11.5 — Steps of multiple plastic stent placement. A. Bismuth type-3 biliary stricture following laparoscopic cholecystectomy. B. Initial treatment with the placement of two 8.5Fr plastic stents. C. Three months later, stents are removed, and cholangiography shows significant Bismuth type-3 biliary stricture. D. Re-insertion of multiple 10Fr plastic stents via ERCP after three months. E. Multiple plastic stents under endoscopic view. F. Cholangiography after stent removal at one year.
The number of plastic stents placed gradually increases, with replacement every three to four months for a period of 12 months, which has become the gold standard for the treatment of benign biliary strictures. Each time a stent is replaced, all previously placed stents are removed, and larger-diameter plastic stents are inserted, including progressive dilation of fibrotic biliary strictures. The placement of ERCP plastic stents is repeated until cholangiography confirms that the stricture has been completely resolved. Treatment typically takes one year, with a good long-term outcome and a success rate of 80%–100%.
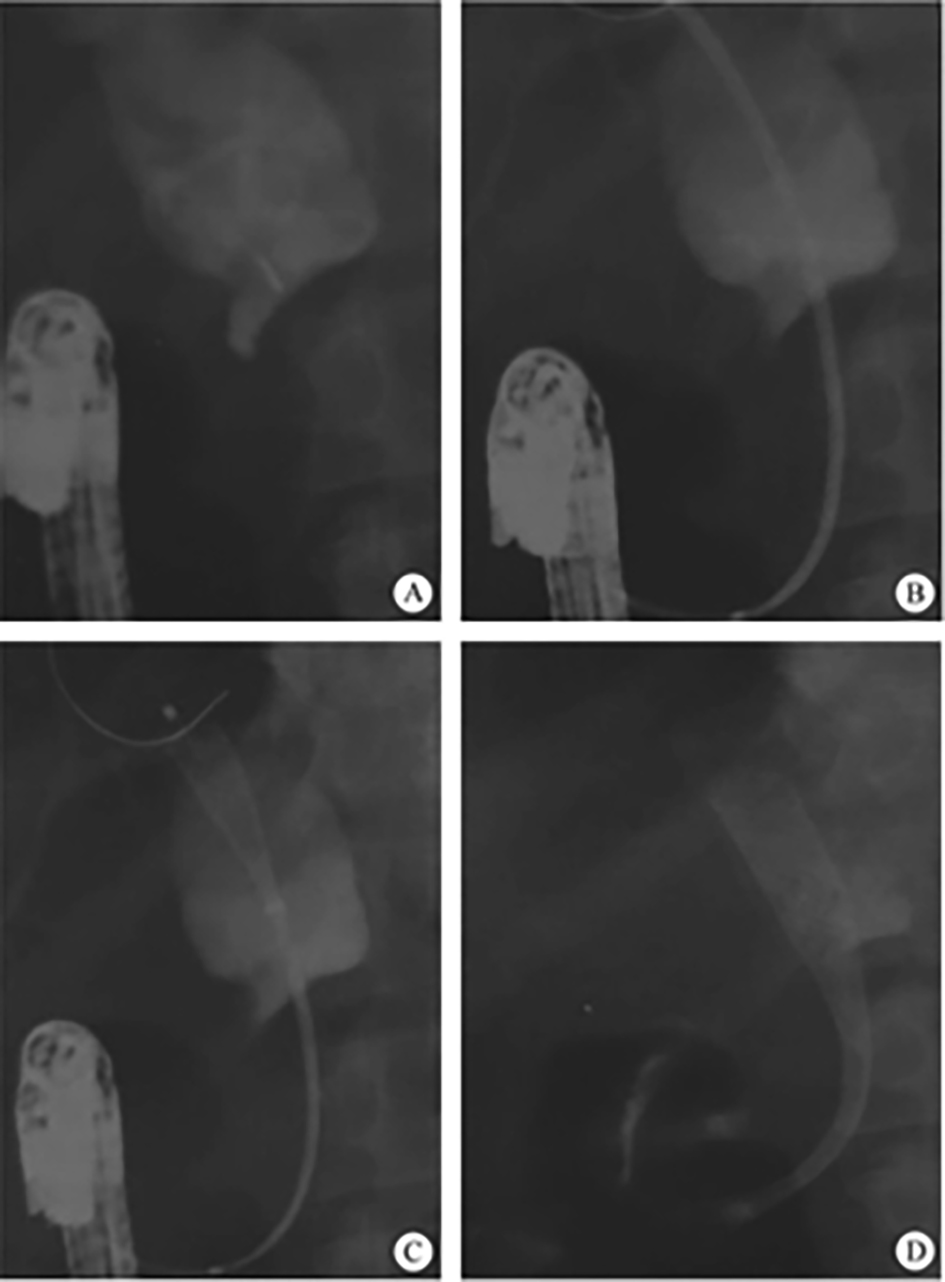

FIG. 11.6 — SEMS placement. A. Cholangiography shows stenosis of the distal bile duct with significant dilation of the proximal bile duct. B. The SEMS delivery system guide wire is inserted into the bile duct, with the proximal end of the stent positioned above the stenosis. C. The outer sheath of the stent begins to be withdrawn. D. The SEMS is fully deployed, and fluoroscopy reveals the formation of a waist at the stent due to the stenosis.
(III) Key procedural steps for endoscopic metallic stent placement (figure 11.6)
1. Guidewire Selection. The guidewire is an essential device for traversing biliary strictures, facilitating catheter advancement, and maintaining biliary access during device exchanges. A 0.035-inch or 0.025-inch guidewire with a stiff shaft and hydrophilic tip is typically preferred to optimize device maneuverability. For hilar biliary strictures, parallel placement of bilateral stents in a Y configuration is required to relieve obstruction. This necessitates advancing multiple guidewires into specific segmental bile ducts for targeted drainage.
2. Stent size and positioning. A 10-mm diameter SEMS is the most commonly used, with the length determined by the length and location of the stenosis. The choice of the length of the stent should also take into account whether the stent is placed above or across the papilla. After deployment, the fully expanded SEMS should span at least 10 mm beyond both ends of the stenosis to prevent tumor ingrowth at the ends of the stent. Some stents may shorten slightly after complete expansion, and this should be considered when calculating the required stent length. When the stent is placed above the papilla, the sphincter of Oddi remains intact, which, if sphincterotomy has not been performed, may reduce the risk of duodenal contents refluxing into the bile duct. This placement is most commonly used for strictures at the hepatic hilum or common hepatic duct, where the length of the SEMS is insufficient to span across the papilla. However, if the stricture is too long, multiple stents can be stacked. A potential drawback of placing the SEMS above the papilla is that it may complicate future access to the stent lumen for reinsertion in the case of stent occlusion. Placement of the SEMS across the papilla is typically used for common bile duct obstruction, with the stent extending from 3 to 10 mm into the duodenal lumen to facilitate future access for stent manipulation. However, if the stent extends too far into the duodenum, it may increase the mechanical risk of damaging the papillary side of the duodenal wall, potentially causing ulcers, bleeding, or even perforation. Care should be taken to avoid this complication. Regardless of whether the SEMS is placed above or across the papilla, sphincterotomy is not always necessary. Due to the small diameter of the SEMS delivery system and the radial expansion force of the stent, it can be fully expanded within 48 h, eliminating the need for prior dilation of the stenotic segment.
3. Deployment of self-expanding metal stents under endoscopic and fluoroscopic guidance. In addition to high-quality cholangiography to determine the length and location of the biliary obstruction, MRCP or CT is also important for preoperative evaluation of patients with suspected hilar biliary obstruction, as it helps guide drainage planning (e.g., unilateral, bilateral, or multisegmental drainage). The port of the stent delivery system (depending on the stent design) should be lubricated with a saline solution to facilitate stent advancement along the guide wire and removal of the outer sheath. The tip of the duodenoscope should be positioned as close as possible to the papilla to prevent inadvertent displacement of the guidewire when inserting the rigid delivery catheter into the papillary orifice. Endoscopists and assistants must coordinate to ensure that the guidewire remains stable while advancing the delivery catheter. The fluoroscopic markers at both ends of the SEMS usually indicate the stent’s position before and after deployment. When the stent is placed across the papilla, continuous endoscopic monitoring allows for tracking of the distal edge of the SEMS. Once the SEMS delivery system reaches the target position, the assistant gradually withdraws the outer sheath or traction string to release the stent. The stent is deployed by “withdrawing the outer sheath,” which pushes the stent out of the delivery system. As the stent expands, its length shortens accordingly. The physician should gradually retract the delivery catheter to precisely adjust the stent position. As the outer sheath is withdrawn, the proximal end of the stent gradually expands. The delivery system can be retracted to adjust the stent position distally. If the stent needs to be moved proximally, the outer sheath can be advanced to recapture the partially deployed stent. After the outer sheath is fully withdrawn, the delivery catheter and guide wire should also be removed. If the SEMS crosses a very tight stricture, it may not immediately fully expand radially, and withdrawing the delivery catheter may cause the stent to shift. To prevent this, the outer sheath should be advanced while withdrawing the delivery catheter to prevent the catheter’s tip from catching on the stent mesh, which could impede smooth removal. Once the stent is deployed, its position cannot be adjusted proximally, but if adjustment or removal is necessary distally, a foreign body forceps can be used to manipulate the stent.
(Ⅳ) Hot-spot issues and research progress in the consensus
1. Treatment of benign biliary strictures. The most effective approach for managing biliary strictures is the gradual increase in the number of plastic stents inserted, supported for more than one year (with stent replacement every three to four months). The multi-stent insertion strategy has a high efficacy, but it requires multiple ERCP sessions and depends on patient compliance. FCSEMS (fully covered self-expanding metal stents) is another option. Compared to the multi-plastic-stent approach, it avoids multiple ERCP procedures and the need for continuous insertion and replacement of plastic stents. However, it is essential to avoid placing uncovered metal stents, as tissue ingrowth into the stent mesh can cause stent occlusion and make removal impossible.
Biodegradable biliary stents hold great potential in the treatment of biliary strictures. These stents require only a single ERCP procedure for placement and do not need to be removed. Currently, several different biodegradable materials are being tested (such as polylactic acid, polycaprolactone, and poly(ester amide)), but their clinical application is still under observation. The primary drawbacks of these stents are the gradual decrease in radial expansion force as the stent degrades, and the potential for inflammatory foreign body reactions leading to hyperplasia.
2. Stent selection for malignant biliary strictures. The selection of metal versus plastic stents and whether to use unilateral or bilateral drainage remains a controversial issue. Compared to plastic stents, metal stents offer longer patency, higher success rates, and a lower incidence of cholangitis in the treatment of hilar or proximal bile duct cancers. However, metal stents are more expensive than plastic ones. For patients with malignant biliary obstruction and a life expectancy exceeding four months, metal stents are often more cost-effective than plastic stents because they require fewer interventions. However, for patients with a life expectancy of less than four months, plastic stents may be more cost-effective.
Due to the risk of obstruction of the intrahepatic bile ducts on the same side or the contralateral side by the covered metal stents, bare metal stents should be placed in cases of hilar and proximal bile duct lesions. Various stent types and techniques can be used. The most commonly used technique involves placing guide wires in each bile duct intended for drainage and sequentially inserting SEMS in parallel along these guide wires. Another option is to place a Y-shaped stent at the hepatic hilum, where one metal stent is inserted through the mesh of another stent into the contralateral bile duct.
Metal stent occlusion is the most significant issue in patients with advanced cholangiocarcinoma. The primary causes of occlusion include tumor ingrowth at both ends of the stent, tissue hyperplasia, and blockage due to bile sludge or debris. Current methods for managing SEMS occlusion include placing a plastic stent within the SEMS or stacking additional metal stents.
11.2.4 Biliary Stricture: Diagnosis and Treatment with Peroral Cholangiopancreatoscopy (POC) Under Direct Visualization
(I) Overview
The use of cholangioscopy for direct visualization of the bile ducts in the management of biliary strictures is becoming increasingly prevalent. This includes the evaluation of IBS (inflammatory biliary strictures), cholangioscopic biopsy, and wire-guided cannulation under direct visualization. Compared to conventional ERCP, peroral cholangiopancreatoscopy (POC) allows for direct observation of lesions within the bile ducts, considerably improving the accuracy of the diagnosis.
(II) Application of peroral cholangiopancreatoscopy in unexplained biliary strictures
IBS can be categorized into benign and malignant strictures. The former is primarily caused by various inflammatory biliary and pancreatic diseases, congenital anomalies, and post-surgical biliary injury. The latter is mainly associated with malignant tumors such as pancreatic cancer, cholangiocarcinoma, and gallbladder cancer. For most benign biliary strictures, endoscopic or medical treatments can provide relief. In contrast, for malignant biliary strictures, curative surgical intervention must be considered. Therefore, distinguishing between benign and malignant IBS is crucial for clinical decision-making.
However, standard ERCP has relatively low sensitivity for diagnosing malignant IBS (cell brush cytology: 23%–56%, biopsy: 33%–65%). The clinical application of peroral cholangiopancreatoscopy provides a new approach for differentiating IBS. Unlike the blind biopsy technique of ERCP, peroral cholangiopancreatoscopy allows for targeted biopsy under direct visualization. A multicenter randomized controlled trial has demonstrated that, compared to ERCP brushing, peroral cholangiopancreatoscopy-directed biopsy increases the sensitivity of the diagnosis (68.2% vs. 21.4%). An observational study published in Gastrointestinal Endoscopy (GIE) showed that single-operator cholangioscopy has high diagnostic value for IBS, with an overall accuracy of 89.5%, a sensitivity of 89.1%, and a specificity of up to 90%. Cholangioscopy also proves to be valuable in differentiating benign and malignant strictures in cases of primary sclerosing cholangitis. A retrospective study revealed that cholangioscopy has significantly higher sensitivity (92% vs. 66%, P = 0.25), specificity (93% vs. 51%, P < 0.001), and accuracy (93% vs. 55%, P < 0.001) compared to standard cholangiography.
Previous studies have identified four key features of malignant IBS: tumor vasculature (irregularly dilated and tortuous mucosal blood vessels), papillary projections, nodular/polyploid masses, and infiltrative lesions. However, the ideal diagnostic criteria for distinguishing IBS lesions through imaging remain to be further refined. Recently, Chinese scholars, based on high-definition images, proposed new diagnostic criteria for differentiating IBS lesions during peroral cholangiopancreatoscopy (table 11.1, figure 11.7), with the aim of enhancing the diagnostic capabilities of cholangioscopy in IBS cases.
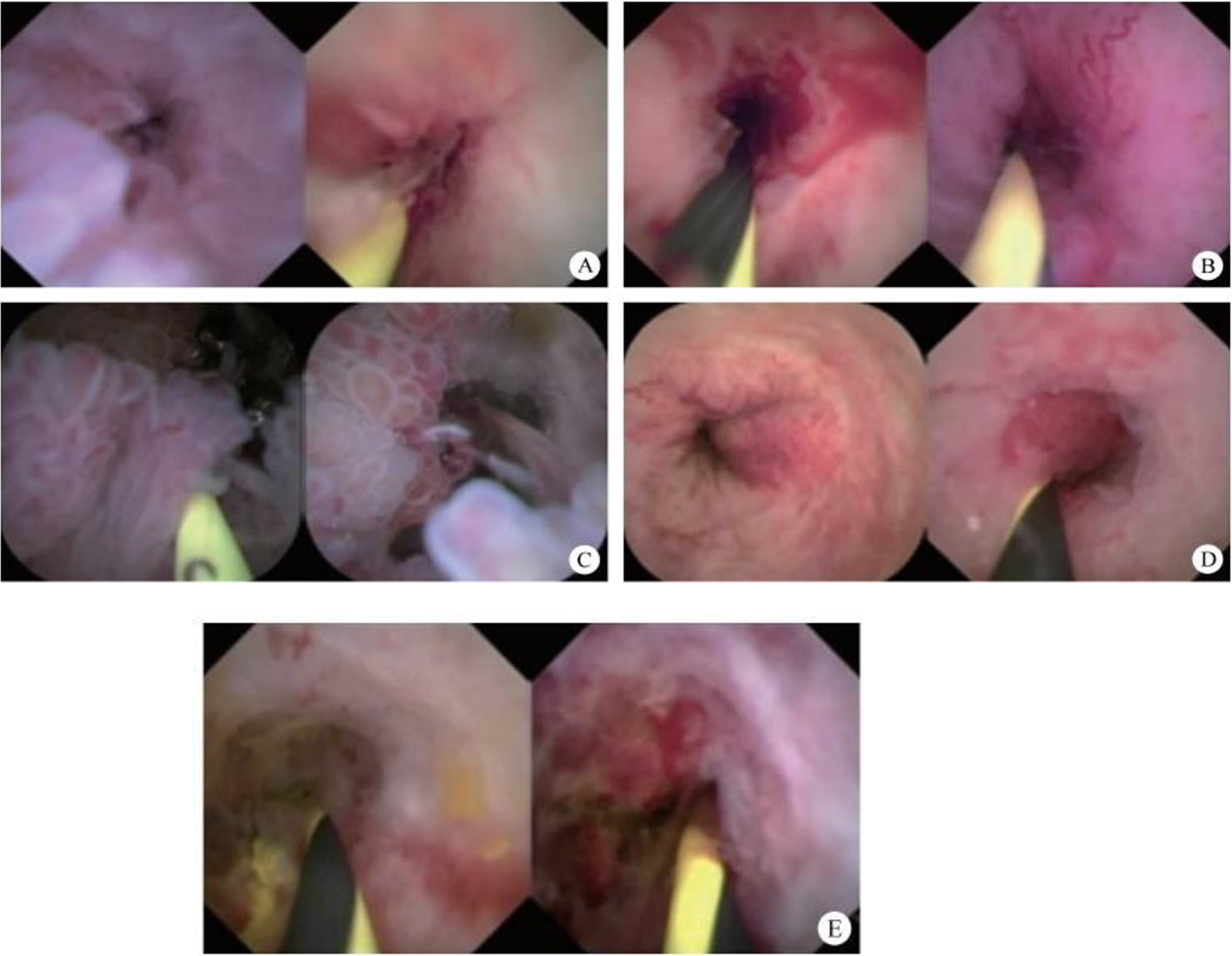
TAB. 11.1 — New criteria for differentiating the nature of IBS lesions in peroral cholangiopancreatoscopy (POC). Super minimally invasive diagnostic procedure.
| No. | Tumor-like IBS Features |
| 1 | Microvillus structures (figure 11.7A) |
| 2 | Irregular blood vessels with easy bleeding (figure 11.7B) |
| 3 | Lobular papillary structures (figure 11.7C) |
| 4 | Dam-like elevations (figure 11.7D) |
| 5 | Mucosal erosion and rough texture (figure 11.7E) |
| Diagnostic criteria | If one of the above five features is present, the lesion can be classified as a tumor-like lesion. If none of the features are present, the lesion can be classified as non-tumor-related. |

FIG. 11.7 — Typical manifestations of the new criteria for differentiating the nature of IBS lesions in peroral cholangiopancreatoscopy (POC) Super minimally invasive diagnostic procedure. A. Microvillus structures visible in the bile duct under cholangioscopy. B. Irregular blood vessels visible in the bile duct under cholangioscopy. C. Lobular papillary structures visible in the bile duct under cholangioscopy. D. Dam-like elevations visible in the bile duct under cholangioscopy. E. Mucosal erosion visible in the bile duct under cholangioscopy.
(III) Application of peroral cholangiopancreatoscopy (POC) in assisting wire cannulation for biliary strictures
The key procedure in ERCP for the treatment of biliary strictures is the passage of a guidewire through the stricture, which is essential for subsequent balloon dilation or stent placement. Studies show that 60%–80% of biliary strictures can be successfully treated with stent drainage via ERCP. For those with failure, research indicates that peroral cholangiopancreatoscopy (POC) can overcome this challenge by locating the small stricture opening under direct visualization and passing the guidewire through it. For patients with difficulties in wire cannulation, the success rate of guidewire cannulation under direct visualization with POC remains from 60% to 73.9%. Bokemeyer et al. reported a 70% success rate in 30 cases of difficult biliary strictures where the conventional ERCP method failed, with benign strictures having a significantly higher success rate than malignant strictures (86.2% vs. 44.2%).
(IV) Procedural steps
The peroral duodenoscope is advanced to the descending part of the duodenum. Standard techniques, such as conventional cannulation and double guidewire methods, are used to achieve cannulation of the common bile duct. Once selective common bile duct cannulation is successful, duodenal papillotomy can be performed as needed. After entering the common bile duct along the guide wire, a suitable amount of saline solution is used to irrigate the duct, providing a clear view for evaluating the biliary stricture and intrahepatic bile ducts. If the guidewire cannot pass through the stricture under ERCP fluoroscopy, cholangioscopy can be used to guide the placement of the guide wire. The method for placing the guide wire under direct cholangioscopic visualization depends on the nature of the stricture: ① If the stricture is a pinhole-like narrowing, which is continuous, the scope is adjusted to align with the stricture. The guidewire is then passed through the stricture while maintaining a clear view. ② If the stricture is completely occluded (figure 11.8A, B), the guide wire may be difficult to pass even under direct visualization (figure 11.8C). In such cases, a stiffer guide wire with a hard tip is used to target the occluded area, and under fluoroscopic guidance, the stricture can be breached (figure 11.8D). After overcoming the stricture, a softer guide wire is used to navigate into the intrahepatic bile ducts, and cholangiography is performed to confirm successful breakthrough (figure 11.8E). After completing the cholangioscopic procedure, a plastic stent or fully covered self-expanding metal stent is placed across the stricture, depending on the type and location of the biliary stricture (figure 11.8F).
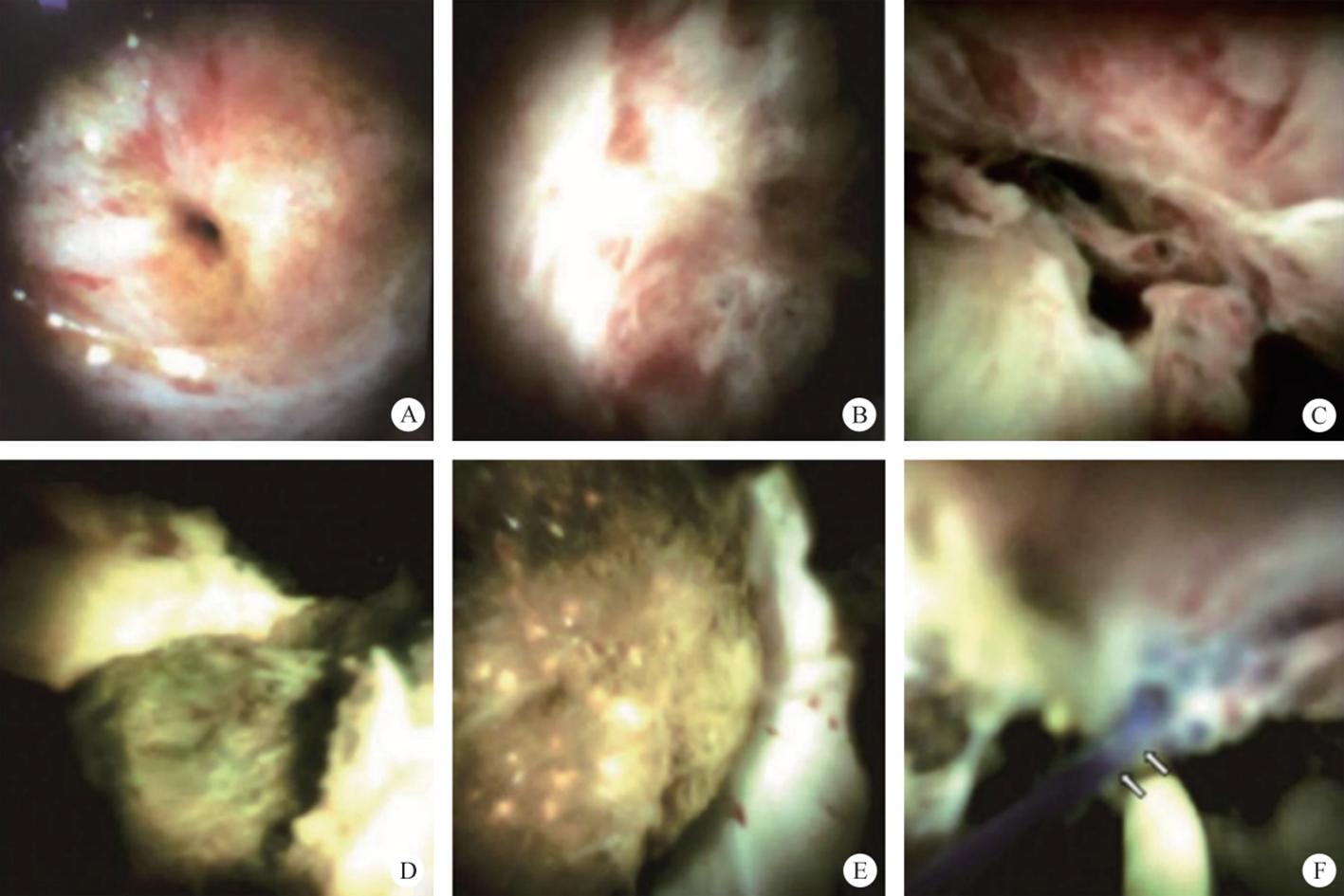

FIG. 11.8 — Endoscopic findings of different types of biliary strictures after liver transplantation. A. Pinpoint anastomotic stricture. B. Completely narrowed anastomotic stricture. C. Non-anastomotic stricture with extensive inflammatory hyperplasia and mild vascular proliferation. D. Non-anastomotic stricture with extensive ulceration and necrosis. E. Stone formation observed at the stricture’s proximal side. F. Non-absorbable sutures visible at the anastomotic stricture site (indicated by arrows).
(V) Hot-spot issues and research progress in the consensus
1. Diagnostic accuracy of cholangioscopy in unexplained biliary strictures. Peroral cholangiopancreatoscopy (POC) can provide a direct diagnosis through visual characteristics under direct observation. However, the ideal diagnostic criteria for IBS image differentiation are still under further development. To date, the Monaco standard, CRM standard, and Mendoza standard have been proposed. Kahaleh et al. conducted a study comparing the diagnostic efficacy of the Monaco standard and CRM standard. The results showed diagnostic accuracies of 61% and 57%, respectively. Another study confirmed a diagnostic accuracy of 77% for the Mendoza standard. Although the image quality of peroral cholangiopancreatoscopy has continually improved, it still lags behind that of gastrointestinal endoscopy, which is the main reason for the current limitations in the diagnostic accuracy of the aforementioned criteria.
2. Endoscopic super minimally invasive snare electrocoagulation resection under direct visualization. For biliary strictures caused by biliary polypoid lesions, endoscopic resection can restore biliary patency. Currently, Professor Enqiang Ling’s team is conducting exploratory research on Super minimally invasive snare electrocoagulation resection under direct visualization with peroral cholangiopancreatoscopy. This technique allows the damaged tissue to be removed under cholangioscopic visualization, and its efficacy and feasibility have been experimentally verified in an in vitro simulated biliary and pancreatic duct fluid environment. This research lays the foundation for the clinical practice of super minimally invasive snare electrocoagulation resection under direct visualization with peroral cholangiopancreatoscopy in the future.
11.3 Super Minimally Invasive Drainage for Biliary Fistulae
(I) Overview
Biliary fistula (biliary leaks) is the leakage of bile or bile-containing fluids from a breach in the biliary system into the abdominal cavity or externally. The abnormal accumulation of bile outside the biliary system is termed a biloma. According to the International Study Group of Liver Surgery (ISGLS), the definition of biliary fistula includes a post-operative period of three days or more, with the bilirubin concentration in the drainage fluid being at least three times higher than the normal plasma bilirubin concentration, or requiring interventional or surgical treatment due to bile accumulation or biliary peritonitis.
Common causes of biliary fistulas include hepatobiliary surgeries, trauma, inflammation, and tumors. Once a biliary fistula occurs, timely intervention is essential; otherwise, complications such as infection and electrolyte imbalances may arise, with mortality rates reaching 40%–50% in severe cases. Treatment options for biliary fistula include conservative medical management, interventional therapy, and traditional surgical procedures. However, surgical treatment is more invasive, with a mortality rate as high as 6.67%. With the introduction of the super minimally invasive concept and the development of corresponding techniques, there have been significant advancements in the endoscopic diagnosis and treatment of biliary fistulas. Super minimally invasive endoscopic drainage procedures for biliary fistulas include endoscopic nasobiliary drainage (ENBD), endoscopic retrograde biliary drainage (ERBD), and endoscopic ultrasound-guided biloma drainage (EUS–BLD).
(II) Diagnosis of biliary fistula
The diagnosis of biliary fistula requires a combination of medical history, clinical presentation, and relevant auxiliary examinations. In cases where abdominal drainage tubes or T-tubes are placed post-surgery, or in patients with lesions penetrating the abdominal wall, bile leakage may be directly observed, confirming the diagnosis. For patients with a history of abdominal surgery or trauma, but with atypical clinical presentations, prompt auxiliary examinations should be performed for definitive diagnosis. Non-invasive imaging techniques, such as abdominal ultrasonography, are the preferred diagnostic methods for biliary fistula. Cholangiography remains the gold standard for diagnosing biliary fistulas. For hepatic or perihepatic cystic lesions, aspiration biopsy helps determine whether the lesion is a biloma.
Based on over 400 cases of biliary fistula treated at the First Medical Center of the PLA General Hospital over the past decade, and focusing on the relationship between bilomas and the biliary system via imaging, we proposed a clinical imaging classification for biliary fistulas. The classification divides biliary fistulas into the three following types. Type A (communicating with the bile duct): the bile duct above the fistula communicates directly with the bile duct or the digestive system below the fistula. This type is further subdivided into three subtypes (A1, A2, and A3) based on the location of the fistula. Type B (distal bile duct disruption): the bile duct above the fistula communicates with the bile duct or the digestive system below the fistula via a ‘biloma.’ Type C (isolated type): the bile duct above the fistula is completely disconnected from the bile duct or the digestive system below the fistula, with no communication. Figure 11.9 illustrates these three types.

FIG. 11.9 — Classification of biliary fistulae. Type A: Communicating with the bile duct; type B: distal bile duct disruption; type C: isolated type.
(III) Indications and contraindications for super minimally invasive biliary fistula drainage
In biliary fistula patients, the bile duct system is typically not dilated, making percutaneous transhepatic biliary drainage (PTBD) treatment more challenging. Therefore, PTBD is not considered a first-line treatment for biliary fistula, but may be used in cases where endoscopic or surgical treatments are not feasible or have failed. Percutaneous liver puncture drainage guided by ultrasound or CT is the traditional treatment for bilomas. Relative contraindications for PTBD include coagulation disorders, contrast agent allergies, and massive ascites.
Surgical treatment is indicated when endoscopic or interventional treatments fail, or in cases of worsening symptoms with diffuse peritonitis, bile duct rupture with active bleeding, or bile duct transection. Surgery should only be performed when secondary infections from the biliary fistula have been controlled.
(IV) Surgical procedures and techniques
1. Endoscopic nasobiliary drainage (ENBD, figure 11.10). In ENBD, the distal end of the nasobiliary catheter is inserted above the biliary fistula using a duodenoscope, with the external end exiting through the nasal cavity. This method provides continuous negative-pressure drainage of bile, reduces bile leakage, and allows for monitoring and recording the quantity and characteristics of the bile drainage. Through nasobiliary ductography, the healing process of the biliary fistula can be assessed, potentially avoiding the need for repeated ERCP. ENBD is suitable for the treatment of biliary fistulas in critically ill patients or those who cannot tolerate surgery. However, it is not recommended for patients with hepatic encephalopathy, infectious toxic encephalopathy, severe gastrointestinal varices, or those requiring long-term drainage.
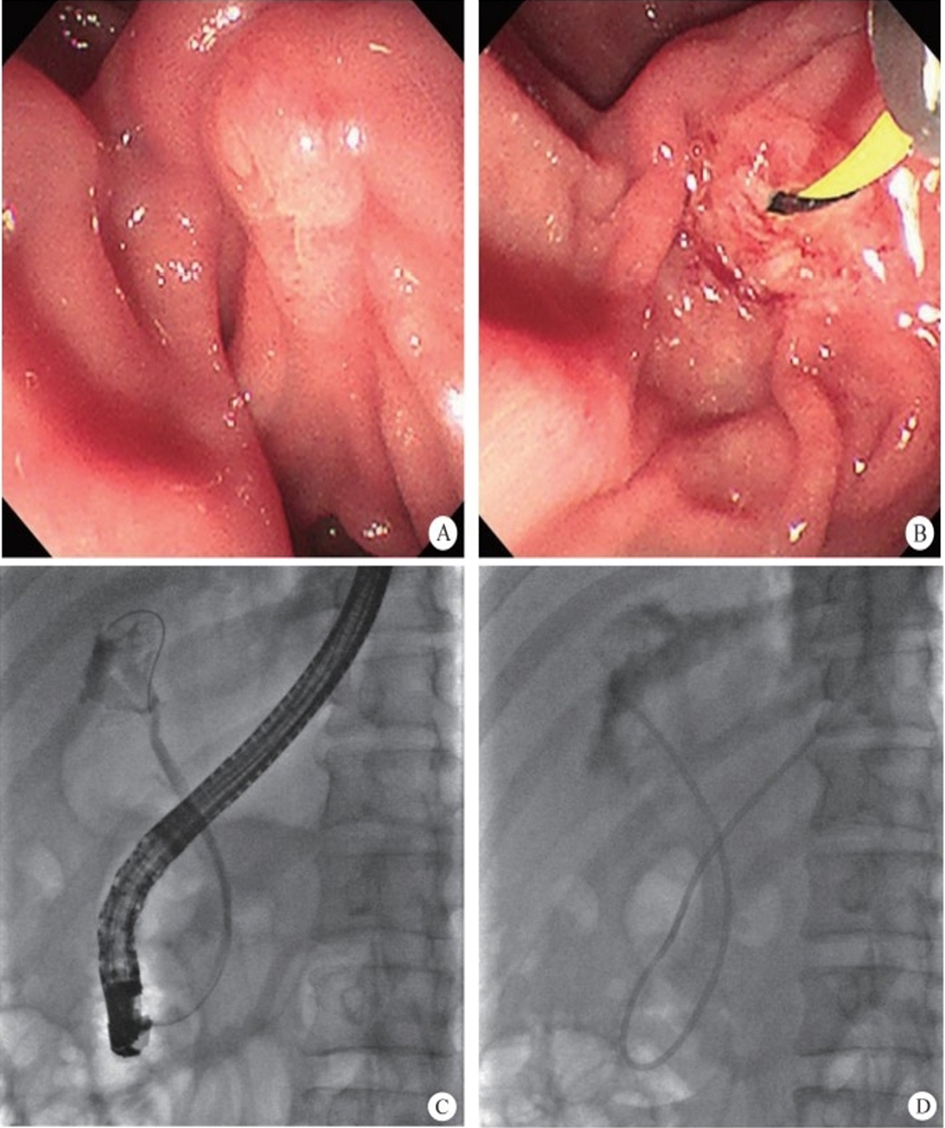

FIG. 11.10 — Super minimally invasive drainage for biliary fistulae – ENBD. A. The duodenal papilla is visible under endoscopy. B. A curved knife is used to guide the guidewire for selective biliary cannulation. C. After successful selective biliary cannulation, cholangiography shows the contrast agent diffusing from the left hepatic duct into the abdominal cavity. D. A nasobiliary drainage catheter is inserted along the guide wire.
2. Endoscopic retrograde biliary drainage (ERBD, figure 11.11). Under endoscopy, a plastic or metal stent is placed within the bile duct to support drainage, reduce bile leakage, and prevent secondary biliary strictures. This procedure is used in patients with biliary fistulas complicated by biliary strictures.
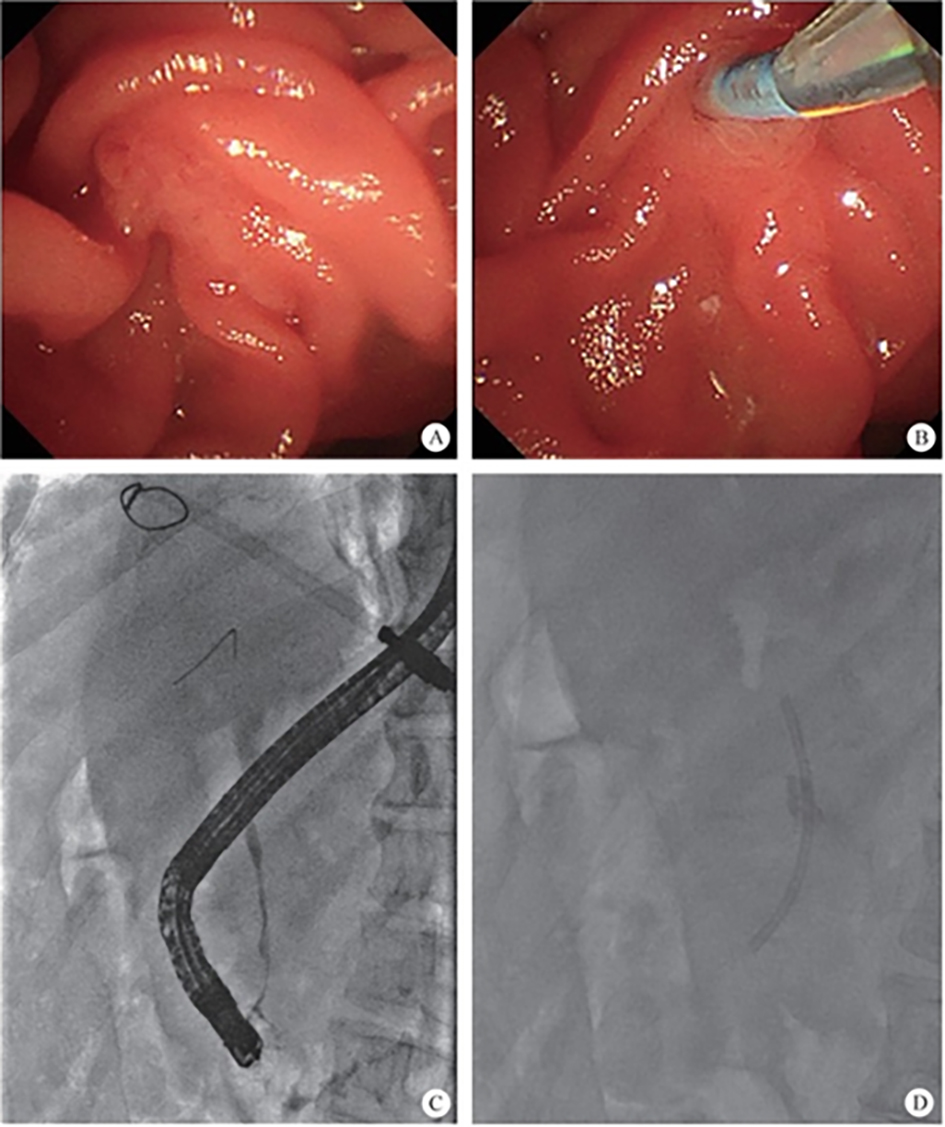

FIG. 11.11 — Super minimally invasive biliary fistula drainage – ERBD. A. Endoscopic view of the duodenal papilla. B. The sphincterotome carries the guide wire to super-select the common bile duct. Contrast shows localized extravasation of contrast in the lower bile duct. C. A plastic bile duct stent is placed along the guide wire. D. A plastic stent (8.5F, 5 cm) is placed along the guide wire.
3. EUS-guided biloma drainage (EUS–BLD). Asymptomatic or small bilomas (less than 5 cm in diameter) can generally be treated conservatively. However, for bilomas that are symptomatic or larger than 5 cm, drainage treatment is required. Compared to the traditional ultrasound or CT-guided percutaneous hepatic puncture drainage, EUS–BLD offers the following advantages: internal drainage is more aligned with physiological characteristics and avoids the increased infection risk caused by the movement of external drainage tubes; puncture and drainage in the hepatic tail lobe are easier.
(V) Hot-spot issues and research progress in the consensus
1. The New classification of biliary leaks and its importance in endoscopic treatment. The new classification of biliary leaks (types A, B, and C) is important for guiding endoscopic treatment options. Type A is mostly caused by iatrogenic factors or trauma, and treatment involves ENBD, ERBD, or PTBD to lower biliary pressure and promote healing. For leaks near the papilla, EST can also be considered. Early intervention is recommended for this type. Type-B treatment options include EUS–BLD, PTBD, or, if necessary, re-biliary-enteric anastomosis. Type-C is usually caused by trauma, battlefield injuries, or post-surgical complications. Asymptomatic cases can be observed, while symptomatic cases or those with larger bilomas (over 5 cm) should undergo EUS–BLD or puncture drainage guided by CT or ultrasound.
2. EUS–BLD: a promising new treatment for type-C biliary leaks. EUS–BLD has a 100% technical success rate for treating bilomas with a median diameter of 68.5 mm, and a clinical success rate of 83.3% (5/6). In one case, endoscopic necrotic tissue removal was required, but the final clinical success rate remained 100%. There were no recurrences or treatment-related complications during a median follow-up period of 83.5 days. Most cases used double-pigtail stent drainage, and in recent years, metal stents such as double mushroom-head metal stents and FCSEMS have been applied, showing good prospects.
3. The impact of super minimally invasive Drainage on the removal rate of external drainage tubes in biliary leaks. A multicenter retrospective study from 13 institutions enrolled 99 patients with biliary leaks from April 2014 to March 2019. The study reviewed 66 cases of biliary leaks post-cholecystectomy (n = 17) or liver resection (n = 49). The results showed that for biliary leaks after cholecystectomy, 100% of patients successfully removed their external drainage tubes after the first endoscopic intervention (16/17 cases). In contrast, after liver resection, the external drainage removal rate was 44.9% (22/49), with all 22 patients eventually avoiding further drainage. A lower body mass index (BMI) was the only significant factor related to avoiding external drainage after liver resection for biliary leaks.
11.4 Super Minimally Invasive Surgery for Gallbladder Stones and Cholecystitis
11.4.1 Overview
Gallstones are a common biliary tract disease, and their formation is associated with various factors, such as improper dietary habits, obesity, and diabetes. Gallstones can lead to both acute and chronic cholecystitis, with primary symptoms including right upper abdominal pain, nausea, vomiting, and jaundice. Currently, laparoscopic cholecystectomy and medication are the main treatment options.
Laparoscopic cholecystectomy is an effective treatment method, allowing for minimally invasive removal of the diseased gallbladder, thus curing the disease. However, cholecystectomy may lead to post-cholecystectomy syndrome and other complications, which can affect the patient’s digestive function. Medication treatment typically involves oral ursodeoxycholic acid or other stone-dissolving drugs. However, a drug treatment takes a long time, and some patients may not be able to completely dissolve the stones.
Over 80% of patients with gallstones are asymptomatic. When the gallbladder outlet becomes obstructed by stones, persistent abdominal pain can occur. The pain typically manifests in the right upper quadrant but can also occur in the upper abdomen, behind the breastbone, or in the left upper quadrant. This pain often radiates to the same-side shoulder blade and may be accompanied by vomiting, bloating, indigestion, and abdominal distension. In cases of cholecystitis, serum transaminase levels may be elevated, and there may be an increase in neutrophil and white blood cell counts.
Abdominal ultrasound has good sensitivity and specificity for detecting gallstones larger than 1.5 mm. It can provide information on the size, load, and mobility of the gallstones, as well as the gallbladder volume, wall thickness (which may indicate acute or chronic cholecystitis, or diffuse thickening of the gallbladder wall greater than 3 mm after meals), and bile duct diameter.
Endoscopic ultrasound (EUS) is useful in detecting small gallstones that might not be identified by other methods, such as microstones that abdominal ultrasound cannot detect.
11.4.2 Gallbladder Stones: Super Minimally Invasive Stone Removal
1. Overview
The super minimally invasive stone removal technique via the duodenal papilla for gallbladder stones, also known as super minimally invasive gallbladder stone extraction, refers to the endoscopic removal of gallstones through the oral cavity. This procedure not only avoids cholecystectomy but also preserves the function of the sphincter of Oddi, offering a novel surgical approach.
While ERCP (Endoscopic retrograde cholangiopancreatography) has become an established technique for treating bile duct stones, it has limitations when it comes to treating gallstones, primarily due to the difficulty in selecting a guide wire under fluoroscopic guidance to access the cystic duct. The advent of the peroral cholangioscopy technique has significantly reduced the difficulty of guide wire selection under direct visualization, improving the success rate of cystic duct cannulation. Furthermore, theoretically, peroral cholangioscopy enables stone extraction using both aspiration and basket retrieval techniques under direct visualization. Thus, the development of peroral cholangioscopy and related instruments has made it possible to perform gallbladder stone extraction via the duodenal papilla approach, offering a promising alternative to traditional methods.
2. Indications and contraindications
(1) Indications: ① gallbladder sludge-like stones; ② small single gallstones; ③ patients with gallstones with a strong desire to preserve the gallbladder; ④ patients with gallstones with poor health, unsuitable for cholecystectomy; ⑤ patients with gallstones with a normal-sized and shaped gallbladder.
(2) Contraindications: ① During acute cholecystitis; ② abnormal gallbladder morphology; ③ patients with gallstones highly suspected of having gallbladder cancer; ④ larger and multiple gallbladder stones.
3. Preoperative preparation
(1) Gastrointestinal preparation. A liquid diet must be started one to two days before surgery, followed by fasting for 6 h and abstaining from water for 2 h before anesthesia. For colorectal surgery, oral laxatives or enemas may be used to cleanse the bowel.
(2) Preoperative examination. Abdominal CT must be performed to clarify the location and nature of the stones. Complete blood count, blood biochemistry, coagulation function, electrocardiogram, echocardiogram, and other relevant tests should be completed.
(3) Discontinuation of medications. Medications that affect coagulation must be discontinued.
aspirin: stop seven days before surgery; low-molecular-weight heparin: stop three days before surgery;
heparin: stop 6–12 h before surgery; warfarin: stop three days before surgery.
4. Surgical steps and techniques (figure 11.12)
(1) First, perform biliary cannulation using a double guidewire technique. Cholangiography can reveal common bile duct (CBD) stones. Then, place a papillary dilator (12 mm in diameter, 30 mm in length) over the guidewire at the lower end of the bile duct. A large amount of bile and sludge-like stones will be seen flowing out from the opening of the papillary dilator under endoscopic guidance (figure 11.12A).
(2) Balloon dilation of the papillary dilator is performed, followed by the insertion of an oral choledochoscope over the guidewire into the CBD (figure 11.12B).
(3) Further inspection is carried out. If no stones are found in the left or right hepatic ducts or CBD, it is confirmed that the CBD stones have been cleared into the duodenum following the placement of the papillary dilator and endoscopic aspiration.
(4) A specially designed basket is inserted through the working channel of the oral choledochoscope into the CBD, and its opening and closing are verified. This ensures the basket can successfully remove the CBD stones under direct visualization.
(5) Perform a cholangiogram again to confirm that no stones remain in the CBD.
(6) Next, another thinner oral choledochoscope is inserted sequentially through the fine guidewire (0.25 mm) into the CBD, cystic duct, and ultimately into the gallbladder. Multiple sludge-like stones are found in the gallbladder, which are then successfully aspirated and removed (figure 11.12C, D).
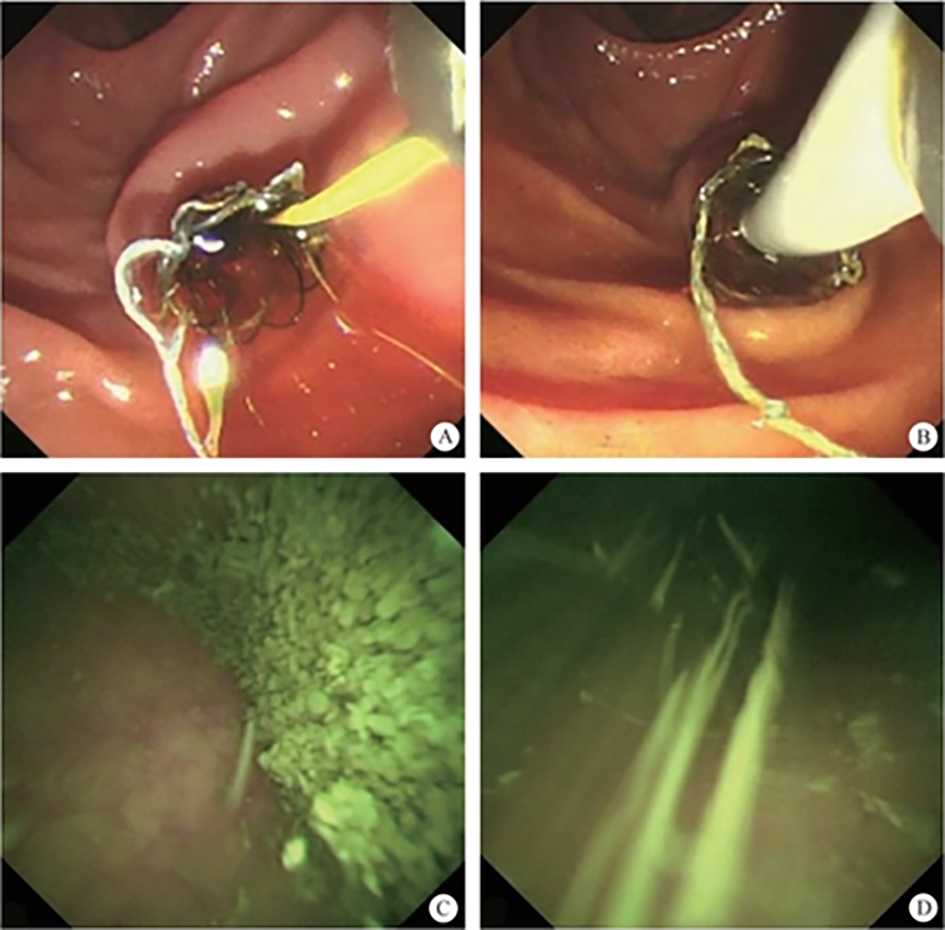

FIG. 11.12 — Procedure of gallbladder stone removal assisted by oral choledochoscopy. A. The papillary dilator (12 mm in diameter, 30 mm in length) is placed at the lower end of the bile duct over the guide wire. Under endoscopic guidance, a large amount of bile and sludge-like stones can be seen flowing out from the opening of the papillary dilator. B. The oral choledochoscope is inserted through the guide wire and placed into the common bile duct (CBD) via the papillary dilator. C. The oral choledochoscope reveals numerous sludge-like stones in the gallbladder. D. Using aspiration techniques under direct visualization via the oral choledochoscope, the sludge-like stones are successfully removed.
5. Postoperative management
(1) Closely monitor the patient’s vital signs and abdominal physical examination, as well as the presence of hematemesis or melena. Blood tests should be performed at 24 h and 72 h postoperatively, including complete blood count, serum amylase, and lipase levels. If clinical assessment suggests gastrointestinal bleeding, perforation, or post-endoscopic retrograde cholangiopancreatography (PEP) complications, timely abdominal CT and/or endoscopic examination should be conducted to clarify the condition.
The patient should fast from food and water for six days postoperatively. If there are no abnormal abdominal signs and postoperative laboratory results show no significant abnormalities, the patient can gradually switch from liquid to semi-liquid diet. At six days postoperatively, proton pump inhibitors (PPIs) and antibiotics are administered. If postoperative pancreatitis or gastrointestinal bleeding occurs, the duration of the treatment may be extended accordingly. Furthermore, if pancreatitis develops, somatostatin therapy should be promptly initiated. It should be noted that for gastrointestinal bleeding, if conservative treatment fails, timely endoscopic, surgical, or interventional measures should be employed to control the bleeding.
6. Postoperative follow-up
It is recommended to closely monitor adverse events through telephone and outpatient follow-up. Patients will be informed and encouraged to return for endoscopic re-examination at three months, six months, one year, two years, three years, four years, and five years postoperatively.
7. A classic case
Patient history: the patient, a middle-aged man presented with “abdominal pain for over one month.” Abdominal CT revealed sludge-like stones in the gallbladder.
Diagnosis: multiple gallstones in the gallbladder.
After thorough preoperative evaluation and signing of the informed consent form, the patient underwent SMIS choledochoscopy and stone extraction for gallbladder sludge-like stones (Refer to video 11.2, Gallbladder stone removal via super minimally invasive stone fragmentation and extraction at the duodenal papilla (See online resources)).
Surgical procedure: the patient was placed in the supine position under general anesthesia for examination. The surgery was performed using a duodenoscope and an oral cholangioscope.
(1) Cholangiocath insertion was performed. (2) The oral cholangioscope was then advanced along the guide wire into the common bile duct (CBD). Under direct visualization, the angle was adjusted to allow the guide wire to selectively enter the cystic duct, while another guidewire was placed into the common bile duct. (3) A dilation catheter (10Fr) was advanced along the guide wire to perform cystic duct dilation. (4) Plastic drainage stents were placed into both the cystic duct and the common bile duct along the guide wires. A significant amount of sandy-like stones was observed flowing out from the cystic duct. (5) Six days later, another oral cholangioscope examination was performed, and the plastic stents from the common bile duct and cystic duct were removed. (6) A cholangiocath was inserted again, and the oral cholangioscope was advanced along the guide wire into the common bile duct. The angle was adjusted under direct visualization to allow the guide wire to selectively enter the cystic duct. (7) The oral cholangioscope advanced along the guide wire into the gallbladder, where sandy-like stones were observed. After thorough irrigation and suction, the stones were successfully removed.
8. Hot-spot issues and research progress in the consensus
Gallbladder stones are a common condition, with an incidence rate of approximately 5%–22% among adults in Western countries and 10%–15% in China, making it one of the most prevalent digestive diseases worldwide. Currently, laparoscopic cholecystectomy is considered the standard treatment for gallbladder stones. However, the gallbladder plays several important roles in the human body, and its removal may lead to adverse events such as gastroparesis, gut microbiota disruption, dysfunction of the sphincter of Oddi, and post-cholecystectomy syndrome. More importantly, cholecystectomy may not be suitable for patients with poor health condition. Therefore, it is essential to develop a technique for stone removal from the gallbladder via the duodenal papilla while preserving the gallbladder.
ERCP can be used under fluoroscopy with metal baskets or balloons to remove common bile duct stones. However, due to the limitations of X-ray indirect visualization, traditional ERCP faces difficulties in selectively cannulating the cystic duct, resulting in a lower success rate. The peroral cholangioscope, when used under direct visualization, facilitates better selective cannulation of the cystic duct and improves the success rate of this procedure. The safety and feasibility of removing sandy-like stones from the gallbladder using oral cholangioscopy under direct visualization have been preliminarily evaluated as part of research. For smaller, solitary gallbladder stones, we will also attempt to perform basket stone removal from the gallbladder under direct visualization using the oral cholangioscope.
Peroral cholangioscope-assisted gallbladder stone removal has the potential to provide a direct-visualization approach for treating gallbladder stones. The potential advantages of this technique are as follows. 1. Compared to traditional ERCP, where X-ray fluoroscopy is used, direct-visualization cholangioscopy allows easier and more accurate selective cannulation of the cystic duct by adjusting the cholangioscope angle. 2. With clear visualization, the stone basket can be deployed at the most appropriate position, enabling timely and effective stone retrieval. 3. The operator can promptly detect adverse events, such as perforation, bleeding, or injury to the cystic duct. 4. Postoperative examination can confirm whether there are any residual bile duct stones. 5. The function of the gallbladder can be preserved, avoiding post-cholecystectomy syndrome and other adverse events related to the loss of the gallbladder. 6. The per-duodenal papillary approach is associated with less trauma.
11.4.3 Percutaneous Ultrasound-Endoscopic Guided Aspiration for Acute Suppurative Cholecystitis
1. Overview
Acute suppurative cholecystitis is an acute purulent inflammation of the gallbladder caused by bacterial infection, involving the entire thickness of the gallbladder wall. This leads to purulent exudates within the gallbladder lumen, and in severe cases, it can result in life-threatening complications such as gallbladder gangrene, gallbladder perforation, biliary fistula, and acute peritonitis. The most common cause is bacterial infection occurring on the basis of gallbladder obstruction, usually due to gallstones lodged in the neck or cystic duct. Typical symptoms include right upper abdominal pain, fever, nausea, and vomiting, with severe cases potentially leading to septic shock and acute peritonitis symptoms.
Ultrasound-endoscopic-guided aspiration for acute suppurative cholecystitis, also known as ultrasound-endoscopic guided fine needle aspiration of the gallbladder, is a technique in which a percutaneous approach is used to aspirate pus from the gallbladder under the guidance of ultrasound and endoscopy. The procedure involves scanning the gallbladder through the stomach or duodenum using an ultrasound-endoscope to identify the puncture site, followed by aspiration of the purulent fluid using a needle. This technique is one of the treatments for acute suppurative cholecystitis and is classified as a super minimally invasive surgery, which removes the disease while preserving the anatomical structure of the organ.
2. Diagnosis of acute suppurative cholecystitis
Clinically, acute suppurative cholecystitis presents with pain in the right upper abdomen or epigastrium, often accompanied by fever. Physical examination typically reveals tenderness in the right upper abdomen, with a palpable enlarged gallbladder and a positive Murphy’s sign. Laboratory tests show elevated white blood cell count, C-reactive protein, procalcitonin, and other markers of systemic inflammation and bacterial infection. Abdominal ultrasound often shows an enlarged gallbladder, thickened gallbladder wall, a ‘double-wall sign,’ and the presence of mixed echogenic foci within the lumen, which may shift with changes in body position or pressure. CT and MRI scans, in addition to evaluating gallbladder lesions, provide excellent diagnostic value for complications. Endoscopic ultrasound (EUS) is not affected by intestinal gas and offers clearer observation of gallbladder pathology.
3. Indications and contraindications for ultrasound-endoscopic guided fine needle aspiration of the gallbladder in acute suppurative cholecystitis
(1) Indications. A confirmed diagnosis of acute suppurative cholecystitis through medical history, laboratory tests, and imaging studies.
(2) Contraindications. 1) Unstable vital signs that make the patient unable to tolerate the procedure. 2) Severe coagulopathy. 3) Upper gastrointestinal obstruction preventing access to the duodenal papilla via endoscopy. 4) Presence of gallbladder gangrene, perforation, biliary fistula, or intraperitoneal infection.
4. Surgical steps and techniques
(1) Anesthesia and positioning
The procedure is performed under intravenous sedation without endotracheal intubation. For patients at high risk of aspiration or those with compromised cardiopulmonary function, general anesthesia with endotracheal intubation may be used. The patient is positioned in the left lateral decubitus position. It is important to ensure warmth, adjust the room temperature, and regulate the infusion temperature to prevent pressure sores.
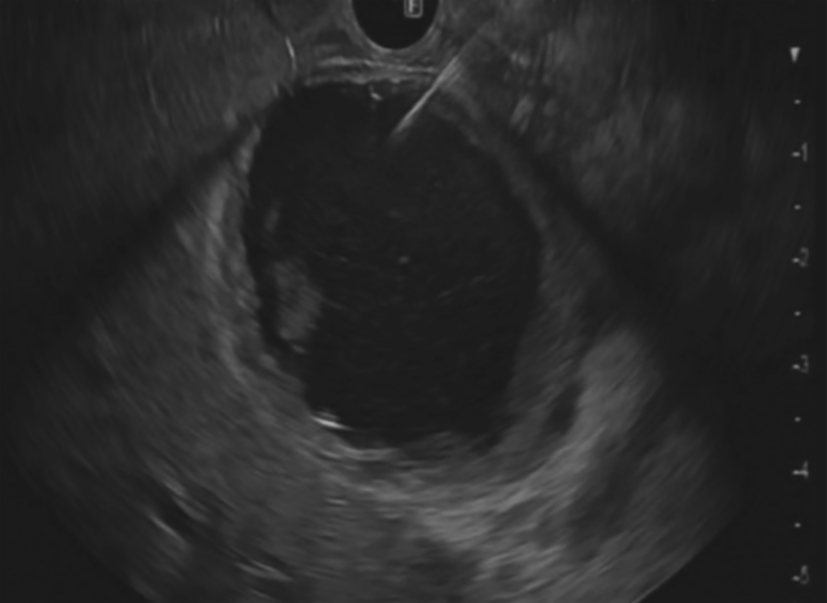

FIG. 11.13 — Gallbladder puncture drainage.
(2) Surgical procedures
(a) Selection of the puncture path: use linear array EUS to scan the gallbladder and surrounding tissues and organs in the gastric antrum and duodenal bulb respectively, observe the size, location, wall thickness, contents and relationship with the surrounding structure of the gallbladder, avoid blood vessels and other organs, and select the best puncture site.
(b) Insertion of the puncture needle: insert a 19G puncture needle into the working channel of the endoscope and secure it; use forceps to adjust the needle to the selected puncture path. Advance the outer sheath until it is in contact with the wall of the digestive tract; use color Doppler mode to ensure that there are no blood vessels along the puncture path.
(c) Puncturing the gallbladder: measure the distance from the tip of the outer sheath to the lumen of the gallbladder and fix the inner core lock to this distance. While maintaining continuous suction, perform the puncture of the gallbladder along the chosen path (figure 11.13).
(d) Aspiration of purulent fluid: remove the inner core, and use a vacuum syringe to aspirate the purulent bile. Saline irrigation may be used to flush the gallbladder until the fluid becomes clear.
(e) Removal of the puncture needle.
5. Postoperative management
The patient should be kept nil by mouth (NPO) and receive acid suppression, fluid resuscitation, and nutritional support. Antibiotics should continue to be administered. Monitor for any complications such as abdominal pain, fever, biliary fistula, peritonitis, gastrointestinal bleeding, etc. Routine laboratory tests including complete blood count (CBC), C-reactive protein (CRP), procalcitonin (PCT), and blood biochemistry should be reviewed. Additionally, follow-up imaging such as abdominal ultrasound, CT, or MRI should be performed to assess the patient’s recovery.
6. Hot-spot issues in the consensus
Acute cholecystitis can be classified into three grades based on its severity. Grade I (mild): mild inflammatory changes in the gallbladder. Grade-II (moderate): acute cholecystitis with complications such as white blood cell count >18 × 10
Currently, there are several minimally invasive treatment methods for acute cholecystitis in clinical practice. Percutaneous transhepatic gallbladder drainage (PTGBD) is the most widely used approach. In patients with poor circulatory stability or severe illness, the success rate and clinical relief efficiency of PTGBD are high, with technical and clinical success rates of 98% and 90%, respectively, and a low incidence of surgical adverse events at 3.7%. The main limitation of PTGBD is that it is merely a temporary measure, allowing for temporary drainage of the gallbladder. Inflammation is alleviated by observing and adjusting the drainage flow rate. It is not designed as a long-term solution, and prolonged placement of the drainage tube is associated with the risks of recurrent cholecystitis, catheter displacement, patient discomfort, and quality of life issues. Long-term use carries the risk of fistula formation. In contrast, endoscopic ultrasound-guided gallbladder drainage (EUS–GBD), which provides internal drainage without an external catheter, not only controls acute inflammation but also prevents recurrence, especially in case of acute suppurative cholecystitis. EUS-guided fine needle aspiration drainage avoids external drainage pathways, better utilizes the internal anatomy of the gallbladder, and allows for internal fistula formation. Compared to PTGBD, the advantages of EUS–GBD include internal bile flow, avoiding the risk of recurrent cholecystitis and bleeding after percutaneous catheter removal, and reducing postoperative pain.
Endoscopic transpapillary gallbladder drainage includes both endoscopic nasobiliary drainage and endoscopic gallbladder stent placement. The specific steps are as follows: after successful insertion of the endoscopic papillary catheter, a 0.025-in or 0.035-in guidewire is selectively advanced into the cystic duct and into the gallbladder, while the cystic duct is dilated and the guidewire is maintained in position. A pig-tail nasobiliary catheter or a plastic stent is then placed in the gallbladder. In cases of Mirizzi syndrome or gallbladder duct stones, the guidewire is used to guide the stone retrieval balloon or basket to push the stones into the gallbladder or pull them into the common bile duct for removal. The drainage options currently available include nasobiliary catheter drainage and endoscopic gallbladder stent placement, but both are temporary and carry risks such as stent blockage. When transpapillary catheter insertion fails, or when the cystic duct is in poor condition (such as narrowing, tortuosity, gallbladder neck stone obstruction, or poor imaging), it becomes difficult to pass the guidewire and drainage materials through the cystic duct, making EUS–GBD an effective supplement to endoscopic transpapillary gallbladder drainage. Based on the puncture site, EUS–GBD can be categorized into EUS-guided gastric approach and EUS-guided duodenal approach. According to the drainage method, it can be further divided into EUS-guided nasobiliary drainage, EUS-guided gallbladder stenting, and EUS-guided gallbladder aspiration. For acute suppurative cholangitis, there is an urgent need to drain pus and relieve the condition. Therefore, EUS-guided gallbladder puncture and aspiration have unique significance.
The 2018 Tokyo Guidelines for Acute Cholecystitis and the 2020 WSGE guidelines update suggest that EUS–GBD should be routinely recommended for larger ERCP centers. The relatively mature endoscopic techniques significantly reduce risks such as perforation and worsening infection during the EUS–GBD procedure. For patients with acute suppurative cholecystitis who are not candidates for surgery, EUS–GBD should be considered a safe and effective alternative to PTGBD. In terms of technical feasibility and efficacy, EUS–GBD has been compared with PTGBD in patients with acute, high-risk, or advanced cholecystitis who did not respond to initial medical treatment and were not suitable for laparoscopic cholecystectomy. Both EUS–GBD and PTGBD demonstrated similar technical success rates (97% vs. 97%, P = 0.001) and clinical success rates (100% vs. 96%, P = 0.0001), with comparable complications (7% vs. 1%, P = 0.492), indicating that EUS–GBD is a safe alternative to PTGBD for patients who are not suitable for surgery.
11.5 Endoscopic Super Minimally Invasive Resection of Biliary Polyps
1. Overview
With advancements in imaging technology and the widespread use of endoscopic retrograde cholangiopancreatography (ERCP) for direct visual diagnosis, the detection rate of polypoid lesions in the common bile duct, hepatic duct, and gallbladder has gradually increased. Biliary polyps have a certain risk of malignant transformation, and patients often face a dilemma between over-treatment with surgical resection and missed opportunities for optimal surgery due to follow-up observation. The development of direct visual endoscopic techniques for the diagnosis and treatment of biliary and gallbladder polypoid lesions has provided a potential solution. However, due to limitations in equipment development, this technology has not been successfully implemented. In response, the Department of Gastroenterology at the PLA General Hospital developed an electrocoagulation and snare device for use in oral cholangioscopy, laying the technical foundation for performing cholangioscopic mucosal resection (CA–EMR). Subsequently, the research team successfully practiced the CA–EMR technique in vitro, in animal models, and in human cases, preliminarily validating its feasibility and safety.
2. Diagnosis of biliary polyps
Most patients with biliary polyps are asymptomatic. Occasionally, patients may present with intermittent upper abdominal pain, nausea, or vomiting. ① Ultrasound can directly show the location, size, and number of polypoid lesions and can further characterize certain lesions. ② EUS eliminates interference from factors such as intestinal gas, obesity, or bile viscosity, providing clear visualization of the bile duct wall structure. EUS plays an important role in the differential diagnosis of cholesterol polyps, adenomas, and gallbladder cancer but cannot differentiate between adenomas and adenocarcinomas. ③ CT is less sensitive than ultrasound in detecting small gallbladder polyps. However, CT is superior to ultrasound in terms of lesion characterization, providing detailed visualization of the bile duct, gallbladder, liver, hepatic hilum, and their anatomical relationships.
3. Indications and contraindications
(1) Indications: ① Symptomatic bile duct polyps; ② bile duct polyps complicated by bile duct stones; ③ rapidly growing bile duct polyps with a diameter ≥ 1 cm; ④ bile duct polyps with atypical hyperplasia.
(2) Contraindications: ① Patients with high suspicion of bile duct cancer; ② during acute cholangitis; ③ patients with poor overall health status, and moderate to severe ascites.
4. Preoperative preparation
(1) Gastrointestinal preparation. A liquid diet should begin one to two days before surgery. Fasting for 6 h and water deprivation for 2 h before anesthesia. For colorectal surgery, patients may take laxatives or undergo enema for bowel cleaning.
(2) Preoperative examination. Preoperative abdominal CT scan should confirm the location and nature of any stones. Routine laboratory tests should be conducted, including complete blood count, blood biochemistry, coagulation function, as well as ECG and echocardiogram. Medications should be discontinued: anticoagulants affecting coagulation (aspirin seven days before, low-molecular-weight heparin three days before, heparin 6–12 h before, warfarin three days before).
5. Surgical procedure Procedure performed under anesthesia, as follows:
(1) Peroral cholangioscopy is performed, identifying a broad-based polypoid lesion in the common bile duct (figure 11.14A).
(2) A peroral cholangioscope with a 1.8-mm working channel is inserted into the common bile duct and, under direct vision at the base of the polypoid lesion, a submucosal injection is made using an injection needle (figure 11.14B).
(3) An electrocautery snare is inserted through the working channel of the cholangioscope into the common bile duct.
(4) Under direct cholangioscopic view, the snare is used to successfully resect the polypoid lesion, with the wound appearing pale (figures 11.14C–E).
(5) After successfully capturing the lesion using a basket under peroral cholangioscopy, the lesion is removed orally (figure 11.14F).
(6) Finally, a nasobiliary drainage tube is placed under duodenoscopy.
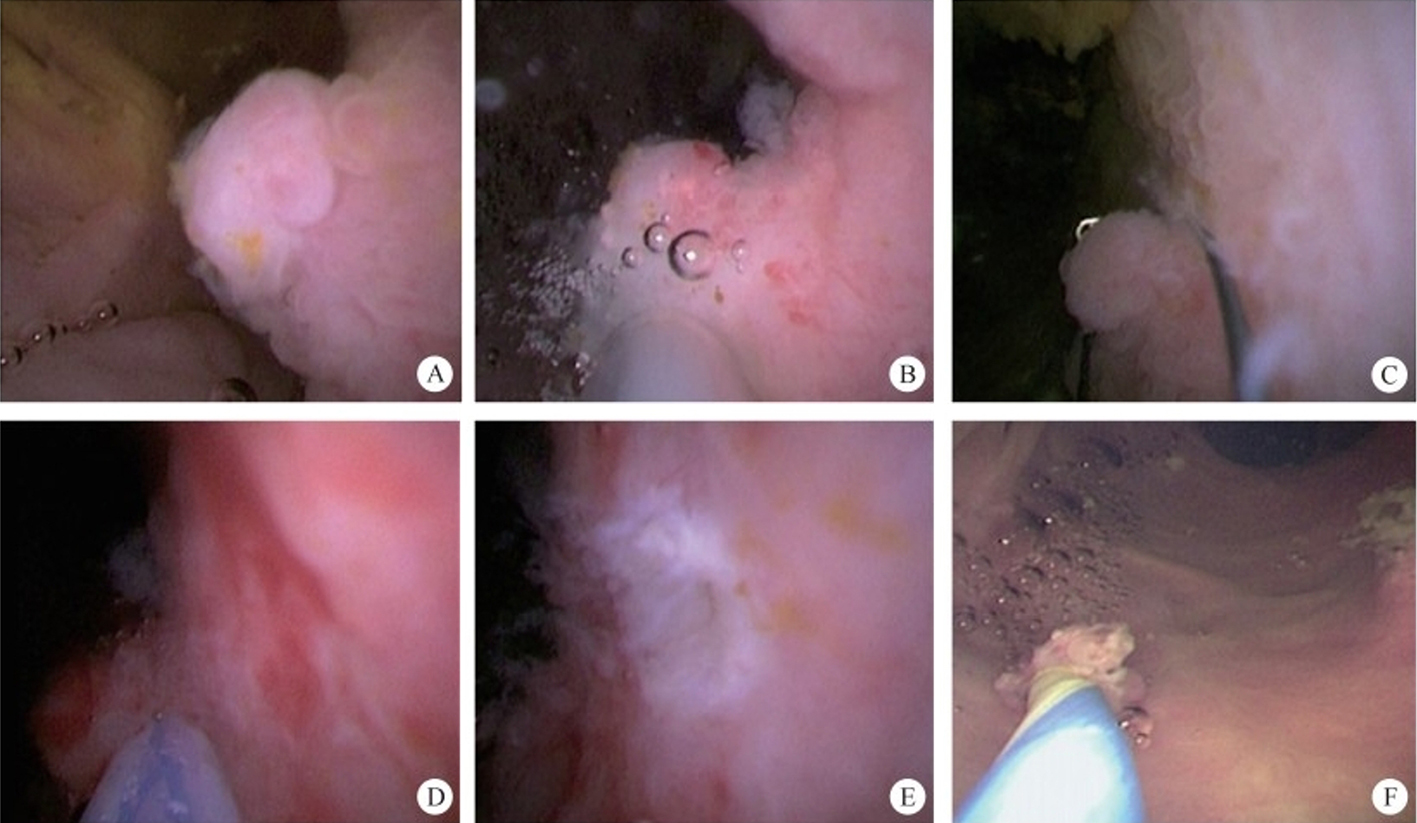

FIG. 11.14 — Clinical procedure of CA–EMR in a human being. A. Insertion of the peroral cholangioscope with a 1.8-mm working channel into the common bile duct, revealing a broad-based polypoid lesion in the distal CBD. B. Submucosal injection using an injection needle under direct vision at the base of the polypoid lesion. C, D. Snaring of the lesion under direct cholangioscopic view. E. The wound appears pale after resection. F. Successful oral extraction of the lesion using a basket under direct peroral cholangioscopic view.
6. Postoperative management
(1) The patient’s vital signs and abdominal symptoms must be closely monitored, as well as for the presence of hematemesis or melena. Blood tests, including complete blood count, venous amylase, and lipase, should be performed at 24 h and 72 h postoperatively. If clinical assessment suggests biliary bleeding, post-ERCP pancreatitis (PEP), biliary perforation, gastrointestinal bleeding/perforation, or other related adverse events, abdominal CT and/or endoscopic examination should be conducted promptly to confirm the condition.
(2) Patients should refrain from food and water for three days postoperatively. If no abnormal abdominal symptoms are present and postoperative laboratory results show no significant abnormalities, a gradual transition to liquid and semi-liquid diets can be initiated. Proton pump inhibitors (PPIs) and antibiotics should be administered for three days postoperatively. If postoperative complications such as pancreatitis, biliary bleeding, or gastrointestinal bleeding occur, the duration of treatment should be extended accordingly. Additionally, for postoperative pancreatitis, somatostatin therapy should be promptly administered. It should be noted that in case of gastrointestinal bleeding, if conservative management fails, timely intervention with endoscopy, surgery, or interventional methods should be considered for hemostasis.
7. Postoperative follow-up
We recommend close telephone and outpatient follow-up to monitor the occurrence of any adverse events. Additionally, patients will be informed and strongly advised to return for endoscopic re-examination at three months, six months, one year, two years, three years, four years, and five years postoperatively.
8. A case example
The patient, a 61-year-old man, presented with common bile duct stones and underwent ERCP at our hospital. After stone removal, a peroral cholangioscopy was performed to confirm whether any residual stones remained after basket extraction. During the procedure, a broad-based polypoid lesion was identified in the common bile duct. The lesion was biopsied under cholangioscopic guidance, and the pathological results indicated mild atypical hyperplasia. After fully informing the patient and his family about the risks associated with CA–EMR and obtaining informed consent, the procedure was carried out.
Refer to video 11.3, Peroral super minimally invasive resection of biliary polyps (See online resources).
9. Hot-spot issues and research progress in the consensus
Up to now, peroral cholangiopancreatoscopy under direct vision has been widely applied in the diagnosis and treatment of biliary and pancreatic duct stones, tumors, and unexplained strictures. However, progress in the use of this technique for treating polypoid lesions in the biliopancreatic duct system remains limited, primarily due to bottlenecks in equipment development. This study demonstrates the excellent performance of the electrocautery snare device developed for use with peroral cholangioscopy under direct vision. The effectiveness and feasibility of the CA–EMR procedure have been preliminarily validated through ex vivo experiments, in vivo animal studies, and clinical practice in humans. Moving forward, our research team plans to conduct large-scale clinical studies to systematically verify the safety and feasibility of this device and technique.
References
[1] Biliary Surgery Group of the Surgical Branch of the Chinese Medical Association, Biliary Surgery Committee of the Surgical Physicians Branch of the Chinese Medical Doctor Association. (2019) Expert consensus on application of choledochoscope in diagnosis and treatment of hepatolithiasis (2019 edition), Chin. J. Dig. Surg. 18(7), 611–615. DOI: 10.3760/cma.j.issn.1673-9752.2019.07.001.
[2] Manes G., Paspatis G., Aabakken L., et al. (2019) Endoscopic management of common bile duct stones: European Society of Gastrointestinal Endoscopy (ESGE) guideline, Endoscopy 51(5), 472–491.
[3] Zhang H., Gao J., Sun Z., et al. (2023) Diagnostic accuracy of updated risk assessment criteria and development of novel computational prediction models for patients with suspected choledocholithiasis, Surg. Endosc. 37(9), 7348–7357.
[4] Luo H., Zhao L., Leung J., et al. (2016) Routine pre-procedural rectal indometacin versus selective post-procedural rectal indometacin to prevent pancreatitis in patients undergoing endoscopic retrograde cholangiopancreatography: a multicentre, single-blinded, randomised controlled trial, Lancet 387(10035), 2293–2301.
[5] Morris S., Gurusamy K. S., Sheringham J., et al. (2015) Cost-effectiveness analysis of endoscopic ultrasound versus magnetic resonance cholangiopancreatography in patients with suspected common bile duct stones, PLoS One 10(3), e0121699.
[6] Gurusamy K. S., Giljaca V., Takwoingi Y., et al. (2015) Endoscopic retrograde cholangiopancreatography versus intraoperative cholangiography for diagnosis of common bile duct stones, Cochrane Database Syst. Rev. (2), CD010339.
[7] Jun C. H., Park C. H., Jeon J., et al. (2015) Feasibility of self-expandable metal stents for preservation of sphincter of Oddi function in patients with common bile duct stones: a pilot study, Gastrointest. Endosc. 82(4), 719–723. doi: 10.1016/j.gie.2015.01.034.
[8] Cho E., Park C. H., Jun C. H., et al. (2018) Self-expandable metal stents for the extraction of common bile duct stones in patients receiving dual antiplatelet agents: a pilot study, Surg. Endosc. 32(2), 1077–1081. doi: 10.1007/s00464-017-5743-2.
[9] Cotton P. B., Lehman G., Vennes J., et al. (1991) Endoscopic sphincterotomy complications and their management: an attempt at consensus, Gastrointest. Endosc. 37(3), 383–393. doi: 10.1016/s0016-5107(91)70740-2.
[10] Geenen J. E., Toouli J., Hogan W. J., et al. (1984) Endoscopic sphincterotomy: follow-up evaluation of effects on the sphincter of Oddi, Gastroenterology 87(4), 754–758.
[11] Pereira-lima J. C., Jakobs R., Winter U. H., et al. (1998) Long-term results (7 to 10 years) of endoscopic papillotomy for choledocholithiasis. Multivariate analysis of prognostic factors for the recurrence of biliary symptoms, Gastrointest. Endosc. 48(5), 457–464. doi: 10.1016/s0016-5107(98)70085-9.
[12] Escourrou J., Cordova J. A., Lazorthes F., et al. (1984) Early and late complications after endoscopic sphincterotomy for biliary lithiasis with and without the gall bladder ‘in situ’, Gut 25(6), 598–602. doi: 10.1136/gut.25.6.598.
[13] Yan X. E., Zheng W., Zhang Y. P., et al. (2021) Endoclip papillaplasty restores sphincter of Oddi function: pilot study, Dig. Endosc. 33(6), 962–969.
[14] Fan X., Li X., Chang H., et al. (2019) Endoclip papilloplasty for a patulous and incompetent biliary papilla, VideoGIE 4(7), 331–333.
[15] Huang Y., Wang K., Zhang H., et al. (2018) A preliminary study of sphincter-preserving effect of SureClip from MicroTech on duodenal papilla occlusion, Chin. J. Dig. Endosc. 35(11), 823–827.
[16] Wang Y., Chang H., Zhang Y., et al. (2021) Endoscopic endoclip papilloplasty preserves sphincter of oddi function, Eur. J. Clin. Invest. 51(3), e13408. doi: 10.1111/eci.13408.
[17] Zhang D., Peng B., Liu J., et al. (2024) Utilization of endoclip papilloplasty in endoscopic retrograde cholangiopancreatography, China J. Endosc. 30(06), 1–7.
[18] Yıldırım A. E., Konduk B. T. (2021) Multicenter randomized trial of endoscopic papillary large balloon dilation without sphincterotomy versus endoscopic sphincterotomy for removal of bile duct stones: MARVELOUS trial, Endoscopy 53(2), 212. doi: 10.1055/a-1288-0772
[19] Mosler P. (2011) Diagnosis and management of acute cholangitis, Curr. Gastroenterol. Rep. 13, 166.
[20] ERCP Group of the Chinese Society of Digestive Endoscopy, Chinese Medical Association, Biliary and Pancreatic Group of the Chinese Medical Doctor Association Branch of Gastroenterologists, National Clinical Research Center for Digestive Diseases. (2018) Chinese guidelines for ERCP (2018), J. Clin. Hepatol.
[21] Du C. (2022) Clinical exploration of ENBD combined with LCBDE and one-stage suture in the treatment of AOSC with extrahepatic biliary tract stones. North Sichuan Medical College.
[22] Sun R., Jin H., Han S. (2020) Application effect comparison of two drainage schemes in emergency treatment of acute obstructive suppurative cholangitis, J. Clin. Emerg. 21(08), 603–607.
[23] Zhang K., Yan J. (2020) Progress in the diagnosis and treatment of acute obstructive suppurative cholangitis, J. Clin. Pathol. Res. 40(07), 1902–1907.
[24] Mukai S., Itoi T. (2023) Preoperative endoscopic ultrasound-guided biliary drainage for primary drainage in obstructive jaundice, Expert Rev. Gastroenterol. Hepatol. 17(12), 1197–1204. doi: 10.1080/17474124.2023.2293813.
[25] Zhang W., Ding H., Li Z., Linghu E. (2023) Laparoscopic common bile duct exploration through the cystic duct using flexible cholangioscopy combined with cholecystectomy for managing cholecysto-choledocholithiasis, Endoscopy 55(S 01), E659–E661. doi: 10.1055/a-2067-4587.
[26] Cotton P. B. (2018) Fifty years of ERCP: a personal review, Gastrointest. Endosc. 88(2), 393–396.
[27] Arvanitakis M., Devière J. (2009) Endoscopic retrograde cholangiopancreatography (ERCP), Endoscopy 41(10), 890–894.
[28] Freeman M. L., Nelson D. B., Sherman S., et al. (1996) Complications of endoscopic biliary sphincterotomy, N. Engl. J. Med. 335(13), 909–918. doi: 10.1056/nejm199609263351301.
[29] Frimberger E. (1998) Long-term sequelae of endoscopic papillotomy, Endoscopy 30(9), A221–A227.
[30] Hawes R. H., Cotton P. B., Vallon A. G. (1990) Follow-up 6 to 11 years after duodenoscopic sphincterotomy for stones in patients with prior cholecystectomy, Gastroenterology 98(4), 1008–1012.
[31] Han S., Shah R. J. (2020) Cholangioscopy-guided basket retrieval of impacted stones, VideoGIE 5(9), 387–388.
[32] Fejleh M. P., Thaker A. M., Kim S., et al. (2019) Cholangioscopy-guided retrieval basket and snare for the removal of biliary stones and retained prostheses, VideoGIE 4(5), 232–234.
[33] Ruemmele P., Hofstaedter F., Gelbmann C. M. (2009) Secondary sclerosing cholangitis, Nat. Rev. Gastroenterol. Hepatol. 6(5), 287–295.
[34] Zhang Y., Li Z. (2022) Research progress in endoscopic diagnosis of malignant biliary stricture, Chin. J. Dig. Endosc. 39(3), 245–248.
[35] Nakai Y., Isayama H., Wang H. P., et al. (2020) International consensus statements for endoscopic management of distal biliary stricture, J. Gastroenterol. Hepatol. 35(6), 967–979.
[36] Kulaksiz H., Weiss K. H., Gotthardt D., et al. (2008) Is stenting necessary after balloon dilation of post-transplantation biliary strictures? Results of a prospective comparative study, Endoscopy 40(9), 746–751.
[37] Zhang C. C., Rupp C., Exarchos X., et al. (2023) Scheduled endoscopic treatment of biliary anastomotic and nonanastomotic strictures after orthotopic liver transplantation, Gastrointest. Endosc. 97(1), 42–49.
[38] Kapoor B. S., Mauri G., Lorenz J. M. (2018) Management of biliary strictures: state-of-the-art review, Radiology 289(3), 590–603.
[39] Cantwell C. P., Pena C. S., Gervais D. A., et al. (2008) Thirty years’ experience with balloon dilation of benign postoperative biliary strictures: long-term outcomes, Radiology 249(3), 1050–1057.
[40] Im B .S., Gwon D. I., Chu H. H., et al. (2022) Percutaneous transhepatic treatment of benign bile duct strictures using retrievable covered stents: long-term outcomes in 148 patients, Korean J. Radiol. 23(9), 889–900.
[41] Siiki A., Sand J., Laukkarinen J. (2018) A systematic review of biodegradable biliary stents: promising biocompatibility without stent removal, Eur. J. Gastroenterol. Hepatol. 30(8), 813–818.
[42] Song G., Zhao H. Q., Liu Q., Fan Z. (2022) A review on biodegradable biliary stents: materials and future trends, Bioact. Mater. 17, 488–495.
[43] Dumonceau J. M., Tringali A., Papanikolaou I. S., et al. (2018) Endoscopic biliary stenting: indications, choice of stents, and results: European Society of Gastrointestinal Endoscopy (ESGE) Clinical Guideline - updated October 2017, Endoscopy, 50(9), 910–930.
[44] Yu J. F., Zhang D. L., Wang Y. B., Hao J. Y. (2022) Digital single-operator cholangioscopy for biliary stricture after cadaveric liver transplantation, World J. Gastrointest. Oncol. 14(5), 1037–049.
[45] Jang S., Stevens T., Kou L., et al. (2020) Efficacy of digital single-operator cholangioscopy and factors affecting its accuracy in the evaluation of indeterminate biliary stricture, Gastrointest. Endosc. 91(2), 385–393.e1.
[46] Fung B. M., Fejleh M. P., Tejaswi S., Tabibian J. H. Cholangioscopy and its role in primary sclerosing cholangitis, Eur. Med. J. Hepatol. 8(1), 42–53.
[47] Chinese Society of Digestive Endoscopy. (2023) Interpretation of Chinese expert consensus on peroral biliary-pancreaticoscopy super minimally invasive technique (2023, Beijing), Chin J. Gastrointest. Endosc. (Electron. Ed.) 10(04), 217–239.
[48] Zhang W., Chai N., Linghu E. (2023) A new criterion for the differential diagnosis of IBS during the peroral cholangioscopy super minimally invasive examination technique, Chin J. Gastrointest. Endosc. (Electron. Ed.) 10(1), 11–13.
[49] Facciorusso A., Rosca E. C., Ashimi A., et al. (2018) Management of anastomotic biliary stricture after liver transplantation: metal versus plastic stent, Ann. Gastroenterol. 31(6), 728–734.
[50] Raghavapuram S., Girotra M., Robertson J. D., et al. (2016) Successful management of ischemic cholangiopathy after failed endoscopic/percutaneous interventions, facilitated by digital cholangioscopy, Gastrointest. Endosc. 84(6), 1053–1054.
[51] Bokemeyer A., Gross D., Brückner M., et al. (2019) Digital single-operator cholangioscopy: a useful tool for selective guidewire placements across complex biliary strictures, Surg. Endosc. 33(3), 731–737.
[52] Li Y., Hao J., Liu X., et al. (2022) Application of SpyGlass peroral choledochoscopy to the diagnosis and treatment of biliary stricture after liver transplantation, Chin. J. Dig Endosc. (12), 998–1003.
[53] Sethi A., Tyberg A., Slivka A., et al. (2022) Digital Single-operator Cholangioscopy (DSOC) Improves Interobserver Agreement (IOA) and accuracy for evaluation of indeterminate biliary strictures: the Monaco classification, J. Clin. Gastroenterol. 56(2), e94-e97.
[54] Robles-Medranda C., Valero M., Soria-Alcivar M., et al. (2018) Reliability and accuracy of a novel classification system using peroral cholangioscopy for the diagnosis of bile duct lesions, Endoscopy 50(11), 1059–1070.
[55] Kahaleh M., Gaidhane M., Shahid H. M., et al. (2022) Digital single-operator cholangioscopy interobserver study using a new classification: the Mendoza Classification (with video), Gastrointest. Endosc. 95(2), 319–326.
[56] Kahaleh M., Raijman I., Gaidhane M., et al. (2022) Digital cholangioscopic interpretation: when north meets the south, Dig. Dis. Sci. 67(4), 1345–1351.
[57] Zhang W., Zhang B., Wang J., et al. (2023) A pilot exploration of super minimally invasive electrocision using snare under peroral biliary-pancreaticoscopy, Chin J. Gastrointest. Endosc. (Electron. Ed.) 10(03), 190–191.
[58] Osanai M., Itoi T., Igarashi Y., et al. (2013) Peroral video cholangioscopy to evaluate indeterminate bile duct lesions and preoperative mucosal cancerous extension: a prospective multicenter study, Endoscopy 45(8), 635–642. doi: 10.1055/s-0032-1326631.
[59] Alameel T., Bain V., Sandha G. (2013) Clinical application of a single-operator direct visualization system improves the diagnostic and therapeutic yield of endoscopic retrograde cholangiopancreatography, Can. J. Gastroenterol. 27(1), 15–19. doi: 10.1155/2013/278758.
[60] Ramchandani M., Reddy D. N., Gupta R., et al. (2011) Role of single-operator peroral cholangioscopy in the diagnosis of indeterminate biliary lesions: a single-center, prospective study, Gastrointest. Endosc. 74(3), 511–519. doi: 10.1016/j.gie.2011.04.034.
[61] Hartman D. J., Slivka A., Giusto D. A., et al. (2012) Tissue yield and diagnostic efficacy of fluoroscopic and cholangioscopic techniques to assess indeterminate biliary strictures, Clin. Gastroenterol. Hepatol. 10(9), 1042–1046. doi: 10.1016/j.cgh.2012.05.025.
[62] Bang J. Y., Navaneethan U., Hasan M., et al. (2020) Optimizing outcomes of single-operator cholangioscopy-guided biopsies based on a randomized trial, Clin. Gastroenterol. Hepatol. 18(2), 441–448.e441. doi: 10.1016/j.cgh.2019.07.035.
[63] Ogura T., Imanishi M., Kurisu Y., et al. (2017) Prospective evaluation of digital single-operator cholangioscope for diagnostic and therapeutic procedures (with videos), Dig. Endosc. 29(7), 782–789. doi: 10.1111/den.12878.
[64] Shah R. J., Raijman I., Brauer B., et al. (2017) Performance of a fully disposable, digital, single-operator cholangiopancreatoscope, Endoscopy 49(7), 651–658. doi: 10.1055/s-0043-106295.
[65] Manta R., Frazzoni M., Conigliaro R., et al. (2013) SpyGlass single-operator peroral cholangioscopy in the evaluation of indeterminate biliary lesions: a single-center, prospective, cohort study, Surg. Endosc. 27(5), 1569–1572. doi: 10.1007/s00464-012-2628-2.
[66] Real-Noval H., Fernández-Fernández J., Soler-Dorda G. (2019) Predicting factors for the diagnosis of gangrene acute cholecystitis, Cir Cir 87(4), 443–449.
[67] Menéndez-Sánchez P., León-Salinas C., Amo-Salas M., et al. (2019) Association of laboratory and radiologic parameters in the diagnosis of acute cholecystitis, Rev Gastroenterol Mex 84(4), 449–454.
[68] (2018) Tokyo Guidelines 2018: management strategies for gallbladder drainage in patients with acute cholecystitis (with videos), J. Hepatobiliary Pancreat. Sci. 25(1), 87–95.
[69] Kiriyama S., Kozaka K., Takada T., Strasberg S. M., Pitt H. A., Gabata T., Hata J., Liau K. H., Miura F., Horiguchi A., et al. (2018) Tokyo Guidelines 2018: diagnostic criteria and severity grading of acute cholangitis (with videos), J. Hepatobiliary Pancreat. Sci. 25(1), 17–30. doi: 10.1002/jhbp.512.
[70] Sobani Z. A., Ling C., Rustagi T. (2021) Endoscopic ultrasound-guided gallbladder drainage, Dig. Dis. Sci. 66(7), 2154–2161.
[71] Minaga K., Yamashita Y., Ogura T., Takenaka M., Shimokawa Y., Hisa T., Itonaga M., Kato H., Nishikiori H., Okuda A., et al. (2019) Clinical efficacy and safety of endoscopic ultrasound-guided gallbladder drainage replacement of percutaneous drainage: A multicenter retrospective study, Dig. Endosc. 31(2), 180–187.
[72] Ahmed O., Ogura T., Eldahrouty A., Khalaf H., Mohammed E., Okasha H., Sameer A., Abdelaal U., Higuchi K. (2018) Endoscopic ultrasound-guided gallbladder drainage: Results of long-term follow-up, Saudi J. Gastroenterol. 24(3), 183–188.
[73] Pisano M., Allievi N., Gurusamy K., Borzellino G., Cimbanassi S., Boerna D. Coccolini F., Tufo A., Di Martino M., Leung J., et al. (2020) 2020 World Society of Emergency Surgery updated guidelines for the diagnosis and treatment of acute calculus cholecystitis, World J. Emerg. Surg. 15(1), 61.
[74] Okamoto K., Suzuki K., Takada T., Strasberg S. M., Asbun H. J., Endo I., Iwashita Y., Hibi T., Pitt H. A., Umezawa A., et al. (2018) Tokyo Guidelines 2018: flowchart for the management of acute cholecystitis, J. Hepatobiliary Pancreat. Sci. 25(1), 55–72.
[75] Chinese Society of Digestive Endoscopy, Branch of Endoscopic Physicians of Chinese Medical Doctor Association, Beijing Branch of Chinese Society of Digestive Endoscopy. (2020) Chinese expert consensus on the endoscopic diagnosis and treatment of biliary fistula (2020, Beijing), Chin. J. Gastrointest. Endosc. (Electron. Ed.) 7(3), 108–116.
[76] Cassis P., Shah-Khan S. M., Nasr J. (2019) EUS-guided drainage of a 20-cm biloma by use of a lumen-apposing metal stent, VideoGIE 5(1), 20–21. doi: 10.1016/j.vgie.2019.09.009. PMID: 31922075; PMCID: PMC6945231.
[77] Yonamine K., Koshita S., Kanno Y., Ogawa T., Ito K. (2023) EUS-guided transluminal drainage for a huge perihepatic biloma covering the right hepatic lobe (with video), Endosc. Ultrasound 12(2), 284–285. doi: 10.4103/EUS-D-22-00061.
[78] Ulla-Rocha J. L., Lopez-Piñeiro S., Dominguez-Comesaña E. (2016) EUS-guided transgastric drainage of perihepatic biloma after laparoscopic liver metastasectomy from colon cancer, J. Gastrointest. Cancer 47(4), 468–469. doi: 10.1007/s12029-015-9772-6. PMID: 26500100.
[79] Editorial Board of Chinese Journal of Digestion. (2019) Consensus on diagnosis and treatment of chronic cholecystitis and gallstones in China (2018), Chin. J. Dig. 39(2), 73–79.
[80] Zhang W., Chai N., Zhai Y., et al. (2023) Cholangioscopy-assisted extraction of choledocholithiasis and partial sediment-like gallstones through papillary support: A pilot exploration for super minimally invasive surgery, Endoscopy 55(S 01), E274–E275.
[81] Tieu A. H., Kumbhari V., Jakhete N., et al. (2015) Diagnostic and therapeutic utility of SpyGlassDig. Endosc. 27(4), 479–485. doi: 10.1111/den.12405.
[82] Minami H., Mukai S., Sofuni A., et al. (2021) Clinical outcomes of digital cholangioscopy-guided procedures for the diagnosis of biliary strictures and treatment of difficult bile duct stones: a single-center large cohort study, J. Clin. Med. 10(8), 1638. doi: 10.3390/jcm10081638.
[83] Almadi M. A., Itoi T., Moon J. H., et al. (2020) Using single-operator cholangioscopy for endoscopic evaluation of indeterminate biliary strictures: results from a large multinational registry, Endoscopy 52(7), 574–582. doi: 10.1055/a-1135-8980.
[84] Yan S., Tejaswi S. (2019) Clinical impact of digital cholangioscopy in management of indeterminate biliary strictures and complex biliary stones: a single-center study, Ther. Adv. Gastrointest. Endosc. 12, 2631774519853160. doi: 10.1177/2631774519853160.
[85] Pereira P., Santos S., Morais R., et al. (2020) Role of peroral cholangioscopy for diagnosis and staging of biliary tumors, Dig. Dis. 38(5), 431–440. doi: 10.1159/000504910.
Chapter 12 Super Minimally Invasive Surgery for Pancreatic Diseases
12.1 Super Minimally Invasive Sclerotherapy with Polidocanol
12.1.1 Overview
Pancreatic cystic neoplasms (PCNs) are cystic tumor lesions originating from the pancreatic ductal epithelium and/or stromal tissue, characterized primarily by the proliferation of ductal or acinar epithelium and the accumulation of secretions, resulting in cyst formation. Previously considered a rare type of pancreatic tumor, the incidence of PCNs has steadily increased with the advancement of medical imaging techniques and the growing awareness of health check-ups. Studies have reported an 8% annual increase in detection rates from 1995 to 2010. The reported detection rates of PCNs vary, with CT and MRI scans showing detection rates ranging from 0.7% to 8.7% and 2.4% to 49.1%, respectively. Autopsy studies have found detection rates as high as 50%. However, these figures may underestimate the true incidence, as approximately 3.6%–36.7% of PCN patients are asymptomatic, and up to 50% of diagnoses are made postoperatively. As the detection rate of PCNs increases, there is growing attention to their diagnosis and treatment. PCNs primarily include mucinous cystic neoplasms (MCNs), intraductal papillary mucinous neoplasms (IPMNs), serous cystic neoplasms (SCNs), solid pseudopapillary neoplasms (SPNs), and cystic neuroendocrine tumors (cNETs). These different types of lesions exhibit significant differences in biological behavior, clinical manifestations, and risk of malignant transformation, leading to varying management approaches. With the increasing detection rate of PCNs, accurate diagnosis has garnered greater attention. Traditional imaging techniques have limited accuracy in diagnosing PCNs, but the advent of endoscopic ultrasound (EUS) and its guided diagnostic technologies has significantly improved diagnostic accuracy, with multiple diagnostic techniques often used in combination to complement each other’s strengths.
For the management of pancreatic cystic neoplasms (PCNs), traditional approaches primarily include surgical treatment and regular monitoring. However, surgical treatment is associated with significant trauma and a high rate of complications. Regular monitoring, on the other hand, imposes economic and psychological burdens on patients and may potentially lead to delays in treatment. As a result, the exploration of minimally invasive treatment options for PCNs has become a major research focus. Since 2005, reports have emerged globally regarding the use of endoscopic ultrasound (EUS)-guided ablation techniques for minimally invasive treatment, marking a shift in the traditional management of PCNs. EUS-guided ablation includes both chemical and physical ablation methods. Chemical ablation agents primarily consist of anhydrous ethanol, polidocanol, paclitaxel, gemcitabine, and others, while physical ablation is mainly performed using radiofrequency ablation. According to current research, preliminary results suggest that EUS-guided ablation can enable a significant proportion of patients to avoid surgery, leading to a marked improvement in postoperative quality of life. The treatment concept of ablation differs from traditional surgical approaches. While surgery removes cystic tumors, it often requires the removal of part of the organ, which directly results in numerous complications that negatively affect quality of life. In contrast, EUS-guided ablation eliminates the cystic tumor without altering the anatomical structure of the pancreas, as it does not involve removing any normal tissue, thereby preserving the integrity of the body’s anatomical structure. EUS-guided ablation aligns with the concept of super minimally invasive surgery and is consistent with the trend in medicine towards more minimally invasive techniques. This innovative treatment modality has great potential in the management of pancreatic cystic neoplasms and is expected to play an increasingly important role in the future.
12.1.2 Diagnosis
1. Abdominal ultrasound. Abdominal ultrasound is the preferred initial screening tool, but it is often influenced by abdominal gas.
2. CT scan. Thin-slice CT can provide detailed observations of tumor size, location, cystic or solid nature, presence of septa, and calcification features.
3. MRI. MRI is the preferred method for diagnosing pancreatic cystic neoplasms (PCN). MRI offers high contrast and multi-sequence imaging, making it particularly suitable for observing cystic cavities, cyst wall nodules, internal septa, surrounding pancreatic parenchyma, and the pancreatic duct system. Magnetic resonance cholangiopancreatography (MRCP) primarily visualizes the pancreatic duct, the bile duct, and their branching structures, making it the best sequence for evaluating the relationship between PCNs and the pancreatic duct.
4. Endoscopic ultrasound (EUS) and its guided diagnostic techniques. EUS allows for real-time, dynamic, and close-up examination of PCNs, enabling detailed visualization of internal structures such as septa, wall nodules, and blood flow patterns. It can also detect finer internal structures to assist in diagnosis. With the continuous development of endoscopic techniques, equipment, and accessories, EUS now facilitates contrast-enhanced EUS (CE–EUS) and EUS–guided fine-needle aspiration (EUS–FNA), among other diagnostic techniques. These include cyst fluid analysis, cystoscopy, confocal laser endomicroscopy (CLE), and EUS-guided through-the-needle biopsy (EUS–TTNB). Compared to other imaging techniques, EUS and its guided diagnostic methods provide more information for diagnosing PCNs, aiding in differential diagnosis and significantly improving diagnostic accuracy. Both Chinese and international guidelines recommend EUS as an important supplementary diagnostic tool for PCNs.
5. Endoscopic retrograde cholangiopancreatography (ERCP). ERCP is used to assess the morphology of the pancreatic duct, determine the presence of obstructions, and collect samples for cytological analysis.
6. Pancreatoscopy. The unique advantage of pancreatoscopy lies in its ability to directly visualize the main pancreatic duct and perform histological biopsy. Current pancreatoscopy systems can be inserted through the working channel of a duodenoscope, and the pancreatoscope itself is equipped with a biopsy channel, enabling direct tissue sampling using biopsy forceps under direct visualization.
12.1.3 Indications and Contraindications for Endoscopic Polidocanol Ablation of Pancreatic Cystic Neoplasms
1. Indications The optimal indication is for low-risk mucinous PCN patients that have not yet undergone malignant transformation.
2. Contraindications
(1) Absolute contraindications: contraindications to endoscopic examination; short life expectancy; pregnancy; coagulation disorders; active pancreatitis; pancreatic necrosis or signs of malignancy.
(2) Relative contraindications: benign or low-grade malignant cysts (e.g., serous cystic neoplasm [SCN]); main pancreatic duct dilation >5 mm with clear communication with the main pancreatic duct; enhanced wall nodules, septations >6, thickened cyst walls, thickened septations, or significant solid components within the cyst; history of acute pancreatitis.
12.1.4 Preoperative Preparation
(1) The patient and the family members must be informed thoroughly about the risks and benefits of the ablation procedure, and obtain informed consent.
(2) The necessary preoperative examinations, including blood routine tests, blood biochemistry, coagulation function, serum 8-item test, blood type, electrocardiogram (ECG), chest X-ray or CT, abdominal MRI or CT, and anesthesia evaluation, as well as any other required endoscopic examinations and tests must be completed, and the results verified.
(3) The use of anticoagulants, antiplatelet agents, or drugs with blood-activating effects must be verified, and their use adjusted or discontinued according to the patient’s condition.
(4) The patient should fast for 6–8 h before surgery, and abstain from drinking water for 4–6 h prior to the procedure.
(5) Anesthesia preparation: left lateral position, intravenous anesthesia.
12.1.5 Surgical Procedure and Techniques (Figure
(1) EUS assessment: the location, number, size, shape, thickness of the cyst wall, thickness of septa within the cyst, size and number of daughter cysts, size of solid components within the cyst, size of wall nodules, presence of calcification, and relationship with the pancreatic duct must be determined. CDFI (Color Doppler Flow Imaging) is used to observe the presence of blood flow within the lesion. In cases when solid components are found within the cystic lesion during EUS examination but it is difficult to distinguish between wall nodules, necrotic tissue, or debris, contrast-enhanced ultrasound (CEUS) can assist in differentiating these by observing whether the solid components enhance.
(2) EUS-guided aspiration: under EUS guidance, a 19G or 22G biopsy needle is used to puncture and aspirate cystic fluid. The volume of aspirated fluid is recorded, and its characteristics, viscosity are observed. The collected sample should be sent for biochemical, tumor markers, cytological, or molecular biomarker testing.
(3) If conditions allow, EUS-guided cystoscopy or fine-needle-type confocal laser endomicroscopy is performed to assist in the assessment of the lesion. If the cyst fluid is turbid, lavage with saline solution may be performed before re-evaluation.
(4) If conditions permit, a cystic puncture biopsy is performed. The puncture needle can be used to aspirate the solid components or cyst wall to obtain tissue samples for diagnosis. Alternatively, a small biopsy forceps can be introduced through the puncture needle into the cyst for direct biopsy of the cyst wall or septa, solid components, etc., to obtain sufficient specimens for histopathological examination.
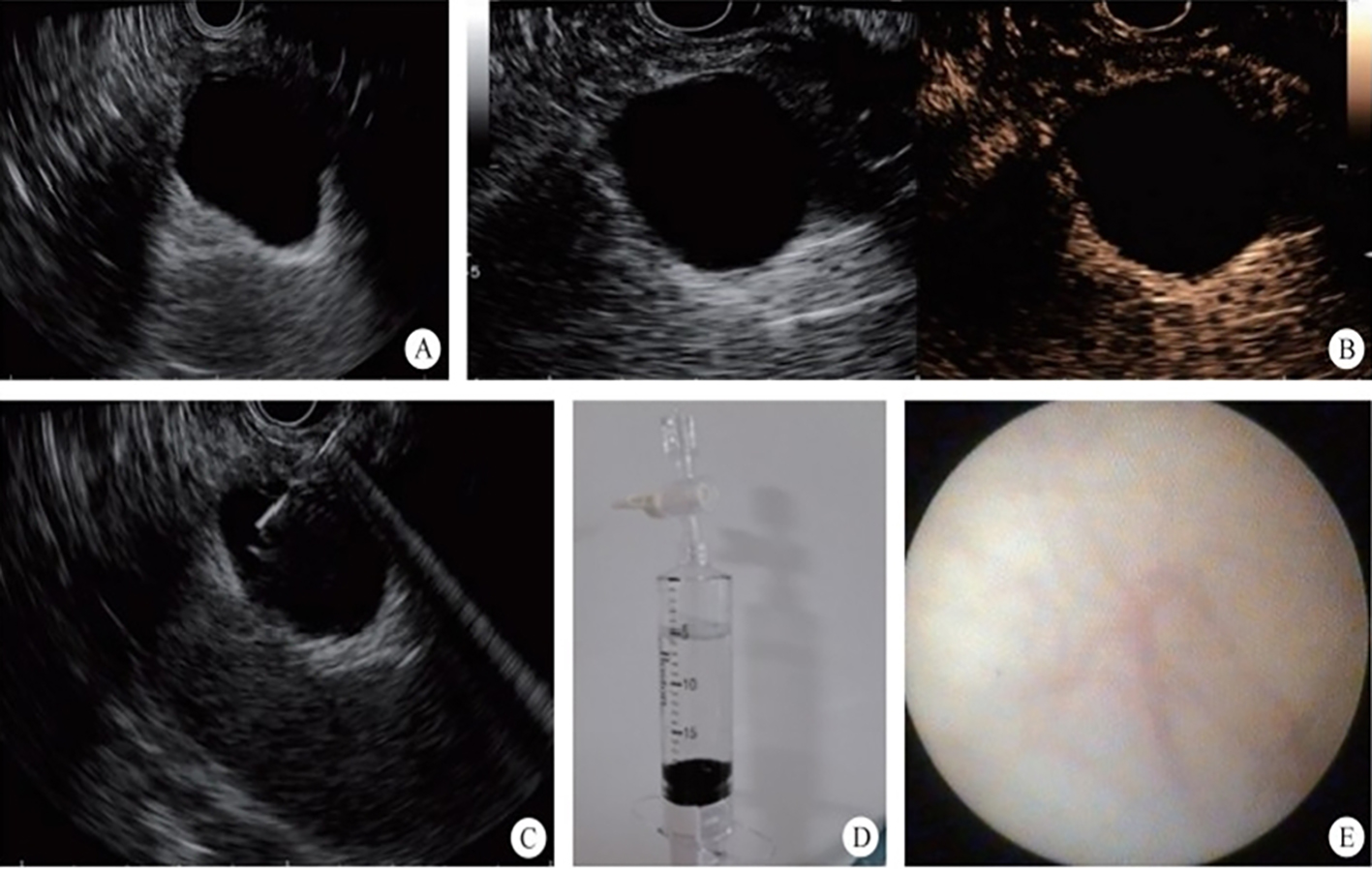
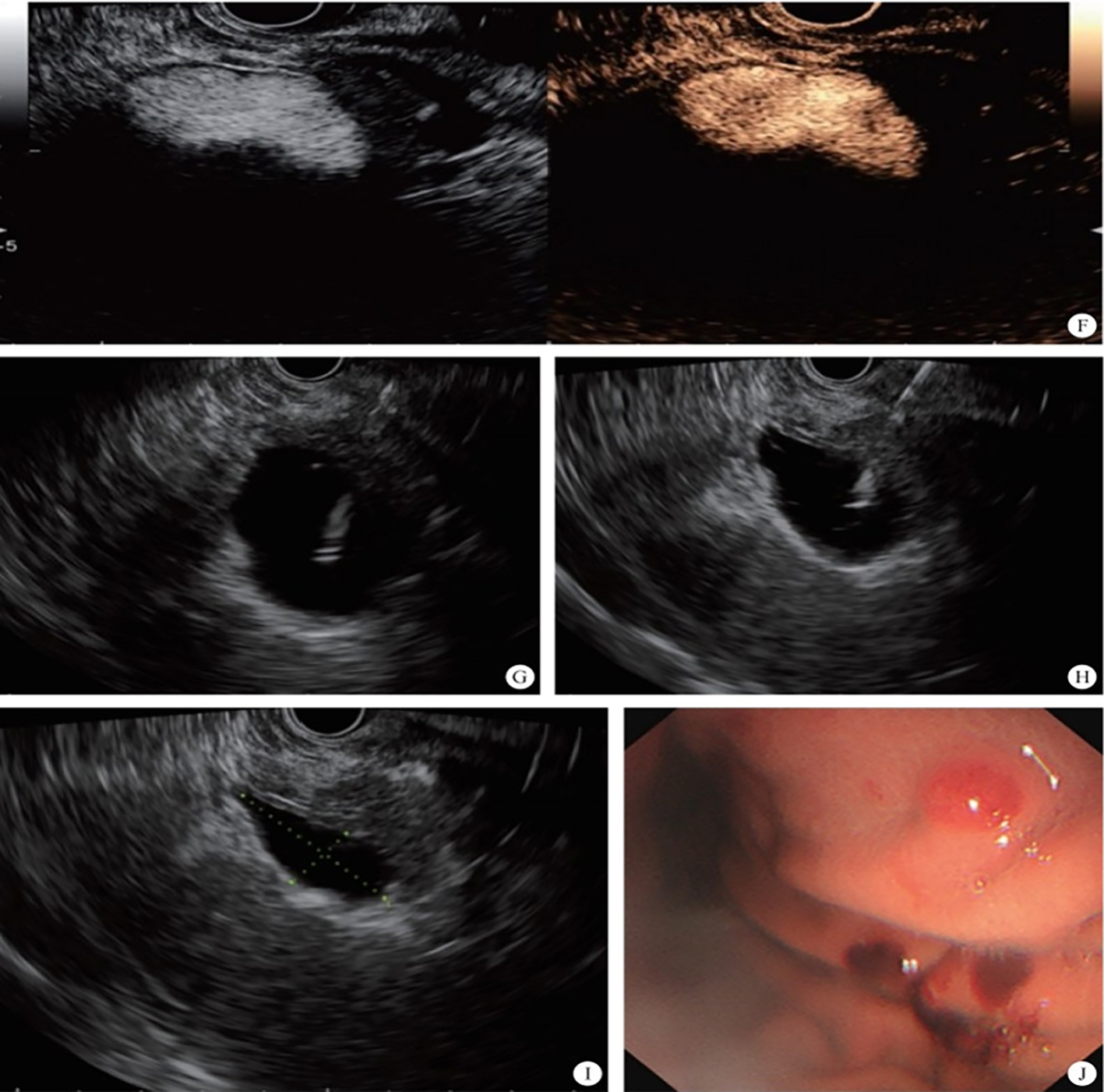

FIG. 12.1 — Surgical steps of EUS-guided polidocanol ablation. A. EUS Assessment: A anechoic structure approximately 3.75 cm × 3.16 cm is observed in the tail of the pancreas, with small septal-like structures inside and no papillary structures. B. EUS Contrast imaging: the cyst wall shows significant enhancement, while the septa are minimally enhanced. C. EUS-guided fine needle puncture: the puncture needle is seen positioned within the cyst. D. Aspiration of a clear, colorless, transparent cyst fluid: No particulate matter is observed. E. Introduction of Spyglass via 19G needle: a tree-like vascular network is visualized within the cyst. F. Contrast imaging: it is confirmed that the cystic cavity is not connected to the pancreatic duct. G. Aspiration of cystic contents: efforts are made to aspirate as much gas and fluid as possible from the cyst cavity. H. Injection of an ablation agent (polidocanol): the ablation agent is injected through the puncture needle, followed by three lavage cycles. I. Retention of approximately two-thirds of polidocanol in the cyst: the appropriate amount of ablation agent is left within the cyst. J. Removal of the puncture needle: the puncture site is observed for any signs of bleeding, confirming no hemorrhage.

Refer to video 12.1, Super minimally invasive polidocanol ablation for pancreatic cystic tumors via a puncture channel (See online resources).
(5) Assessment of the feasibility of polidocanol ablation: Polidocanol ablation is performed. Prior to ablation, as much cystic fluid as possible must be aspirated. An equal volume of polidocanol to the aspirated cystic fluid should be used for ablation, followed by three lavage cycles, each lasting approximately 3 min. Finally, polidocanol is left within the cyst.
(6) Removal of the puncture needle: observe the puncture site for any signs of bleeding, and then retract the endoscope.
12.1.6 Postoperative Management
Postoperative fasting for three days, with water restriction for one day. On the following morning, blood routine tests and pancreatic function assessments must be completed. Routine intravenous administration of proton pump inhibitors and antibiotics must be carried out for three days. If symptoms such as abdominal pain occur, blood amylase and lipase levels must be promptly rechecked, and, if necessary, abdominal imaging must be performed. If pancreatitis or hyperamylasemia develops, pancreatic enzyme inhibitors for symptomatic support should be administered. If an infection is present, the duration of antibiotic therapy must be extended or the antibiotics adjusted as needed. After three days, the patient switches to a normal diet.
12.1.7 Postoperative Follow-Up
(1) Follow-up of patients who have undergone ablation: they should undergo regular follow-up monitoring. Follow-up is necessary both to assess the efficacy and complications of the ablation procedure and to monitor the potential development of pancreatic cancer. Pancreatic CT and/or MRI should be performed three months after surgery, with subsequent follow-up at intervals of six months and one year.
(2) Efficacy assessment after ablation: the effectiveness of ablation is mainly judged based on the degree of tumor shrinkage observed in postoperative imaging.
Complete disappearance: no visible lesion on imaging. Nearly complete disappearance: volume reduction ≥95%. Significantly effective: volume reduction between 94% and 75%. Effective: volume reduction between 74% and 25%. Stable: volume reduction between 24% and 0%. Progressive: volume increase. CR (Complete Response): complete or nearly complete disappearance of the tumor. PR (Partial Response): significantly effective and effective results. Non-response: stable or progressive disease.
12.1.8 Hot-Spot Issues and Research Progress in the Consensus
Traditional imaging techniques have limited diagnostic accuracy for pancreatic cystic neoplasms (PCN). The advent of endoscopic ultrasound (EUS) and its guided diagnostic technologies has significantly improved the diagnostic accuracy for PCN. Moreover, various diagnostic techniques can be combined to complement each other’s advantages. The accuracy of EUS diagnosis is closely related to the experience of the operator, with significant regional differences in diagnostic levels within the country. Future efforts should focus on standardized training to enhance the overall level of EUS diagnosis. In PCN, the choice of EUS-related diagnostic techniques should be individualized based on factors such as the patient’s clinical characteristics, patient preferences, available hardware, and the operator’s experience. The standardization and guidelines for each diagnostic method require further research, and in the future, comprehensive health economics studies should be conducted to develop standardized protocols for EUS diagnosis. With the continuous advancement of endoscopic equipment and accessories, the value of EUS in the diagnosis and treatment of PCN will continue to expand.
The development of PCN ablation techniques has been relatively slow since the first report in 2005, with few researchers involved and a potential bias in the reported cases. The number of reported cases is relatively small, and there is no consensus on the evaluation of the effectiveness of ablation. The credibility of the conclusions regarding ablation remains insufficient, and prospective, multicenter, large-sample, long-term follow-up studies are needed for further validation. Currently, there is a lack of long-term follow-up studies on the ablation of lesions, and the long-term efficacy and safety of ablation are still unclear. Most studies have not set up control groups, and comparative studies on the effectiveness of different ablation agents need further improvement. The operational guidelines for EUS-guided ablation also require further research and validation.
12.2 Super Minimally Invasive Removal of Pancreatic Duct Stones
12.2.1 Overview
Pancreatic duct stones are formed due to the precipitation of calcium ions or proteins in the pancreatic juice, resulting in mixed crystals or protein plugs within the pancreatic duct. These stones can occur in the main duct or its branches and are often secondary to chronic pancreatitis, leading to upstream ductal obstruction and recurrent episodes of pancreatic inflammation and abdominal pain. In patients with chronic pancreatitis, the incidence of pancreatic stones increases progressively from 50 to 100% over a period of five to fourteen years following the onset of the disease.
The treatment principle for pancreatic duct stones is to relieve obstruction, remove the stones completely, ensure unobstructed drainage, and alleviate the pain caused by the obstruction. Patients with pancreatic duct stones commonly present with non-specific symptoms such as recurrent persistent upper abdominal pain, diarrhea, indigestion, and weight loss. Ultrasound (US) can correctly diagnose the condition in the majority of cases, typically showing ductal dilation and hyperechoic foci within the duct. Magnetic Resonance Cholangiopancreatography (MRCP) has now become a non-invasive diagnostic method for pancreatic duct stones, fully displaying the condition of the pancreatic duct, identifying the location and number of stones, and revealing a ‘string of beads’ appearance for multiple small stones. Computed Tomography (CT) can also diagnose the disease, typically showing uneven enlargement of the pancreas, pancreatic duct stones, and ductal dilation.
12.2.2 Endoscopic Super Minimally Invasive Removal of Pancreatic Duct Stones via the Duodenal Papilla + Extracorporeal Shockwave Lithotripsy (ESWL)
(I) Overview
The 2020 International Consensus Guidelines on Chronic Pancreatitis established extracorporeal shock wave lithotripsy (ESWL) as the first-line treatment for pancreatic duct stones, with endoscopic treatment recommended for the removal of residual small or fragmented stones after ESWL. After ESWL, pancreatic duct stones can be treated in combination with endoscopic Super Minimally Invasive removal via the duodenal papilla. Several studies have demonstrated that the use of ESWL for pancreatic duct stones leads to a high stone clearance rate, significant improvement in postoperative pain, and a marked increase in the quality of life of patients.
ESWL is based on the physical principles of an electromagnetic pulse generator, using X-rays or ultrasound to precisely locate the pancreatic duct stones. It allows for a non-invasive treatment that passes through body tissues and directly delivers high-pressure, high-energy shock waves to the stones. The stones are shattered by thousands of shock waves within a few hours, facilitating their expulsion from the body. Early ESWL devices used a water-bath structure, which had the disadvantages of large size, imprecise positioning, complex operation, and increased patient discomfort. With technological advancements, the introduction of water-bag-based lithotripsy machines has significantly reduced patient pain during treatment and lowered the incidence of related complications, leading to widespread adoption. Currently, ESWL technology has matured, causing minimal tissue damage, offering a broader treatment range, and demonstrating effective results for stones located in deeper regions.
Initially, ESWL was mainly used for treating kidney stones. In 1987, ESWL was first applied by Sauerbruch et al. for the treatment of pancreatic duct stones, marking the beginning of its use in pancreatic duct stone therapy. Over the past 30 years, it has been regarded as a safe, effective, and non-invasive treatment method. In recent years, the application of ESWL in pancreatic duct stone treatment has increased significantly, achieving remarkable progress, and it has now become a first-line treatment option. The European Society of Gastrointestinal Endoscopy (ESGE) in 2018 recommended that the indications for ESWL in pancreatic duct stones include obstructive main pancreatic duct stones greater than 5 mm in the head/body of the pancreas, or cases where endoscopic stone removal fails due to pancreatic duct stones. With the optimization of the technology and the expansion of its indications, ESWL can also be applied to some special types of pancreatic duct stones. At Shanghai Changzhai Hospital’s Department of Gastroenterology, ESWL treatment for pancreatic duct stones was pioneered, with the combination of ESWL and endoscopic retrograde cholangiopancreatography (ERCP) achieving a 72% complete clearance rate of main pancreatic duct stones and a 90% successful drainage rate for the main pancreatic duct. Furthermore, ESWL has also proven effective for pancreatic duct stones after pancreatic surgery.
(II) Indications and contraindications of ESWL in the treatment of pancreatic duct stones.
1. Indications
Stone size: ESWL is primarily indicated for pancreatic duct stones with a diameter greater than 5 mm. For larger stones, traditional endoscopic stone removal methods often face difficulty in extracting the stones directly. ESWL, however, effectively fragments these stones into smaller, more easily expulsed particles.
Stone location: ESWL is suitable for pancreatic duct stones located in the head or the body of the pancreas. Stones in these areas are deeper, making traditional endoscopic retrieval techniques more difficult. ESWL, with its shockwave energy, can penetrate tissues and reach the stone site for fragmentation.
Associated pancreatic duct stricture: For patients with pancreatic duct stones complicated by ductal stricture, the combination of ESWL and endoscopic therapy offers better treatment outcomes. After effective stone fragmentation via ESWL, endoscopic retrograde cholangiopancreatography (ERCP) and other endoscopic interventions, such as ductal dilation and stent placement, can be performed to relieve the stricture and facilitate the passage of stone fragments.
Failure of ERCP stone removal: for patients in whom stone removal fails via ERCP, ESWL can serve as an adjunctive treatment. While ERCP is minimally invasive and has a fast recovery time, it may be challenging to extract certain complex stones directly. In such cases, ESWL can be used to fragment the stones, followed by ERCP to remove the fragments, improving stone clearance rates and patient outcomes.
2. Contraindications
The main contraindications for ESWL include pancreatic and related malignant diseases, the presence of other calcifications along the shockwave transmission path, severe cardiopulmonary dysfunction, and pregnancy.
(III) Surgical procedure and techniques
1. Preoperative preparation. To improve the efficacy and safety of ESWL, the International Consensus Guidelines on Endoscopic Intervention for Chronic Pancreatitis (2020) offers the following recommendations: preoperative preparation should include determining the stone burden, location, and nature; for stones that are radiolucent, a nasopancreatic duct catheter may be placed for auxiliary localization.
2. Surgical Process (figures 12.2 and 12.3)
Energy settings: one should begin with a lower energy setting and gradually increase the power to minimize the risk of renal injury.
Post-fragmentation management: for stones that do not pass spontaneously after fragmentation or can still be extracted, further stone retrieval can be performed via endoscopic retrograde cholangiopancreatography (ERCP);
Treatment sequence: typically, the treatment follows a sequence from the head to the tail of the pancreas. Each shockwave session should not exceed 5000 pulses, with a frequency generally between 90 and 120 pulses per minute. The duration of each treatment session is usually 60–90 min.
(IV) Hot-spot issues and research progress in the consensus
With the continuous advancement of medical technology, ESWL equipment is expected to undergo further optimization and improvements. Newer lithotripters will offer higher precision, lower tissue damage rates, and a broader range of applications. Additionally, emerging technologies such as digital pancreatic ductoscopy-assisted lithotripsy will provide more treatment options for pancreatic duct stones. As each patient with pancreatic duct stones presents with unique conditions and stone characteristics, individualized treatment will become a future trend. Physicians will tailor treatment plans based on the specific condition of each patient, including the selection of appropriate treatment methods and setting optimal lithotripsy parameters, in order to improve treatment outcomes and enhance the quality of life of patients. The treatment of pancreatic duct stones involves multiple medical specialties, including gastroenterology, hepatobiliary surgery, and radiology. In the future, multidisciplinary collaboration will become a key model for treating pancreatic duct stones. Through the joint efforts of a multidisciplinary team, more comprehensive and precise treatment plans can be provided, improving both treatment outcomes and patient satisfaction.

FIG. 12.2 — Endoscopic super minimally invasive removal of pancreatic duct stones via the duodenal papilla + extracorporeal shock wave lithotripsy (ESWL). A. X-ray localization of pancreatic duct stones. B. Pancreatic duct stent placement via ERCP after ESWL.

FIG. 12.3 — Endoscopic super minimally invasive removal of pancreatic duct stones via the duodenal papilla. A. Small stone in the head of the pancreas in a patient with chronic pancreatitis. B. Balloon sweeping of pancreatic duct stones.
Multiple studies have confirmed the efficacy of ESWL in the treatment of large pancreatic duct stones. A meta-analysis revealed that the complete stone fragmentation rate with ESWL is 86.3%, the complete pancreatic duct clearance rate is 69.8%, and the complete pain relief rate during follow-up is 64.2%. Although ESWL is successful in the majority of patients, approximately 25% of patients experience stone recurrence during long-term follow-up. Factors such as isolated stones in the pancreatic head, low stone density, placement of a pancreatic duct stent prior to ESWL, and the use of pancreozymin are predictive factors for successful pancreatic duct clearance after ESWL. Severe complications from ESWL are rare, with the most common adverse event being postoperative pancreatitis, occurring in approximately 4% of cases. Additionally, ESWL may cause mild complications such as hematuria or skin bruising, which generally resolve on their own. For patients with contraindications, ESWL should be strictly avoided to prevent unnecessary harm.
1. ESWL combined with ERCP for pancreatic duct stone treatment. Although ESWL has shown significant efficacy in treating pancreatic duct stones, it often cannot achieve complete stone clearance when used alone, especially for larger or harder stones. After ESWL, further stone removal through ERCP is often necessary. ERCP allows for retrograde cannulation and contrast injection, providing a clear view of the pancreaticobiliary system for precise stone retrieval. Therefore, the combination of ESWL and ERCP has become one of the standard treatment options for pancreatic duct stones. This combination offers advantages such as minimal trauma, simplicity, and fewer complications. ESWL can fragment large stones into smaller pieces, making them easier to retrieve with ERCP, which can then directly clear residual stones after fragmentation, improving stone clearance rates. Furthermore, combined treatment can effectively alleviate pancreatic duct hypertension, improve pancreatic microcirculation, and help mitigate pancreatic tissue damage.
2. Application of ESWL in the treatment of pancreatic duct strictures. Pancreatic duct stricture is a common complication of pancreatic duct stones, often caused by chronic inflammation or stone compression. Strictures can lead to impaired pancreatic fluid drainage, elevated ductal pressure, and further pancreatic damage. Therefore, treatment of pancreatic duct stones should also address pancreatic duct stricture. For patients with pancreatic duct strictures, ESWL can indirectly improve ductal stenosis by fragmenting the stones and alleviating ductal compression. Additionally, combining ESWL with ERCP enables procedures like sphincterotomy, probing or balloon dilation, and pancreatic duct stenting to further improve stricture conditions. Pancreatic duct stenting is a critical approach for treating ductal strictures. Plastic stents, which are widely used, can be tailored in diameter and length based on the patient’s condition. After stent placement, the stricture and obstruction are effectively relieved, improving pancreatic fluid drainage. Stent placement is also important for draining residual stones after ESWL, especially in patients with pancreatic duct stricture and downstream stone obstruction in the pancreatic head.
12.2.3 Pancreatic Duct Stones and Super Minimally Invasive Stone Removal via Pancreatoscopy
(I) Overview
Pancreatic duct stones commonly occur as a sequela of chronic pancreatitis, with an incidence as high as 90% in patients with alcoholic chronic pancreatitis. Conventional ERCP techniques, including sphincterotomy, balloon or basket stone extraction, and mechanical lithotripsy, are effective for treating smaller (≤5 mm) pancreatic duct stones. However, for larger (>5 mm) or multiple pancreatic duct stones, achieving complete stone removal presents technical challenges.
Oral pancreatoscopy-guided lithotripsy (LL/EHL) provides a novel treatment option for difficult pancreatic duct stones. A study investigating main pancreatic duct stones compared the therapeutic outcomes of ERCP with or without oral pancreatoscopy-guided lithotripsy. The results demonstrated that the combination of ERCP and oral pancreatoscopy-guided lithotripsy significantly improved the technical success rate compared to conventional ERCP stone extraction (98.8% vs. 87.6%, P < 0.001). Notably, the treatment group using the combined therapy had larger stones, more stones, and a higher proportion of impacted stones.
(II) Indications and contraindications
1. Indications: ① large and dense pancreatic duct stones; ② pancreatic duct stones complicated by ductal stenosis; ③ pancreatic duct stones in the pancreatic tail; ④ obstructive pancreatic duct stones without complications.
2. Contraindications: ① suspected mass in the pancreatic head; ② patients with a native papilla and very distal stones in the pancreatic duct; ③ poor general health, with moderate or severe ascites; ④ during acute cholangitis.
(III) Preoperative preparation
1. Gastrointestinal preparation. A liquid diet should be started one to two days before surgery. The patient should fast for 6 h and abstain from water for 2 h prior to anesthesia. For colorectal surgeries, laxatives or enemas may be used to clean the intestines.
2. Preoperative examinations. Preoperative abdominal CT scans should be performed to determine the location and the nature of the stones. Complete blood count, blood biochemistry, coagulation function tests, as well as electrocardiogram and echocardiography, should be conducted.
3. Discontinuation of medications. Medications affecting coagulation: aspirin should be stopped seven days before surgery, low-molecular-weight heparin should be stopped three days before surgery, heparin should be stopped 6–12 h before surgery, and warfarin should be stopped three days prior.
(IV) Surgical steps and techniques
The patient is placed in the left lateral decubitus position under general anesthesia. The pancreatoscope is inserted through the duodenoscope into the pancreatic duct (PD). Electrohydraulic lithotripsy (EHL) and laser lithotripsy (LL) are delivered via a water medium, with the probe tip in direct contact with the stone or within 1–2 mm of the stone. The EHL settings (1.9F fiber; Autolith, Northgate Technologies, Elgin, IL) are 10–20 pulses per second, with a power range of 50–100 W. For LL (200-μm, 272-μm, or 365-μm fibers, Versa Pulse Power Suite 20-W Holmium laser, New Star, Roseville, CA), the energy range is 0.8–2.5 J, frequency is 8–15 Hz, and power is 9–30 W. Both forms of intracorporeal lithotripsy can be performed during the same treatment session. After lithotripsy, residual stone fragments smaller than 5 mm are removed using a retrieval balloon, a basket, or a combination of both methods.
(V) Postoperative management
Postoperatively, the patient’s vital signs and abdominal signs, as well as any occurrence of hematemesis or melena, must be closely monitored. Complete blood count, intravenous amylase, and lipase should be tested at 24 h and 72 h after surgery. If clinical assessment suggests the possibility of gastrointestinal bleeding, perforation, post-endoscopic pancreatitis (PEP), or other adverse events, abdominal CT and/or endoscopic examination should be performed promptly to clarify the condition.
The patient should remain fasting for six days postoperatively. If there are no abnormal abdominal signs and if postoperative laboratory results are within normal limits, the patient can gradually transition to a liquid diet and then soft foods. On postoperative day 6, proton pump inhibitors (PPIs) and antibiotics are administered. If pancreatitis or gastrointestinal bleeding occurs postoperatively, the duration of treatment should be extended accordingly. Additionally, if pancreatitis develops, somatostatin therapy should be promptly initiated. It should be noted that for gastrointestinal bleeding, if conservative treatment is ineffective, endoscopic, surgical, or interventional methods should be promptly employed for hemostasis.
(VI) Postoperative follow-up
Postoperatively, we recommend close monitoring through telephone and outpatient follow-up to document any adverse events. Additionally, patients are advised and encouraged to return for endoscopic re-examination at three months, six months, one year, two years, three years, four years, and five years after surgery.
(VII) Hot-spot issues and research progress in the consensus
Between 50% and 90% of patients with chronic pancreatitis develop pancreatic duct stones, which can lead to pancreatic duct hypertension and severe pain. This pain is typically managed with opioid medications, which not only pose a risk of dependence but also increase hospitalization rates. Standard methods for pancreatic duct stone removal include endoscopic retrograde cholangiopancreatography (ERCP) combined with sphincterotomy, distal stricture dilation, balloon and/or basket stone extraction, and extracorporeal shock wave lithotripsy (ESWL) for larger stones.
Although ERCP techniques have been developed and refined, they are still limited by the fact that endoscopists can only observe structures indirectly via fluoroscopy. This indirect imaging is particularly limited in cases of larger pancreatic duct stones and uncertain ductal strictures. Additionally, due to the small diameter of the pancreatic duct, ERCP often fails to completely remove or clear stone fragments, including those located in the pancreatic tail, in cases with ductal stricture, and large stones embedded within the duct. On the other hand, the disadvantages of ERCP are as follows. ① Blind spots during cholangiopancreatic duct exploration, where imaging is limited to indirect angiography, making it difficult to directly assess lesions. ② Low stone removal rates and poor treatment efficiency for complex stones, particularly those located in the pancreatic tail with associated ductal stricture and large diameter stones, or stones embedded in the pancreatic duct. ③ Low positive rates for cytology brushings from lesions, making it difficult to meet clinical diagnostic and therapeutic needs. ESWL is widely used in Europe and Asia; however, it has certain limitations, including limited effectiveness for high-density or radiolucent stones, a restricted ability to treat multiple stones in a single session, and ineffectiveness in treating downstream pancreatic duct strictures, which can lead to hematoma formation, reduced stone fragment clearance, and may be associated with stone recurrence. Therefore, both ERCP and ESWL may be limited by imprecision, availability, and reduced effectiveness in removing large impacted stones. The introduction of oral pancreatoscopy has made targeted treatment of pancreatic duct stones possible, including electrohydraulic lithotripsy (EHL) and laser lithotripsy (LL). Previous studies have shown that pancreatoscopy-guided lithotripsy for difficult pancreatic duct stones achieves complete stone clearance rates of 79%–100%. Brewer Gutierrez et al. conducted a retrospective multicenter study involving 109 patients with symptomatic pancreatic duct stones, most of whom had previously undergone conventional ERCP stone extraction or ESWL. The results showed a complete stone clearance rate of 89.9%, with a single-session success rate of 73.5%, and the therapeutic efficacy of EHL and LL was comparable (complete stone clearance: 94.1% vs. 100%, P = 0.243; single-session success rate: 77.1% vs. 70%, P = 0.5), though EHL had a longer average procedure time (74.4 vs. 53.8 min, P < 0.001).
The goal of pancreatic duct stone lithotripsy is to alleviate clinical symptoms such as abdominal pain and improve the patient’s quality of life. Related research has shown that pancreatoscopy-guided lithotripsy for pancreatic duct stones results in clinical symptom relief rates ranging from 72% to 100%, with no significant difference between EHL and LL in terms of clinical relief rates (88% vs. 88.9%, P = 1). However, there is still limited research on the super minimally invasive stone removal technique using oral pancreatoscopy, and further studies are needed to evaluate its safety, efficacy, and broader applicability.
12.3 Pancreatic Fistula – Super Minimally Invasive Drainage
12.3.1 Overview
Pancreatic fistula (PF) refers to a series of clinical syndromes caused by rupture of the main pancreatic duct and/or its branches, leading to abnormal communication between the pancreatic ductal epithelium and other epithelial tissues, resulting in the leakage of enzyme-rich fluid. This condition is also commonly referred to as disconnected pancreatic duct syndrome (DPDS). Clinically, the primary causes of pancreatic fistula are acute and chronic pancreatitis, pancreatic and peripancreatic surgical procedures, and pancreatic trauma. In case of acute necrotizing pancreatitis (ANP), the incidence of PF is approximately 18%–46%. Pancreatic fistulas often lead to pancreatic ascites, pseudocysts, or encapsulated necrosis, which may result in severe intra-abdominal infections, pseudoaneurysm rupture, bleeding, and other life-threatening complications.
Currently, the treatment options for pancreatic fistula include conservative drug therapy, percutaneous catheter drainage (PCD), endoscopic super minimally invasive drainage, and surgical intervention. Among these, endoscopic super minimally invasive drainage, which primarily includes ERCP-guided drainage via the duodenal papilla and EUS-guided transgastric/transduodenal drainage, has become a preferred method for treating pancreatic fistulas. When necessary, endoscopic debridement can be performed during the procedure. With the rapid development of super minimally invasive concepts and techniques, along with ongoing clinical research, endoscopic super minimally invasive drainage has gradually become the first-choice intervention for pancreatic fistula treatment.
12.3.2 Diagnosis of Pancreatic Fistula
Clinically, the diagnostic methods for pancreatic fistula primarily include CT, MRI, MRCP, EUS, ERCP, and ductography. Among these, ERCP and ductography can dynamically display the location, size of the fistula, outflow tract, and communication with other structures, making them the gold standard for diagnosing pancreatic fistulas. Compared to ERCP, MRCP is a non-invasive examination that provides detailed imaging of the pancreatic ductal anatomy and peripancreatic fluid collection, with an accuracy rate greater than 90%, making it an ideal initial diagnostic approach for pancreatic fistulas. Secretin-enhanced MRCP (sMRCP) further improves the accuracy of diagnosing pancreatic fistulas.
Currently, the diagnostic criteria for pancreatic fistulas associated with acute necrotizing pancreatitis (ANP) include: ① The long diameter of pancreatic necrosis greater than 2 cm; ② Viable pancreatic tissue at the distal end of the necrotic area; ③ Contrast extravasation observed during ERCP. For postoperative pancreatic fistulas (POPF), the diagnosis mainly relies on the surgical history, the amount and nature of postoperative drainage fluid, and other factors. The diagnostic criteria for postoperative pancreatic fistula, as defined by the International Study Group of Pancreatic Surgery (ISGPS), are: fluid output from the abdominal drainage tube for three days or more postoperatively, with amylase concentration exceeding three times the upper limit of normal serum amylase. Based on the clinical severity of the fistula, POPF is classified into three grades. Grade A: biochemical fistula, with no clinical adverse consequences. Grade B: affects postoperative recovery and requires clinical treatment or intervention, such as infection, need for percutaneous or endoscopic drainage, blood transfusion, or embolization for bleeding; Grade C: associated with organ failure and severe life-threatening complications, often requiring surgical intervention. In 2020, the Chinese Society of Gastroenterology, Endoscopy Branch proposed an endoscopic classification standard based on fistula location, fistula type, and prior pancreatic surgery history. This classification helps in guiding endoscopic treatment strategies.
12.3.3 Indications and Contraindications for Endoscopic Super Minimally Invasive Drainage of Pancreatic Fistula
1. Indications. The primary indications for endoscopic super minimally invasive drainage are pancreatic fistula patients with significant symptoms (such as abdominal pain, infection, obstruction, etc.) who have not responded to conventional conservative treatment.
2. Contraindications. Contraindications to endoscopic examination; short life expectancy; pregnancy; coagulation disorders; active pancreatitis; pancreatic necrosis or signs of malignancy.
12.3.4 Surgical Methods and Procedures
1. ERCP-guided drainage via duodenal papilla. This includes ERCP-guided nasopancreatic duct placement and pancreatic duct stent placement. The key to this technique is bridging the pancreatic duct across the fistula, allowing thorough drainage of the upstream pancreatic fluid. It is primarily applicable to patients with main pancreatic duct injury or stenosis, with an overall treatment success rate of about 65%. However, for patients with complete main pancreatic duct rupture or those with gastrointestinal bypass, ERCP-guided pancreatic duct drainage is more challenging, with a success rate of less than 30%. The ERCP stent should bridge the fistula and the stenotic segment, typically remaining in place for four to eight weeks before follow-up and replacement until the fistula completely heals. If conventional ERCP pancreatic duct cannulation is difficult or the guidewire cannot be passed retrogradely through the pancreatic fistula or stenotic segment, EUS-guided pancreatic duct rendezvous combined with ERCP-guided pancreatic stent placement may be attempted. The specific procedural steps of ERCP pancreatic stent placement are not detailed here.
2. EUS-guided transgastric/transduodenal through-the-wall drainage. This procedure is primarily aimed at draining peripancreatic fluid collections caused by pancreatic fistula, and when necessary, endoscopic debridement can be performed under direct visualization. This technique has been widely used for the treatment of ANP with peripancreatic fluid collections and has gradually become the preferred treatment approach. Clinically, the commonly used through-the-wall drainage stents are the double-pigtail plastic stents and the newer lumen-apposing metal stents (LAMS).
12.3.5 Hot-Spot Issues in the Consensus
EUS-guided through-the-wall drainage for the treatment of pancreatic fistula and peripancreatic fluid collections follows a similar treatment principle, timing, and procedure as for ANP with peripancreatic fluid. The timing for treatment should be delayed until approximately four weeks after onset, unless the condition worsens, at which point fluid encapsulation and cyst wall formation are complete. The overall clinical success rate for treating ANP with pancreatic fistula and peripancreatic fluid collections is above 80%, while the overall clinical success rate for postoperative pancreatic fistula is approximately 88.9% (ranging from 79% to 100%). The following aspects should be noted regarding the procedure.
1. Puncture site. The puncture site should be selected near the junction of the stomach or duodenal wall and the cyst wall, ideally within 1 cm, avoiding vascular structures. Additionally, puncture through the gastric wall is preferred for drainage.
2. Stent selection. The first choice is the LAMS (lumen-apposing metal stent) due to its large lumen, good apposition, and ability to provide a channel for debridement intervention. It is recommended in several relevant guidelines. If LAMS is unavailable or if the peripancreatic fluid is homogenous with less necrotic material, a double-pigtail plastic stent (10 Fr in diameter) may be used as an alternative. Clinical outcomes, stent displacement, and recurrence rates are similar between single and multiple plastic stents.
3. Stent placement duration. It is currently recommended to leave a double-pigtail plastic stent in place in the long term, replacing it as needed, until the fluid collection resolves and the fistula heals. Once the fluid collection is resolved with the LAMS stent, it is advisable to continue with the double-pigtail plastic stent after removing the LAMS stent.
4. Debridement selection. If there is a significant amount of necrotic tissue in the peripancreatic fluid collection, and drainage alone is insufficient, endoscopic debridement should be performed. Debridement can be carried out immediately after LAMS stent placement, and should be performed as needed, depending on clinical symptoms and infection control. After debridement, acid-suppressive drugs should be avoided to enhance the natural self-clearing action of stomach acid on the necrotic tissue.
12.4 Pancreatic Walled-Off Necrosis: Super Minimally Invasive Drainage
12.4.1 Overview
Walled-off necrosis (WON) commonly results from acute pancreatitis, chronic pancreatitis, pancreatic trauma, and pancreatic duct obstruction. It is characterized by the accumulation of necrotic tissue from pancreatic fluid, blood leakage, and pancreatic autodigestion, which cannot be absorbed and leads to the formation of local tissue necrosis. Clinically, WON is often associated with symptoms such as abdominal pain, bloating, masses, infection, jaundice, bleeding, and splenic infarction.
WON can be treated through super minimally invasive drainage via a gastric puncture channel, where a stent is placed. Endoscopic necrosectomy (DEN) under direct visualization can be performed concurrently to achieve therapeutic goals. This is one of the endoscopic treatment methods for pancreatic walled-off necrosis, with a short-term clinical success rate (three to six months) ranging from 86.3% to 90%, and a complication rate of approximately 9.3%–11.3%. Currently, DEN has become one of the first-line treatment options for pancreatic walled-off necrosis.
12.4.2 Diagnosis of WON
The diagnosis of Walled-off Necrosis (WON) requires two conditions: a history of acute pancreatitis lasting more than four weeks. CT or MRI imaging showing the following criteria: mixed fluid and non-fluid components at varying degrees. A mature, fully encapsulated cyst wall. Located inside and/or outside the pancreas.
12.4.3 Indications and Contraindications
1. Indications. This procedure is indicated in patients with a history of pancreatitis and Walled-off Necrosis (WON) with a diameter ≥6 cm, accompanied by any of the following symptoms: refractory abdominal pain; gastric outlet or biliary obstruction; persistent systemic illness, anorexia, and weight loss; rapidly expanding WON in the short term; infected WON.
2. Contraindications. ① Patients in poor general condition who cannot tolerate endoscopic examination, such as those with severe pulmonary or cardiac diseases, coagulation disorders, or bleeding tendencies, or those with contraindications for endoscopic drainage, such as after gastrointestinal reconstruction surgery, acute upper gastrointestinal inflammation, ulcers, or upper gastrointestinal strictures or obstructions preventing endoscopic access. ② Cysts located near major blood vessels, especially with accompanying aneurysms, cystic bleeding, cyst rupture, or varices in the gastroesophageal junction, where there is no ideal puncture site. ③ Suspected malignancy. ④ A distance > 1 cm between the cyst wall and the gastric wall. ⑤ Patients with a history of allergy to nickel-titanium (NiTi). ⑥ Pregnant or breastfeeding women.
12.4.4 Surgical Procedures and Techniques
(I) Stent placement
(1) Anesthesia and positioning: all patients undergo general anesthesia. For larger lesions, tracheal intubation should be considered. Patients are positioned in the left lateral decubitus position. Attention should be paid to maintaining body temperature, adjusting room temperature, and warming infusion fluids to prevent hypothermia and pressure ulcers.
(2) The lesion’s location, size, blood flow, and intracystic contents are evaluated using endoscopic ultrasonography. A suitable puncture site is selected, and a 19G puncture needle is used for the puncture. A guidewire is then inserted, ensuring the guidewire forms a loop within the PPC (pancreatic pseudocyst) or WON (walled-off necrosis) (figure 12.4).

FIG. 12.4 — Steps of ultrasound-guided puncture in super minimally invasive drainage of walled-off pancreatic necrosis (WON) via gastric puncture channel. A. Endoscopic ultrasonographic evaluation of the WON. B. Puncture of the lesion using a 19G puncture needle. C. Insertion of the guidewire with coiling within the cyst.
(3) An electrosurgical knife is inserted along the guidewire to incise the gastric wall and the cyst wall.
(4) A drainage stent is placed and the cyst-end flange of the stent is deployed within the WON under the guidance of an endoscopic ultrasound (EUS).
(5) Switch to the white-light endoscopic view to release the gastric-end flange of the stent and observe the drainage of cyst fluid. The ‘one-step’ hot stent allows for direct puncture and stent deployment without the need for initial puncture with a needle, followed by guidewire insertion and subsequent stent delivery system exchange (figure 12.5).
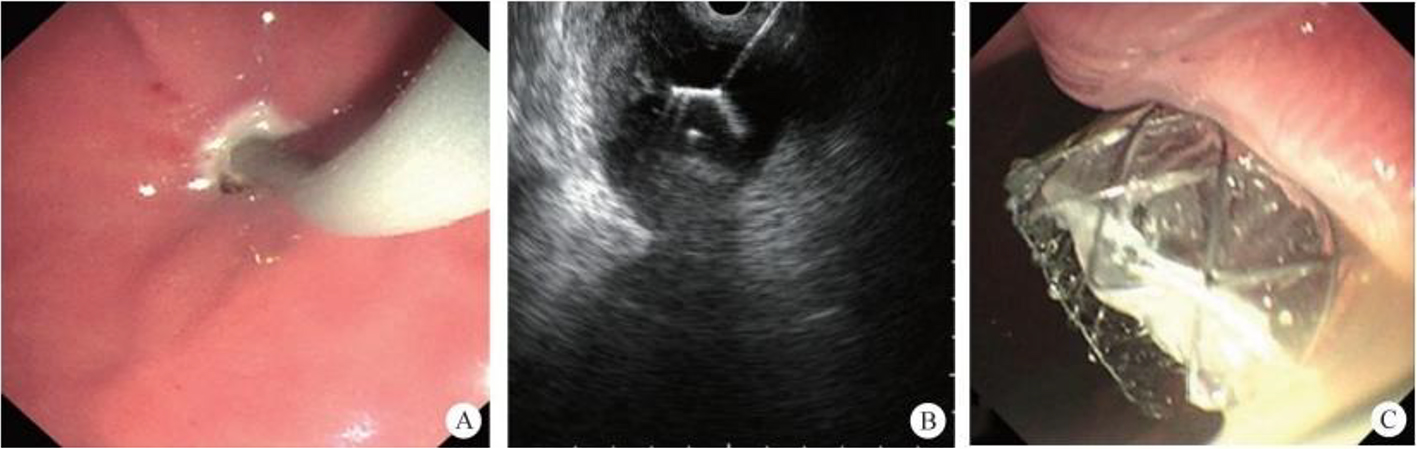

FIG. 12.5 — Steps of stent placement in super minimally invasive drainage of pancreatic walled-off necrosis via a gastric puncture pathway. A. Incision of the gastric wall and cyst wall. B. Deployment of the cyst-end flange. C. Deployment of the gastric-end flange.
Technical tips: when deploying the cyst-end flange of the stent, ensure under EUS guidance that the flange marker reaches the interior of the cyst before releasing. For the deployment of the gastric-end flange, confirm under white-light endoscopic visualization that the second release marker is visible before proceeding with deployment to avoid stent deployment failure.
(II) Direct endoscopic debridement (figure 12.6).
(1) The procedure is performed under general or intravenous anesthesia with the patient in the left lateral decubitus position.
(2) A therapeutic gastroscope is introduced into the stomach to check the proper positioning of the stent, and the gastric-end flange of the stent is flushed.
(3) The gastroscope is advanced through the stent into the cyst cavity, where a normal saline solution is used for flushing and suction. The solid necrotic tissue within the cyst is removed using snares, baskets, or similar devices.

FIG. 12.6 — Steps of super minimally invasive debridement of pancreatic walled-off necrosis via gastric puncture pathway. A. Flushing and removal of necrotic material from the gastric-end flange. B. Entry into the cyst cavity for removal of necrotic material.
Technical tips: the necrotic tissue should be removed under a clear visual field with gentle maneuvers to avoid massive bleeding. For cyst cavities with septations, thorough debridement is necessary to prevent recurrence. When there is a large amount of necrotic tissue that is difficult to remove, devices such as a DualKnife or snare can be used to divide the tissue into smaller pieces, which can then be removed with the assistance of a transparent cap.
12.4.5 Postoperative Management
Patients should be kept nil per os (NPO) with acid suppression, enzyme inhibition, and fluid replacement for one day. Prophylactic anti-infection measures, such as third-generation cephalosporins combined with ornidazole, should be administered for 24 h. Gradual dietary progression is recommended: liquid diet → semi-liquid diet → regular diet. Digestive enzyme therapy should be continued for two weeks postoperatively. Regular monitoring of blood glucose, blood gas, and biochemistry is necessary to promptly correct metabolic disturbances.
12.4.6 A Classic Case
Case history: A young male patient was admitted with a primary complaint of “intermittent abdominal pain for more than three years.” He had a prior history of pancreatitis for over four years. CT imaging revealed multiple cystic low-density lesions in the head, neck, and body of the pancreas, with atrophy of the pancreatic body and tail (figure 12.7).
Diagnosis: pancreatic walled-off necrosis (WON)

FIG. 12.7 — Contrast-enhanced CT and MRI reveal heterogeneous signals within the cyst, with a lesion size of approximately 7.8 cm × 4.5 cm.
After stent placement, two sessions of direct endoscopic debridement were performed. Endoscopic findings showed a large amount of necrotic tissue and exposed blood vessels (figure 12.8).

FIG. 12.8 — First session of direct endoscopic debridement.
After the second debridement, the necrotic tissue within the cyst was almost completely removed (figure 12.9). A follow-up CT scan one month postoperatively showed that the WON had largely resolved, and the stent was subsequently removed under endoscopic guidance.
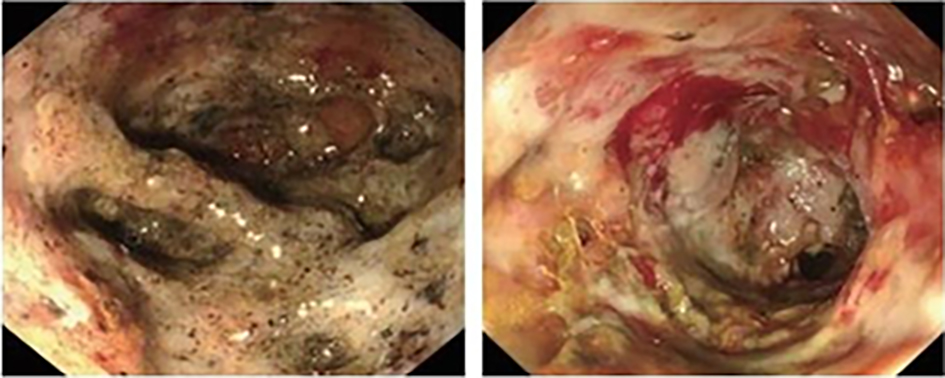

FIG. 12.9 — Second session of direct endoscopic debridement.
Refer to video 12.2, Super minimally invasive drainage of pancreatic walled-off necrosis via a gastric puncture pathway (See online resources).
12.4.7 Hot-Spot Issues and Research Progress in the Consensus
1. Which stent is safer and more effective for super minimally invasive drainage via gastric wall puncture? Endoscopic drainage primarily utilizes plastic and metal stents. Current studies suggest that metal stents provide better drainage efficacy and fewer complications compared to plastic stents. Regarding the choice of metal stent, the ‘one-step’ hot stent is simpler to operate than the self-expanding metal stent but is more expensive. Further research is needed to compare and confirm the effectiveness and safety of these two types of stents.
2. Need for direct endoscopic necrosectomy (DEN) after stent placement. Studies have shown that most cases of walled-off necrosis (WON) achieve effective drainage after metal stent placement without requiring additional endoscopic debridement. Following endoscopic drainage and stent placement, it takes several days for the fistula to mature and the stent to fully expand to allow endoscope passage. Performing endoscopic debridement too early may require additional dilation of the fistula, increasing costs and potentially raising the risks of stent displacement, WON recurrence, and intra-abdominal infection. Therefore, most endoscopists prefer to perform endoscopic debridement two to three days after stent placement. Whether debridement is necessary should be based on the patient’s clinical condition and the effectiveness of the drainage.
References
[1] Li Z., Jin Z., Li X. (2023) Chinese guidelines for the diagnosis and treatment of pancrestic cystic neoplasm(2022), J. Clin. Hepatol. 39(2), 290–298.
[2] Gardner T. B., Park W. G., Allen P. J. (2024) Diagnosis and management of pancreatic cysts, Gastroenterology.
[3] Smith Z. L., Satyavada S., Simons-Linares R., et al. (2022) Intracystic glucose and carcinoembryonic antigen in differentiating histologically confirmed pancreatic mucinous neoplastic cysts, Am. J. Gastroenterol. 117(3), 478–485.
[4] Cho S. H., Song T. J., Seo D. W., et al. (2022) Efficacy and safety of EUS-guided through-the-needle microforceps biopsy sampling in categorizing the type of pancreatic cystic lesions, Gastrointest Endosc. 95(2), 299–309.
[5] Gaujoux S., Brennan M. F., Gonen M., et al. (2011) Cystic lesions of the pancreas: changes in the presentation and management of 1424 patients at a single institution over a 15-year time period, J. Am. Coll. Surg. 212(4), 590–600, discussion -3.
[6] Stark A., Donahue T. R., Reber H. A., et al. (2016) Pancreatic cyst disease: a review, Jama 315(17), 1882–93.
[7] Cahalane A. M., Purcell Y. M., Lavelle L. P., et al. (2016) Which is the best current guideline for the diagnosis and management of cystic pancreatic neoplasms? An appraisal using evidence-based practice methods, Eur. Radiol. 26(9), 3121–3128.
[8] Yoon J. G., Smith D., Ojili V., et al. (2021) Pancreatic cystic neoplasms: a review of current recommendations for surveillance and management, Abdom. Radiol. (NY) 46(8), 3946–3962.
[9] Du C., Chai N. L., Linghu E. Q., et al. (2020) Endoscopic ultrasound-guided injective ablative treatment of pancreatic cystic neoplasms, World J. Gastroenterol. 26(23), 3213–3224.
[10] Cho S. H., Seo D. W., Oh D., et al. (2024) Long-Term outcomes of endoscopic ultrasound-guided ablation vs. surgery for pancreatic cystic tumors, Clin. Gastroenterol. Hepatol.
[11] Vege S. S., Ziring B., Jain R., et al. (2015) American gastroenterological association institute guideline on the diagnosis and management of asymptomatic neoplastic pancreatic cysts, Gastroenterology 148(4), 819–822.
[12] Basar O., Brugge W. R. (2017) Pancreatic cyst guidelines: which one to live by?, Gastrointest. Endosc. 85(5), 1032-1035.
[13] Elta G. H., Enestvedt B. K., Sauer B. G., et al. (2018) ACG clinical guideline: diagnosis and management of pancreatic cysts, Am. J. Gastroenterol. 113(4), 464–479.
[14] Pancreas T. E. S. G. O. C. T. O. T. (2018) European evidence-based guidelines on pancreatic cystic neoplasms, Gut. 67(5), 789–804.
[15] Milovic V., Wehrmann T., Dietrich C. F., et al. (2011) Extracorporeal shock wave lithotripsy with a transportable mini-lithotripter and subsequent endoscopic treatment improves clinical outcome in obstructive calcific chronic pancreatitis, Gastrointest. Endosc. 74(6), 1294–1299.
[16] Ammann R. W., Hkovbiantz A., Largiader F., et al. (1984) Course and outcome of chronic panereatitis. Longitudinal study of a mixed medicalsurgical series of 245 patients, Gastroenterology 86(5 Pt 1), 820–828.
[17] Wu B., Yu S., Li X., et al. (2022) Application value of laparoscopic pancreatic duct lithotomy and pancreatic duct T drainage for chronic pancreatitis complicated with pancreatic duct stones, China J. Endosc. 28(6), 72–77.
[18] Kitano M., Gress T. M., Garg P. K., et al. (2020) International consensus guidelines on interventional endoscopy in chronic pancreatitis. Recommendations from the working group for the international consensus guidelines for chronic pancreatitis in collaboration with the international association of pancreatology, the american pancreatic association, the japan pancreas society, and european pancreatic club, Pancreatology 20(6), 1045–1055.
[19] Geusens D., van Malenstein H. (2021) The role of extracorporeal shock wave lithotripsy in the treatment of chronic pancreatitis, Acta Gastro.-Ent. Belg. 84(4), 620–626.
[20] Gerges C., Pullmann D., Schneider M., et al. (2019) Pancreatoscopy in endoscopic treatment of pancreatic duct stones: a systematic review, Minerva. Chir. 74(4), 334–347.
[21] van Huijgevoort N. C. m., Veld J. V., Fockens P., et al. (2020) Success of extracorporeal shock wave lithotripsy and ERCP in symptomatic pancreatic duct stones: A systematic review and meta-analysis, Endosc. Int. Open 8(8), E1070–E1085.
[22] Yamamoto S., Inui K., Katano Y., et al. (2022) Pancreatic stones: clinical outcomes with nonsurgical treatment in a Japanese single-center study, Pancreas 51(2), 205–211.
[23] Sauerbruch T., Holl J., Sackmann M., et al. (1987) Disintegration of pancreation duct stone with extracorporeal shock waves in a patient with chronic pancreatitis, Endoscopy 19, 207–208.
[24] Dumonceau J. M., Delhaye M., Tringali A., et a1. (2019) Endoscopic treatment of chronic pancreatitis: European Society of Gastrointestinal Endoscopy (ESGE)Guideline—Updated August 2018, Endoscopy 51(2): 179–193. doi: 10.1055/a0822-0832.
[25] Wang D., Hu L. (2019) Extracorporeal Shock Wave Lithotripsy (ESWL) Combined with Endoscopic Retrograde Cholangiopancreatography (ERCP) for Pancreatic Duct Stones, J. Hepatobiliary Surg. 27(2), 150–152. doi: 10.3969/j.issn.1006-4761. 2019.02.022.
[26] Wang D., Ji J. T., Xin L., et a1. (2018) Extracorporeal shock wave lithotripsy for chronic pancreatitis patients with stones after pancreatic surgery, Pancreas 47(5), 609–616. doi: 10.1097/MPA.0000000000001042.
[27] Tandan M., Nageshwar Reddy D., Talukdar R., Vinod K., Kiran S. V. V. S., Santosh D., Gupta R., Ramchandani M., Lakhtakia S., Rakesh K., Manohar Reddy P., Basha J., Nabi Z., Jagtap N., Rao G. V. (2019 Oct) ESWL for large pancreatic calculi: report of over 5000 patients, Pancreatology 19(7), 916–921.
[28] Sharzehi K. (2019 Dec 4) Management of pancreatic duct stones, Curr. Gastroenterol. Rep. 21(11), 63.
[29] Choi E.K., Lehman G.A. (2012) Update on endoscopic management of main pancreatic duct stones in chronic calcific pancreatitis, Korean J. Intern. Med. 27, 20–29. doi: 10.3904/kjim.2012.27.1.20.
[30] Han S., Shah R. J., Brauer B. C. et al. (2019) A Comparison of endoscopic retrograde pancreatography with or without pancreatoscopy for removal of pancreatic duct stones, Pancreas 48, 690–697. doi: 10.1097/mpa.0000000000001317.
[31] Alatawi A., Leblanc S., Vienne A., et al. (2013) Pancreatoscopy-guided intracorporeal laser lithotripsy for difficult pancreatic duct stones: a case series with prospective follow-up (with video), Gastrointest. Endosc. 78, 179–183. doi: 10.1016/j.gie.2013.02.015.
[32] Attwell A. R., Patel S., Kahaleh M., et al. (2015) ERCP with per-oral pancreatoscopy-guided laser lithotripsy for calcific chronic pancreatitis: a multicenter U.S. experience, Gastrointest. Endosc. 82, 311-318. doi: 10.1016/j.gie.2015.01.020.
[33] Ogura T., Okuda A., Imanishi M., et al. (2019) Electrohydraulic lithotripsy for pancreatic duct stones under digital single-operator pancreatoscopy (with Video), Dig. Dis. Sci. 64, 1377–1382. doi: 10.1007/s10620-018-5374-z.
[34] Brewer Gutierrez O. I., Raijman I., Shah R. J., et al. (2019) Safety and efficacy of digital single-operator pancreatoscopy for obstructing pancreatic ductal stones, Endosc. Int. Open 7, E896–E903. doi: 10.1055/a-0889-7743.
[35] Gerges C., Albers D., Schmitz L., et al. (2023) Digital single-operator pancreatoscopy for the treatment of symptomatic pancreatic duct stones: a prospective multicenter cohort trial, Endoscopy 55, 150–157. doi: 10.1055/a-870-3403.
[36] Bekkali N. L., Murray S., Johnson G. J., et al. (2017) Pancreatoscopy-directed electrohydraulic lithotripsy for pancreatic ductal stones in painful chronic pancreatitis using SpyGlass, Pancreas 46, 528–530. doi: 10.1097/mpa.0000000000000790.
[37] vander Wiel S. E., Stassen P. M. C., Poley J. W., et al. (2022) Pancreatoscopy-guided electrohydraulic lithotripsy for the treatment of obstructive pancreatic duct stones: a prospective consecutive case series, Gastrointest. Endosc. 95, 905–914.e902. doi: 10.1016/j.gie.2021.11.047.
[38] Gerges C., Pullmann D., Bahin F., et al. (2019) SpyGlass DS-guided lithotripsy for pancreatic duct stones in symptomatic treatment-refractory chronic calcifying pancreatitis, Endosc. Int. Open 7, E99–E103. doi: 10.1055/a-0808-4499.
[39] Bassi C., Dervenis C., Butturini G., et al. (2005) Postoperative pancreatic fistula: an international study group (ISGPF) definition, Surgery 138, 8–13.
[40] Le Moine O., Matos C., Closset J., et al. (2004) Endoscopic management of pancreatic fistula after pancreatic and other abdominal surgery, Best Pract. Res. Clin. Gastroenterol. 18, 957–975.
[41] Bassi C., Marchegiani G., Dervenis C., et al. (2017) The 2016 update of the International Study Group (ISGPS) definition and grading of postoperative pancreatic fistula: 11 years after, Surgery 161, 584–591.
[42] Zhai Y., Chai N., Linghu E., et al. (2019) Postoperative pancreatic fistula: a new indication for endoscopic intervention, Chin. J. Gastrointest. Endosc. (Electron. Ed.) 6, 77–81.
[43] Nahm C. B., Connor S. J., Samra J. S., et al. (2018) Postoperative pancreatic fistula: a review of traditional and emerging concepts, Clin. Exp. Gastroenterol. 11, 105–118.
[44] Xu X., Sun M. (2008) Endoscopic nasopancreatic drainage for pancreatic fistula: three case reports, Chin. J. Pancreatol. 411–412.
[45] Halttunen J., Weckman L., Kemppainen E., et al. (2005) The endoscopic management of pancreatic fistulas, Surg. Endosc. 19, 559–562.
[46] Cicek B., Parlak E., Oguz D., et al. (2006) Endoscopic treatment of pancreatic fistulas, Surg. Endosc. 20, 1706–1712.
[47] Goasguen N., Bourrier A., Ponsot P., et al. (2009) Endoscopic management of pancreatic fistula after distal pancreatectomy and enucleation, Am. J. Surg. 197, 715–720.
[48] Telford J. J., Farrell J. J., Saltzman J. R., et al. (2002) Pancreatic stent placement for duct disruption, Gastrointest. Endosc. 56, 18–24.
[49] Jurgensen C., Distler M., Arlt A., et al. (2019) EUS-guided drainage in the management of postoperative pancreatic leaks and fistulas (with video), Gastrointest. Endosc. 89, 311–319 e311.
[50] Cooper S. T., Malick J., McGrath K., et al. (2010) EUS-guided rendezvous for the treatment of pancreaticopleural fistula in a patient with chronic pancreatitis and pancreas pseudodivisum, Gastrointest. Endosc. 71, 652–654.
[51] Minami K., Iwasaki E., Itoi T., et al. (2017) Successful EUS-guided nondilated pancreatic duct stent placement in patient with refractory pancreatic fistula after pancreaticoduodenectomy. VideoGIE 2, 315–316.
[52] Irani S., Gluck M., Ross A., et al. (2012) Resolving external pancreatic fistulas in patients with disconnected pancreatic duct syndrome: using rendezvous techniques to avoid surgery (with video), Gastrointest. Endosc. 76, 586–593. e581–e583.
[53] Futagawa Y., Imazu H., Mori N., et al. (2017) The effectiveness and feasibility of endoscopic ultrasound-guided transgastric drainage of postoperative fluid collections early after pancreatic surgery, Surg. Laparosc., Endosc. Percutan. Tech. 27, 267–272.
[54] Onodera M., Kawakami H., Kuwatani M., et al. (2012) Endoscopic ultrasound-guided transmural drainage for pancreatic fistula or pancreatic duct dilation after pancreatic surgery, Surg. Endosc. 26, 1710–1717.
[55] Haseeb A., Abu Dayyeh B. K., Levy M. J., et al. (2016) Endoscopic ultrasound-guided treatment of pancreaticocutaneous fistulas, ACG Case Rep. J. 3, e105.
[56] Banks P. A., Bollen T. L., Dervenis C., et al. (2013) Classification of acute pancreatitis--2012: revision of the Atlanta classification and definitions by international consensus. Gut. 62, 102–111.
[57] Sharaiha R. Z., Tyberg A., Khashab M. A., et al. (2016) Endoscopic therapy with lumen-apposing metal stents is safe and effective for patients with pancreatic walled-off necrosis, Clin. Gastroenterol. Hepatol. 14(12), 1797–1803.
[58] Siddiqui A. A., Kowalski T. E., Loren D. E., et al. (2017) Fully covered self-expanding metal stents versus lumen-apposing fully covered self-expanding metal stent versus plastic stents for endoscopic drainage of pancreatic walled-off necrosis: clinical outcomes and success, Gastrointest. Endosc. 85(4), 758–765.
[59] Arvanitakis M., Dumonceau J. M., Albert J. et al. (2018) Endoscopic management of acute necrotizing pancreatitis: European Society of Gastrointestinal Endoscopy (ESGE) evidence-based multidisciplinary guidelines, Endoscopy 50, 524–546.
[60] Baron T. H., DiMaio C. J., Wang A. Y., et al. (2020) American gastroenterological association clinical practice update: management of pancreatic necrosis, Gastroenterology 158, 67–75.e1.
[61] Hocke M., Burmeister S., Braden B., et al. (2022) Controversies in EUS-guided treatment of walled-off necrosis, Endosc. Ultrasound 11, 442–457.
Chapter 13 Super Minimally Invasive Surgery of Colorectal Diseases
13.1 Resection of Early Colorectal Cancer by Super Minimally Invasive Surgery
13.1.1 Overview
Colorectal cancer (CRC) originates from the mucosal epithelial lining of the colon and rectum and is one of the most common malignant tumors of the digestive tract. Studies have found that colorectal cancer accounts for approximately one-tenth of all newly diagnosed cancers and cancer-related deaths globally. It ranks second among the most common cancers in women and third in men. As residents in China experience changes in lifestyle, colorectal cancer has emerged as the most common cancer within the digestive system in the country. Early detection, prompt diagnosis, and proactive treatment are essential for reducing both the incidence and mortality associated with colorectal cancer. Early colorectal cancer (ECC) is defined as any size of colorectal epithelial tumor with infiltration limited to the mucosa and submucosa, regardless of lymph node metastasis. When tumor infiltration is confined to the mucosa, it is classified as mucosal cancer (M-stage cancer). Infiltration into the submucosa without invasion of the muscularis propria is classified as submucosal cancer (SM-stage cancer). Further refinement includes the following stages: tumors confined to the mucosal epithelium are classified as M1-stage cancer; tumors invading the basement membrane and into the mucosal propria layer are classified as M2-stage cancer; tumors infiltrating into the muscularis mucosa are classified as M3-stage cancer. If the tumor invades the upper, middle, or lower third of the submucosa, they are classified as SM1, SM2, and SM3-stage cancers, respectively. Early colorectal cancers with invasion of no more than the upper third of the submucosa (1000 μm) can generally be treated endoscopically.
(I) Indications and contraindications
1. Indications
Endoscopic resection for early colorectal cancer is primarily indicated for lesions with a low risk of lymph node metastasis, where the size and location of the tumor have been evaluated and can be fully excised endoscopically. The indications for endoscopic treatment in the current Chinese consensus guidelines mainly refer to those in the Japanese guidelines.
According to the Japanese Society for Cancer of the Colon and Rectum (2022 edition), the indications for endoscopic resection of early colorectal cancer include: ① mucosal cancer or mildly invasive submucosal cancer; ② no size restriction for the lesion; ③ any macroscopic type.
The latest Chinese consensus in China recommends that for lesions with a maximum diameter >20 mm, which are difficult to completely resect in one session with endoscopic mucosal resection (EMR), lesions with negative lift sign, and EMR residuals >10 mm or lesions that are difficult to treat with EMR during follow-up, endoscopic submucosal dissection (ESD) should be considered.
2. Contraindications: ① Preoperative assessment reveals deep submucosal invasion, involvement of the muscularis propria, lymph node metastasis, or distant metastasis; ② patients unable to cooperate with the procedure; ③ severe cardiopulmonary diseases that cannot tolerate endoscopic treatment; ④ patients with hematologic diseases, coagulopathy, or those on anticoagulant therapy, where coagulation function has not been corrected; ⑤ unstable vital signs; ⑥ lesions located in areas with high technical difficulty, such as around the intestinal lumen or those involving multiple folds, with a high risk of perforation; ⑦ presence of other organ malignancies with a poor prognosis; ⑧ acute active intestinal inflammation, such as active ulcerative colitis.
(II) Preoperative preparation for endoscopic resection of early colorectal cancer
(1) The patient’s overall condition should be assessed and the absence of contraindications for anesthesia and endoscopic treatment re-confirmed. A detailed explanation must be provided to the patient and the family regarding the endoscopic resection procedure, the expected outcomes, the potential complications, etc., and written informed consent obtained.
(2) All patients should undergo electrocardiographic monitoring. Intravenous propofol anesthesia may be administered based on the patient’s condition.
(3) Adequate bowel preparation should be performed preoperatively. For patients with insufficient bowel preparation, repeated bowel irrigation should be carried out before the procedure to remove residual feces and fluids.
(III) Postoperative management
(1) Handling of postoperative specimens: after the tissue is excised, the endoscopist should flatten the entire mucosal specimen, fix it with a fine stainless steel needle on a flat surface with the mucosal side facing up, and distinguish the oral and anal ends of the resection. For detailed procedures and specifics, refer to the latest Expert Consensus on Routine Histopathological Processing of Specimens from Endoscopic Submucosal Dissection/Endoscopic Mucosal Resection.
(2) Postoperative day 1 management: no food intake is allowed on the first postoperative day. Vital signs including blood pressure, pulse, respiration, and body temperature must be closely monitored. Signs of abdominal pain or distension must be carefully observed, and the liver dullness boundary checked. Necessary laboratory tests, including complete blood count and C-reactive protein, should be performed. If no abnormalities are detected in clinical symptoms and related tests, a liquid diet may be introduced on the second postoperative day for two to three days, followed by a low-residue semi-liquid diet. Most patients are discharged five to seven days postoperatively.
(3) Antibiotic use: routine antibiotic use is not recommended after surgery. For patients with extensive resection, prolonged procedure duration, poor bowel preparation, or high risk of perforation, prophylactic antibiotics, such as second- or third-generation cephalosporins combined with nitroimidazoles, may be considered. The duration of antibiotic therapy should not exceed 72 h. In patients at higher risk of bleeding, hemostatic agents may be administered as appropriate.
(4) Management of complications: perforation during the procedure should first be managed with early endoscopic clipping. For delayed postoperative bleeding, endoscopic hemostasis is the preferred treatment. A mucosal defect greater than 90% of the circumferential area after colorectal ESD is an independent risk factor for postoperative stricture. Endoscopic balloon dilation can effectively treat most cases of stricture.
(IV) Technical methods for super minimally invasive resection in early colorectal cancer
Early colorectal cancer can be cured through super minimally invasive surgery (SMIS), primarily utilizing peranal natural orifice access and per-tunnel access for endoscopic treatment. Both super minimally invasive non-full-thickness resection and full-thickness resection techniques can be performed. The technical methods include peroral super minimally invasive resection for early colorectal cancer, which is the technique of locally and completely resecting early colorectal cancer under peranal endoscopy. Another method is per-tunnel super minimally invasive resection for early colorectal cancer, which is the technique of locally and completely resecting early colorectal cancer under tunnel endoscopy.
The current technical methods applied in super minimally invasive surgery for early colorectal cancer are listed along with their previous nomenclature in table 13.1.
TAB. 13.1 — Technical methods for super minimally invasive surgery in early colorectal cancer and their previous nomenclature.
| No. | Super Minimally Invasive Surgery (SMIS) | Technical Method | Previous Nomenclature |
| 1 | Early colorectal cancer peroral super minimally invasive resection | Peranal endoscopic local resection | Endoscopic submucosal dissection (ESD) traction-assisted endoscopic submucosal dissection |
| Cap-assisted endoscopic mucosal resection endoscopic mucosal resection (EMR) | |||
| 2 | Peroral super minimally invasive resection for early colorectal cancer | Super minimally invasive full-thickness resection | Endoscopic submucosal full-thickness resection |
| 3 | Per-tunnel super minimally invasive resection for early colorectal cancer | Super minimally invasive non-full-thickness resection | Traction-assisted endoscopic full-thickness resection |
| Endoscopic tunnel technique for submucosal dissection | |||
| Traction-assisted endoscopic submucosal dissection |
13.1.2 Peroral Super Minimally Invasive Non-Full-Thickness Resection for Early Colorectal Cancer
I. Endoscopic submucosal dissection (ESD)
Peroral super minimally invasive non-full-thickness resection for early colorectal cancer can be achieved through endoscopic submucosal dissection (ESD). ESD is a new technique developed based on endoscopic mucosal resection (EMR). This method is suitable for lesions of varying locations, sizes, and depths of invasion. The specific procedure involves injecting a solution into the submucosa, followed by the use of specialized electrosurgical instruments, such as the IT knife, DualKnife, or Hook knife, to gradually dissect the tissue between the mucosal layer and the muscularis propria, thereby achieving complete removal of the diseased mucosa and submucosa.
(I) Surgical procedure and techniques (figure 13.1)
1. Marking. Argon plasma coagulation (APC) or an electrosurgical knife is used to perform coagulation marking 5–10 mm from the edge of the lesion. (Note: since the margins of most colorectal tumors are clearly visible before and after submucosal injection, this marking step can often be omitted.)
2. Submucosal injection. Multiple submucosal injections are performed at the outer edge of the lesion marking. The injection solution is a mixture of saline solution, adrenaline, indigo carmine, or methylene blue. Injections can be repeated until the lesion is sufficiently lifted.
3. Incision of the surrounding mucosa. The mucosa is incised using a single-use mucosal incision knife (DualKnife) along the outer edge of the marking. The incision generally starts from the anal side of the lesion.
4. Submucosal dissection. The submucosal layer beneath the lesion is dissected using an IT knife or a DualKnife, or a combination of both.
5. Wound management. After complete resection of the lesion, bleeding points on the surface of the wound are healed using high-frequency coagulation forceps, argon gas, or metal titanium clips.

FIG. 13.1 — Operational steps of peroral super minimally invasive non-full-thickness resection for early colorectal cancer. A. Observation of an LST lesion in the transverse colon. B. Identification of the lesion’s boundaries after staining. C. Dissection of the lesion after submucosal injection. D. Inspection of the wound after complete resection of the lesion.
(II) Hot-spot issues and research progress in the consensus
1. Comparison of treatment outcomes between endoscopic submucosal dissection (ESD) and endoscopic mucosal resection (EMR). Endoscopic submucosal dissection (ESD) and endoscopic mucosal resection (EMR) are both commonly used endoscopic treatments for early colorectal cancer. Numerous studies have compared the treatment outcomes of the two methods. According to the Expert Consensus on the Management Strategy for Colorectal Cancer Precursors and Pre-cancerous States in China, ESD is generally recommended as the treatment of choice for early colorectal cancer (CRC) and/or high-grade intraepithelial neoplasia, as it is considered to have superior overall treatment outcomes compared to EMR. In addition, the Chinese Consensus on the Endoscopic Diagnosis and Treatment of Colorectal Cancer and Precancerous Lesions (2023, Guangzhou) recommends the use of EMR for lesions that are suitable for complete resection in one session, such as flat-type lesions (types IIa, IIb, IIc) and some type-Is lesions. EMR is recognized for its high safety and efficacy, particularly for flat lesions or sessile lesions that cannot be removed directly with a snare. To reduce the incomplete resection rate and post-operative recurrence risk associated with piecemeal resection, whole-block resection EMR is recommended for managing these lesions. According to previous chinese guidelines, the maximum lesion diameter for whole-block resection EMR should be 20 mm. For lesions larger than 20 mm in diameter, those that are difficult to resect completely in one session with EMR, those with negative lifting signs, and lesions with EMR residuals or recurrence that are difficult to treat with subsequent EMR, ESD is recommended. The 2019 JGES Guidelines for Colorectal EMR/ESD suggest that en bloc resection is ideal for the endoscopic treatment of early colorectal cancer. However, for certain adenomas and “adenomas with focal carcinoma,” piecemeal EMR is permissible in appropriate cases, though magnifying endoscopy should be used for careful evaluation before treatment, ensuring that cancerous areas are not resected piecemeal. Magnifying endoscopy, which allows the margins of the lesion and the base of ulcers after resection to be observed, can help to reduce the local residual/recurrent rate. A follow-up colonoscopy should be performed approximately six months after the treatment. The occurrence of T1 (SM) cancer increases with tumor size. Piecemeal resection complicates pathological reconstruction of the tumor, making histological evaluation more difficult and leading to a higher local residual/recurrent rate. For lesions larger than half the circumference of the colon, piecemeal EMR should be avoided, and ESD should be performed by skilled endoscopists. Surgical treatment should only be considered as an alternative when ESD is not feasible.
2. Endoscopic submucosal dissection (ESD) for the treatment of larger colorectal lesions. The Chinese Consensus on the Endoscopic Diagnosis and Treatment of Colorectal Cancer and Precancerous Lesions (2023, Guangzhou) recommends the use of endoscopic submucosal dissection (ESD) for lesions with a long axis greater than 20 mm, lesions that are difficult to completely resect with EMR in one session, lesions with negative lifting signs, and lesions with residual or recurrent disease after EMR treatment for which further EMR is difficult. However, the evaluation of the safety and efficacy of ESD for larger colorectal lesions still requires further research and validation. A Japanese research team enrolled 1740 patients with colorectal epithelial tumors larger than 20 mm who received ESD treatment. By analyzing long-term outcomes such as five-year overall survival, disease-specific survival, and bowel preservation rates, they concluded that ESD provides good long-term prognosis and could be considered a standard treatment for colorectal epithelial tumors. For colorectal tumors with a long axis greater than 50 mm, a study analyzed 177 cases of large colorectal tumors treated with ESD (the median tumor size was 61 mm). The study found a 96.2% en bloc resection rate and an 80.5% cure rate. The ESD resection speed using the IT-nano knife was significantly faster than when only using the DualKnife. However, these findings require further clinical studies for validation.
II. Traction-assisted endoscopic submucosal dissection (ESD)
Traction-assisted endoscopic submucosal dissection (ESD) is an endoscopic technique that improves the surgical field by determining the lesion’s boundaries, performing marking, submucosal injection, and adequately lifting the lesion, while applying external traction forces to the lesion. This method facilitates better intraoperative visibility. During the procedure, the electrosurgical knife gradually dissects the tissue between the lesion’s mucosa and the muscularis propria, achieving complete removal of the diseased mucosa and submucosa, followed by wound management.
A clear surgical field is essential for ensuring the safety of endoscopic procedures. When performing ESD to treat colorectal lesions, the operator must carefully evaluate the submucosal layer, as blind operations increase the risk of perforation and bleeding. Traditional ESD methods typically involve using a transparent cap to stabilize the submucosal layer during dissection. To further expose the surgical field, gravity is often employed to allow the dissected tissue to prolapse in the direction of dissection. However, in some difficult locations, larger lesions, or cases with scar adhesions, these methods may be ineffective. To address these challenges, various endoscopic traction methods based on different forces, such as gravity, magnetic force, mechanical tension, and elasticity, have been developed. These include techniques such as double-endoscope traction, percutaneous traction, weight-assisted traction, external forceps traction, dental floss traction, magnetic traction, and robotic-assisted methods. This chapter will introduce effective traction-assisted techniques commonly used in colorectal ESD.
(I) Surgical technique and skills
1. Extraluminal dental floss traction. Dental floss traction has significant advantages in shortening ESD procedure time, reducing complications such as bleeding and perforation, and improving en bloc and complete resection rates. Studies have shown that extraluminal dental floss traction can provide adequate exposure of the submucosal layer through lateral or same-direction tension applied opposite to the direction of dissection. This creates optimal conditions for lesion dissection and allows traction in multiple directions, thereby providing proper direction and sufficient pulling force for a clearer surgical field. Additionally, the dental floss used in these studies can be inserted through the biopsy channel, making the procedure simple to perform. Moreover, dental floss is inexpensive and does not increase the patient’s financial burden.
However, a limitation of the commonly used extraluminal dental floss traction in ESD is the need for frequent retraction and insertion of the endoscope. This technique is more convenient for lesions in the left colon, but for right-sided lesions, the repeated insertion and withdrawal of the endoscope can lead to bowel spasms, making re-insertion more difficult, extending procedure time, and increasing the risk of surgical complications.
Specific Operating Steps (figure 13.2):
1) Marking and submucosal injection: using an argon plasma coagulation (APC) device, electrocautery marking is performed at the anal side edge of the raised lesion. After marking, submucosal injections with a mixture of saline solution, indigo carmine, and adrenaline are made at multiple points on the outer side of the marking point. The electrosurgical knife is then used to cut until part of the submucosal tissue of the lesion is separated. The endoscope is then reversed, and the anal side of the lesion is clearly visible.
2) Submucosal injection and floss placement: the procedure is repeated at the raised lesion edge, with submucosal injection and electrocautery to separate the submucosal layer up to about one-quarter of the lesion’s circumference. The endoscope is withdrawn, and preparation is made to insert the dental floss. First, a titanium clip is inserted into the endoscope. The opening and closing functions of the titanium clip are checked. After opening the clip, one side of the clip is used to secure the dental floss, with the shorter end of the floss cut off to avoid obstructing the view. The longer end of the floss is left attached to the outside of the endoscope, and it is pulled by the operator or assistant.
3) Floss traction and lesion dissection: after reinserting the endoscope, the procedure continues as described. The endoscope is reversed to observe the anal side of the lesion, and a titanium clip is placed at the cutting edge of the lesion to secure the dental floss to the lesion. The floss is then pulled, which results in the anal edge of the lesion being elevated, exposing the submucosal layer. The dissection continues until the lesion is completely removed.
4) Post-resection: after the lesion is fully resected, the surgical field is examined to ensure the wound is intact and there is no bleeding. The specimen is then dragged out of the body using the dental floss.

FIG. 13.2 — Operating steps of tissue clip-dental floss traction-assisted dissection in per-rectal super minimally invasive resection for early rectal cancer. A. Marked early rectal cancer: the lesion is marked after initial inspection. B. Circumferential mucosal incision: the lesion is circumferentially incised around its edges. C. Dental floss-tissue clip fixed on the dissected mucosal side: the dental floss and tissue clip are attached to the dissected mucosal side. D. Dental floss-tissue clip lifting the lesion: the floss and the tissue clip are used to lift the lesion. E. Traction separates the mucosal and muscular layers: the traction separates the mucosal and muscular layers clearly, allowing continued submucosal dissection. F. Postoperative wound: the wound is checked after the lesion has been completely removed.
2. Rubber-band and clip traction. Rubber band and clip facilitate endoscopic submucosal dissection. (RAC–ESD) is a technique in which the mucosal layer is lifted during the procedure to fully expose the submucosal layer. This method eliminates the need for continuous endoscope pressure on the dissected surface, thus preventing the exposure of the surgical field from being insufficient, which could lead to deviations in the range or depth of the resection. It ensures that the horizontal and vertical margins of the lesion are sufficiently distant from the tumor tissue, avoiding interference with post-surgical pathological evaluation. The use of direct visualization throughout the procedure reduces complications such as intraoperative bleeding and perforation, which may occur due to poor visibility or blind cutting. Additionally, RAC–ESD maintains tension on the lesion tissue during the procedure, reducing the difficulty of the operation and shortening the overall surgical time. Studies have shown that the RAC–ESD group achieves a significantly larger area of dissection per unit time compared to the conventional ESD group. For larger lesions, if traction from a single rubber band cannot maintain submucosal visibility throughout the complete dissection of the lesion, another tissue clip can be applied with a second rubber band to traction the opposite side of the remaining lesion, continuing until the surgery is completed.
Specific operating procedure (figure 13.3):
Marking, submucosal injection, and circumferential resection: follow the previous steps for marking, injection, and circumferential mucosal incision;
Installation of a rubber band-tissue clip traction device: one end of the rubber band is fixed to the partially dissected mucosal layer, while the other end is attached to the opposite side of the lesion using a tissue clip to secure it to the distal mucosal surface. This action helps separate the submucosal layer from the muscular layer. Based on the size of the lesion, one or more traction devices can be used.
After traction, submucosal injection is continued. Once a clear view of the submucosal layer is obtained, the lesion is dissected further until it is completely separated from the wound;
The wound is closed after careful hemostasis with a hemostatic forceps.

FIG. 13.3 — Operating steps of tissue clip-dental floss traction-assisted dissection in super minimally invasive resection of early cancer of the transverse colon via peroral approach. A. Early cancer of the transverse colon. B. Circumferential incision of the lesion. C. One set of rubber band and tissue clip used for unidirectional traction. D. Two sets of rubber band and tissue clip used for multidirectional traction. E. Postoperative wound. F. Tissue clip used to close the wound.
3. Magnetic anchor traction. Magnetic anchoring technique (MAT) has traditionally been applied in surgical procedures, where an external anchoring magnet is used to pull the internal target magnet and the instruments it carries to the desired position, thereby enhancing the minimally invasive and precise nature of the surgery. Since 2004, studies have explored the use of MAT to assist in endoscopic submucosal dissection (ESD), with some promising results. However, the large size of the external devices has limited the further clinical application of MAT equipment. In 2018, an improved version of MAT was utilized in gastric ESD, with research reporting successful en bloc resection of lesions in 50 patients.
In colorectal ESD, particularly for lesions in the right colon, conventional traction methods require repeated advancement and withdrawal of the endoscope. This increases intestinal stimulation, leading to recurrent peristalsis, which complicates submucosal injection and dissection. Additionally, repeated traction can cause damage to the mucosa at the lesion site. By using MAT, the procedure becomes simpler. It not only eliminates the need for repeated advancement and withdrawal but also allows for thorough exposure of the submucosal layer, providing multi-angle traction. This technique significantly reduces complications such as bleeding and perforation while shortening surgery time. Furthermore, MAT does not increase the patient’s financial burden and can be easily promoted in grassroots hospitals.
Specific Operating Procedure (figure 13.4)
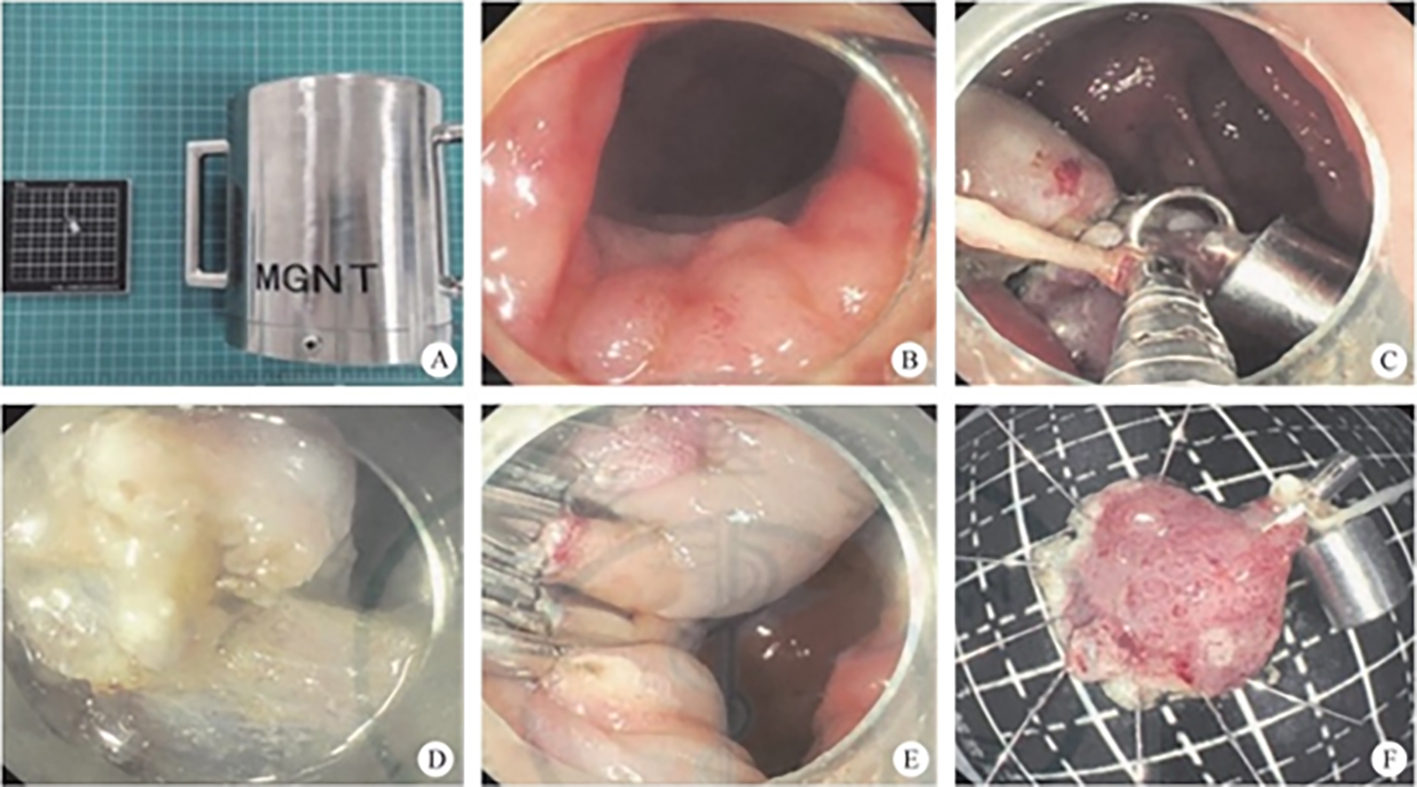
Delivery of the target magnet to the body: the titanium clip is delivered through the endoscopic biopsy channel, and its front end is used to grasp the tail structure of the target magnet, which is pre-attached with dental floss; the target magnet is then released at the lesion site, and the titanium clip is withdrawn.
Lesion marking: after determining the boundaries of the lesion, a submucosal injection is performed.
Circumferential incision of the mucosa: a DualKnife is used to perform a circumferential incision around the lesion.
Fixation of the anchoring magnet: the titanium clip is fixed at the oral-side edge of the lesion. The direction of the target magnet within the colon is controlled by adjusting the external anchoring magnet.
Lesion dissection: under multi-angle traction provided by the target magnet, the submucosal layer is thoroughly exposed. Submucosal injection and dissection are performed until the lesion is completely separated.
Hemostasis and wound closure: after adequate hemostasis, the titanium clip is used to close the wound. The lesion is completely removed and sent for pathological examination.

FIG. 13.4 — Operating steps of magnetic anchor traction in peroral super minimally invasive resection of early colorectal cancer. A. Image of the external anchoring magnet and the target magnet inside the colon (left: target magnet, right: anchoring magnet). B. Mixed-type LST near the cecum. C. The colon target magnet is fixed on the lesion mucosa with a harmonic clamp. D. The submucosal layer is fully exposed after traction by magnetic anchoring. E. A titanium clip is used to close the wound. F. Surgical specimen.
(II) Classic cases
Case 1: peroral super minimally invasive resection of a colorectal cancer precursor lesion
Medical history: the patient, a young man, was admitted with the chief complaint of “discovery of a colon polyp 18 days ago.” Colonoscopy revealed an irregularly elevated lesion in the sigmoid colon (approximately 30 cm from the anal verge), occupying about half of the lumen, with three biopsy samples taken. Pathological diagnosis: (Sigmoid) tubular adenoma of the colonic mucosa with moderate atypical hyperplasia in some glands. The patient had a history of gastroesophageal reflux disease (GERD).
Diagnosis: precursor lesion of colorectal cancer.
Following thorough preoperative assessment, nutritional optimization, and signed informed consent, the patient underwent peranal super minimally invasive resection of the laterally developing sigmoid colon lesion. Refer to video 13.1, Peranal super minimally invasive full-thickness resection of laterally developing lesion in the colon (See online resources).
Surgical procedure: the endoscope was advanced to 20 cm from the anal verge, where a laterally developing lesion was found in the sigmoid colon, with a congested, erythematous mucosal surface, occupying about one-third of the lumen. Mixtures of methylene blue/adrenaline/saline solution and hyaluronic acid/saline solution were injected around the lesion’s submucosa, making mucosal lifting possible. A DualKnife was used to circumferentially cut the surrounding mucosa. Mucosal dissection was performed with the electroknife (Nano knife, triangle knife) while submucosal fluid was injected. During the dissection, dental floss was used to assist with traction, and the lesion was completely removed. Hemostasis was achieved using hemostatic forceps, and several titanium clips were applied to close the wound. Biological protein glue was sprayed onto the wound, and the specimen was sent for pathological examination. The size of the ex vivo specimen was approximately 3.5 cm × 3.0 cm.
Case 2: Peranal super minimally invasive resection of early rectal cancer
Medical history: the patient, a middle-aged woman, was admitted with the chief complaint of “rectal bleeding for six months.” Six months before, the patient had begun experiencing rectal bleeding at the end of bowel movements, bright red in color, with small amounts of blood, without associated abdominal pain, bloating, or fever. Normal eating habits. The symptoms appeared intermittently. Colonoscopy suggested possible rectal cancer, and pathology confirmed (rectum) high-grade intraepithelial neoplasia. The patient had a history of appendicitis surgery and hemorrhoids.
Diagnosis: high-grade intraepithelial neoplasia of the rectum.
After comprehensive preoperative evaluation, nutritional optimization, and signing the surgical informed consent form, the patient underwent peranal super minimally invasive resection of early rectal cancer. Refer to video 13.2, Peranal super minimally invasive full-thickness resection of early rectal cancer (See online resources).
Surgical procedure: a flat elevated lesion was found 5–7 cm from the anal verge, with a red surface, nodular irregularities, central depression, fragile texture, and easy bleeding upon palpation. Methylene blue, adrenaline, and saline solution were injected submucosally around the lesion to facilitate mucosal lifting. A DualKnife was used to circumferentially cut the surrounding mucosa. The lesion was retracted toward the anal side using dental floss, while mixtures of methylene blue/adrenaline/saline solution, on the one hand, and of hyaluronic acid/methylene blue/adrenaline/saline solution, on the other hand, were injected sequentially. The lesion was dissected step-by-step using the DualKnife, IT2, and a triangle knife. During dissection, a network of blood vessels was observed, and hemostasis was achieved through electrocoagulation with hemostatic forceps. Finally, biological protein glue was sprayed onto the wound after electrocoagulation to secure hemostasis.
(III) Hot-spot issues and research progress in the consensus
1. Advantages and disadvantages of various traction techniques Traction techniques in endoscopic submucosal dissection (ESD) can be classified into two categories: those that require additional devices and those that do not, depending on the specific situation. Traction provided by distal cap attachments is generally considered in ESD due to its advantages in exposing the submucosal layer. Traction techniques that do not require additional devices primarily rely on tissue gravity, tunneling, bridging, and the tension provided by the oral pocket. In early March 2023, the European Society of Gastrointestinal Endoscopy (ESGE) published a technical review in the journal Endoscopy, detailing the advantages and disadvantages of various traction techniques.
Common device-assisted external traction techniques. ① Clips and threads: Easy to use and widely applied, but they can only provide traction in one direction. ② Clips and snare: capable of both traction and pushing, widely used; however, they complicate the endoscopic view and their effective range can interfere with each other. ③ External forceps: can both pull and push, widely applied, but difficult to insert and prone to mucosal damage; their effect range can also interfere with that of other tools. ④ Double-lumen endoscope: allows for dynamic and real-time triangulation; however, it suffers from light artifact issues from two light sources, requiring two endoscope platforms and operators.
Common device-assisted internal traction techniques. ① Double-clip traction: widely used, adjustable during the procedure, with traction force modifiable via insufflation; however, it can damage the specimen.② Internal traction lines: easy to use and place, with adjustable clips, but they are more cumbersome in the ESD field and limited for larger lesions.③ Magnet-assisted traction: adjustable during the procedure, allowing for traction force modulation through insufflation, but it carries a risk of damage to the specimen.
2. New auxiliary traction techniques
(1) External magnetic traction-assisted ESD (MAG–ESD):
A research team has developed the external magnetic traction-assisted ESD technique for endoscopic treatment of tumors at different sites in the gastrointestinal tract. The procedure involves binding a magnetic ring with dental floss and placing it on the side of the colonoscope lens. The colonoscope with the magnetic ring is then inserted into the body, and a metal clip is used to clamp the mucosa at the lesion’s apex. A cylindrical permanent magnet is placed on the abdominal wall, and its position is adjusted to provide effective traction to the lesion. This traction causes the mucosa to elevate, thereby clearly exposing the submucosal layer. The success rate of traction, complete lesion resection rate, traction time, operation time, and the incidence of complications such as bleeding and perforation were analyzed and evaluated. High traction success rates, high complete resection rates, low complication rates, and shorter operation times all confirm the feasibility of the MAG–ESD traction method.
(2) New pulling clip-assisted traction:
A new pulling clip with a built-in spring mechanism at the tip has been designed to assist in traction during the dissection process. The new pulling clip is inserted through the endoscopic biopsy channel. After reaching the desired location, the two arms of the clip open, releasing the spring component, which then grabs the mucosa at the lesion’s edge. The clip is released, and another metal clip is inserted through the endoscopic biopsy channel. The arms of the clip open, with one arm catching the spring component’s circular ring. The clip is then fixed at the appropriate position on the mucosa of the opposite bowel wall. Forty-two patients with colorectal lesions were included in the study, and the results showed that the new pulling clip-assisted colorectal ESD is safe and effective, significantly shortening the ESD procedure time and reducing the patient’s postoperative hospital stay.
Using reasonable and effective traction techniques during ESD can better expose the submucosal layer, ensure a clear operative field, and provide excellent assistance for endoscopic operations. Future developments may include more traction methods to facilitate smoother completion of ESD procedures.
13.1.3 Peranal Super Minimally Invasive Full-Thickness Resection of Colorectal Cancer
(I) Overview
With the development of reliable endoscopic closure techniques and tools, endoscopic full-thickness resection (EFTR) is becoming a treatment option for challenging lesions that are difficult to treat adequately and/or safely with endoscopic mucosal resection (EMR) or endoscopic submucosal dissection (ESD). To address the issue of complications such as pneumoperitoneum, which are common in traditional EFTR, our team has invented a modified EFTR technique, which involves performing full-thickness resection and simultaneously closing the defect until the lesion is completely removed. This modified technique has been used to excise large gastrointestinal stromal tumors (GISTs). Due to the complexity of the structure, this technique poses greater challenges in colon lesions. Professor Enqiang Ling’s team successfully applied this modified EFTR technique in the treatment of early-stage colorectal cancer. The peranal super minimally invasive full-thickness resection of colorectal cancer is performed using this EFTR technique.
(II) Surgical procedure and techniques
1. Diagnostic assessment. The lesion is first identified using white-light endoscopy. The surface mucus and fecal matter are repeatedly flushed using a water pump to clearly display the lesion. Narrow band imaging (NBI) is then used to observe the glandular openings. Next, small-probe endoscopic ultrasound (EUS) is performed to determine the lesion’s depth. Indigo carmine is sprayed onto the surface to clearly delineate the lesion’s boundaries, glandular structures, and morphology, and the findings are recorded.
2. Surgical procedure (figures 13.5and 13.6)
The lesion must be fully exposed, and an Argon Plasma Coagulation (APC) device is used to mark the lesion’s edges.
Submucosal injection is performed around the lesion to separate the mucosal layer from the submucosal layer.
The DualKnife is used to circumferentially cut the lesion’s edge.
The lesion is retracted towards the opposite side using an 8-shaped loop, and the lesion is gradually excised using a DualKnife, IT knife, and triangle knife.
Full-thickness resection is performed to excise the lesion, with tissue clips used to close the incision during the resection process. The entire procedure involves simultaneous full-thickness dissection and closure of the perforated wound.
After complete resection, tissue clips are again used to close the wound.
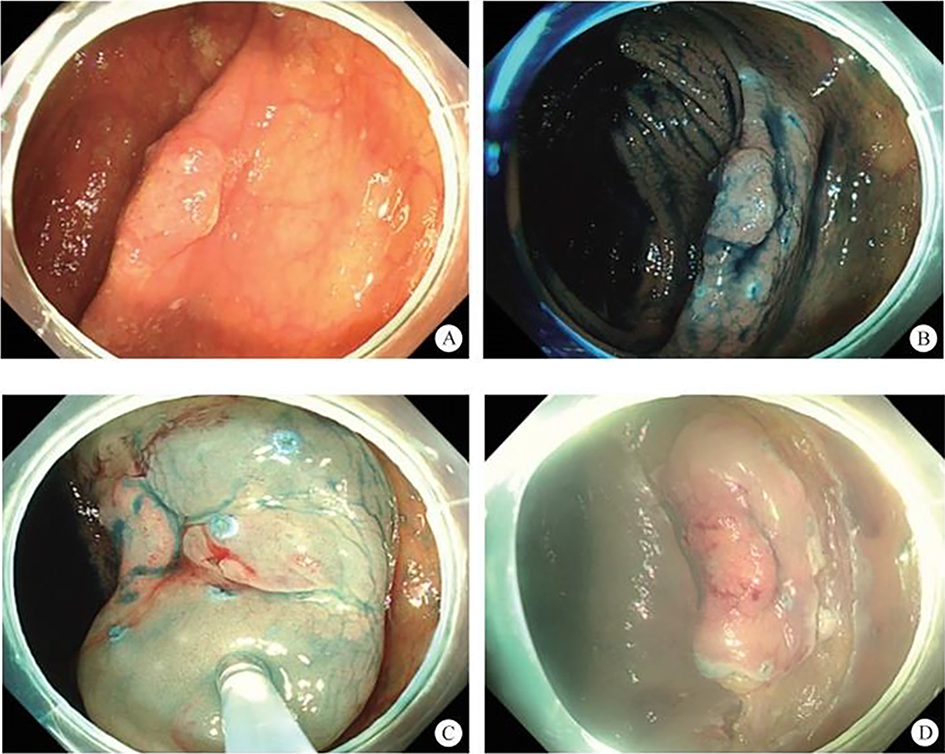
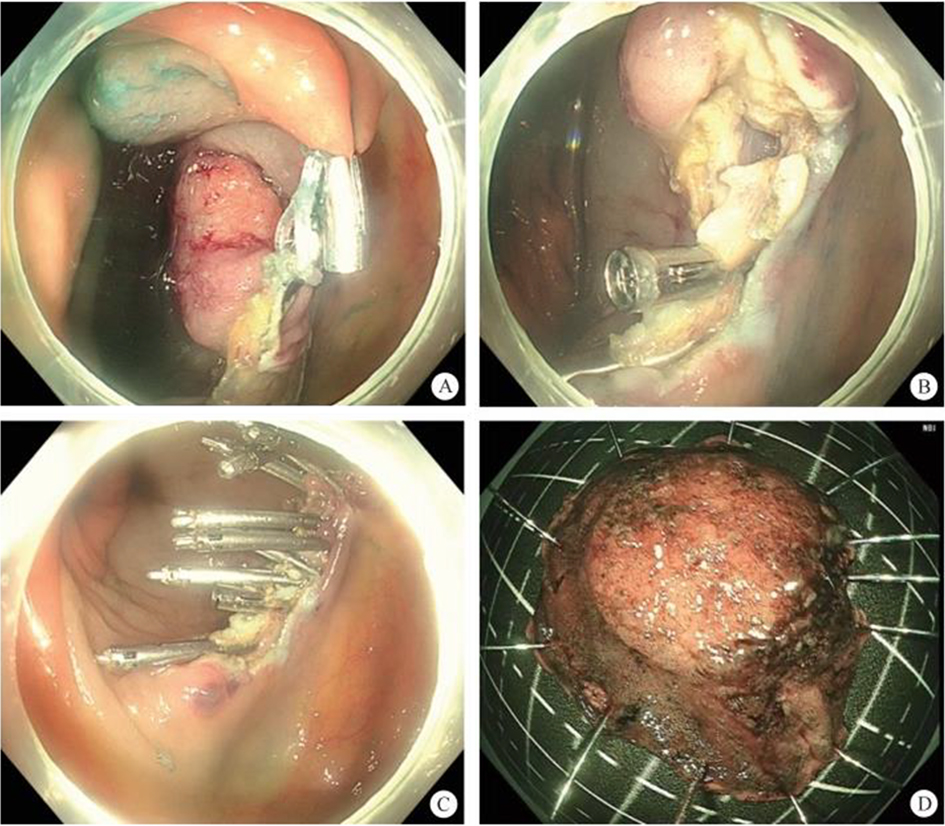

FIG. 13.5 — Steps of circumferential resection in peranal super minimally invasive full-thickness resection of ascending colon cancer. A. Early cancer features revealed under white-light endoscopy. B. The lesion’s edges are marked with APC, followed by spraying indigo carmine to further define the lesion’s boundaries. C. Submucosal injection is performed to separate the mucosal layer from the submucosal layer around the lesion. D. A circumferential submucosal incision is made to fully expose the lesion.

FIG. 13.6 — Steps of full-thickness resection by peranal super minimally invasive full-thickness resection of ascending colon cancer. A. The lesion is retracted towards the opposite side using an 8-shaped loop. B. A full-thickness resection of the lesion is performed, with simultaneous use of clips to close the muscular layer defect. C. The lesion is completely resected, and the submucosal layer defect is sutured. D. The lesion’s morphology is displayed using narrow-band imaging (NBI).
(III) Classic cases
Case 1: Early cancer at the appendix orifice treated by peranal super minimally invasive full-thickness resection
Medical history: the patient, an elderly woman, was admitted for “intermittent abdominal pain for four years, worsened for one year.” The patient experienced abdominal pain four years ago, without obvious triggers. The pain was paroxysmal and dull, relieved by positional changes (right lateral position), without acid reflux or heartburn. However, in the past year, the symptoms and their frequency had worsened. Two months ago, colonoscopy revealed a peduncle-like polypoid protrusion approximately 1 cm × 1 cm at the appendiceal orifice. Pathological examinations suggested: (appendiceal orifice) likely a villous adenoma, with moderate atypical hyperplasia observed in some glands. The patient had a history of hypertension, cholecystectomy, and benign paroxysmal positional vertigo. No abnormalities were found during physical examination upon admission.
Diagnosis: colonic adenoma (possible malignancy).
After thorough preoperative assessment, nutritional improvement, and signing of the informed consent form, the patient underwent cecal adenoma peranal super minimally invasive full-thickness resection (SMIS–EFTR). Refer to video 13.3, Peranal super minimally invasive full-thickness resection for early cecal cancer (See online resources).
Surgical procedure: at the appendiceal orifice, a peduncle-like polypoid protrusion measuring approximately 1 cm × 1 cm was visible and appeared soft upon extraction using foreign body forceps. Submucosal injection was performed, elevating the surrounding mucosa. The lesion’s edges were circumferentially incised with a DualKnife. The lesion was retracted using an 8-shaped loop, and the DualKnife, IT knife, and triangle knife were used to gradually dissect the lesion. Full-thickness resection was performed to excise the local lesion, with simultaneous resection and closure of the defect. Tissue clips were applied again to close the mucosal layer.
Case 2: Early cancer of the ascending colon treated by peranal super minimally invasive full-thickness resection
Medical history: the patient, a young woman, was admitted for “recurrent, shapeless stools for more than four months.” The patient began experiencing loose stools approximately four months before, which were yellow and watery in appearance, without any obvious triggers. Colonoscopy revealed a mass near the ileocecal valve in the ascending colon, approximately 2.0 cm × 1.5 cm in size, with a rough surface that easily bled upon contact, and hard to the touch upon biopsy. Pathology suggested: (ascending colon) adenocarcinoma (moderately differentiated). The patient had a history of cough variant asthma and a cesarean section.
Diagnosis: early-stage colorectal cancer.
After thorough preoperative assessment, nutritional improvement, and signing of the informed consent form, the patient underwent peranal super minimally invasive full-thickness resection (SMIS–EFTR) of early-stage ascending colon cancer. Refer to video 13.4, Peranal super minimally invasive full-thickness resection for early ascending colon cancer (See online resources).
Surgical procedure: a disc-shaped protrusion was observed on the opposite side of the ileocecal region in the ascending colon. The surface was covered with a coating, and the mucosa was rough. After irrigation, spontaneous bleeding was observed, and the lesion felt tough upon palpation. After marking the lesion’s edges with APC, indigo carmine was sprayed to further define the lesion’s extent. Submucosal injection was performed, elevating the surrounding mucosa. The lesion’s edges were incised with a DualKnife, and the lesion was retracted using an 8-shaped loop. The lesion was gradually dissected using the DualKnife, IT knife, and triangle knife. Full-thickness resection was performed for the local lesion, followed by the application of two doses of biological protein glue.
Case 3: Residual tumor after radiotherapy and chemotherapy for rectal cancer treated by super minimally invasive full-thickness resection
Medical history: the patient, a middle-aged woman, was admitted for “abdominal pain accompanied by tenesmus and blood in stool for over two years.’ Two years ago, the patient experienced shapeless stools, which were black and frequent, approximately four times per day. In November 2023, a gastrointestinal endoscopy showed rectal cancer and colon polyps treated by electrocautery and polypectomy. The patient subsequently visited the Fourth Hospital of Hebei Medical University, where she was diagnosed with rectal adenocarcinoma (T3N1M0) and recommended for chemoradiotherapy. From December 26, 2023, to March 2024, she completed four cycles of chemotherapy using the XELOX regimen combined with immunotherapy (Oxaliplatin 175 mg IV, Capecitabine 1000 mg in the morning, 1500 mg in the evening PO, days 1-14, and Toripalimab 200 mg IV every 21 days) along with phase-I radiotherapy. Follow-up colonoscopy revealed a scar at 5 cm from the anus, where a mucosal elevation was noted. Magnifying endoscopy did not reveal any atypical tumor blood vessels, and pathology indicated chronic inflammation with foam cell aggregation in the lamina propria and focal pale-staining acellular areas. PET–CT and rectal MRI showed no evidence of tumor. After multidisciplinary consultation involving oncology, general surgery, gastroenterology, and radiology, the tumor was classified as having achieved clinical complete remission (cCR). The patient had previously undergone external hemorrhoidectomy and cesarean section.
Diagnosis: post-treatment rectal cancer (cCR).
After thorough preoperative evaluation, nutritional optimization, and signing the informed consent form, the patient underwent peranal super minimally invasive full-thickness resection (SMIS–EFTR) for rectal cancer after treatment (cCR). Refer to video 13.5, Rectal cancer post-chemoradiotherapy treated by peranal super minimally invasive full-thickness resection (See online resources).
Surgical procedure: a scar was seen at 5 cm from the anus in the rectum after chemoradiotherapy. The lesion was marked 1 cm from the tumor using argon plasma coagulation (APC). After submucosal injection of a mixture of saline solution, methylene blue, and adrenaline, the surrounding mucosa was elevated. The lesion’s edge was then incised with a DualKnife and circumferentially marked. Submucosal dissection was performed, and where the scar tissue failed to elevate, the lesion was retracted using the 8-shaped loop and tissue clips. After the rectal wall was elevated, the lesion was completely excised with a triangle knife. Hemostasis was achieved using hemostatic forceps, and the muscular layer was closed with sutures. A dose of biological protein glue was sprayed to reinforce the closure. The specimen size was measured at 2.5 cm × 2.0 cm. Postoperative pathological examinations revealed a chronic inflammation of the (rectal) colonic mucosa, with some glands showing mild to moderate atypical hyperplasia, focal fibrous tissue proliferation in the mucosa and submucosa, with no definite cancer seen.
(IV) Hot-spot issues and discussions in the consensus
Endoscopic full-thickness resection (EFTR) has become an important super minimally invasive technique for tumor removal, including the full thickness of the gastrointestinal wall. EFTR represents a technological advancement in the treatment of tumors of the digestive tract and offers a feasible option for lesions involving the mucosa and submucosa. It is primarily used for early gastrointestinal tumors, such as gastrointestinal stromal tumors (GISTs), gastric-like tumors, and early gastric cancers.
EFTR has shown high technical success rates and safety in the treatment of colorectal tumors. For instance, a study conducted in three UK institutions using the FTRD system on 11 cases of colon EFTR showed an 82% technical success rate, with no intraoperative or delayed bleeding or perforation events. Additionally, experiences from two Swiss tertiary referral centers suggest that the EFTR technique with the FTRD system is feasible and effective for benign tumors with a diameter of up to 30 mm, despite some complication risks. A recent review suggests that EFTR could potentially become a strong competitor as an alternative standard for the treatment of locally advanced rectal cancer. A meta-analysis summarizing studies before December 2016 comparing open versus closed management after full-thickness resection of rectal tumors found no significant difference in the overall complication rates between the two groups.
However, the closure of the rectal wall defect remains a contentious issue, as EFTR involves intentional perforation and the creation of wall defects during tumor resection.
Recently, several new suturing tools and techniques have emerged to enhance the safety, efficiency, and healing of these defects. For example, a new disposable circular stapler (FS5L-1, Olympus) simplifies the procedure by cutting and closing irregularly shaped mucosal defects. This technique has been successful in completely closing defects up to 90 mm in length. The double-sleeve suturing technique, which uses two end-loops and metal clips through a single-channel endoscope, has also proven effective in repairing large gastric defects, making it a safe and reliable method for closing full-thickness perforations. Additionally, the X-Tack device has shown promise in closing large mucosal defects, providing durable sealing for full-thickness perforations.
Endoscopic full-thickness resection (EFTR) has garnered significant attention as a super minimally invasive surgery option, especially for gastrointestinal tumors, including colorectal cancers. One of the major areas of research is expanding the indications for EFTR, particularly for locally advanced rectal cancers, as an alternative to the standard total mesorectal resection (TME).
Currently, TME is considered the gold standard for rectal cancer surgery. However, TME is a technically demanding procedure, requiring long operative times and high expertise from the surgeon. Postoperative complications, such as anastomotic leaks, bleeding, infections, and potential damage to blood vessels and nerves, can affect the patient’s functional outcomes, including sexual function. The importance of organ preservation has become more pronounced with the development of super minimally invasive techniques, leading to growing interest in EFTR for rectal cancer patients, particularly those who achieve clinical complete response (cCR) or near-complete response after neoadjuvant therapy. Recent studies have investigated the feasibility and safety of EFTR for rectal cancer patients who are in near-complete remission following neoadjuvant therapy. One such study focused on a 74-year-old woman diagnosed with cT3N0M0 stage rectal adenocarcinoma. After receiving neoadjuvant chemoradiotherapy, the patient was evaluated as being in near-complete remission and subsequently underwent EFTR. The postoperative pathology showed good local control, and the patient had a satisfactory recovery. During a one-year follow-up, there was no evidence of recurrence, and anal function was preserved. This suggests that EFTR can be a safe and viable organ-preserving treatment option for patients with near-complete remission, though larger-scale studies are needed to validate these findings.
In summary, EFTR has demonstrated high technical success and safety, particularly in the treatment of early-stage gastrointestinal tumors, and emerging studies suggest its potential in the management of locally advanced rectal cancer, especially in cases of near-complete remission after neoadjuvant treatment. Although EFTR involves intentional perforation and the creation of wall defects, the advent of new suturing tools and techniques, such as the disposable circular stapler, double-sleeve suturing method, and X-Tack device, has enhanced the safety and efficacy of the procedure. These advancements support the expansion of EFTR’s indications and suggest it may become a viable alternative to traditional rectal cancer surgery. Future research should focus on validating the long-term safety and efficacy of EFTR in larger populations of patients and continue to refine the surgical indications and techniques to optimize treatment outcomes, reduce complications, and improve the quality of life of the patients.
13.1.4 Early Rectal Cancer: Endoscopic Submucosal Tunnel Dissection
(I) Overview
Early rectal cancer endoscopic submucosal tunnel dissection (ESTD), formerly known as tunnel technique endoscopic submucosal dissection, is a minimally invasive procedure performed endoscopically to excise early rectal cancers. ESTD is a technique in which the boundaries of the lesion are identified endoscopically, with markings made on both its oral and anal sides. After submucosal injection, the mucosa on the oral and anal sides of the lesion is incised, and the dissection is carried out in the submucosal layer, creating a tunnel between the mucosal muscle layer and the muscularis propria. The entire lesion is dissected laterally within the tunnel, and the specimen is retrieved, with the wound treated accordingly.
Traditional endoscopic submucosal dissection (ESD) is technically challenging, particularly in the colon and rectum, due to several anatomical factors. These include the thin walls of the colon, frequent peristalsis, the angulated folds, and limited working space within the lumen. Additionally, as the dissection progresses, the lesion gradually retracts, and the previously dissected edges may invert, obscuring the surgical field and increasing the difficulty of the procedure. In 2009, Professor Enqiang Linghu developed the ‘endoscopic tunnel technique,’ which involves creating a channel between the mucosa and the muscle layer through endoscopic submucosal dissection. This channel serves as a working space for endoscopic treatment, and the technique was named endoscopic submucosal tunnel dissection (ESTD). Initially, this method was applied mainly to the treatment of achalasia of the cardia. Subsequently, it was gradually adopted for the treatment of extensive early esophageal cancers and precancerous lesions. In recent years, with the further development of ESTD, there has been an increasing number of reports on its application in the treatment of early gastric and colorectal cancers, as well as submucosal tumors.
(II) Surgical procedures and techniques
1. Routine ESTD
(1) Diagnostic evaluation: a white light endoscope is used to identify the lesion. The surface mucus and fecal matter are repeatedly washed using a water pump to clearly visualize the lesion, and NBI (narrow-band imaging) is applied to observe the ductal openings. Subsequently, small-probe EUS (endoscopic ultrasound) is performed to assess the lesion’s depth. Indigo carmine is sprayed onto the surface to clearly delineate the lesion’s boundaries, ductal structures, and morphology, and images are recorded.
(2) Tunnel technique (figure 13.7): due to the unique anatomical structure of the intestine, the tumor’s boundaries are relatively well-defined, eliminating the need for marking. The procedure is performed under direct endoscopic visualization.
1) Submucosal injection: a mixture of saline solution, methylene blue, and adrenaline is injected into the submucosa to adequately lift the mucosal layer.
2) Creation of the tunnel opening: a DualKnife or Hook knife is used to laterally incise the superficial mucosa and create the tunnel opening, followed by dissection.
3) Tunnel creation: the submucosal dissection is performed along the long axis of the lesion.
4) Expansion of the tunnel: the dissection is expanded laterally along the previously created tunnel, reaching the lesion’s boundaries.
5) Tunnel perforation: the mucosa on the deeper side of the tunnel is cut, and the presence of a ‘perforation sign’ is checked inside the tunnel.
6) Separation of mucosal edges: the mucosal edges on both sides of the tunnel are separated.
7) Wound management: the wound is treated accordingly.
8) Specimen retrieval and documentation: the specimen is retrieved, photographed, and fixed for further examination.
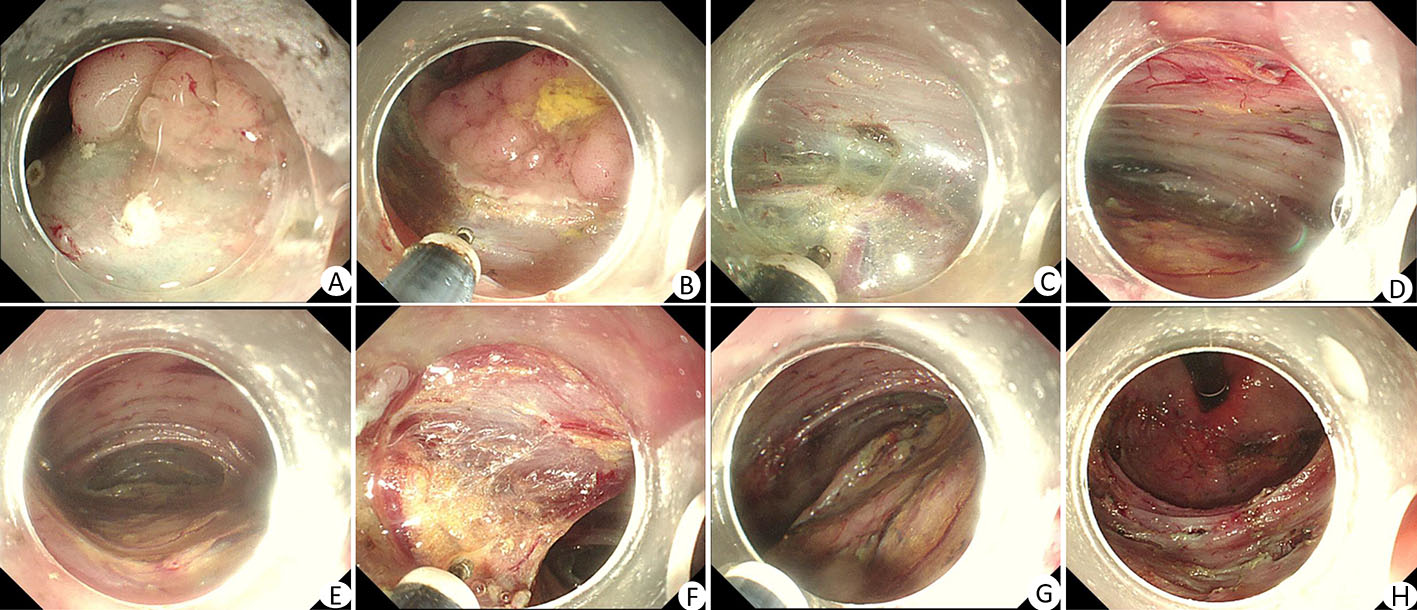

FIG. 13.7 — Endoscopic submucosal dissection (ESD) procedure using the tunnel technique. A. Good elevation sign after submucosal injection. B. Lateral incision of the tunnel opening. C. Establishment of the tunnel. D. Expansion and separation of the internal tunnel. E. Perforation sign indicating tunnel perforation. F. Separation of both sides of the mucosa under the effect of gravity. G. The lesion appears ‘suspended’ after dissection. H. Fully dissected wound site.
1. Pushing-type ESTD
Due to the tortuosity of the colon wall and the influence of the colonic folds, it is not possible to construct a straight submucosal tunnel as in the esophagus. Therefore, Professor Enqiang Leng’s team developed a modified ESTD technique based on the submucosal tunnel for endoscopic treatment of large colonic lesions. This technique involves performing colonoscopy from the anal side toward the oral side of the lesion, following a sequence of tunnel establishment, incision of tunnel edges, re-establishment of the tunnel, and re-incision of the tunnel edges, and is named ‘pushing endoscopic submucosal tunnel dissection’ (PESTD). In brief, PESTD involves simultaneous tunnel creation and incision of edges, which not only resolves the issue of fluid retention in the submucosal layer but also overcomes the difficulty of folding over the colonic folds within the submucosal tunnel as seen in traditional ESTD techniques.
Specific operation method (figure 13.8):
(1) Treatment begins with submucosal injection of saline solution from the anal side. The lesion is adequately elevated, and the DualKnife is used to make an arcuate incision along the lesion’s boundary.
(2) The endoscope is then inverted at the oral side of the lesion, and after submucosal injection of a liquid cushion, an arcuate incision is made on the mucosa.
(3) The endoscope proceeds with submucosal dissection from the anal side to establish a tunnel of 0.5–1 cm in length. The mucosa on both sides of the tunnel is incised along the lesion’s boundary, and the tunnel is further extended. Another 0.5–1 cm of submucosal dissection is performed, followed by incision of the mucosa on both sides of the tunnel. The procedure is performed sequentially from the anal side to the oral side: tunnel creation, edge incision, tunnel creation, edge incision, and ultimately complete dissection of the lesion.
(4) After removal of the lesion, hemostatic forceps are used to treat exposed bleeding points and small vessels at the wound site. A careful inspection is performed to ensure that there is no perforation at the wound and no residual lesion at the periphery.
(5) The resected lesion specimen is retrieved and sent for pathological examination.
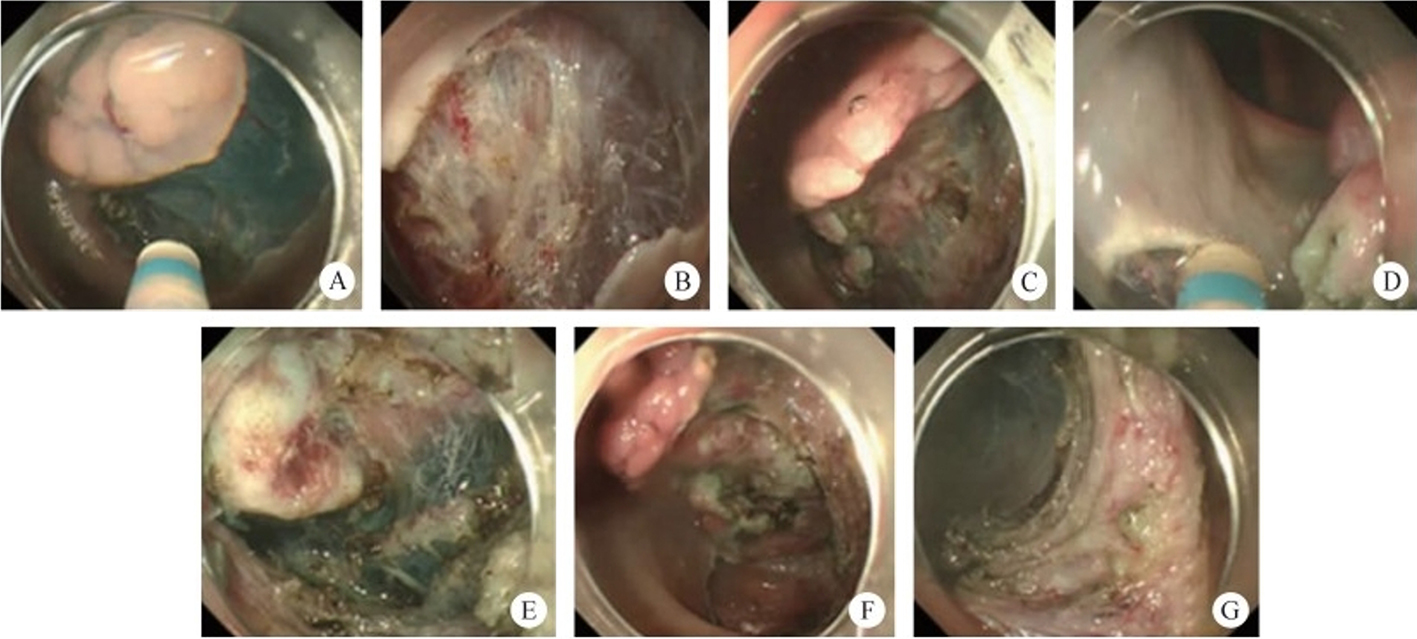

FIG. 13.8 — Pushing endoscopic submucosal tunnel dissection (PESTD) procedure. A. Arcuate incision of the mucosa along the lesion boundary on the anal side. B. Submucosal dissection and tunnel establishment. C. Tunnel length of 1–2 cm. D. Incision of the mucosa around the tunnel. E. Re-establishment of the tunnel. F. Further incision of the mucosa around the tunnel, with the procedure advancing from the anal side toward the lesion’s oral side. G. Final complete removal of the lesion.
(III) A classic case
Medical history: the patient is an elderly woman who was admitted due to “a rectal mass found during a routine examination two weeks ago.” Colonoscopy revealed a laterally growing mass in the rectum with an uneven, nodular surface, occupying about half of the lumen. Pathological examinations indicated high-grade dysplasia and carcinoma (well to moderately differentiated adenocarcinoma) of the rectal mucosa. The patient has a history of hypertension and old cerebral infarction.
Diagnosis: early-stage rectal cancer.
After a thorough preoperative evaluation, nutritional status improvement, and obtaining informed consent for surgery, the patient underwent peranal super minimally invasive resection (SMIS–ESTD) for the rectal lesion. Refer to video 13.6, Super minimally invasive resection of early rectal cancer through a tunnel channel (See online resources).
Surgical procedure: an 8.0-cm mucosal elevation was observed in the rectum, with the anal side of the lesion located 1 cm from the dentate line. NBI–ME: small focal microvascular enlargement and mild tortuosity, with a well-preserved arrangement and direction. After marking with a DualKnife, submucosal injection was performed, and the lesion’s circumferential mucosa was incised down to the submucosal layer. An ‘I’-shaped incision was made on the anal side, through which the submucosal layer was progressively dissected to establish a submucosal tunnel. Dissection was performed while injecting fluid, and the lesion was completely removed. Hemostasis was achieved using hemostatic forceps. The muscularis propria was found to be thin in some areas of the wound, and tissue clips were used to secure it. The pathological specimen was retrieved, and the endoscope was withdrawn. The excised lesion measured 8.0 cm × 4.5 cm.
(IV) Hot-spot issues and discussions in the consensus
1. Advantages of ESTD. The Expert Consensus on Endoscopic Submucosal Tunnel Dissection (ESTD) in GastroColorectal Diseases (2017, Beijing) indicates that due to the non-straight tubular shape of the stomach and colon, constructing a complete submucosal tunnel is challenging, and related reports are rare. The rectal lumen is relatively straight, and although some literature reports using ESTD for resecting circumferential or large mucosal lesions in the rectum, these studies are limited by small sample sizes and are currently used only in clinical case reports. However, the inherent advantages of ESTD make its role in gastrointestinal disease treatment undeniable. A study on the clinical application of endoscopic submucosal dissection (ESD) in large lateral spreading tumors of the colon and rectum points out the following advantages of ESTD. ① Good visibility: the physiological space between the mucosal, submucosal, and muscular layers provides a clear visual field, aiding in accurate knife handling and anatomical layer identification. ② Predictable vascular handling: adequate space ensures clear display of submucosal blood vessels and tissue layers. Coarse blood vessels and the perforating vessels of the muscular layer can be treated with thermal biopsy forceps and moderate electrocautery; finer vessels can be directly treated with slow contact electrocautery using an electrosurgical knife. Complete electrocautery is necessary to avoid delayed bleeding from residual vessel stumps. ③ Control of dissection depth: dissection along the lower third of the submucosal layer is easy under direct vision, with few blood vessels in this layer, resulting in minimal intraoperative bleeding. It also reduces muscular layer damage and lowers the risk of perforation. ④ Use of gravity: when first establishing the tunnel and performing dissection, begin from the higher point of gravity to facilitate quick access to the tunnel and reduce the need for excessive gas injection. When opening the edges, cut the mucosa at the lower gravity point first and then move to the higher point using the ‘suspension technique’ based on gravity principles, with the entire process conducted under direct vision. ⑤ High Complete Resection Rate: clear operation steps, good visibility, stable anatomical layer and knife handling, and predictable vascular treatment all significantly improve the complete resection rate. ⑥ Increased surgical efficiency: after successfully establishing the tunnel, utilizing the transparent cap, a CO
2. Selection of ESTD treatment candidates. Currently, there is no clear definition or guideline for ESTD indications. In clinical practice, the decision to use ESTD mainly relies on the surgeon’s experience and the lesion’s location. Based on previous reports, two main criteria should guide ESTD treatment selection: (1) the lesion must be in a location that allows easy tunnel creation, such as the rectum, esophagus, large curvature of the gastric antrum, and the greater curvature of the gastric body; (2) the establishment of the tunnel should facilitate the rapid and safe dissection of the lesion. Based on these criteria, ESTD demonstrates safety and efficiency in the dissection of large early colorectal cancer lesions or giant colonic adenomas, providing advantages in lesion removal.
3. Difficulties and key points in ESTD operations. ① The tunnel entrance should be large, with transverse cuts near the lesion’s horizontal diameter, and deep dissection should reach the lower third of the submucosal layer to facilitate subsequent steps. In special areas such as the sigmoid colon and ascending colon, the endoscope may need to be reversed for increased stability or may require positional changes during operation. ② During tunneling dissection, the endoscope’s rotary knob and right-hand rotation of the endoscope handle should be used to follow the arc-shaped muscle plane for dissection. ③ To determine the length of the tunnel, the endoscope should be withdrawn and observed from outside the tunnel. If dissection exceeds two-thirds of the longitudinal length, the mucosa opposite the tunnel entrance should be incised to facilitate tunnel continuity. Injecting a slightly higher concentration of methylene blue solution into the opposite normal mucosa can assist in localization.
13.2 Super Minimally Invasive Resection of Colorectal Subepithelial Lesions
13.2.1 Overview
Colorectal subepithelial lesions (SELs) are protruding lesions originating from the mucosal muscle layer, submucosal layer, or muscularis propria, and can also present as extraluminal lesions. Typically, SELs smaller than 2 cm do not exhibit significant clinical symptoms and are often discovered incidentally during endoscopic examinations. However, some specific types or lesions located in certain areas may develop symptoms such as abdominal pain, bleeding, and obstruction as the disease progresses and the lesion size increases.
(I) Diagnosis of colorectal subepithelial lesions
1. White-light endoscopy. Routine white-light endoscopy is the fundamental and initial method for detecting SELs, providing information on lesion location, size, mobility, mucosal color, shape, and signs of erosion or bleeding. However, it cannot determine the nature or origin of the lesion, particularly for SELs exhibiting an exophytic growth pattern, where diagnosis cannot be made based solely on routine white-light endoscopy.
2. Endoscopic ultrasound (EUS). EUS is the first-line examination for diagnosing SELs, offering information on lesion origin, intramural or extramural location, size and shape, echogenicity, vascularization, and associated lymph node involvement. These features allow for the evaluation of the lesion’s histology and even its malignant potential. EUS has a sensitivity of 92% for distinguishing extraluminal compression from intraluminal lesions, with an accuracy rate of 77%–89% for diagnosing gastrointestinal stromal tumors (GIST).
3. Other imaging techniques. Other imaging methods, including CT and MRI, also play a crucial role in diagnosing SELs. These techniques can directly reveal the tumor’s location, growth pattern, size, shape, lobulation, density, homogeneity, enhancement degree, and boundary characteristics. Additionally, they can assess whether the gastrointestinal wall is thickened and the extent of thickening. More importantly, these imaging techniques can detect local invasion of adjacent structures, as well as metastasis to the surrounding peritoneum, lymph nodes, and other organs, making them vital for clinical tumor staging, treatment planning, and prognosis evaluation.
4. Histopathological diagnosis. Histopathological examination remains the gold standard for diagnosing SELs. Since SELs are covered by normal mucosa, routine endoscopic biopsy techniques often fail to obtain sufficient tissue samples. Multiple biopsies from the same site may retrieve deeper tissues but increase the risk of bleeding, and the obtained tissue may still be insufficient for a definitive diagnosis. Notably, all preoperative methods for obtaining pathological samples are invasive and may damage the mucosa or cause adhesion to the submucosal tissue, thereby complicating surgery. These procedures also carry risks such as bleeding, perforation, or tumor dissemination. Therefore, preoperative biopsy is not always necessary, especially for SELs that can be diagnosed via routine endoscopy combined with EUS, such as lipomas, cysts, and ectopic pancreas, where tissue sampling may not be required. Moreover, for potentially malignant SELs that meet the criteria for endoscopic resection, an experienced endoscopist may directly perform endoscopic removal after preoperative evaluation, without requiring pathological diagnosis. In cases where malignancy is suspected or when routine endoscopy combined with EUS cannot assess the benign or malignant nature of the lesion, techniques like EUS-guided fine needle aspiration/biopsy (EUS–FNA/FNB) or mucosal incision-assisted biopsy (MIAB) can be used for tissue sampling to facilitate preoperative pathological assessment.
(II) Indications and contraindications
1. Indications
(1) For tumors suspected or pathologically confirmed as having malignant potential based on preoperative examinations, endoscopic resection is primarily used for colorectal SELs, including neuroendocrine tumors (NETs) and lipomas, according to the ESGE guidelines.
(2) For colorectal NETs, the indications for endoscopic treatment are as follows, based on the 2022 Chinese Consensus on Gastrointestinal, Pancreatic, and Neuroendocrine Tumors: Colonic NENs: endoscopic resection can be considered for tumors <2 cm, without lymph node metastasis, and with pathological grading of G1 or G2. For positive resection margins, salvage radical surgery (colonic resection + regional lymph node dissection) is required. Rectal NENs: for rectal NETs < 1 cm, staged as T1, without lymph node metastasis, and with pathological grading of G1 or G2, endoscopic resection is recommended.
(3) SELs with symptoms (e.g., bleeding, obstruction).
(4) For tumors suspected or pathologically confirmed as benign but for patients who are unable to follow up regularly, or for those whose tumors rapidly enlarge during the follow-up period and have a strong desire for endoscopic treatment.
2. Contraindications
(1) Tumors with confirmed lymph node or distant metastasis.
(2) For some SELs with confirmed lymph node or distant metastasis, large tissue biopsy for pathological diagnosis may be considered a relative contraindication.
(3) Patients with poor general condition who are unable to tolerate endoscopic surgery after detailed preoperative evaluation.
(III) Postoperative management
Fasting and intravenous fluids for two to three days. For full-thickness resection during surgery, or in cases with systemic infections, perforation, or immunocompromised states, antibiotics should be used according to relevant guidelines. Gradual transition to oral diet: liquids → soft diet → regular diet. Postoperatively, routine monitoring of blood counts and biochemistry is essential to promptly correct any metabolic disorders.
(IV) Technical methods for super minimized invasive resection of colorectal subepithelial tumors
Colorectal subepithelial tumors can be cured through super minimally invasive surgery (SMIS), primarily using natural orifices, tunnel, or multi-cavity approaches for endoscopic treatment:
Peroral super minimally invasive resection of subepithelial lesions is a technique in which the subepithelial tumor of the colon or rectum is completely resected locally via a peranal endoscopic approach;
Per-tunnel super minimally invasive resection of subepithelial lesions is a technique in which the subepithelial tumor of the colon or rectum is completely resected locally via a tunnel endoscopic approach;
Per-multiple-cavity super minimally invasive resection of subepithelial lesions is a technique involving both abdominal skin puncture and the peranal natural orifice, with the combined use of digestive endoscopy and laparoscopy, either concurrently or sequentially, for local resection of colorectal subepithelial tumors.
The technical methods currently applied in super minimally invasive surgery for colorectal subepithelial tumors and their previous names are listed in table 13.2.
TAB. 13.2 — Technical methods for super minimally invasive surgery of colorectal subepithelial tumors and their previous names.
| No. | Super Minimally Invasive Surgery (SMIS) | Technical Method | Previous Name |
| 1 | Peranal super minimally invasive submucosal tumor resection of colorectal epithelium | Super minimally invasive non-total layer resection | Endoscopic submucosal dissection (ESD), traction-assisted endoscopic submucosal dissection (TA-ESD) |
| 2 | Peranal super minimally invasive submucosal tumor resection of colorectal epithelium | Super minimally invasive full-thickness resection | Cap-assisted endoscopic mucosal resection (EMR), endoscopic mucosal resection (EMR), endoscopic submucosal tumor excavation, endoscopic full-thickness resection, traction-assisted endoscopic full-thickness resection |
| 3 | Peranal super minimally invasive tunnel resection of colorectal submucosal tumors | Super minimally invasive non-total layer resection | Endoscopic tunnel submucosal dissection (ETSD) |
| 4 | Peranal super minimally invasive multi-lumen channel resection of colorectal submucosal tumors | Multi-lumen channel super minimally invasive resectionTraction-assisted endoscopic submucosal dissection | Multi-endoscope combined therapy |
13.2.2 Peranal Super Minimally Invasive Resection of Colorectal Submucosal Tumors
I. Ligation-assisted endoscopic mucosal resection (LAEMR)
Peranal super minimally invasive resection of colorectal submucosal tumors can be achieved through ligation-assisted endoscopic mucosal resection (LAEMR). LAEMR is a technique where the lesion’s boundaries are identified under endoscopy, followed by marking, submucosal injection, and adequate lifting of the lesion. A ligator with an attached ligation ring is used to directly suck and ligate the lesion, after which the lesion is excised using a snare at the base of the ligation ring and retrieved. Finally, the wound is treated.
Studies around the world have confirmed that endoscopic mucosal resection (EMR) for the resection of superficial endoscopic lesions (SELs) smaller than 2 cm is safe and effective. The risk of bleeding is between 4% and 13%, while the perforation risk ranges from 2% to 7%.
(I) Surgical procedure and techniques (figures 13.9 and 13.10)
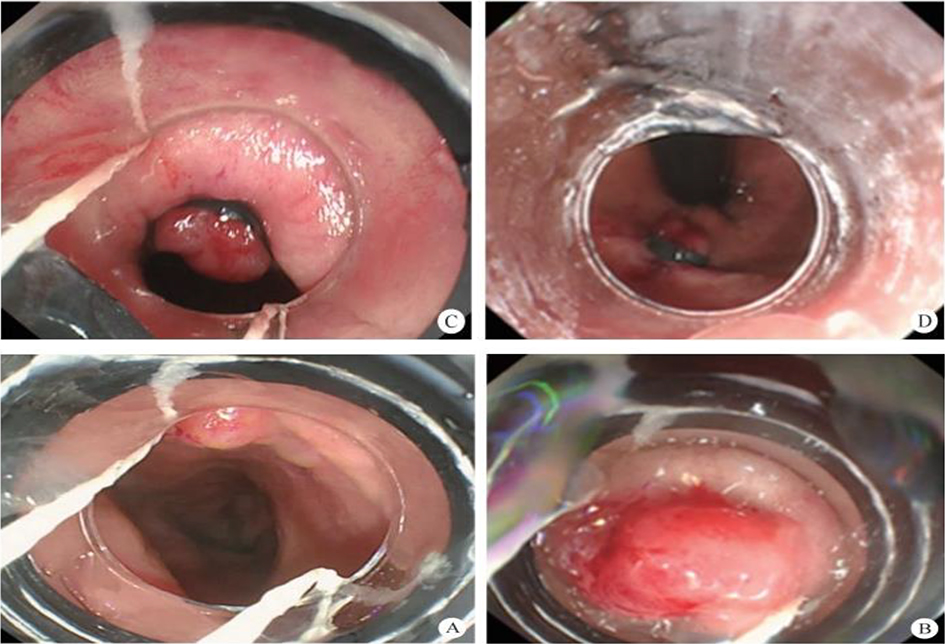
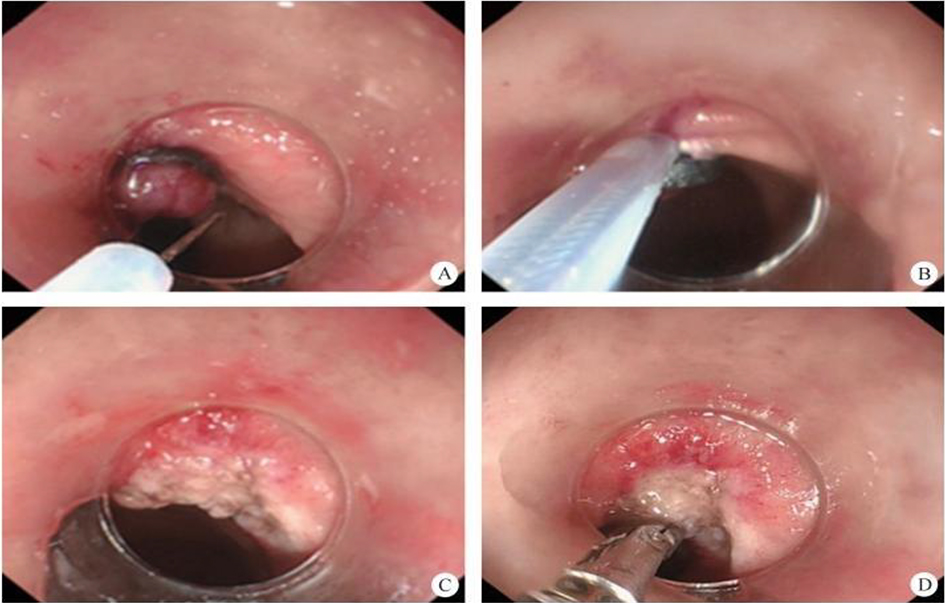
After fully exposing the lesion, the ligation ring is used to ligate the base of the lesion.
The snare is used to directly ligate and excise the lesion. For larger lesions, segmental resection may be necessary.
If there is minimal oozing from the wound, rinse with norepinephrine solution and use an argon plasma coagulator (APC) for hemostasis. If there is significant arterial bleeding, use a hot biopsy forceps or titanium clips for hemostasis. Routine closure of the wound is performed with titanium clips.

FIG. 13.9 — Steps of the placement of the rubber band by peranal super minimally invasive ligation-assisted mucosal resection of colorectal submucosal tumors. A. Rectal mucosal lesion (NET), preparation for ligator-assisted resection device. B. Position the ligator close to the lesion, ensuring the lesion is centered in the field of view for suction. C. Release the rubber band, fully ligating the lesion. D. Use reverse endoscopy to assess the effectiveness of the ligation.

FIG. 13.10 — Steps of ligation and resection in peranal super minimally invasive ligation-assisted mucosal resection of colorectal submucosal tumors. A. Insert the snare. B. Tighten the snare at the base of the rubber band to capture the lesion. C. Perform electrocoagulation to resect the lesion. D. Apply prophylactic hemostasis to the wound.
Refer to video 13.7, Peranal super minimally invasive non-full-thickness layer resection of rectal subepithelial tumors (See online resources).
(II) Hot-spot issues and research progress in the consensus
1. Indications of ligation-assisted endoscopic mucosal resection (LAEMR) The ESGE Guidelines on Endoscopic Treatment of Submucosal Lesions, including neuroendocrine tumors, recommend modified EMR as the first choice for resecting colorectal neuroendocrine tumors (NETs) smaller than 10 mm. For lesions smaller than 20 mm, ESD (Endoscopic Submucosal Dissection) is preferred. A meta-analysis indicated that for rectal neuroendocrine tumors smaller than 16 mm, the complete resection rate for ESD was 89%, with an adverse event rate of approximately 4%, and a local recurrence rate of less than 1%. The complete resection rate of ESD is comparable to modified EMR techniques, including ligation-assisted EMR, double-channel EMR, cap-assisted EMR, and pre-cut EMR, all of which significantly outperform conventional EMR.
The ACG Clinical Guidelines: Diagnosis and Management of Gastrointestinal Submucosal Lesions recommend EMR or ESD for type-1 or type-2 gastrointestinal neuroendocrine tumors (gNETs). However, for small low-grade type-3 gNETs, where EUS or imaging shows no signs of lymphatic metastasis, ESD is preferred, and EMR is not recommended. A retrospective study comparing clinical outcomes in 50 patients with type-3 gNETs treated with EMR or ESD found that the pathological margin positivity rate was 7/41 (14.6%) in the EMR group and 1/9 (11.1%) in the ESD group, although the difference was not statistically significant (P = 0.249). Furthermore, research on type-1 gNETs suggests that ESD may have a higher R0 resection rate compared to EMR. Therefore, EMR is not recommended for type-3 gNETs.
In contrast, the Chinese Expert Consensus on Endoscopic Diagnosis and Treatment of Gastrointestinal Submucosal Tumors (2023 Edition) suggests that for relatively superficial lesions, where preoperative EUS and CT scans confirm the lesion protrudes into the lumen and can be completely resected with a snare, endoscopic snare resection may be performed.
2. Post-endoscopic treatment medication guidelines for SELs. For cases involving full-thickness resection during surgery, or those complicated by systemic infections, perforation, or immunocompromised conditions, antibiotics may be administered according to relevant antibiotic guidelines. Reports on bacteremia or similar infections following endoscopic treatment of SELs are rare. Early foreign literature has recommended the prophylactic use of antibiotics for cystic lesions prior to EUS–FNA, although large prospective clinical studies are lacking. A recent chinese study indicated that for patients undergoing gastric ESD or ESE without perforation or serious comorbidities, the use of antibiotics is generally not necessary for fever post-surgery. The primary purpose of postoperative antibiotic use is to prevent infections in the mediastinum, retroperitoneum, or free abdominal cavity around the surgical wound, as well as potential systemic infections that may occur after surgery. For patients with a larger resection scope, longer procedural duration, or complications such as gastrointestinal perforation and significant bleeding, or those who develop systemic infections or have weakened immune systems, antibiotics may be administered based on the patient’s condition, in accordance with relevant antibiotic guidelines.
II. Endoscopic submucosal excavation (ESE)
Peranal super minimally invasive resection of colorectal submucosal tumors can be achieved through endoscopic submucosal excavation (ESE). ESE refers to the technique where the lesion’s boundaries are identified under endoscopy, marked, submucosal injection is performed, and the lesion is adequately lifted. The submucosal layer is then incised to expose the tumor, and the lesion is carefully peeled off along its capsule and retrieved. Finally, the wound is treated. Literature reports indicate that the complete resection rate for ESE can reach 92%. For tumors ≤1.5 cm, the complete resection rate can be 100%, while for tumors >1.5 cm, the complete resection rate can still reach 77.8%.
(I) Surgical procedure and techniques (figures 13.11 and 13.12)
(1) Marking: an electrosurgical knife or argon gas is used to perform electrocoagulation marking at the edge of the lesion.
(2) Submucosal Injection: multiple submucosal injections are performed outside the marked point at the edge of the lesion.
(3) Incising the mucosa at the lesion’s outer edge: the mucosa is incised along the marked point at the lesion’s edge.
(4) Excavating the lesion: submucosal dissection is performed beneath the lesion to excise the tumor. Multiple submucosal injections are made during the excavation process.
(5) Wound treatment: after tumor resection, any visible small blood vessels on the wound should be treated with APC coagulation. If necessary, metal hemostatic clips can be used to suture the wound.
(II) Hot-spot issues and research progress in the consensus
1. Preoperative diagnosis of SELs. Currently, the preoperative diagnosis of SELs still relies on endoscopic ultrasound (EUS), with some cases requiring additional imaging techniques such as CT scans to assess the potential nature of the lesion and guide treatment decisions. Although endoscopic treatment techniques have become more advanced, it is crucial, in the author’s opinion, to ensure a thorough preoperative diagnosis. Proceeding with endoscopic treatment before completing the necessary diagnostic procedures may result in treatment failure or delay the progression of the disease. Given the technical limitations of EUS, artificial intelligence (AI) may play a role in advancing this field. AI has already been widely applied in medicine, with notable success in areas like quality control in gastrointestinal endoscopy, polyp detection, and early cancer diagnosis. Applying AI to white-light endoscopy and endoscopic ultrasound in the preoperative assessment of SEL could potentially improve diagnostic accuracy and reduce the technical requirements associated with these procedures.
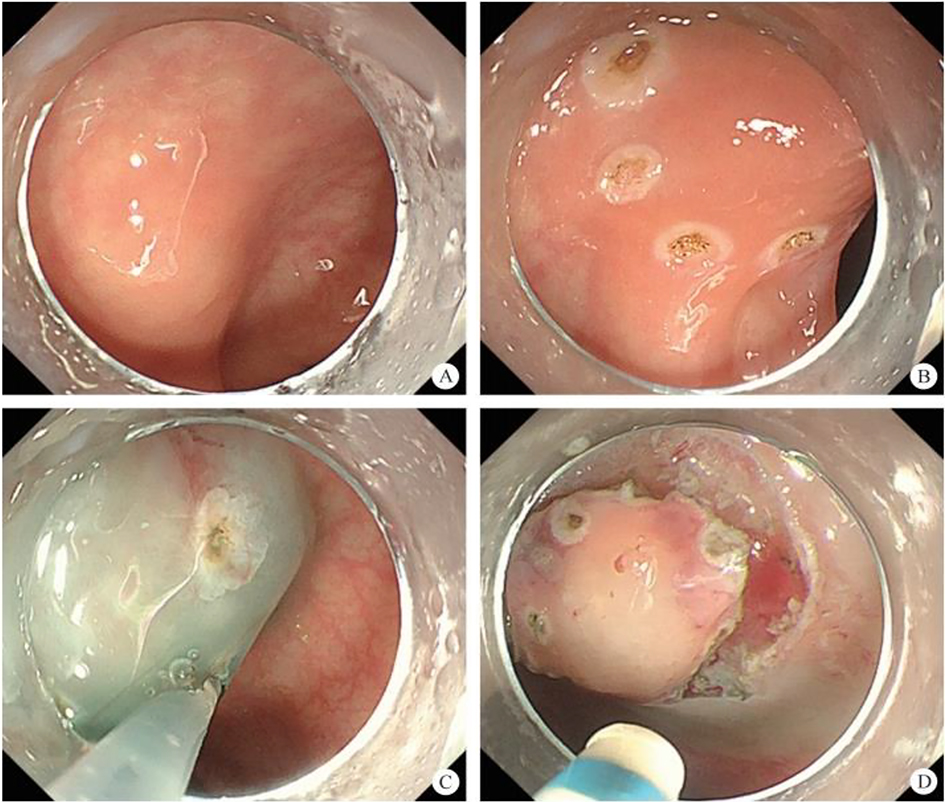
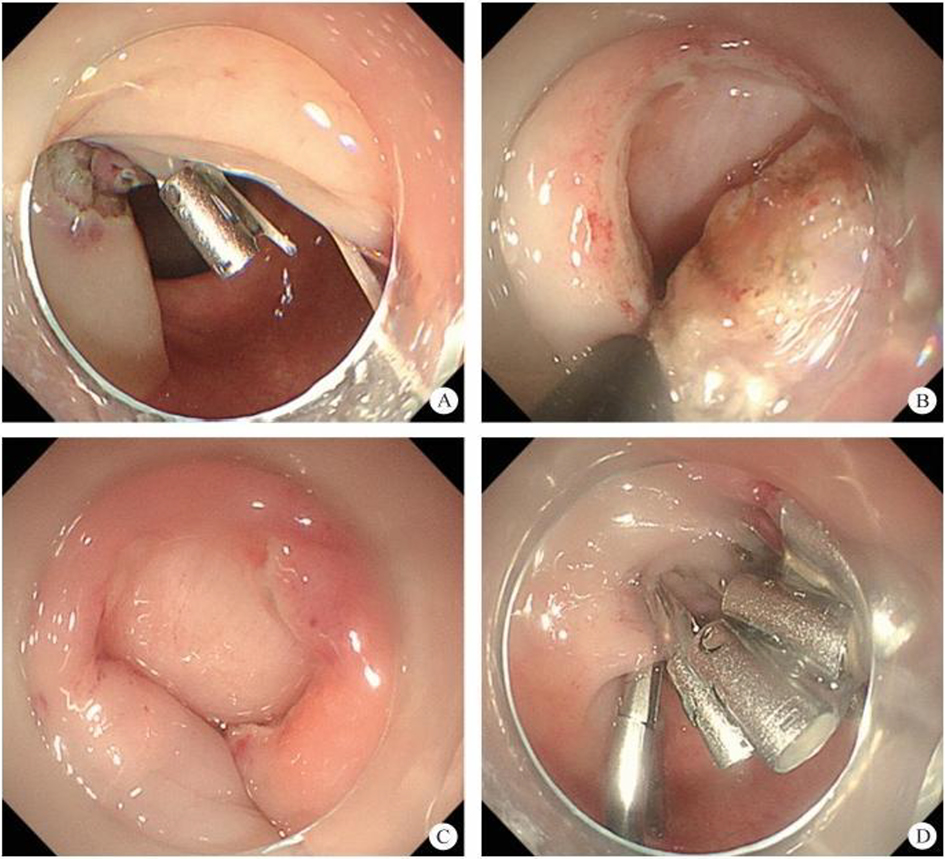

FIG. 13.11 — Steps of circumferential incision in peranal super minimally invasive full-layer resection of colorectal submucosal tumors. A. Fully expose the lesion. B. Perform electrocoagulation marking at the edge of the lesion. C. Perform submucosal injection. D. Incise the mucosa along the marked point at the edge of the lesion.

FIG. 13.12 — Steps of full-layer resection in peranal super minimally invasive full-layer resection of colorectal submucosal tumors. A. Use a tissue grasper and dental floss to traction the lesion, exposing it. B. Perform full-layer resection of the lesion. C. Manage the perforation wound after full-layer resection. D. Use tissue graspers to suture the wound.
Currently, the window procedure is used for preoperative biopsy of SELs, allowing direct biopsy of the target lesion, which improves the accuracy of preoperative diagnosis and reduces unnecessary treatments. However, with continuous advancements in endoscopic technology and increasing health demands from patients, the necessity of performing a preoperative window biopsy for SELs is a topic worth careful consideration. Preoperative EUS is often sufficient to establish the decision for endoscopic treatment. Except in certain specific cases, preoperative biopsy of the lesion is not necessary to avoid overdiagnosis, conserve medical resources, and minimize harm to patients. Nonetheless, further research is needed to confirm the accuracy of preoperative EUS.
2. ESE for gastrointestinal submucosal tumor resection. The Chinese Expert Consensus on Endoscopic Diagnosis and Treatment of Gastrointestinal Submucosal Tumors (2023 Edition) recommends that for SELs with a long diameter ≥2 cm, or those with tumors protruding into the lumen identified via preoperative EUS and CT imaging, which are difficult to remove with endoscopic mucosal resection (EMR), ESE may be performed. ESE incorporates techniques from both endoscopic submucosal dissection (ESD) and endoscopic mucosal resection, commonly using a circumferential ‘flap’ incision around the tumor to remove the mucosa covering the SEL, thereby fully exposing the tumor. This approach preserves the tumor’s integrity, improves the radicality of the surgery, and reduces intraoperative complications. For tumors ≤1.5 cm, ESE can achieve a 100% complete resection rate.
Different anatomical locations of SELs require different techniques for resection. Lesions located in the gastric antrum, the lesser curvature of the mid-body of the stomach, and the posterior wall of the lower part of the stomach are relatively easier to operate on under direct vision, making them suitable for beginners. However, lesions in the gastric fundus, the greater curvature of the upper to mid-body of the stomach, and the lesser curvature of the lower body may require mirror inversion and reverse operation, making the procedure more challenging. Studies have shown that ESE is more prone to perforation when resecting lesions in the gastric fundus.
In the esophagus, the narrow lumen, limited space, and thin wall with no serosal layer make ESE suitable only for resection of esophageal SELs originating from the mucosal or muscularis mucosa layer. For SELs originating from the submucosal or muscularis propria layers, submucosal tunneling endoscopic resection (STER) should be considered. Compared to the rectum, the colon has a thinner and more flexible wall, with greater variability in the direction of the bowel and fixed position, making it more challenging to operate on and prone to complications.
With the continuous development of endoscopic treatment concepts and techniques, new technologies for SEL endoscopic treatment continue to emerge. The scope of SELs that can be resected endoscopically is constantly expanding, from the previous mucosal and submucosal layers, to the muscularis propria and even the serosal layer and extraluminal tumors around the gastrointestinal wall. This is an area that warrants attention. However, research on new techniques for SEL endoscopic treatment is still limited, and consensus has not yet been reached. More clinical practice and evidence are needed to clarify these techniques and further improve them.
13.2.3 Neoadjuvant Therapy Followed by Peranal Super Minimally Invasive Resection of Rectal Stromal Tumors
Gastrointestinal stromal tumors (GISTs) originate from mesenchymal tissues and have potential malignancy. The diagnosis of gastrointestinal stromal tumors is similar to that of other gastrointestinal malignancies. Recently, magnetic resonance imaging (MRI) has been used for the preoperative evaluation of rectal GISTs, showing diagnostic efficacy comparable to that of endoscopic ultrasound (EUS). Aggressive and complete surgical resection remains the most definitive treatment approach. However, there are cases of inoperable gastrointestinal stromal tumors, for which preoperative treatment with imatinib (a tyrosine kinase inhibitor) remains a first-line strategy after obtaining a definitive diagnosis and molecular subtyping through EUS-guided biopsy.
For low-lying rectal GISTs, traditional surgical resection, which involves abdominoperineal resection (APR) and permanent colostomy, significantly impacts postoperative quality of life. However, after neoadjuvant therapy, where the tumor shrinks or softens internally, it may become amenable to local resection, or even super minimally invasive surgery, thus providing an opportunity for ‘sphincter-preserving surgery’ in such patients. This approach of combining neoadjuvant treatment with super minimally invasive surgery has gained attention as it offers the possibility to remove the tumor with minimal invasiveness, preserving the rectal function and improving the patient’s quality of life after surgery. The goal is to achieve a balance between effective oncological control and preserving organ function, making it a promising option for patients with rectal GISTs, particularly in low-lying tumors that would otherwise require extensive resection.
(I) Preoperative preparation
Rectal tumors in male patients are often located adjacent to the prostate. Preoperative instructions should remind the surgeon to carefully monitor the frequency and intensity of electrocautery during surgery to avoid excessive electrocautery, which could result in prostate damage and subsequent urinary difficulties. In male patients, consideration should be given to the placement of a urinary catheter before surgery to prevent postoperative urinary retention.
(II) Indications and contraindications
1. Indications: (1) Gastrointestinal stromal tumors (GISTs) without distant metastasis as assessed by CT, MRI, and endoscopic ultrasound; (2) Tumors that have shrunk sufficiently for endoscopic resection after neoadjuvant therapy, such as imatinib.
2. Contraindications: (1) GISTs with distant metastasis; (2) Contraindications to endoscopic treatment.
(III) Surgical procedure and techniques
1. Anesthesia and positioning. General intravenous anesthesia is administered, and the patient is positioned in the left lateral position.
2. Surgical process (figure 13.13)
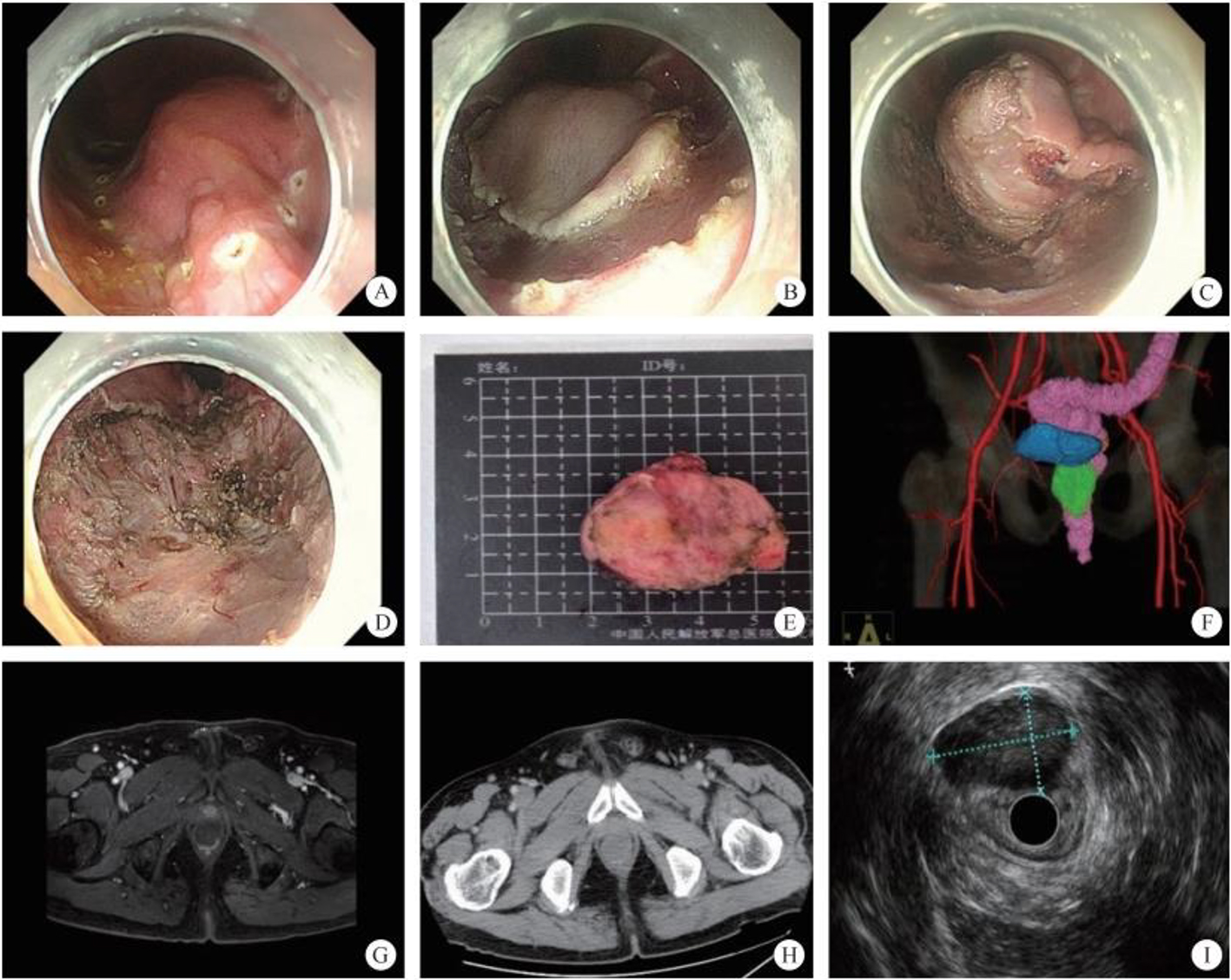
Marking: electrocautery is used with a needle-shaped scalpel to mark the edges of the lesion.
Submucosal injection: multiple submucosal injections are made at points laterally outside the marked lesion borders.
Circumferential mucosal incision: a circumferential incision is made in the mucosal layer, followed by dissection of the submucosal layer to expose the tumor growing in the muscularis propria.
Dissection: dissect along the tumor mass until the tumor is detached from the wound. The decision to perform full-thickness or partial-thickness resection is based on the tumor’s depth.
Treatment of the wound: for full-thickness resection wounds, muscle-to-muscle tissue approximation is performed using tissue forceps. For partial-thickness resection wounds, electrocautery is used for hemostasis, and no closure treatment is necessary.

FIG. 13.13 — Operational steps of peranal super minimally invasive resection of rectal gastrointestinal stromal tumors (GISTs) after neoadjuvant therapy. A. Endoscopic marking of the lesion. B. Circumferential mucosal incision. C. Dissection after tumor exposure. D. Wound site after tumor removal. E. Gross specimen of rectal GIST. F. 3D reconstructed image. G. MRI images via the rectum. H. Pelvic CT images. I. Endoscopic ultrasound image showing a hypoechoic tumor in the muscularis propria of the rectum.
(IV) A classic case
Medical history: the patient, a middle-aged man, was admitted with the chief complaint of “rectal mass detected during a physical examination five months ago.” Endoscopic ultrasound revealed a hypoechoic mass within the fourth layer of the intestinal wall at the site of rectal protrusion, measuring approximately 42 mm × 34 mm. The mass exhibited uniform internal echogenicity with a clear border, most of it protruding into the intestinal lumen. Endoscopic ultrasound-guided fine needle aspiration was performed, and pathology indicated a spindle cell tumor. Immunohistochemistry was positive for CD117 (+), DOG1 (+), CD34 (+), and negative for S-100 (–), SMA (–), Desmin (–), Ki-67 (10%), SDHB (+), confirming the diagnosis of gastrointestinal stromal tumor (GIST). Risk assessment classified the tumor as low risk. The patient was prescribed 0.5 g imatinib once daily for six months. The patient also had a history of hypertension.
Diagnosis: rectal GIST (post-immunotherapy).
After comprehensive preoperative evaluation, improvement of nutritional status, and obtaining informed consent for surgery, the patient underwent peranal super minimally invasive resection of rectal GIST. Refer to video 13.8, Peranal super minimally invasive full-thickness resection of a GIST in the retum (See online resources).
Surgical process: a submucosal mass was visible from the anus to 4 cm from the anal verge. The surface was smooth with a white scar at the local site. Electrocautery was used to mark the lesion circumferentially. After submucosal injection, the lesion was lifted, and electrocautery was used to circumferentially incise around the lesion. The tumor was then dissected layer by layer until fully exposed and detached from the wound site. There was partial loss of the muscularis propria, and meticulous hemostasis was achieved using hemostatic forceps. Two vials of biological protein glue were applied. The specimen size measured externally was 4.5 cm × 4.0 cm.
(V) Research progress
Gastrointestinal stromal tumors (GISTs) typically do not invade adjacent tissue layers, thus large-scale resections are generally not necessary. Lymph node metastasis occurs infrequently (approximately 10%), and therefore, extensive lymph node dissection is not required. If adjacent organs are involved, complete resection should be performed when feasible. Tumor rupture during the resection process should be avoided, as it is closely associated with tumor recurrence. Endoscopic resection of GISTs is technically feasible, but further studies are needed to assess tumor integrity during the procedure.
Preoperative evaluation using multi-omics imaging can assist in the smooth execution of surgery and ensure complete resection of GISTs. CT or MRI three-dimensional reconstruction techniques can clearly provide information on the relationship between the tumor and surrounding structures, including whether vascular penetration is present, the relationship between rectal lesions and the prostate in male patients, and the tumor’s relative position to the intestinal wall. These imaging techniques provide critical reference information for the surgical procedure.
Due to the low malignant potential of GISTs, and because current adjuvant therapies do not significantly improve local or distant recurrence rates or survival, long-term follow-up is essential for all patients with GISTs, as metastasis may occur decades after diagnosis.
13.3 Super Minimally Invasive Resection of Laterally Spreading Tumors in the Colon and Rectum
13.3.1 Overview
Laterally spreading tumors (LSTs) of the colon and rectum are flat tumors that grow laterally along the intestinal wall with a diameter of ≥10 mm. Based on their morphological characteristics, LSTs are generally categorized into granular type LST (LST-G) and non-granular type LST (LST-NG). The granular type LST is further divided into homogeneous granular type (LST-G-H) and nodular mixed type (LST-G-M), while the non-granular type LST is subdivided into flat elevated type (LST-NG-F) and pseudo-depressed type (LST-NG-PD). Compared to other types of colorectal polypoid adenomas, LSTs have distinct morphological features, unique growth patterns, and higher malignant potential.
Studies have shown that the probability of colorectal cancer (CRC) co-occurring with LSTs ranges from 8.4% to 52.5%. Even benign LSTs have the potential to progress to advanced colorectal cancer within three years. Therefore, early diagnosis and treatment of LSTs before malignant transformation is crucial to reduce the mortality associated with colorectal cancer. Endoscopic treatment offers several advantages over surgical resection, including minimal trauma, faster healing, shorter hospital stays, and better postoperative quality of life. As a result, it has become the preferred treatment for colorectal LSTs, with the main approaches being endoscopic mucosal resection (EMR) and endoscopic submucosal dissection (ESD).
(I) Endoscopic diagnosis
Colonoscopy is the gold standard for diagnosing colorectal lesions, as it allows direct observation of the location, morphology, and size of the lesions. However, due to the unique growth pattern of laterally spreading tumors (LSTs), they are often difficult to detect under conventional white-light endoscopy, particularly for LST-NG lesions, which are relatively flat and harder to identify, leading to a high risk of missed diagnosis. Therefore, for mucosal flat-type lesions detected during routine colonoscopy, additional endoscopic techniques should be used to assist in diagnosis and improve the detection rate of LSTs.
In clinical practice, LSTs are often observed using chromoendoscopy with magnification to examine the morphology of glandular openings. The depth of tumor infiltration is initially assessed based on the Kudo pit pattern classification, which is used to evaluate the lesion’s boundary. Additionally, techniques such as chromoendoscopy and endoscopic ultrasound (EUS) have proven valuable in the diagnosis of LSTs, and these methods should be used in combination to observe and assess the lesions.
(II) Technical methods and previous terminology of LST super minimally invasive resection
1. Endoscopic mucosal resection (EMR). EMR refers to the removal of a mucosal lesion in whole or in pieces via endoscopy. It is a commonly used resection method in clinical practice, with advantages including simplicity, short operation time, and few complications. Current international guidelines recommend EMR for treating colorectal lesions larger than 10 mm that are sessile or flat. However, when the lesion exceeds 20 mm in size, the rate of complete resection significantly decreases. Studies show that compared to LSTs with diameters ≤ 20 mm, those with diameters > 20 mm have lower rates of en bloc and complete resection, and longer operation times. Therefore, for LST lesions smaller than 20 mm, EMR is considered the first choice for endoscopic resection. For larger, flat lesions ≥20 mm, endoscopic piecemeal mucosal resection (EPMR) can be an alternative treatment approach, which involves dividing the lesion into sections and removing them in multiple steps.
2. Endoscopic submucosal dissection (ESD). Compared to EMR, ESD achieves higher rates of en bloc and complete resection, with lower recurrence rates after resection, and provides complete specimens for pathological diagnosis, which is crucial for more accurate evaluation of cancerous cells at the resection margins. However, factors such as the thinness of the colorectal wall, some segments being relatively mobile, and limited space within the intestinal lumen make ESD technically challenging for colorectal lesions. Factors that increase the difficulty of colorectal ESD include lesions ≥30 mm in size, lesions involving ≥2/3 of the circumferential wall, non-granular LST lesions, lesions located in the cecum, bends (e.g., hepatic flexure, splenic flexure, rectosigmoid junction), or at the dentate line, all of which contribute to significantly prolonged operation times. Prolonged surgery increases the risk of complications and reduces resection efficiency. Additionally, non-granular LST lesions, particularly the pseudo-depressed type, are associated with high rates of submucosal infiltration, multifocal infiltration, and deeper infiltration, making them more prone to submucosal fibrosis. Careful attention to operating protocols is essential, and en bloc resection should be performed whenever possible for these types of lesions.
3. Precutting endoscopic mucosal resection (precutting-EMR). Precutting-EMR involves making a circumferential incision around the lesion’s mucosal border before performing EMR. The advantage of this approach is that it allows en bloc resection of the lesion without the need for submucosal dissection, which reduces technical difficulty and operation time compared to ESD. For LST lesions between 20–30 mm in size, Precutting-EMR can be an effective alternative treatment option when ESD is technically difficult.
4. Endoscopic submucosal tunnel dissection (ESTD). ESTD involves injecting fluid into the submucosa around the lesion, then making a circumferential incision on both the oral and anal sides of the lesion. The submucosal layer is then accessed from the anal side, and dissection is performed from the anal to the oral side, establishing a submucosal tunnel. The entire lesion is then dissected along both sides of the mucosa until the lesion is completely separated. Compared to conventional ESD, ESTD offers several advantages: (1) it provides better surgical visibility by creating a tunnel, making it easier to identify blood vessels and muscle layers, which reduces intraoperative complications; (2) the tension on the lesion is maintained, preventing it from retracting and allowing for a clearer view and increased dissection space, thus reducing technical difficulty and improving efficiency; (3) the liquid cushion maintained after submucosal injection reduces the need for repeated injections, shortening operation time; (4) the use of a transparent cap on the endoscope’s tip helps in bluntly separating the submucosal layer, speeding up the dissection process.
LSTs are most commonly found in the rectum. Because the rectal lumen is straight and lacks colonic valves, it is theoretically possible to establish an effective submucosal tunnel. In the treatment of rectal LSTs, studies have shown that compared to traditional ESD, ESTD offers faster dissection speeds and significantly reduced operation times. In treating large colorectal LSTs (with the shortest lesion diameter > 4 cm), ESTD also achieves high rates of en bloc resection, complete resection, and curative resection. Some scholars suggest that for larger lesions located in the rectum or near the rectosigmoid junction, ESTD should be considered the first choice. However, for lesions outside these locations, using tunnel technology is more difficult, and ESTD is not recommended as the first choice. Additionally, ESTD may result in larger or even circumferential resection sites, with postoperative stenosis being a significant concern. Studies have shown that over 80% of surrounding mucosal defects are risk factors for postoperative stenosis. To date, the indications for ESTD in treating colorectal LSTs remain unclear and require validation through multi-center, large-sample studies to confirm its efficacy and safety.
5. Endoscopic full-thickness resection (EFTR). EFTR is an endoscopic technique based on ESD that targets full-thickness resection of the gastrointestinal wall. It enables the acquisition of complete specimens for pathological evaluation and provides an alternative surgical option for complex lesions that cannot be resected by conventional endoscopic methods, such as those with severe submucosal fibrosis, lesions involving anatomical areas like the appendix or diverticula, recurrent lesions at postoperative anastomoses, or those with negative lifting signs. A recent large-sample prospective study evaluated the effectiveness of EFTR in treating complex colorectal lesions, showing a success rate of 83.9%, a complete resection rate of 82.4%, and a full-thickness resection rate of 83.2%. The overall complication rate was 9.3%, including bleeding, perforation, and appendicitis, with an emergency surgery rate of 2.7%. Additionally, EFTR has been successfully used to resect LSTs around the appendix and those that have recurred after ESD.
However, in clinical practice, EFTR is still in its early stages and has not yet fully matured. Its efficacy and safety require more extended observation and a greater number of cases. Currently, EFTR faces limitations, including difficulties in infection prevention, poor surgical visibility, the lack of specialized surgical platforms, complex instrumentation, and inconsistent quality control standards. Full-thickness resection may also lead to defects in the colorectal wall, and timely and effective repair of these defects during surgery is crucial to the success of the treatment. Although numerous Chinese and international suturing devices and techniques are available, most are still in development or are clinically applied but do not yet match the precision of laparoscopic or open surgery.
13.3.2 Endoscopic Super Minimally Invasive Resection of Laterally Spreading Tumors (LST) in the Rectum
I. Piecemeal endoscopic mucosal resection (EPMR)
Endoscopic super minimally invasive resection of laterally spreading tumors (LST) in the rectum can be achieved through endoscopic piecemeal mucosal resection (EPMR). EPMR refers to the technique of removing a lesion that cannot be resected in a single piece under endoscopy. The lesion’s boundaries are identified, marked, and then, after submucosal injection, the lesion is sufficiently elevated. It is resected piece by piece starting from one side using a snare, and the excised lesion is retrieved, followed by the management of the wound. This technique is developed from traditional endoscopic mucosal resection (EMR), but due to limitations with EMR snare devices, EPMR involves resecting the lesion in multiple stages.
(I) Indications and contraindications
1. Indications. Traditional EMR is suitable for resecting lesions of the digestive tract that are less than 2 cm in diameter, involving the mucosal layer and part of the submucosal or muscularis mucosa layer. EPMR has a broader range of indications compared to EMR, but other conditions are largely the same. This includes lesions confined to the mucosal epithelial layer (M1), the mucosal propria layer (M2), the muscularis mucosa layer (M3), or the upper third of the submucosal layer (SM1).
2. Contraindications. EPMR should be avoided in patients with severe heart or lung diseases, blood disorders, coagulation dysfunctions, or those who are taking anticoagulant medications. These conditions pose a higher risk for complications, including bleeding, and may interfere with the patient’s ability to safely undergo the procedure.
(II) Preoperative preparation
1. Surgical instruments: Gastrointestinal endoscope, transparent cap, snare, high-frequency electrosurgical generator, argon beam coagulator, injection needle, hemostatic clips, etc.
2. Patient preparation: A preoperative bowel cleansing must be carried out, and the patient must refrain from eating and drinking for 8 h on the day of surgery.
(III) Surgical operation and techniques
1. Determination of the extent of the lesion: Lugol’s solution or 0.4% indigo carmine solution must be used for staining so as to identify the lesion’s boundaries.
2. Marking: multiple point markings must be made along the lesion’s margin with APC (Argon Plasma Coagulation) within 3–5 mm.
3. Submucosal injection: a methylene blue solution must be injected submucosally at multiple points around the lesion to raise the lesion uniformly and completely.
4. Segmental resection: multiple resections must be performed along one side of the lesion until it is completely excised.
5. Specimen handling: all excised specimens must be retrieved, fixed in sequence, and the lesion’s full appearance restored for pathological examination.
(IV) Postoperative complications and management
1. Bleeding: bleeding is the most common complication of EPMR (endoscopic piecemeal resection), typically occurring during the procedure or within 24 h postoperatively. Intraoperative bleeding can be controlled using electrosurgical coagulation, hemostatic clips, or metal clips. For minor postoperative bleeding, close observation is required, and non-surgical treatment may be sufficient for spontaneous cessation. If necessary, endoscopic hemostasis can be performed. In cases of major bleeding where endoscopic hemostasis is not feasible, surgical or interventional hemostasis may be required.
2. Perforation: small perforations can be closed using titanium clips, with strict fasting and observation. Alternatively, a combination of titanium clips and nylon rings can be used for endoscopic perforation closure. If endoscopic treatment fails, the patient must be transferred for surgical intervention.
3. Postoperative stricture: stricture is commonly seen when the esophageal lesion exceeds 2/3 of the circumference after EPMR. Preventive treatment with intraoperative stent placement or postoperative balloon dilation can be employed to reduce the risk of stricture.
4. Infection: in patients with large lesions or prolonged surgery times, prophylactic antibiotic use may be considered to prevent infection.
5. Residual lesions and recurrence: regular endoscopic follow-up at three, six, and twelve months postoperatively is recommended. If residual lesions or recurrence are detected, early-stage lesions may be treated with endoscopic EMR, EPMR, or ESD. For advanced-stage lesions, surgical resection or adjuvant therapies such as radiotherapy or chemotherapy may be required.
(V) Hot-spot issues and research progress in the consensus
1. LST lateral growth characteristics and mechanism of progression to colorectal cancer. The specific mechanism by which LST progresses to colorectal cancer remains unclear. Current studies on the pathogenesis of LST have identified several potential factors, including:
GSK-3β: as an important negative regulator in the Wnt signaling pathway, the increased expression of phosphorylated GSK-3β may be one of the potential factors for LST carcinogenesis;
C-myc: the expression level of C-myc, a key downstream target gene in the Wnt pathway, is significantly increased in LST. C-myc can activate the telomerase of cells to induce its transcription, thereby enabling cellular ‘immortality’ and promoting neovascularization within the tumor;
BCL2-L1: an apoptosis-related gene responsible for lateral growth, whose overexpression leads to the horizontal spread of LST;
αPCKλ/ι: a critical regulator of cell polarity, associated with tumor pathogenesis and progression. The high expression of αPCKλ/ι may be related to the lateral spread of LST.
2. Treatment options and prognosis for LST. Endoscopic resection remains the preferred treatment for LST, with most patients having a favorable prognosis and low incidences of perforation, bleeding, and recurrence. Recurrence is associated with factors such as lymph node metastasis, submucosal invasion depth, histological grade, incomplete resection, and tumor size ≥40 mm. The probability of submucosal invasion exceeding 1000 μm is significantly higher in LST-G-M compared to LST-NG-FE and LST-NG-PD. The lymph node metastasis rate is higher in LST-NG-FE than in other subtypes. Studies have shown that for low-risk lesions (negative vertical margins, well or moderately differentiated adenocarcinoma, no lymphovascular invasion, invasion depth <1000 μm), the recurrence rate after endoscopic resection is 0.8%. For high-risk lesions (absence of low-risk pathological features), the recurrence rate is 3.6%. Therefore, LST subtypes with higher recurrence rates, especially LST-NG-PD and LST-G-M, require closer follow-up monitoring.
LST lesions are often large, and more than 90% of colorectal ESD (endoscopic submucosal dissection) procedures result in circumferential mucosal defects, which may lead to strictures. However, the incidence and severity of strictures are generally lower compared to ESD in the esophagus, cardia, and pylorus. This may be due to the larger lumen of the intestinal tract and the “self-expanding” effect of solid feces. Most strictures after colorectal ESD are asymptomatic, and local steroid injection during the procedure can effectively prevent them. Symptomatic patients can usually be successfully treated with endoscopic balloon dilation.
In certain special cases, additional surgical intervention and lymph node dissection may be required after endoscopic treatment of LST, such as when the following are observed in the histopathological evaluation of the resected specimen: submucosal invasion depth ≥ 1000 μm, positive vascular invasion, poorly differentiated adenocarcinoma, signet ring cell carcinoma, or mucinous carcinoma.
3. EMPR in the treatment of LST. In clinical practice, when using EMPR (endoscopic mucosal resection) to remove nodular mixed-type LST, the primary lesion (such as a nodule ≥1 cm) should be completely removed first, followed by resection of the remaining lesions, while controlling the number of pieces resected. However, EMPR has several drawbacks, including a low complete resection rate, difficulties in ex vivo reconstruction of fragmented tissue specimens, inability to provide a complete specimen for postoperative pathology, and challenges in evaluating lesion invasion depth and resection margins, which lead to a high risk of local residual and recurrence. Close follow-up is required. Additionally, the procedure can cause vascular injury, leading to intraoperative bleeding or delayed bleeding. Recurrence of LST after EMPR is associated with factors such as the number of excised lesion pieces, lesion diameter ≥ 40 mm, intraoperative bleeding, and highly atypical hyperplasia.
II. Precutting endoscopic mucosal resection (pre-cut-EMR).
Lateral spreading tumors (LSTs) of the colon and rectum can be effectively removed through peranal minimally invasive resection via precutting endoscopic mucosal resection (pre-cut-EMR). Pre-cut-EMR is an endoscopic technique in which the tumor boundaries are identified, marked, and the submucosal injection is performed to fully elevate the lesion. The surrounding mucosal layer is then circumferentially incised with the tip of an electrosurgical knife or snare, without separating the submucosa. The lesion is directly captured using a snare, and complete resection is performed along with retrieval of the lesion, followed by wound management. As a novel endoscopic technique for treating gastrointestinal LSTs, pre-cut-EMR is derived from traditional EMR, and its primary advantage lies in reducing the difficulty of lesion dissection, thereby lowering the risks of perforation and secondary infections.
(I) Surgical procedure and techniques
1. The size and extent of the lesion must be carefully observed, and, if necessary, indigo carmine dye should be applied for better visualization.
2. A submucosal injection must be performed 0.5 cm away from the lesion, starting from the oral side, followed by the lateral margin, and finally the anal side. The volume of submucosal solution injected at each point should be determined based on the degree of lesion elevation.
3. The lesion’s margin must be incised using an electrosurgical knife, carefully controlling the cutting depth to avoid injury to the muscle layer.
4. After having completely incised the lesion’s margin, the lesion must captured with an appropriate snare. The view must be adjusted so as to ensure that the lesion is fully enclosed by the snare. Once secured, the snare must be tighten and the lesion’s tissue excised. For lesions larger than 3.0 cm in diameter, where the snare cannot enclose the lesion in one attempt, further dissection from the margin toward the center of the lesion may be necessary to reduce the lesion size, thereby completing the pre-cut-EMR.
5. The site of the wound must be flushed with a saline solution under endoscopic guidance to check for any residual lesion. In case of perforation, smaller perforations can be closed with a metal clip. For larger perforations, if endoscopic suturing is difficult, surgical laparoscopic repair may be required. Thermal coagulation forceps or APC should be used to cauterize the wound to prevent delayed postoperative bleeding.
6. Fix the lesion’s margin with large-head pins, observe whether the resection margin is complete, measure and document the lesion size, and place the specimen in formalin solution for pathological examination.
Refer to video 13.9, Super minimally invasive non-full-thickness resection for rectal tumors (See online resources).
(II) Hot-spot issues and research progress in the consensus
1. Indications for precutting endoscopic mucosal resection (precutting-EMR). For flat colorectal lesions measuring 20–30 mm, no significant differences were found between the Precutting-EMR group and the endoscopic submucosal dissection (ESD) group in terms of complete resection rate and en bloc resection rate. Postoperative complications and recurrence rates were also not significantly different, and no local recurrence was observed. However, the surgical time in the Precutting-EMR group was significantly shorter than that in the ESD group. Studies on colorectal laterally spreading tumors (LSTs) show that, compared to EMR, Precutting-EMR can effectively remove lesions and improve the en bloc resection rate for larger lesions (21–30 mm). However, since the maximum inner diameter of standard snares is approximately 30 mm, Precutting-EMR should be used for lesions smaller than 30 mm.
2. Advantages of precutting endoscopic mucosal resection (EMR) Although colorectal LSTs have larger lesion areas, literature reports indicate that the incidence of submucosal invasion in these lesions is lower than that in similarly sized colorectal polypoid lesions, making them suitable for endoscopic resection. EMR works by lifting the lesion with submucosal injection and then using a snare to excise the lesion. This procedure is simple, with short surgical time and low complication rates. However, for lesions larger than 20 mm in diameter, en bloc resection is not possible; the lesion must be resected in pieces, making it difficult to obtain accurate pathological assessment postoperatively and increasing the risk of residual lesions and recurrence. EMR-P (precutting EMR) is an improved version of EMR. It first involves making a circumferential mucosal incision along the lesion’s margin, followed by lifting the lesion with submucosal injection and using a snare for resection.
Currently, EMR-P is widely applied in various parts of the gastrointestinal tract. Compared with traditional EMR, it has several advantages: ① It allows for en bloc resection of larger lesions. Literature reports indicate that, compared to traditional EMR, EMR-P significantly improves the en bloc resection rate for LSTs ≤30 mm in diameter. An animal study also confirmed that EMR-P can achieve en bloc resection of flat lesions with a diameter of 40 mm.② It provides high-quality pathological specimens. The circumferential mucosal incision performed in EMR-P ensures accurate lesion margin evaluation. ③ Animal experiments have shown that the submucosal resection depth of specimens obtained by EMR-P is greater than that from traditional EMR, with most extending beyond the SM1 layer, which is beneficial for assessing whether the lesion has been completely resected. ④ For LSTs, which are flat lesions, the difficulty in snaring the lesion is high. The pre-incised circumferential mucosa creates a ‘slot-like’ structure, facilitating lesion snaring. In this study, among eight cases of lesions with diameters between 21 and 30 mm in the EMR-P group, 5 (62.5%) achieved en bloc resection, indicating that EMR-P improves the en bloc resection rate for larger lesions compared to traditional EMR.
13.4 Appendiceal Diseases – Super Minimally Invasive Surgery
13.4.1 Acute Appendicitis – Super Minimally Invasive Drainage via the Anus
(I) Overview
Acute appendicitis (AA) is one of the most common acute abdominal conditions in clinical practice. In Western developed countries, the incidence of AA is 9–10/10 000 people per year, with a higher prevalence in young adults. The lifetime incidence of AA is 7%–8%, with 8.6% in males and 6.9% in females. The main causes of AA are obstruction and infection of the appendiceal lumen, although genetic, racial, environmental, and microbial factors also contribute. Surgical appendectomy is the conventional treatment for AA, but increasing evidence suggests that the appendix is not a redundant organ. The appendix is a lymphoid organ that secretes intestinal hormones and plays a role in regulating immune function. It also plays a significant role in maintaining the homeostasis of the gut microbiota, acting as a regulator and storage organ. Furthermore, studies have shown that surgical appendectomy may increase the risk of Crohn’s disease, gut dysbiosis, and colorectal cancer. In recent years, endoscopic treatment of AA has provided patients with the opportunity to preserve the appendix and its normal physiological function. This approach allows patients to undergo minimal invasive treatment without skin disruption while directly treating the lesion for optimal therapeutic outcomes.
Acute appendicitis can be treated with super minimally invasive drainage via the anus through endoscopic retrograde appendicitis therapy (ERAT). ERAT involves inserting a catheter into the appendiceal lumen through an endoscope, aspirating pus, removing appendiceal concretions (fecal stones), and irrigating the appendiceal lumen to relieve obstruction and eliminate inflammatory materials within the appendix, thereby achieving the therapeutic goal of treating AA.
(II) Diagnosis
The diagnosis primarily relies on clinical presentation and laboratory test results. Some patients, especially the elderly and women of reproductive age, may have a higher rate of misdiagnosis. Imaging studies are recommended for suspected cases to confirm the diagnosis. The first choice for imaging is abdominal ultrasound, which is safe, convenient, and particularly suitable for suspected patients during pregnancy. Enhanced CT scanning has higher sensitivity and specificity and is appropriate for suspected non-pregnant patients with negative ultrasound findings. Abdominal MRI is not routinely recommended but can be used for suspected pregnant patients with negative ultrasound results. Negative imaging findings do not completely exclude acute appendicitis, and a follow-up examination is recommended if the patient’s condition changes. Colonoscopy can be used both as a diagnostic and therapeutic tool. On endoscopy, the appendiceal orifice may appear swollen, or pus may be seen flowing from the appendiceal lumen, or fecal stones may be observed, all of which can diagnose acute appendicitis. Endoscopic retrograde appendicography findings include: an appendiceal lumen dilated >6 mm, localized narrowing, filling defects, and contrast medium extravasation from the lumen.
(III) Indications and contraindications
1. Indications: ① clinically suspected or diagnosed acute appendicitis (Alvarado score ≥ 5), including simple appendicitis and suppurative appendicitis; ② abdominal ultrasound or CT diagnosis of acute appendicitis or inability to rule out acute appendicitis (appendiceal diameter > 6 mm, combined with appendiceal wall thickening, peri-appendiceal edema and/or fluid accumulation, excluding appendiceal perforation or suspected tumor).
2. Contraindications: ① patients with contraindications to endoscopic examination and treatment; ② patients who cannot undergo endoscopic examination and treatment; ③ patients who cannot undergo bowel preparation for any reason; ④ patients with signs of diffuse peritonitis; ⑤ patients highly suspected of having a gastrointestinal perforation based on clinical and imaging examinations; ⑥ patients suspected of having a cecal or appendiceal tumor.
(IV) Preoperative preparation
1. A routine medical history and physical examination must be completed, as well as routine tests such as blood routine, coagulation profile, electrolytes, liver and kidney function tests, and electrocardiogram.
2. A preoperative anesthesia assessment must be made in order to select the anesthesia method based on the patient’s condition and the hospital’s capabilities.
3. Adequate bowel preparation must be performed according to the expert consensus on bowel preparation for colonoscopy.
(V) Surgical procedure and techniques
1. Anesthesia and positioning. The choice of the method of anesthesia should be made based on the patient’s condition and the hospital’s capabilities. The patient is usually positioned in the left lateral decubitus position, which may be adjusted based on the location of the appendiceal orifice or the surgeon’s preference.
2. Surgical steps. (1) The colonoscope is inserted so as to reach the appendiceal orifice, perform appendiceal cannulation, and contrast imaging. (2) Aspiration of pus, irrigation, stone removal, and stent placement:
insert the colonoscope through the rectum to the cecal area, locating the appendiceal orifice;
using the transparent cap on the tip of the endoscope, push aside the Gerlach valve, and insert the appendiceal catheter with a guidewire;
aspiration of pus from the appendiceal cavity to decompress the cavity;
under X-ray guidance, inject a contrast agent into the appendiceal cavity to assess its shape and identify the presence of appendicoliths or any narrowing of the cavity;
repeatedly irrigate the appendiceal cavity until the irrigation fluid becomes clear;
in cases with significant pus or lumen narrowing, a plastic stent may be placed. A follow-up abdominal X-ray should be performed one week later to assess the stent placement and determine if it should be removed.
(VI) Postoperative management
1) Antibiotics: administration of antibiotics can be delayed until the appendiceal cavity obstruction has been relieved and the inflammatory material has been cleared, provided the patient does not exhibit fever or other systemic signs of infection. If the patient shows significant systemic infection or toxicity, empirical antibiotic therapy should be considered, typically with a third-generation cephalosporin combined with nitroimidazole. Antibiotic use should follow the Clinical Application Guidelines for Antimicrobial Agents.
2) Gradual dietary progression postoperatively: clear liquids → soft foods → regular diet.
3) Postoperative monitoring of the patient’s clinical symptoms, body temperature, and infection indicators such as blood routine and CRP levels.
(VII) Hot-spot issues and research progress in the consensus
The optimal timing for ERAT remains controversial. Khan et al. suggested that ERAT treatment should be initiated on day 1 of illness, but they also pointed out that premature intervention might lead to insufficient preoperative antibiotic administration, potentially affecting outcomes. Similarly, Podda et al. recommended early ERAT to maximize patient benefits. More research is still needed to determine the best timing for ERAT implementation.
13.4.2 Super Minimally Invasive Trans-Rectal Removal of Appendicoliths using Endoscopic Retrograde Appendicitis Therapy (ERAT)
(I) Overview
Super minimally invasive removal of appendicoliths via the trans-rectal approach can be achieved through ERAT. After the colonoscope reaches the appendiceal orifice, an Eyemax choledochoscope and stone retrieval basket are introduced for stone removal, drainage, and other procedures. The use of the choledochoscope in conjunction with the endoscope for retrograde appendicitis treatment is an improvement based on the ERAT principles. This method modifies retrograde appendiceal imaging by allowing direct visualization of the appendiceal cavity through the choledochoscope under colonoscopic guidance. It provides a clear view of fecaliths, purulent fluid, pus plaques, appendiceal wall congestion, edema, as well as the curvature, dilation, and narrowing of the appendiceal lumen. This enhances diagnostic precision, eliminates the radiation exposure risks associated with ERAT for both the physician and the patient, and reduces the risk of contrast agent leakage into the peritoneal cavity, which could cause peritonitis in patients with perforated appendicitis. The method has significant advantages, including shorter surgical and recovery times compared to traditional surgery and ERAT, more precise treatment, higher success rates, and a reduction in complications such as perforation and bleeding.
(II) Indications and contraindications
1. Indications. ① Clinically suspected or confirmed acute appendicitis (Alvarado score ≥ 5), including simple appendicitis and suppurative appendicitis. ② Abdominal ultrasound or CT diagnosing acute appendicitis or unable to exclude acute appendicitis (appendix diameter > 6 mm with thickened appendiceal wall, pericolic edema and/or fluid accumulation, excluding appendiceal perforation or suspected tumors).
2. Contraindications. Acute appendicitis with contraindications for trans-rectal super minimally invasive drainage surgery.
(III) Surgical procedure and techniques (figure 13.14)
(1) The colonoscope is inserted to reach the appendiceal orifice, perform appendiceal cannulation, and contrast imaging.
(2) The choledochoscope is introduced and the presence of appendicoliths is observed under direct visualization. Balloons or baskets may be used to remove the appendicoliths. The choledochoscope, guided by a guidewire, is advanced to the appendiceal orifice where the light source is activated. With careful handling, the choledochoscope is introduced into the appendiceal cavity under direct visualization. Gentle manipulation reduces the occurrence of physician-induced abrasions, tears, or other complications.
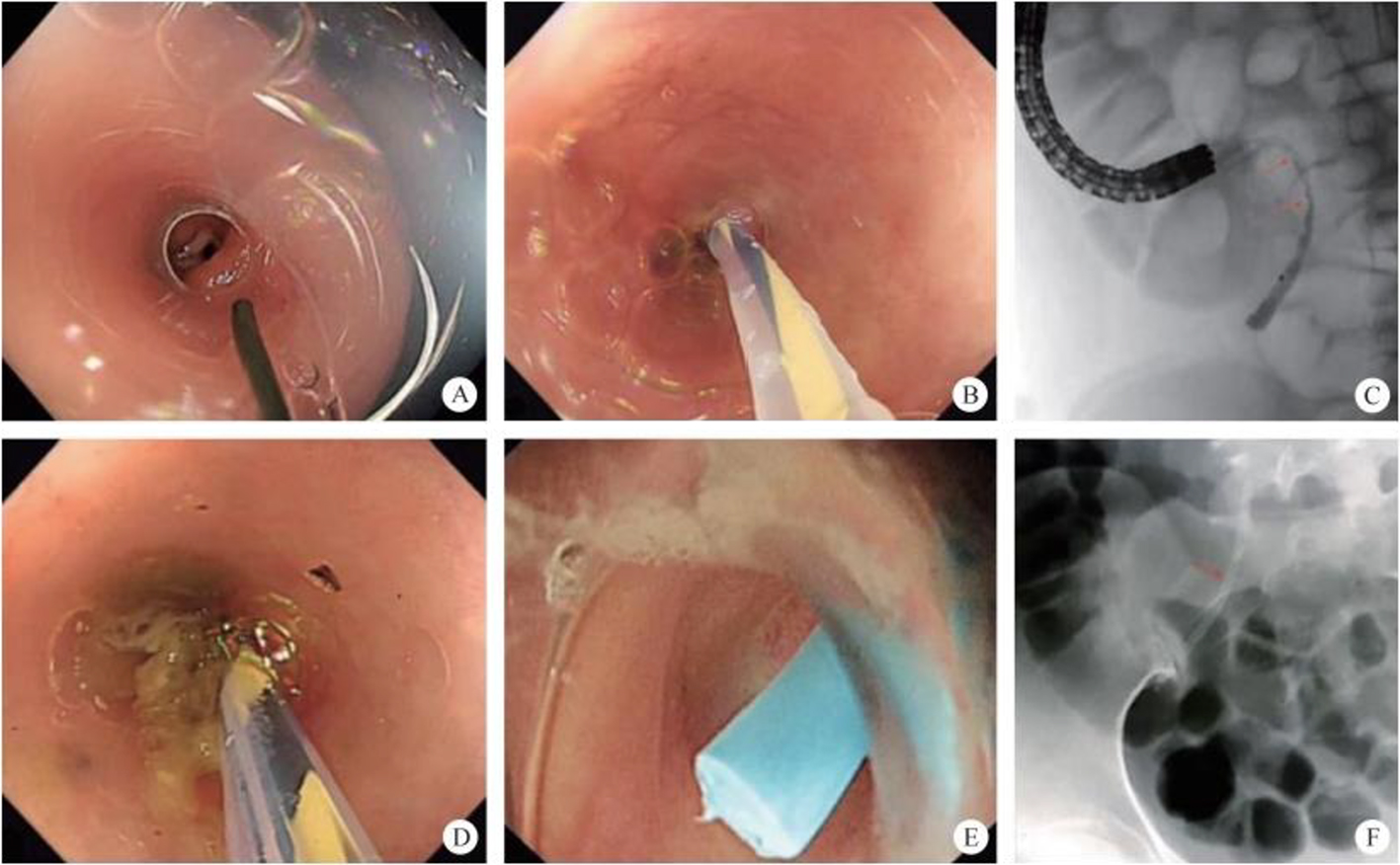

FIG. 13.14 — Operational steps of super minimally invasive removal of appendicoliths via trans-rectal choledochoscope under direct visualization. A. Use of the conical cap to assist in reaching the appendiceal orifice. B. Insertion of the guidewire. C. Appendiceal contrast imaging. D. Visualizing fecaliths under direct choledochoscope guidance. E. Insertion of the drainage tube. F. Repeated contrast imaging to confirm proper positioning of the drainage tube.
(3) Repeatedly irrigate the appendiceal cavity until the irrigation fluid becomes clear. During the antibiotic irrigation procedure with the choledochoscope, suction should be performed simultaneously to prevent excessive pressure in the appendiceal cavity, which could lead to physician-induced perforation.
(4) For patients with significant pus accumulation or lumen narrowing, a plastic stent may be left in place.
(IV) Hot-spot issues and research progress in the consensus
Difficult appendiceal cannulation remains an unresolved challenge. The use of the choledochoscope system in ERAT may provide an effective solution to this problem. The use of peroral choledochoscopes has been effectively validated in ERCP procedures, and some studies have also applied the choledochoscope in ERAT to achieve direct visualization, non-radiographic lavage, drainage of the appendiceal cavity, and stone removal. Just as choledochoscopes are applied in ERCP, the ‘appendiceal choledochoscope’ system could potentially be further utilized in ERAT and may help solve the issue of difficult appendiceal cannulation.
13.4.3 Periappendiceal Abscess – Super Minimally Invasive Trans-Rectal Drainage
(I) Overview
Periappendiceal abscess is one of the common complications of acute appendicitis, typically occurring when acute appendicitis progresses to suppuration, gangrene, or perforation. During this pathological process, the omentum may move to the right lower abdomen, and the omentum and small intestine around the appendix tend to envelop the appendix, leading to adhesions and the formation of an inflammatory mass or periappendiceal abscess. Common symptoms include fever and a palpable mass in the right lower abdomen. The management of periappendiceal abscesses remains a topic of debate,most clinicians advocate for conservative treatment, whereas others recommend primary surgical intervention. Conservative treatment involves a long course and carries the risks of recurrence, abscess rupture leading to acute peritonitis, and other complications. Surgical treatment is often challenging due to adhesions, surrounding tissue edema, and difficulty in excising the lesion, which may result in infection spread, intestinal fistulas, and other complications. With the widespread use of endoscopic techniques, cases of periappendiceal abscess have been increasing in recent years. On colonoscopy, appendicitis is characterized by a deformed and raised appendiceal orifice, irregularity, surrounding mucosal congestion, edema, erosion, purulent discharge, and the presence of protruding mass-like lesions into the intestinal lumen.
Periappendiceal abscess can also be treated through super minimally invasive trans-rectal drainage under endoscopic guidance. The procedure involves mucosal incision at the site where a distinct bulge, fluctuation, or purulent discharge is observed under direct endoscopic visualization. This allows the purulent fluid to drain into the intestinal lumen, thereby achieving the therapeutic goal. This approach is considered a form of super minimally invasive drainage for appendicitis.
(II) Diagnosis
The preferred diagnostic method for periappendiceal abscess is ultrasound, with a diagnostic rate of up to 97%. CT and ultrasound have comparable diagnostic rates for periappendiceal abscesses; however, CT offers clearer identification of the appendix’s morphology and can detect early perforations. CT is also more advantageous in diagnosing ectopic periappendiceal abscesses. For a mass in the right lower abdomen, it is important to consider the possibility of a concurrent colon tumor, intussusception, or Crohn’s disease. Further colonoscopy can assist in diagnosis and serve as an alerting tool. Colonoscopic findings include a deformed, raised, and irregular appendiceal orifice with surrounding mucosal congestion, edema, erosion, purulent discharge, and the presence of mass-like lesions protruding into the intestinal lumen.
(III) Indications and contraindications for super minimally invasive trans-rectal surgery
1. Indications: ① patients diagnosed with periappendiceal abscess or right lower abdomen mass by abdominal ultrasound or CT (Alvarado score ≥ 5); ② patients who may have acute appendicitis or whose diagnosis of acute appendicitis cannot be ruled out.
2. Contraindications: ① contraindications for endoscopic examination and treatment; ② patients unable to undergo endoscopic examination or treatment; ③ patients who cannot undergo bowel preparation for various reasons; ④ patients with signs of diffuse peritonitis; ⑤ patients highly suspected of gastrointestinal perforation based on clinical and imaging findings; ⑥ patients suspected of having cecal or appendiceal tumors.
(IV) Surgical procedure and techniques (figure 13.15)
1. The colonoscope is inserted through the anus to reach the cecum and ileocecal junction, then the appendiceal orifice must be located.
2. The most prominent area or the site with noticeable fluctuation and purulent discharge around the appendiceal orifice is selected. A mucosal incision (window technique) is made with a mucosal incision knife or another instrument, exposing the abscess cavity, allowing the purulent fluid to drain into the intestinal lumen, then the abscess cavity is irrigated with a saline solution.
3. The scope is retracted after ensuring that there is no active bleeding at the incision site.
(V) Postoperative management
1. Postoperatively, antibiotic therapy is generally recommended, using third-generation cephalosporins combined with nitroimidazole drugs. In the event of severe complications, penicillin-β-lactamase inhibitor combinations should be used. If the patient is allergic to these drugs, carbapenems may be considered. Antibiotic use should follow the Clinical Guidelines for the Use of Antimicrobial Drugs.
2. Postoperative fasting should be observed for three days, followed by gradual dietary progression: clear liquids → semi-liquid foods → regular diet.
3. Postoperatively, the patient’s clinical symptoms, body temperature, and infection markers such as blood routine and C-reactive protein (CRP) must be monitored.
(VI) A classic case
The patient, a 40-year-old woman, was admitted due to intermittent abdominal pain and fever for half a month, with exacerbation of symptoms for one day. Upon admission, physical examination revealed tenderness in the middle-upper abdomen and right lower abdomen, with a palpable mass approximately 10 cm × 5 cm in size, tender to palpation, and suspicious rebound tenderness. Laboratory tests upon admission were as follows. White blood cell count 11.04 G/l, with neutrophils at 83.6%, and no other significant abnormalities. Liver and kidney functions, as well as coagulation tests, showed no significant abnormalities. Abdominal ultrasound revealed a heterogeneous mass in the right abdomen, measuring approximately 130 mm × 83 mm × 51 mm, with unclear borders and uneven internal echoes. A portion of the mass exhibited an appendiceal-like echo with a diameter of approximately 22 mm and a wall thickness of 3 mm, of irregular shape. A free anechoic area was observed in the lower part of the abdominal cavity, about 42 mm in depth. Impression: acute suppurative appendicitis with a right abdominal mass, suspected to originate from the intestines. Free fluid in the abdominal cavity. Preliminary diagnosis: acute suppurative appendicitis complicated by the formation of a peri-appendiceal abscess. After sufficient communication with the patient and her family, a colonoscopy was planned, and, based on intraoperative findings, minimally invasive surgery for acute appendicitis and peri-appendiceal abscess drainage was considered. Endoscopic findings: swelling of the mucosa at the cecum, ileocecal valve, and appendiceal orifice, with a mucosal elevation seen at the terminal ileum, showing ulceration at the apex, accompanied by purulent fluid. After discussion with the patient, an appendiceal cavity lavage and drainage were performed, along with an incision and drainage of the mucosal elevation in the peri-appendiceal abscess. A large amount of white purulent fluid was noted to drain from the appendiceal cavity, which was irrigated until clear. Intraoperative ultrasound confirmed no fecaliths were retained in the appendiceal cavity. After incision of the terminal ileum mucosal elevation, a large amount of purulent fluid drained, and the elevation significantly decreased in size. No bleeding was observed, and the endoscope was withdrawn. Postoperative management included fasting, anti-infective therapy, and fluid resuscitation, and the patient was discharged after stabilization. Three months postoperatively, ultrasound showed an appendiceal-like echo with a diameter of approximately 11 mm and a wall thickness of 2.1 mm. Six months postoperatively, a follow-up colonoscopy revealed no significant abnormalities in the cecum or terminal ileum (figure 13.16).

FIG. 13.15 — Operational steps of super minimally invasive trans-rectal drainage of a periappendiceal abscess. A. Large prominence at the cecum–ileum junction. B. Purulent discharge observed at the top of the prominence. C. Yellow-white purulent fluid is visible after the top is incised.
(VII) Hot-spot issues in the consensus
1. Indications for super minimally invasive surgery via the rectum and expanding indications
Advancements in technology and the accumulation of clinical experience, have led to an expanded range of indications for super minimally invasive surgery, now encompassing chronic appendicitis, appendicitis complicated by perforation, appendiceal cysts, and more. Studies have shown that surgery remains the best option for patients with complicated appendicitis; however, super minimally invasive surgery offers patients the opportunity to preserve the appendix and provides a minimally invasive method that reduces disturbance to other organs, leading to faster recovery. Concurrently, further research is necessary to confirm the safety and efficacy of ERAT in these patient populations.
2. Choice of technique for super minimally invasive surgery via the rectum
Studies have indicated that 1%–13% of patients with acute appendicitis have developed peri-appendiceal abscesses by the time they present for treatment. The mechanism is thought to involve acute appendicitis, where fecaliths obstruct the appendiceal lumen, leading to inflammation. This results in inflammatory exudates in the appendiceal wall, which are encapsulated by the omentum, forming adhesions that limit the spread of infection. Therefore, for patients with acute appendicitis complicated by peri-appendiceal abscess, treating the appendicitis is key to addressing the underlying issue. As shown in the special case above, when performing super minimally invasive surgery for peri-appendiceal abscess, super minimally invasive drainage of the appendiceal cavity is also required.
Similar to ERCP, super minimally invasive surgery via the rectum also faces challenges with catheter insertion into the appendiceal cavity. The difficulties in catheter insertion are varied and include issues like appendiceal orifice abscesses, appendiceal polyps, or fecalith impaction. Therefore, depending on the specific case, Super minimally invasive drainage, appendectomy, or stone removal may be considered.
Acute appendicitis is one of the most common acute conditions, with patients presenting in hospitals of all levels. The goal of super minimally invasive drainage surgery for appendicitis is to maintain patency of the appendiceal cavity and to remove or reduce inflammatory material within the cavity. Due to regional differences and varying levels of hospital capabilities, super minimally invasive surgery for acute appendicitis is also influenced by the available equipment and expertise. For simple acute appendicitis, where preoperative examinations show no fecaliths or narrowing of the appendiceal lumen, a simple lavage of the appendiceal cavity to remove purulent material can be performed. In hospitals without X-ray equipment, intraoperative ultrasound monitoring can be employed to ensure the safety and effectiveness of the procedure.
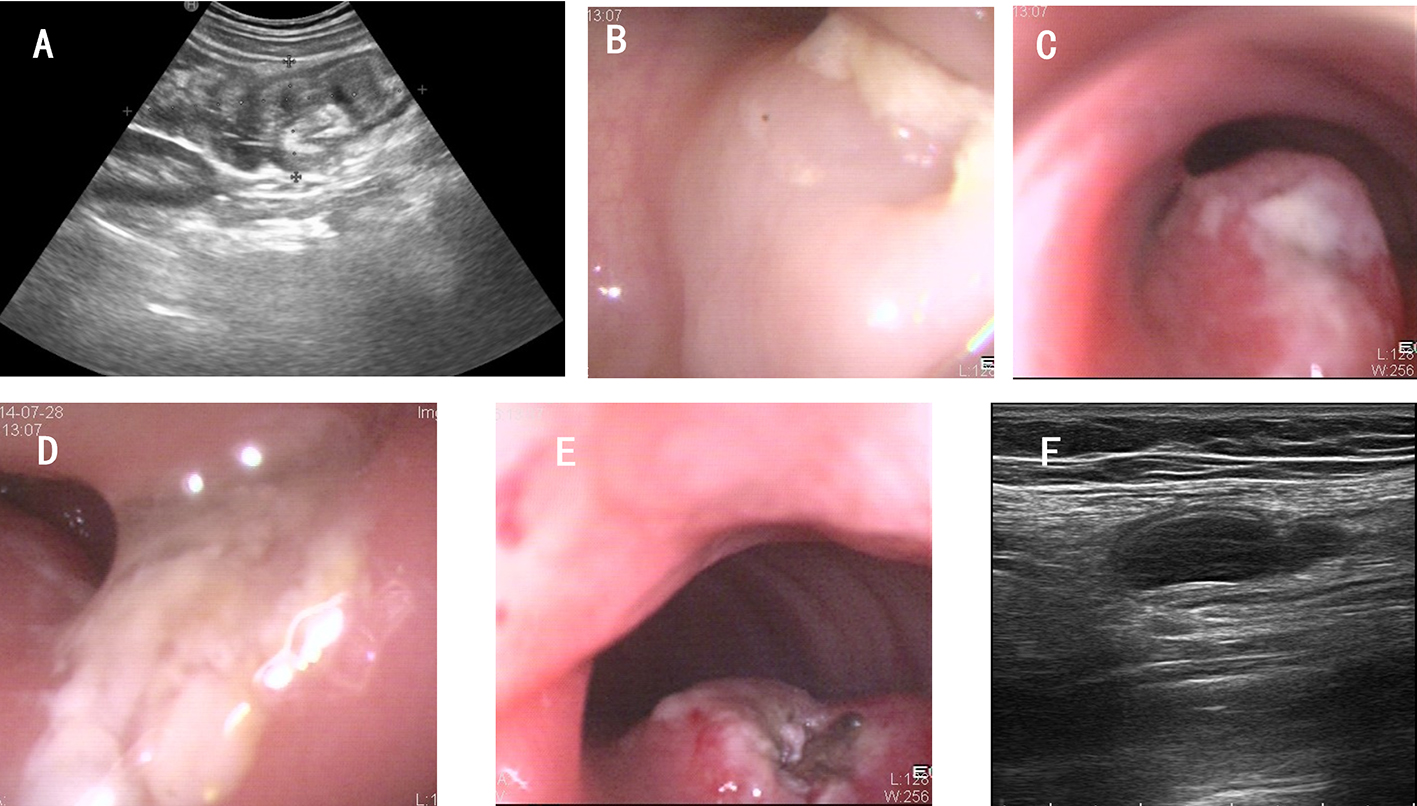

FIG. 13.16 — Postoperative imaging follow-up of super minimally invasive drainage surgery for appendiceal abscess. A. Preoperative ultrasound: heterogeneous mass in the right abdomen. B. Colonoscopy: purulent secretion at the appendiceal orifice, with mucosal swelling in the cecum and ileocecal valve. C. Terminal ileum: mucosal elevation with ulceration at the apex, and purulent material attached. D. Super minimally invasive drainage of the appendiceal cavity: a large amount of purulent secretion drained. E. Incision of the mucosal elevation at the terminal ileum: Purulent fluid flowed out, and the elevation significantly reduced in size. F. Three-month postoperative ultrasound: appendiceal diameter of approximately 11 mm, wall thickness 2.1 mm.
References
[1] Dekker E., Tanis P. J., Vleugels J. L. A., Kasi P. M., Wallace M. B. (2019 Oct 19) Colorectal cancer, Lancet 394(10207), 1467–1480.
[2] Zheng R., Chen R., Han B., et al. (2024) Cancer incidence and mortality in China, 2022, Chin. J. Oncol. 46(03), 221–231.
[3] Watanabe T., Itabashi M., Shimada Y., et al. (2012) Japanese Society for Cancer of the Colon and Rectum (JSCCR) guidelines 2010 for the treatment of colorectal cancer, Int. J. Clin. Oncol. 17(1), 1–29.
[4] Colorectal Group of the Chinese Society of Digestive Endoscopy, Chinese Medical Association. (2023) Expert consensus on endoscopic diagnosis and treatment for colorectal cancer and precancerous lesions in China (2023, Guangzhou), Chin. J. Dig. Endosc. 40(7), 505–520.
[5] Gastrointestinal Pathology Group of the Chinese Society of Pathology, Chinese Medical Association,Pathology Technology Expert Group of the Medical Technicians Committee, Chinese Medical Doctor Association,Standardization Department of the Pathology Equipment Branch, China Association of Medical Equipment (CAME), et al. (2023) Expert consensus on routine preparation of endoscopic submucosal dissection/endoscopic mucosal resection specimens, Chin. J. Pathol. 52(10), 989–994.
[6] Digestive Endoscopy Terminology Examination Subcommittee. (2021) Terminology of digestive endoscopy. Science Press, Beijing.
[7] Tanaka S., Kashida H., Saito Y., et al. (2020 Jan) Japan gastroenterological endoscopy society guidelines for colorectal endoscopic submucosal dissection/endoscopic mucosal resection, Dig Endosc. 32(2), 219–239.
[8] National Clinical Research Center for Digestive Diseases (Shanghai), Chinese Society of Digestive Endoscopy (CSDE), Professional Committee of Tumor Endoscopy of Chinese Anti-Cancer Association, et al. (2022) Expert consensus on management strategies for precancerous lesions and conditions of colorectal cancer in China, Chin. J. Dig. Endosc. 39(01), 1–18.
[9] Ohata K., Kobayashi N., Sakai E., et al. (2022 Nov) Long-term Outcomes after endoscopic submucosal dissection for large colorectal epithelial neoplasms: a prospective, multicenter, cohort trial from Japan, Gastroenterology 163(5), 1423–1434.e2.
[10] Imai K., Hotta K., Yamaguchi Y., et al. (2017 Apr) Endoscopic submucosal dissection for large colorectal neoplasms, Dig Endosc. 29(Suppl 2), 53–57.
[11] Nagata M. (2022 Jan 7) Advances in traction methods for endoscopic submucosal dissection: what is the best traction method and traction direction?, World J. Gastroenterol. 28(1), 1–22.
[12] Cai S., Zhong Y., Zhou P., et al. (2014) Application of dental floss traction-assisted technique in endoscopic submucosal dissection for rectal tumors, Chin. J. Gastrointest. Surg. (6), 612–613.
[13] Liu G., Rong L., Guo X., et al. (2021) Application of rubber band and clip facilitated endoscopic submucosal dissection for colorectal neoplasms (with video), Chin. J. Dig. Endosc. 38(7), 545–550.
[14] Li L., Chai Y., Lv Y., et al. (2021) Magnetic anchor guided endoscopic submucosal dissection for ileocecal laterally spreading tumor: a case report, Chin. J. Dig. Endosc. 38(1), 71–72.
[15] Libânio D., Pimentel-Nunes P., Bastiaansen B., et al. (2023 Apr) Endoscopic submucosal dissection techniques and technology: European Society of Gastrointestinal Endoscopy (ESGE) technical review, Endoscopy 55(4), 361–389.
[16] Li J., Ren M., Yan X., et al. (2023) Clinical application of magnetic anchor-guided endoscopic submucosal dissection, Chin. J. Dig. Endosc. 40(10), 788–792.
[17] Wang Y., Li J., Sun Y., et al. (2023) Application value of a new type of lifting clip-assisted traction in endoscopic submucosal dissection for early colorectal cancer and its precancerous lesions, Chin. J. Dig. Endosc. 40(10), 793–797.
[18] ASGE Technology Committee., Aslanian H. R., Sethi A., Bhutani M. S., et al. (2019 Jun 29) ASGE guideline for endoscopic full-thickness resection and submucosal tunnel endoscopic resection, VideoGIE 4(8), 343–350.
[19] Li X., Zhang W., Gao F., et al. (2023 Dec) A modified endoscopic full-thickness resection for gastrointestinal stromal tumors: a new closure technique based on the instruction of super minimally invasive surgery, Endoscopy 55(S 01), E561–E562.
[20] Mun E. J., Wagh M. S. (2023) Recent advances and current challenges in endoscopic resection with the full-thickness resection device, World J. Gastroenterol. 29, 4009–4020.
[21] Schurr M. O., Baur F. E., Krautwald M., et al. (2015) Endoscopic full-thickness resection and clip defect closure in the colon with the new FTRD system: experimental study, Surg. Endosc. 29, 2434–2441.
[22] Aepli P., Criblez D., Baumeler S., et al. (2018) Endoscopic full thickness resection (EFTR) of colorectal neoplasms with the Full Thickness Resection Device (FTRD): Clinical experience from two tertiary referral centers in Switzerland, United European Gastroenterol. J. 6, 463–470.
[23] Wu Z. W., Ding C. H., Song Y. D., et al. (2022) Colon sparing endoscopic full-thickness resection for advanced colorectal lesions: is it time for global adoption?, Front Oncol. 12, 967100.
[24] Menahem B., Alves A., Morello R., et al. (2017) Should the rectal defect be closed following transanal local excision of rectal tumors? a systematic review and meta-analysis, Tech. Coloproctol. 21, 929–936.
[25] Nomura T., Sugimoto S., Oyamada J., et al. (2023) Application of a new loop cutter for nylon lines and the reopenable clip-over-line method for large defect closure after duodenal endoscopic submucosal dissection, Endoscopy 55, E503–E504.
[26] Liu X., Dou L., Liu Y., et al. (2017) Evaluation of a new closure technique of large defects after endoscopic full-thickness resection, Zhonghua Wei Chang Wai Ke Za Zhi 20, 775–781.
[27] Hernandez A., Marya N. B., Sawas T., et al. (2021) Gastrointestinal defect closure using a novel through-the-scope helix tack and suture device compared to endoscopic clips in a survival porcine model (with video), Endosc. Int. Open 9, E572–E577.
[28] You Y. N., Hardiman K. M., Bafford A., et al. (2020) The american society of colon and rectal surgeons clinical practice guidelines for the management of rectal cancer, Dis. Colon Rectum. 63, 1191–1222.
[29] Zhou W., Yu H. (2018) Submucosal tunneling endoscopic resection for colorectal lesions, China J. Endosc. 24(09), 28–33.
[30] Linghu E. (2011) The creation and prospects of tunneling technology, Chin. J. Laparosc. Surg. (Electron. Ed.) 4(05), 326–327.
[31] Xiong Y., Chai N., Chen Q. et al. (2016) Advances in Endoscopic Submucosal Tunnel Dissection (ESTD) for the Treatment of Giant Lateral Spreading Tumors (LST) in the Colon, Chin. J. Gastrointest. Endosc. (Electron. Ed.). 3(2), 83–84.
[32] Chai N., Xiong Y., Zhai Y. (2017) Expert consensus on digestive endoscopic tunnel technique (2017, Beijing), Chin. J. Gastrointest. Endosc. (Electron. Ed.) 4(04), 145–158.
[33] Zhang Q., Han S., He Y., et al. (2017) Clinical application of tunnel technique on endoscopic submucosal dissection for colorectal laterally spreading tumor, Chin. J. Dig. Endosc. 34(9), 630–634.
[34] Deprez P. H., Moons L. M. G., OʼToole D., et al. (2022 Apr) Endoscopic management of subepithelial lesions including neuroendocrine neoplasms: European Society of Gastrointestinal Endoscopy (ESGE) guideline, Endoscopy 54(4), 412–429.
[35] Surgical Group of the Chinese Society of Digestive Endoscopy, Chinese Medical Association, Natural Orifice Transluminal Endoscopic Surgery (NOTES) Group of the Chinese Society of Digestive Endoscopy, Chinese Medical Association, Digestive Endoscopy Committee of the Endoscopists Branch, Chinese Medical Doctor Association, et al. (2023) Chinese consensus on endoscopic diagnosis and managment of gastrointestinal submucosal tumors(version 2023), Chin. J. Dig. Endosc. 40(4), 253–263.
[36] Chinese Society of Digestive Endoscopy. (2023) Chinese expert consensus on wound pretreatment and antibiotic application in endoscopic super minimally invasive surgery (2023, Beijing) [J/CD], Chin. J. Gastrointest. Endosc. (Electron. Ed.) 10(2), 83–91.
[37] Zhu H., Shi D., Song H., et al. (2020 Sep) Snare-assisted endoscopic resection of gastric subepithelial tumors originating from the muscularis propria layer: a multicenter study, Surg. Endosc. 34(9), 3827–3832.
[38] Geng Y. (2020) Clinical Efficacy of Endoscopic Mucosal Resection (EMR) for Flat Gastrointestinal Mucosal Tumors, Contemp. Med. 26(35), 128–129.
[39] Jacobson B. C., Bhatt A., Greer K. B., et al. (2023 Jan) ACG clinical guideline: diagnosis and management of gastrointestinal subepithelial lesions, Am. J. Gastroenterol. 1, 118(1), 46–58.
[40] Xu H. W., Zhao Q., Yu S. X., et al. (2019) Comparison of different endoscopic resection techniques for submucosal tumors originating from muscularis propria at the esophagogastric junction, BMC Gastroenterol. 19(1), 174.
[41] Okamoto T., Tanaka S., Haruma K., et al. (1996) Clinicopathologic evaluation on colorectal laterally spreading tumor (LST), Nihon Shokakibyo Gakkai Zasshi 93(2), 83–9.
[42] Son D. J., Kweon S. S., Lee J., et al. (2019) Risk factors associated with clinical outcomes of endoscopic mucosal resection for colorectal laterally spreading tumors: A Honam Association for the Study of Intestinal Diseases (HASID) multicenter study, Turk. J. Gastroenterol. 30(4), 350–356.
[43] Li B., Shi Q., Xu E. P., et al. (2021) Prediction of technically difficult endoscopic submucosal dissection for large superficial colorectal tumors: a novel clinical score model, Gastrointest. Endosc. 94(1), 133–144.e3.
[44] Li J., Feng J., Huang X. (2023) Research progress in endoscopic treatment for colorectal laterally spreading tumor, Chin. J. Dig. Endosc. 40(07), 566–570.
[45] Zou J., Chai N., Linghu E., et al. (2021) Efficacy and safety of endoscopic submucosal tunnel dissection for rectal laterally spreading tumors, Surg. Endosc. 35(8), 4356–4362.
[46] Tan Y., Lu J., Lv L., et al. (2020) Current status of endoscopic submucosal tunnel dissection for treatment of superficial gastrointestinal neoplastic lesions, Expert Rev. Gastroenterol. Hepatol. 14(6), 453–462.
[47] Hayashi T., Kudo S. E., Miyachi H., et al. (2017) Management and risk factor of stenosis after endoscopic submucosal dissection for colorectal neoplasms, Gastrointest. Endosc. 86(2), 358–369.
[48] Zwager L. W., Bastiaansen B., Bronzwaer M., et al. (2020) Endoscopic full-thickness resection (eFTR) of colorectal lesions: results from the Dutch colorectal eFTR registry, Endoscopy 52(11), 1014–1023.
[49] Shi L., Wu Y. X., Yu J. H., et al. (2017) Research of the relationship between β-catenin and c-myc-mediated Wnt pathway and laterally spreading tumors occurrence, Eur. Rev. Med. Pharmacol. Sci. 21(2), 252–257.
[50] Minemura S., Tanaka T., Arai M., et al. (2015) Gene expression profiling of laterally spreading tumors, BMC Gastroenterol. 15, 64.
[51] Ichikawa Y., Nagashima Y., Morioka K., et al. (2014) Colorectal laterally spreading tumors show characteristic expression of cell polarity factors, including atypical protein kinase C λ/ι, E-cadherin, β-catenin and basement membrane component, Oncol. Lett. 8(3), 977–984.
[52] Guo X., Cong C., Wang L., Wang H. (2021) Research Progress in Diagnosis and Treatment of Colorectal Laterally Spreading Tumors (LSTs), Mod. Dig. Intervention 26(4), 517–519, 523.
[53] Khashab M. A., Sharaiha R. Z., Saxena P., et al. (2013) Novel technique of auto-tunneling during peroral endoscopic myotomy (with video), Gastrointest. Endosc. 77(1), 119–22.
[54] Ohara Y., Toyonaga T., Tanaka S., et al. (2016) Risk of stricture after endoscopic submucosal dissection for large rectal neoplasms, Endoscopy 48(1), 62–70.
[55] Hashiguchi Y., Muro K., Saito Y., et al. (2020) Japanese Society for Cancer of the Colon and Rectum (JSCCR) guidelines 2019 for the treatment of colorectal cancer, Int. J. Clin. Oncol. 25(1), 1–42.
[56] Ate D. J., Desomer L., Klein A., et al. (2017) Adenoma recurrence after piecemeal colonic EMR is predictable: the Sydney EMR recurrence tool, Gastrointest. Endosc. 85(3), 647–656.e6.
[57] Jin Y., Gong L., Wang X., et al. (2020) Indication analysis of therapeutic effects of pre-cut-endoscopic mucosal resection on colorectal lateral spreading tumors (with video), Chin. J. Dig. Endosc. 37(10), 717–721.
[58] Zou J., Chai N., Zhai Y., et al. (2020) Endoscopic resection for colorectal laterally spreading tumors, Chin. J. Dig. Endosc. 37(03), 169–173.
[59] Wang Z., Yang Y., Li W., Sun g., Peng L., Wang X. (2021) Current status and prospect of endoscopic therapeutic strategy for acute appendicitis, Chin. J. Dig. Endosc. 38(12), 976–s979.
[60] Liu L., Duan S., Meng X., et al. (2021) Meta-analysis of the efficacy comparison of retrograde endoscopic appendicitis in the treatment of acute non-perforated appendicitis, Chin. J. Gastrointest. Endosc. (Electron. Ed.) 8(01), 18–23.
[61] Surgical Branch of the Chinese Medical Association,Committee of Evidence-Based and Translational Medicine for Infectious Diseases, Chinese Research Hospital Association, Editorial Department of Chinese Journal of Surgery. (2021) Expert consensus on multidisciplinary management of intra-abdominal infections, Chin. J. Surg. 59(03), 161–178. DOI: https://doi.org/10.3760/cma.j.cn112139-20201223-00874.
[62] Yang B., Kong L., Ullah S., et al. (2022) Endoscopic retrograde appendicitis therapy versus laparoscopic appendectomy for uncomplicated acute appendicitis, Endoscopy 54(8), 747–754.
[63] Kong L. J., Liu D., Zhang J. Y., et al. (2022) Digital single-operator cholangioscope for endoscopic retrograde appendicitis therapy, Endoscopy 54(4), 396–400.
[64] Shen Z., Sun P., Jiang M., Zhen Z., Liu J., Ye M., Huang W. (2022 Feb 13) Endoscopic retrograde appendicitis therapy versus laparoscopic appendectomy versus open appendectomy for acute appendicitis: a pilot study, BMC Gastroenterol. 22(1), 63. [74] Guiding Principles for the Clinical Application of Antibiotics (2015 Edition) Issued by the National Health and Family Planning Commission of the People’s Republic of China http://www.nhc.gov.cn/ewebeditor/uploadfile/2015/09/20150928170007470.pdf.
[65] Khan S., Ali F. S., Ullah S. (2023 Jan) Endoscopic retrograde appendicitis therapy: is it really a need of the hour?, Ann. Surg. 1, 277(1), e1–e4.
[66] Podda M., Di Saverio S., Agresta F., et al. (2020) Endoscopic retrograde appendicitis therapy: the true proof of the pudding is in the eating, Gastrointest. Endosc. 92, 1278–1279.
[67] Liu B. R., Song J. T., Han F. Y., et al. (2012) Endoscopic retrograde appendicitis therapy:a pilot minimally invasive technique (with videos), Gastrointest. Endosc. 76(4), 862–866.
[68] Feng J., Feng Z., Sun R., et al. (2014) Colonoscopic fenestration and intracavity douching for periappendicular abscess, Chin. J. Gastrointest. Endosc. (Electron. Ed.) 1(1): 23–26.
[69] Pidhorecky I., Cheney R. T., Kraybill W. G. et al. (2000) Gastrointestinal stromal tumors: current diagnosis, biologic behavior, and management, Ann. Surg. Oncol. 7, 705–712. doi: https://doi.org/10.1007/s10434-000-0705-6.
[70] Kim G. H., Park D. Y., Kim S., et al. (2009) Is it possible to differentiate gastric GISTs from gastric leiomyomas by EUS?, World J. Gastroenterol. 15, 3376–3381. doi: https://doi.org/10.3748/wjg.15.3376.
[71] Joensuu H. (2008) Risk stratification of patients diagnosed with gastrointestinal stromal tumor, Hum. Pathol. 39, 1411–1419. doi: https://doi.org/10.1016/j.humpath.2008.06.025.
[72] Nannini M., Rizzo A., Indio V., et al. (2021) Targeted therapy in SDH-deficient GIST, Ther. Adv. Med. Oncol. 13, 17588359211023278. doi: https://doi.org/10.1177/17588359211023278.
[73] Kelly C. M., Gutierrez Sainz L., Chi P. (2021) The management of metastatic GIST: current standard and investigational therapeutics, J. Hematol. Oncol. 14, 2. doi: https://doi.org/10.1186/s13045-020-01026-6.
Chapter 14 Super Minimally Invasive Surgery for Other Systemic Diseases
14.1 Super Minimally Invasive Weight Loss Surgery for Obesity
14.1.1 Overview
Obesity is a chronic disease characterized by abnormal and/or excessive accumulation of body fat, influenced by genetic, behavioral, and environmental factors. It is a major cause of non-communicable chronic diseases, such as cardiovascular diseases and type-2 diabetes. Obesity has become a global epidemic, posing a serious threat to people’s health in both developed and developing countries. Therefore, effective interventions for obesity are urgently needed.
Traditional interventions for obesity primarily include lifestyle modifications, pharmacological treatments, and weight loss surgery (figure 14.1). Lifestyle interventions typically involve exercise, dietary therapy, and behavioral correction. Multiple studies have shown that high-intensity, comprehensive lifestyle programs may lead to up to a 10% reduction in initial body weight within the first four to six months, but the rebound rate is high. Recent studies have shown that pharmacological treatments can achieve better weight loss outcomes, but the rebound rate is also relatively high. Currently, surgical weight loss procedures are the most definitive treatment for weight reduction, but they face challenges such as low patient acceptance, surgical complications, and the risk of postoperative nutritional deficiencies. Endoscopic super minimally invasive weight loss surgery, as an emerging weight loss treatment method, offers advantages such as minimal trauma and fewer complications. In recent years, it has continued to develop and is increasingly accepted by patients.

FIG. 14.1 — Management methods of obesity.
(I) Diagnosis of obesity
1. General indicators. Currently, the primary general indicator for diagnosing obesity is the body mass index (BMI). This index provides an indirect assessment of body fat composition, is easy to perform, and is widely used in clinical practice. However, there is no uniform standard for diagnosing obesity based on BMI. In China, the current standard is as follows: BMI between 24.0 kg/m
2. Metabolic indicators. Metabolic indicators are primarily used to assess the patient’s metabolic status and the presence of obesity-related complications, which, in turn, help in evaluating the degree of obesity and guiding management. These generally include measurements of organ functions (such as heart, lung, liver, etc.), glucose and lipid metabolism indicators, thyroid function, gonadal function, etc. Specific tests should be selected based on the individual case.
(II) Technical methods and previous terminology in super minimally invasive weight loss surgery for obesity
Super minimally invasive weight loss surgery refers to weight reduction procedures performed without altering the anatomical structure. If instruments are used during the process, they must be removable, and after their removal, the body returns to its original state. The technical methods included under this category are commonly referred to by previous names, such as intragastric balloon insertion for weight loss; endoscopic sleeve gastrectomy (ESG); endoscopic vertical gastroplasty (EVG); endoscopic duodenal-jejunal bypass sleeve (DJBS); endoscopic gastric–jejunal bypass stent surgery.
14.1.2 Intragastric Balloon Placement for Weight Loss
(I) Overview
Intragastric balloon (IGB) placement is currently the most widely used endoscopic weight loss procedure for obese patients. This procedure involves the placement of a space-occupying device in the stomach to reduce its volume and delay gastric emptying. The first intragastric balloon was developed in 1985. Presently, various types of gastric balloons are available for clinical use, differing in the materials used, techniques for placement and removal, balloon placement duration, and the number of balloons used. Research findings indicate that the overall weight loss rate with intragastric balloons is approximately 18.4% ± 2.9%.
Commonly used balloons (figure 14.2) include the Orbera balloon, ReShape balloon, Heliosphere balloon, Spatz3 balloon, and Obalon balloon. Among these, the Orbera balloon, ReShape balloon, and Spatz3 balloon are saline solution-filled balloons, while the Heliosphere balloon and Obalon balloon are gas-filled balloons. The Orbera balloon is the most widely used. It is worth mentioning that the Spatz3 balloon is an adjustable balloon, which allows for modifications to the balloon’s volume based on the patient’s condition and post-operative response. Studies have shown that, when combined with lifestyle modifications, it can achieve a better weight loss outcome. Additionally, the Elipse balloon is a special type of balloon that does not require endoscopic operation; instead, it is ingested by the patient and reaches the stomach, where it can self-expel. However, research reports on this balloon are limited.

FIG. 14.2 — Common intragastric balloon (IGB) devices. A. Saline solution-filled balloons that can be placed for six months: (1) Medsil, (2) Orbera, (3) Medicone, (4) Silimed. B. Saline-filled balloons that can be placed for 12 months: (1) Spatz2, (2) Spatz3, (3) Orbera365, (4) Easy life. C. Other types of IGB devices: (1) Obalon, (2) Elipse, (3) Heliosphere (non-surgical gas-filled balloon), (4) Stella.
(II) Indications and contraindications for intragastric balloon placement for weight loss
1. Indications. The indication for intragastric balloon placement is for patients with a BMI between 30 and 40 kg/m
2. Contraindications. Contraindications include peptic ulcers, esophageal-gastric varices, a history of esophageal and gastric surgeries, hiatal hernia, coagulation disorders, mental disorders, pregnancy, and severe cardiopulmonary diseases.
(III) Preoperative preparation
Before surgery, a comprehensive evaluation of the patient’s condition should be conducted, including general health status, psychological and behavioral patterns, as well as detailed preoperative examinations. For endoscopic placement of the intragastric balloon, anesthesia is recommended, and therefore preoperative anesthesia assessment is required. Depending on the specific case, endotracheal intubation may be considered. On the day before the surgery, proton pump inhibitors should be administered, and the patient should switch to a liquid diet. Fasting is required on the day of the surgery. Prophylactic use of antibiotics is not recommended. Anti-emetic treatment is suggested, including the use of corticosteroids during the procedure. During the intragastric balloon treatment period, proton pump inhibitors are recommended, and the use of non-steroidal anti-inflammatory drugs should be avoided.
(IV) Surgical procedure and techniques (figure 14.3)

FIG. 14.3 — Endoscopic procedure for balloon placement. A. Balloon filling. B. After balloon release.
The surgical procedures for different types of balloons vary. However, before the placement of any balloon, a detailed examination and assessment of the esophagus, stomach, and duodenum should be performed to exclude patients who are not suitable candidates for the procedure.
Taking the Orbera balloon as an example, after the balloon is introduced into the gastric cavity via a sheath, 700 ml of saline solution and 10 ml of methylene blue are injected under endoscopic visualization. Once the balloon is properly filled and no leakage is observed, the balloon is released.
After the placement of the intragastric balloon, the patient should adopt a transitional diet to prevent dehydration. A liquid diet should be followed for three days postoperatively, and then gradually transition to a regular diet. During the first week after the intragastric balloon placement, adaptive symptoms such as nausea, vomiting, and abdominal cramping should be actively managed. This is crucial for preventing dehydration or esophageal injury caused by dry heaving. The use of proton pump inhibitors is necessary and should continue for up to six weeks post-surgery. For the first three days after the procedure, anti-emetic medications should be used regularly, rather than waiting for symptoms to appear. Depending on the patient’s condition, anxiolytics may be prescribed before bedtime to help avoid cramping, a symptom that is typically more prominent in the early days following the procedure. During this adaptation period, it is also important to actively use antispasmodic medications.
The patient should be followed up within one week after the intragastric balloon placement to monitor for complications and provide appropriate management. Persistent vomiting after balloon placement is uncommon; however, if it occurs, it is important to assess for electrolyte imbalances, dehydration, gastric outlet obstruction, inappropriate diet, or tolerance issues. Consideration should be given to using relevant medications or removing the balloon if necessary.
(V) Postoperative management
1. Balloon removal. The commonly used intragastric balloons are typically removed after six months of placement, although some are removed after twelve months. After the balloon is removed, it is recommended that the patient follows a liquid diet for at least two days. The balloon removal procedure should be performed under anesthesia, and generally, no antibiotics or prokinetic drugs are required after removal.
2. Complications. The incidence of complications following intragastric balloon placement is approximately 2.5%. The most common complications are abdominal pain and nausea, with occurrence rates of about 33.7% and 29%, respectively. Other symptoms include constipation and dehydration. Severe complications, such as balloon displacement, perforation, and intestinal obstruction, are rare. The incidence of early balloon removal due to patient intolerance is about 2.2%.
(VI) Hot-spot issues and discussions in the consensus
There are limited guidelines and consensus on super minimally invasive intragastric balloon (IGB) placement for weight loss. Currently, there is only one guideline issued by the American Gastroenterological Association (AGA) and two consensuses from Brazil and Spain. Due to the differences in medical policies across countries, these documents have certain limitations.
1. Standardization of IGB indications and specific procedures needs further regulation. Currently, there is no unified minimum age for intragastric balloon placement. The Brazilian consensus considers individuals over 12 as eligible, while the Spanish consensus sets the minimum age at 16. The AGA guideline does not provide detailed descriptions of IGB indications, and the FDA has not approved the use of IGB in adolescents. The minimum BMI requirement for IGB placement also varies. Some consensuses accept a BMI ≥ 25 kg/m
2. Further research is needed on the use of various medications during and after IGB placement. A major issue to address post-IGB placement is improving patient tolerance, particularly managing the main adverse reactions such as nausea, vomiting, and abdominal pain, which are crucial to improving tolerance. Therefore, the formulation of intraoperative anesthesia protocols and the selection of postoperative antispasmodic and antiemetic drugs are key aspects of IGB treatment. However, there is currently no consensus on the anesthesia protocols used during IGB placement. Existing anesthesia options include awake sedation without intubation, deep/general sedation without an anesthesiologist, deep/general sedation with intubation but without an anesthesiologist, and general anesthesia with intubation. The overall anesthesia plan should aim to minimize the incidence of nausea and vomiting, but there is a lack of relevant randomized controlled trials (RCTs) to determine the best anesthesia option. Commonly used antispasmodic and antiemetic drugs include midazolam, ondansetron, and scopolamine. However, there is no consensus on which drug provides the best effect or the optimal duration of use. One RCT comparing the combination of midazolam and ondansetron versus ondansetron alone for postoperative antiemetic effects after IGB placement found that the combination therapy was more effective, with a lower early balloon removal rate. Another RCT demonstrated that after IGB placement, tropisetron had a better antiemetic effect than alizapride, and the combination of fluphenazine and tropisetron did not improve efficacy. Additionally, a study showed that a single preoperative dose of 300 mg of netupitant and 0.5 mg of palonosetron 6 h before surgery effectively relieved postoperative vomiting, nausea, and stomach pain. However, the existing evidence is insufficient, and the development of postoperative antiemetic regimens should be guided by clinical practice and supported by more RCT results.
It has been reported that IGB placement can lead to gastroesophageal reflux or exacerbate pre-existing reflux, thereby increasing the incidence of esophagitis. Additionally, to prevent complications such as gastric ulcers and gastric perforation due to long-term pressure on the stomach wall by the balloon, long-term use of proton pump inhibitors (PPIs) is recommended after IGB placement. However, the optimal dosage, frequency, and duration of PPI administration post-IGB placement remain unclear and require further research for exploration.
14.1.3 Endoscopic Sleeve Gastroplasty (ESG)
(I) Overview
Super minimally invasive endoscopic sleeve gastroplasty (ESG) is an emerging endoscopic weight-loss surgery. It mimics laparoscopic sleeve gastrectomy (LSG) by using full-thickness sutures to fold and reshape the greater curvature of the stomach, reducing its volume and delaying gastric emptying, thus achieving weight loss. ESG is named for its similarity to the laparoscopic sleeve gastrectomy in surgical practice, but unlike the surgical procedure, ESG does not involve the removal of any gastric tissue. It simply reduces the stomach’s volume while preserving the stomach’s original anatomical structure, which results in fewer severe complications compared to LSG. The full-thickness suturing device currently used in ESG is the OverStitch endoscopic suturing system developed by Apollo Endosurgery, which is the only commercially available device approved by the U.S. FDA to perform full-thickness suturing of the gastric body’s greater curvature. The procedure was first reported in 2013, and its efficacy and safety have continuously improved since its development, making it an important weight-loss method today.
Research results show that ESG can reduce patients’ body weight by 15% at six months and 20% at twelve months post-surgery. A recent multi-center RCT study found that, compared to lifestyle changes alone, which resulted in an overall weight loss rate of 0.8% after 52 weeks, ESG combined with lifestyle changes was significantly more effective, with an overall weight loss rate of 13.6%. Compared to LSG, ESG showed a weight loss rate of 4.7%–14.4% at six months post-surgery, while LSG achieved a rate of 18.8%–26.5%. At twelve months post-surgery, ESG achieved a weight loss rate of 4.5%–18.6%, while LSG showed a rate of 28.4%–29.3%. Although ESG’s weight loss effects are slightly less than those of LSG, ESG is less invasive and better tolerated by patients, offering significant potential for clinical application.
As an emerging endoscopic weight-loss surgery, ESG still requires further development of endoscopic treatment instruments and more clinical applications to continuously improve its effectiveness and safety in the treatment of obesity.
(II) Indications and contraindications for ESG
1. Indications: patients with a Body Mass Index (BMI) greater than 30 kg/m
2. Contraindications: these include malignancy, gastric ulcers, esophageal-gastric varices, a family history of gastric cancer, history of gastric surgery, coagulation disorders, pregnancy, and other severe cardiopulmonary diseases.
(III) Preoperative preparation
A detailed assessment of the patient’s general condition should be conducted preoperatively. For patients currently on anticoagulants, adjustments may be required based on individual circumstances. The patient should fast for 12 h before the surgery and antibiotics should be administered to prevent infection. The surgery is performed under anesthesia, and thus, an anesthetic evaluation is necessary before the procedure. Prior to performing suturing, a thorough examination of the esophagus, stomach, and duodenum should be conducted to rule out any contraindications. During the procedure, CO
(IV) Surgical procedure steps
Before beginning suturing, the areas to be sutured should be clearly defined. Suturing is performed along the greater curvature of the stomach, while the antrum should not be sutured. The suturing is typically carried out in a proximal-to-distal sequence, using U-shaped, square, or rectangular patterns for full-thickness suturing (figure 14.4). The U-shaped suturing procedure is as follows: the suturing sequence is from the anterior wall to the greater curvature, then to the posterior wall, followed by suturing in the opposite direction at a distance of 1 cm. After completing the suturing, the suture line is released to complete one full cycle. In the square/rectangular pattern, suturing starts from the anterior wall, moving from proximal to distal. Generally, four to six rounds of suturing are performed to achieve a ‘sleeve-like’ gastric shape. When suturing the gastric fundus, only the distal part of the fundus should be sutured to preserve its functionality (figure 14.5).
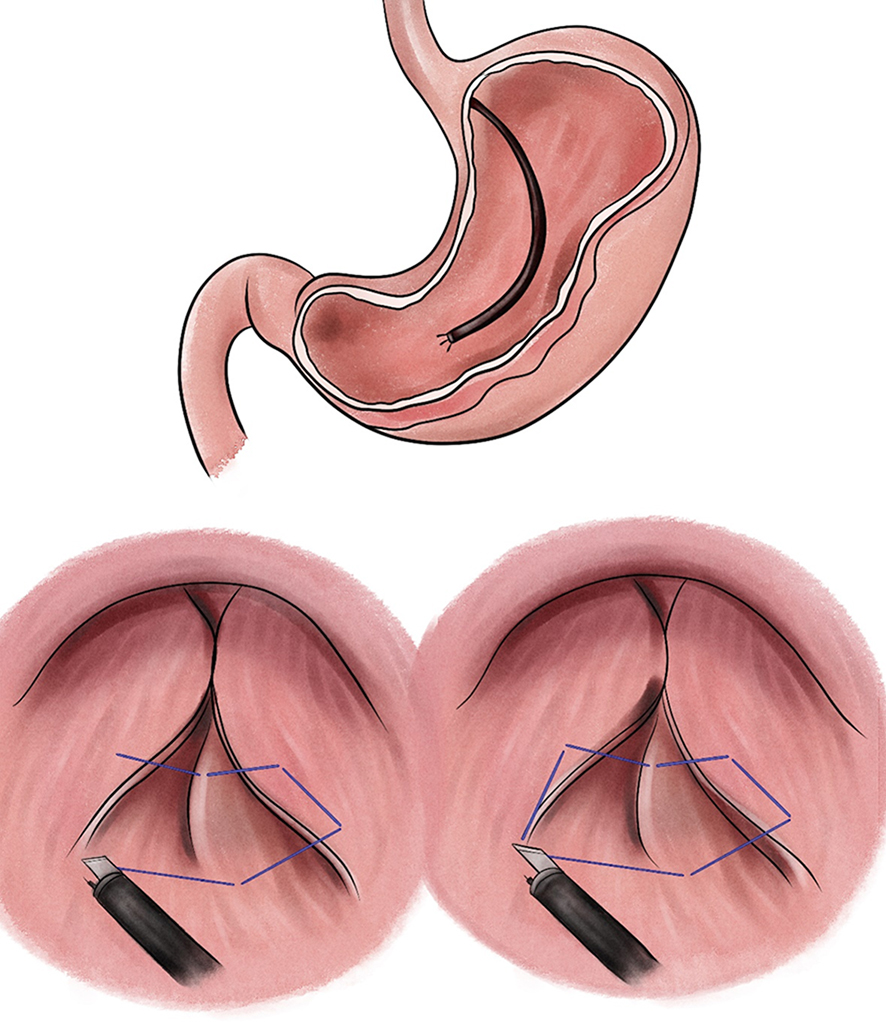
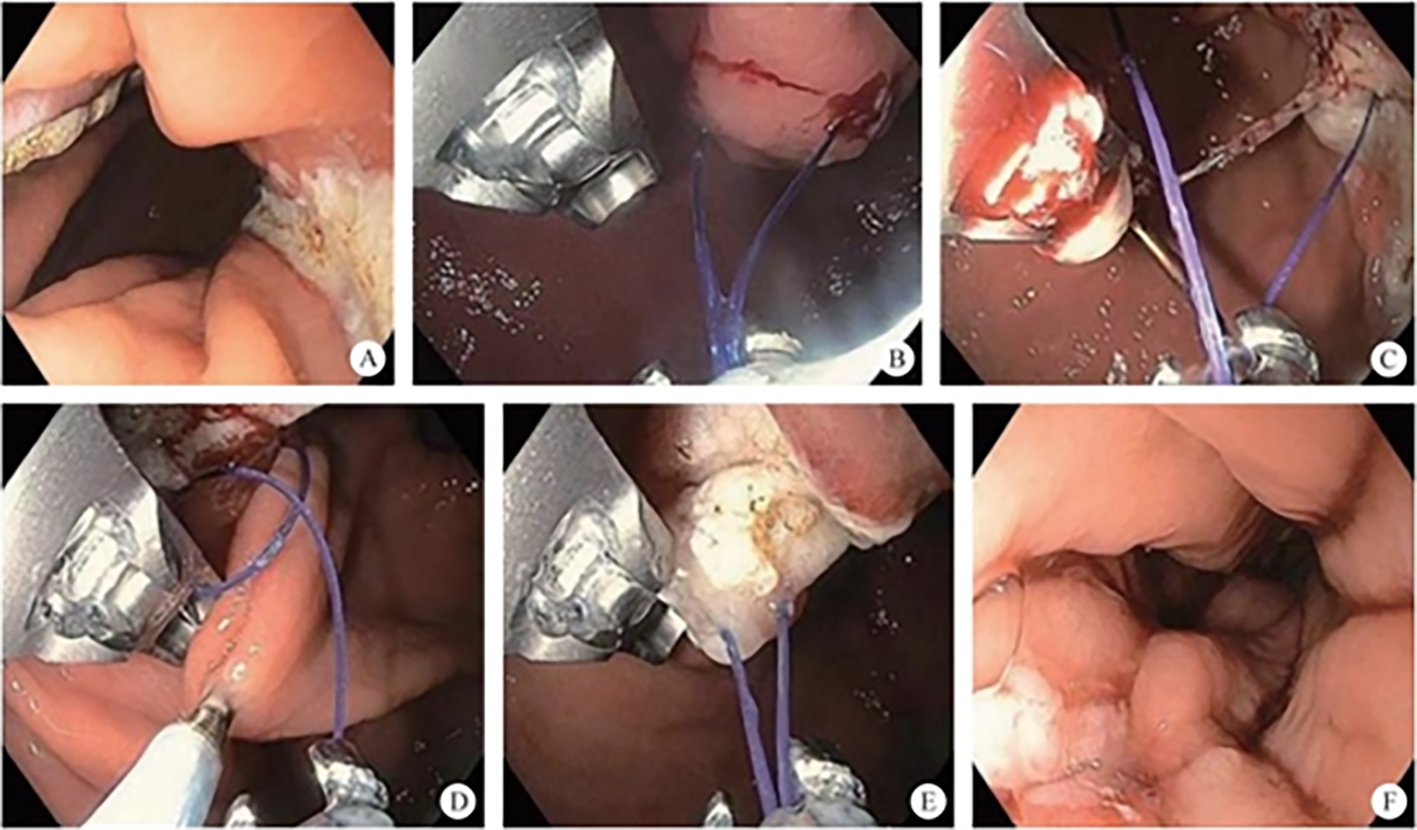

FIG. 14.4 — Comparison of U-shaped and square/rectangular suturing methods.

FIG. 14.5 — ESG surgical procedure flow. A. Marking the suturing sites with argon plasma coagulation (APC). B. First suturing. C. Re-threading the suture through the needle holder. D. Tensioning the tissue with instruments. E. Final suturing. F. Reduction in gastric volume after ESG completion.
(V) Postoperative management
1. Postoperative monitoring: close monitoring of the patient’s condition is essential postoperatively. Proton pump inhibitors (PPIs) should be administered and continued for one to three months after surgery. If the patient experiences symptoms such as nausea or abdominal pain, symptomatic treatment should be provided, including antiemetics and antispasmodics.
2. Complications: the incidence of complications associated with ESG is approximately 1%, including bleeding, peritoneal exudates, venous thrombosis, and others. The majority of complications can be managed symptomatically, with only a small number requiring surgical intervention. Approximately 12 000–15 000 ESG procedures have been performed worldwide, with only one patient reported to have died from deep vein thrombosis complicated by pneumonia.
(VI) Hot-spot issues and research progress in the consensus
At present, ESG has not been recognized by the American Society of Metabolic and Bariatric Surgery (ASMBS). The implementation of ESG varies across different countries and regions, resulting in relatively few procedures being performed. Consequently, there is only one consensus opinion published in Brazil, but its guidance for different countries and regions remains limited.
1. Indications for ESG are yet to be unified and require further research and exploration. Currently, the FDA-approved indications in the United States are for adult obese patients with a BMI ranging from 30 to 50 kg/m
2. There remains considerable controversy regarding the method of endoscopic suturing. ESG suturing methods primarily include U-shaped or square/rectangular techniques. The Brazilian consensus mentions that the majority of suturing is performed using the square/rectangular technique. However, large-scale research on which method is the most effective, provides the most durable results, and causes fewer complications is currently lacking. Therefore, further clinical studies are needed.
3. The long-term effectiveness of ESG requires further investigation. Currently, there are limited reports on the long-term efficacy of ESG, and its durability has not been fully established. Due to factors such as gastric motility, many endoscopic suturing systems face issues such as partial or complete detachment of staples or fixation devices shortly after surgery, leading to suture loosening and potentially affecting the effectiveness and durability of the procedure. In a prospective cohort study involving 216 patients, 149, 68, and 56 patients completed follow-up at one year, three years, and five years, respectively. The mean percentage total weight loss (%TWL) at one year was 15.6%, at three years 14.9%, and at five years 15.9%. However, this study only followed up on patients’ weight changes without rechecking metabolic indicators or performing follow-up endoscopies, and some patients received additional adjunctive treatments postoperatively. Reliable data is needed to confirm the long-term efficacy of ESG.
14.1.4 Endoluminal Vertical Gastroplasty (EVG)
(I) Overview
Endoluminal vertical gastroplasty (EVG) is a type of endoscopic restrictive gastric volume-reducing surgery. EVG utilizes the EndoCinch suturing system (Bard, USA) to perform seven continuous, cross sutures from the proximal gastric fundus to the distal body of the stomach. After the sutures are completed, the sutures are tightened to reduce the gastric volume, achieving the goal of gastric reduction (figure 14.6). The EndoCinch suturing system was originally developed for the treatment of gastroesophageal reflux disease (GERD), but due to its poor durability and incomplete reflux control, its long-term efficacy was found to be suboptimal. Fogel et al. were the first to report the use of the EndoCinch system for EVG in the treatment of obesity, finding that EVG offers advantages such as short operative time, rapid recovery, good effectiveness, and low complication rates.
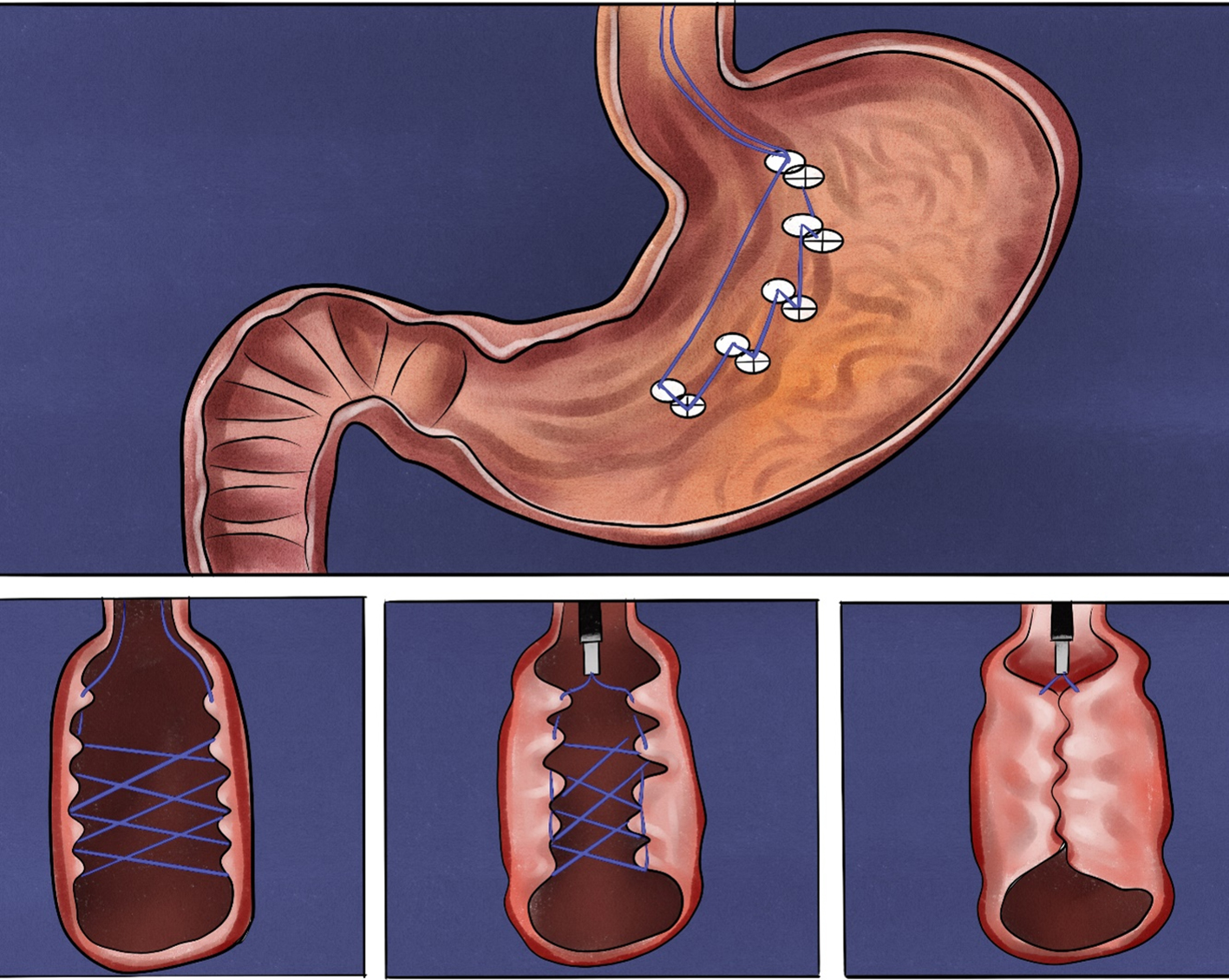

FIG. 14.6 — Diagram of endoluminal vertical gastroplasty. A. Cross-section of the stomach after suturing. B. Endoluminal suturing of the gastric wall. C. Tightening the sutures. D. Fixing the sutures after tightening, completing the procedure.
(II) Indications and contraindications of endoscopic vertical gastroplication
1. Indications: ① Patients with a BMI ≥ 37.5 kg/m
2. Contraindications: ① history of stroke; ② history of myocardial infarction; ③ uncontrolled diabetes; ④ history of previous gastric surgery; ⑤ inappropriate expectations regarding the surgery; ⑥ patients unwilling to assume the potential risks of surgical complications; ⑦ poor compliance, unable to adhere to postoperative lifestyle and dietary changes; ⑧ contraindications to general anesthesia; ⑨ contraindications to gastroscopy or related treatments.
(III) Preoperative preparation
1. Preoperative examination. A thorough review of the patient’s medical history and a comprehensive physical examination should be performed, along with an assessment of their mental and psychological state. Routine gastroscopy should be conducted to assess whether the patient’s gastric anatomy is normal and to identify any history of prior gastric surgery. Helicobacter pylori screening should also be carried out.
2. Informed consent. Detailed information about the procedure, expected outcomes, and potential postoperative changes in diet and lifestyle should be provided to the patient and his/her family. The risks and complications of the surgery should be fully explained, and the patient should sign an informed consent form.
3. Dietary control. The patient should consume easily digestible foods for three days prior to surgery. On the day of the surgery, fasting for at least 6 h and water restriction for 2 h is required.
(IV) Surgical procedure and techniques
1. Anesthesia and positioning. General anesthesia with endotracheal intubation is administered. The patient is positioned in the left lateral decubitus position. It is important to maintain the patient’s warmth and regulate the room temperature as well as the temperature of the intravenous fluids.
2. Surgical steps
Gastroscopy and determining the suturing position: a routine gastroscopy is performed to assess whether there are any abnormalities in the stomach, determine the shape and volume of the stomach preoperatively, and plan the suturing locations.
Suturing the stomach wall: the first suture is placed at the proximal end of the anterior gastric fold near the fundus. Subsequently, a second suture is placed at the most distal part of the anterior gastric fold in the body, usually 10–13 cm away from the first suture. The third suture is placed at the posterior gastric wall 1–2 cm from the proximal end of the second suture, followed by alternating suturing of the anterior and posterior gastric walls towards the proximal end, with a stitch distance of approximately 2 cm.
Tightening and fixing the sutures: the sutures are tightened to bring the anterior and posterior gastric walls together, reducing the gastric volume. The sutures are then secured.
Checking for bleeding and gastric volume reduction: gastroscopy is performed again to ensure there is no active bleeding and adequate hemostasis. The reduction in gastric volume is also evaluated.
(V) Postoperative management
After regaining consciousness from anesthesia, the patient should be monitored for at least 1 h to observe for any complications. The patient is placed on a liquid, sugar-free diet for the first three days, with gradual transition to a semi-solid diet and then to a regular diet. For patients still experiencing reflux or heartburn, treatment with proton pump inhibitors (PPIs) and gastric mucosal protectants should continue.
(VI) Hot-spot issues and research progress in the consensus
Currently, there are only a few reports on endoscopic vertical gastroplication (EVG), and its feasibility and effectiveness still need further validation. The first research study on EVG included 64 patients with an average BMI of 39.9 kg/met al. reported the 18-month follow-up results of 30 obese patients who underwent EVG. At 12 months, the %EWL was 42.8% ± 16.8%, and at 18 months, it was 54.5% ± 27.9%. No serious complications were reported in any of the patients. Another study on 21 adolescents (aged 13–17) with an average BMI of 36.2 kg/m
Subsequently, the improved device, RESTORe (Davol, Murray Hill, NJ), was introduced. It is a single-insertion, multi-needle suturing system that allows for full-thickness suturing and reloading of sutures within the body, eliminating the need for an outer sheath and a second endoscope. This device was studied in a trial involving 18 patients. No significant adverse events were reported. One year later, the patients experienced an average weight loss of 11.0 ± 10 kg, an average waist circumference reduction of 12.6 ± 9.5 cm, and an average BMI reduction of 4.0 ± 3.5 kg/m
In recent years, research on EVG has almost stagnated, awaiting the development of better devices and the validation of multi-center, large-sample clinical studies.
14.1.5 Endoscopic Duodenojejunal Bypass Sleeve (EDJBS)
(I) Overview
Endoscopic duodenojejunal bypass sleeve (EDJBS) is a malabsorptive type of surgery. Its principle is to shorten the time it takes for the chyme to reach the middle segment of the jejunum, thereby altering the secretion of digestive hormones and the neural regulation of the digestive tract. This results in changes to food digestion and absorption, ultimately achieving weight loss. The EDJBS technique is inspired by the principles of duodenojejunal bypass surgery and is facilitated by the EndoBarrier device developed by GI Dynamics (Lexington, Massachusetts, USA). The EndoBarrier device consists of a flexible, endoscopically implantable, and removable fluoropolymer sleeve, approximately 60 cm in length, which is anchored to the proximal duodenum using a self-expanding nickel–titanium alloy (Nitinol) anchor. The sleeve is placed between the duodenum and the jejunum, allowing food to pass through while preventing direct contact between food and the duodenum, biliary tract, and pancreatic secretions. In addition, this mechanism causes delayed digestion and interferes with the body’s metabolic functions, including altering the incretin pathway, reducing weight, and improving diabetes.
(II) Indications and contraindications
1. Indications: currently, there are no clearly defined indications for the procedure.
2. Contraindications: ① patients with unrealistic expectations regarding the surgery; ② patients unwilling to accept the potential risks and complications of the surgery; ③ patients with poor compliance who are unable to follow the postoperative lifestyle and dietary changes; ④ contraindications to general anesthesia; ⑤ contraindications to gastroscopy or related treatments.
(III) Preoperative preparation
1. Preoperative assessment. A thorough review of the patient’s medical history and a comprehensive physical examination should be performed. Additionally, an evaluation of the patient’s lifestyle, presence of stress factors, expectations regarding the surgical outcome, and understanding of the procedure should be conducted. A comprehensive assessment of the patient’s condition is essential to develop a reasonable and appropriate surgical plan.
2. Informed consent. Detailed explanations of the surgical procedure, expected outcomes, and potential post-operative changes in diet and lifestyle should be provided to the patient and the family. All possible risks and complications of the surgery must be thoroughly communicated, and an informed consent form should be signed.
3. Dietary control. The patient should consume easily digestible foods for the three days prior to surgery. On the day of the operation, fasting for at least 6 h and refraining from drinking liquids for 2 h before surgery is required.
(IV) Surgical procedure and techniques
1. Anesthesia and positioning. General anesthesia with endotracheal intubation should be administered. The patient should be positioned in the left lateral decubitus position. Care should be taken to maintain warmth, adjust room temperature, and regulate the temperature of the intravenous fluids, to prevent pressure sores.
2. Surgical steps (figure 14.7)
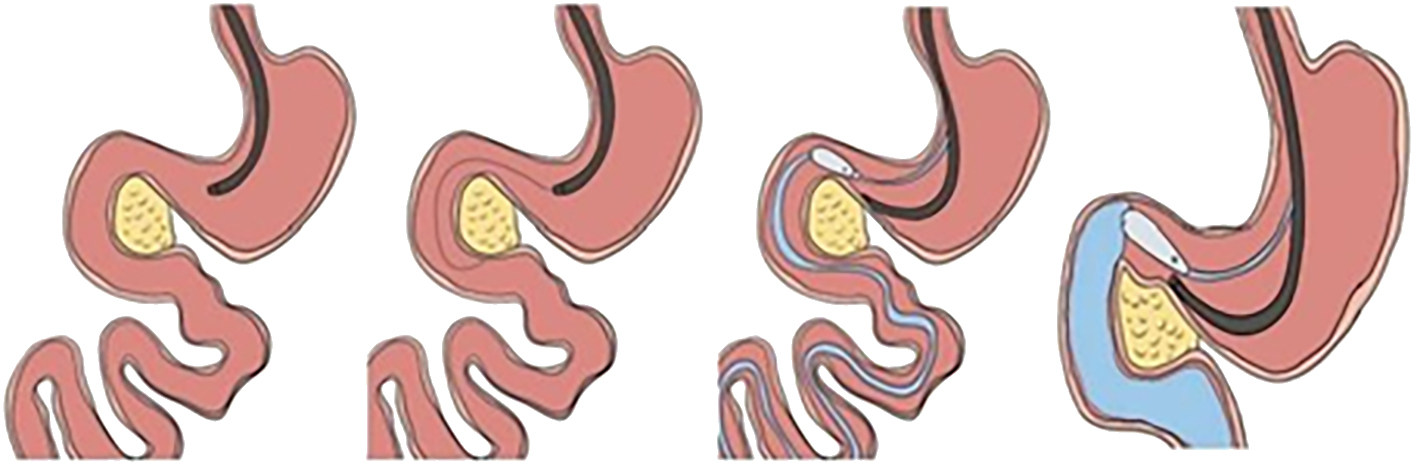

FIG. 14.7 — Schematic of endoscopic duodenal-jejunal bypass sleeve surgery. A. Examination of the gastric and duodenal cavities. B. Insertion of the guidewire into the duodenal lumen. C. Advancement of the device along the guidewire into the duodenum, pushing the internal guidewire and releasing the sheath. D. Release of the anchor, securing it in the duodenal bulb.
(1) Gastroscopy: a thorough examination of the stomach and duodenum should be performed to assess the condition of the upper gastrointestinal tract.
(2) Guidewire insertion and device advancement: under endoscopic guidance, a guidewire is inserted into the duodenum. After withdrawing the endoscope, fluoroscopy should be performed to advance the device into the duodenum following the guidewire. The placement of the device should be confirmed by reintroducing the endoscope.
(3) Sheath release and proximal anchoring: under fluoroscopic guidance, the device’s internal guidewire is advanced, and the end of the device is pushed into the duodenum, connecting it to the cannula with a non-damaging balloon. The sheath should be fully expanded with the assistance of the balloon. Subsequently, the anchor should be placed in the duodenal bulb.
(4) Position confirmation and documentation
A contrast agent is injected to verify the patency of the sheath. Once confirmed, the internal guidewire must be separated from the sheath and from the non-damaging balloon, and the insertion device removed. Another endoscopic examination must be performed to check for any complications, such as bleeding. If no abnormalities are found, the endoscope is withdrawn and the surgical procedure is documented.
(V) Surgery-related complications and postoperative management
Common complications associated with this procedure include pain, obstruction, displacement, bleeding, and infection. In rare cases, patients may experience duodenal perforation, duodenal fistula, cholecystitis, and other complications. Some prospective studies have reported additional complications, such as esophageal mucosal tears, although none of these required surgical intervention.
(VI) Hot-spot issues and research progress in consensus
1. High incidence of adverse events in EDJBS. The issue of a high incidence of adverse events in EDJBS remains unresolved. Currently, EDJBS is still in the clinical trial phase, and the relevant devices have not yet been approved by the FDA. There are no standardized guidelines or consensus on its operation. The first human study applying DJBS was reported in 2008 by Rodriguez-Grunert et al., which included 12 patients. Two patients (16.67%) experienced operation-related adverse events, including mucosal tears in the oropharynx and esophagus. During the period when the sheath was in place, a total of 71 adverse events occurred, primarily nausea, vomiting, and inflammation at the anchoring site. Among these, 55 cases (78%) were considered related to the device itself. Subsequently, several experiments further explored the clinical application of EDJBS. Bark Betzel et al. conducted a systematic review of adverse events related to EDJBS, including 38 studies with a total of 1056 patients. A total of 891 adverse events were reported, with 33 cases (3.7%) classified as severe adverse events, such as liver abscess and esophageal perforation. The study also found that approximately 85% of the adverse events were related to the anchoring device. Improvements to the anchoring device may potentially enhance the safety of EDJBS.
Recently, some improved devices have been introduced. A Chinese study investigated the safety and efficacy of these improved devices. In a cohort of 26 subjects, one case of severe adverse event (upper gastrointestinal bleeding) occurred, but the overall incidence of adverse events significantly decreased. However, long-term follow-up and multicenter, large-sample clinical studies are still needed.
2. Long-term efficacy of EDJBS needs confirmation through multicenter and large-sample studies. Early research on EDJBS mostly had follow-up periods of less than six months, making it difficult to determine long-term outcomes. A subsequent multicenter randomized controlled trial (ENDO) was prematurely terminated by the FDA due to an unexpectedly high rate of liver abscesses, resulting in limited existing clinical data. In recent years, with the advent of improved devices, new studies have been initiated. However, multicenter, large-sample studies with long-term follow-up data are still lacking.
Quezada et al. conducted a follow-up study on the improved second-generation EndoBarrier, lasting up to three years. The study found that although long-term placement of the sheath significantly and continuously led to weight loss in patients, the frequency and severity of adverse events also increased. Furthermore, the weight loss effect during the second and third years was not superior to that of the first year, and serious complications occurred more frequently after 12 months of sheath placement. Therefore, the study recommended that the maximum duration for sheath placement be 12 months. A multicenter randomized controlled trial involving 170 patients reported follow-up data comparing the results of leaving the device in place for 12 months versus removing it after 12 months. With dietary and exercise interventions, the experimental group showed significantly greater weight loss and reductions in systolic blood pressure, serum cholesterol, and alanine aminotransferase (ALT) levels compared to the control group at 12 months after the removal of the device. However, these differences gradually diminished after the device was removed, and by 12 months post-removal, there was no significant difference between the two groups. The study also found no significant difference in blood glucose reduction between the groups.
However, this multicenter study included data from only two centers, and its reproducibility and representativeness should be considered. Therefore, the efficacy and safety of EDJBS still require further clinical validation, and there is anticipation for the development of safer and more effective improved devices.
14.1.6 Endoscopic Gastro-Duodenal-Jejunal Bypass Sleeve Surgery
(I) Overview
Sandler et al. reported a unique intraluminal approach for surgery, involving the endoscopic implantation of a movable gastro-duodenal-jejunal bypass sleeve (figure 14.8). Using endoscopic and laparoscopic techniques, the sleeve is placed at the gastroesophageal junction and extends approximately 120 cm into the small intestine, creating an intraluminal gastro-duodenal-jejunal bypass. The sleeve prevents the absorption of nutrients from the stomach, duodenum, and jejunum, allowing food to directly pass from the esophagus into the intestine. This procedure mimics the mechanism of traditional gastric bypass surgery. The EGDJBS technique is similar to EDJBS but is a hybrid procedure that not only limits gastric intake but also reduces intestinal absorption. Theoretically, this technique should have a more significant weight loss effect compared to the previously mentioned methods; however, reports on the efficacy of this technique are scarce.

FIG. 14.8 — Schematic of the gastro-duodenal-jejunal bypass sleeve and device. A. Schematic of the gastro-duodenal-jejunal bypass sleeve surgery. B. Gastro-duodenal-jejunal bypass sleeve device.
(II) Surgical procedure
The preoperative preparation is the same as for a standard gastroscopy. The EGDJBS procedure is similar to EDJBS, with the principle of mimicking the anatomical changes seen after gastric bypass surgery. The procedure involves the placement of a longer sleeve, approximately 120 cm in length, using endoscopic and laparoscopic assistance. The sleeve is positioned from the gastroesophageal junction to the proximal jejunum, achieving both gastric intake restriction and reduced intestinal absorption.
(III) Surgical complications and postoperative management
The complications of this procedure are similar to those of EDJBS. However, clinical data on this procedure are currently limited, and its short- and long-term weight loss effects require further clinical observation.
(IV) Hot-spot issues and research progress in the consensus
Currently, reports on EGDJBS are scarce, and its short- and long-term safety and efficacy need further research. Since EGDJBS was first reported in 2011, only three studies have been published, with limited clinical data. Moreover, the associated devices have not received FDA approval, and there are no established guidelines or consensus. In the first report, 22 patients underwent EGDJBS, and the study lasted 12 weeks. Five patients (23%) had to have the sleeve removed prematurely due to intolerance. After 12 weeks, the 17 remaining patients showed an average weight loss of 39.7%. Additionally, seven patients with preoperative diabetes had normal blood glucose levels throughout the trial, eliminating the need for diabetes medication. Four patients with elevated hemoglobin levels preoperatively showed improvement by the end of the trial. No serious adverse events occurred during the trial, indicating the short-term efficacy of EGDJBS. Subsequently, the same research team reported a 12-month follow-up for single-center and multicenter studies in 2015 and 2018. In the single-center study, 13 patients were included, with 12 successfully undergoing sleeve placement. Two patients (16.7%) had the sleeve removed prematurely. Among the 10 patients who completed follow-up, six had the device still in place and patent after 12 months, with an average excess weight loss percentage (%EWL) of 54%. Four patients experienced partial sleeve detachment, and their overall weight loss was slightly lower than the remaining six. However, comorbidities such as hypertension, hyperglycemia, and hyperlipidemia improved during the sleeve placement period. Five of the six patients who underwent follow-up 14 months after sleeve removal maintained %EWL at 30%. In the multicenter study, 32 patients were included, all of whom successfully received the sleeve. Four patients (12.5%) had the sleeve removed prematurely. After 12 months, the average %EWL was 44.8%, with an average total weight loss of 17.6%, and an average weight reduction of 20.8 kg. The average BMI reduction was 7.5 kg/m
Based on these results, EGDJBS appears to demonstrate good tolerance, weight loss efficacy, and safety, with significant potential for clinical application. However, since there are currently only a small number of studies, further trials are needed to verify its clinical efficacy and safety.
14.2 Super Minimally Invasive Drainage of Mediastinal and Abdominopelvic Abscesses
14.2.1 Overview
Mediastinal abscess refers to the acute pyogenic infection of the connective tissue in the mediastinum, caused by the invasion of Staphylococcus aureus due to various reasons, leading to tissue necrosis and liquefaction, which results in the accumulation of pus within the mediastinum. Infection is the most common cause of mediastinal abscess. Based on the source of infection, the causes of mediastinal abscess can be classified into the following categories: first, open mediastinal infection caused by trauma; second, esophageal or tracheal perforation due to surgical procedures or foreign bodies leading to mediastinal infection, such as mediastinal abscess caused by esophageal perforation during digestive endoscopy, or infection resulting from esophageal foreign bodies penetrating into the mediastinum; third, intrinsic mediastinal infection, where spontaneous mediastinal emphysema becomes infected, and bacteria invade, causing a mediastinal abscess.
Abdominal abscess refers to the necrosis and liquefaction of tissues in a specific space or region within the abdominal cavity, surrounded by intestinal loops, viscera, abdominal wall, omentum, or mesentery, forming localized pus accumulation. It is mainly classified into subdiaphragmatic abscess, pelvic abscess, and intraluminal abscess. Some scholars consider pelvic and abdominal cavities as distinct regions and classify pelvic abscess separately. Given the similar super minimally invasive drainage surgery techniques for these two types of abscesses, this section will address abdominal and pelvic abscesses as a whole. The main causes of abdominal-pelvic abscess include various diseases leading to secondary peritonitis, abdominal surgery, and trauma.
14.2.2 Clinical Symptoms and Diagnosis
The clinical symptoms of mediastinal abscess are primarily systemic, including infection-related signs such as chills, high fever, and restlessness. Severe retrosternal pain may also occur, which worsens with deep breathing or coughing. The pain may radiate to the neck, behind the ears, the anterior chest, and between the scapulae, and nerve root pain may also develop. If the abscess compresses surrounding organs, symptoms such as hoarseness due to recurrent laryngeal nerve compression, diaphragmatic weakness or paralysis due to phrenic nerve compression, Horner’s syndrome due to compression of the sympathetic stellate ganglion, and tachycardia from vagus nerve compression may appear. If the mediastinal abscess ruptures into the pleura, it can cause an empyema or pneumothorax, leading to respiratory distress and possible hemorrhage, with severe cases potentially progressing to shock. Mediastinal abscess may also complicate with pulmonary infections. For patients with abdominal abscess, if the abscess is located below the diaphragm, the following symptoms may occur: (1) signs of septicemia, such as intermittent or remittent high fever, chills, high fever, reduced appetite, rapid or weak pulse, and hypotension; (2) pain, including upper abdominal pain, percussion tenderness, and palpation pain in the subdiaphragmatic and hypochondriac regions; (3) physical examination may reveal decreased or absent breath sounds at the base of the affected lung. A subdiaphragmatic abscess on the right side may also present with an elevated liver dullness. If the abscess is located in the mesentery, it may manifest as vague abdominal pain and fever. If the abscess is large, a painful mass may be palpable, and systemic toxic symptoms may be present. Pelvic abscess tends to present with milder systemic symptoms compared to abdominal abscess, but its local symptoms are relatively severe. Persistent remittent fever may occur, along with rectal and bladder irritability, such as lower abdominal distension, increased frequency of bowel movements, urgency, tenesmus, and mucus stool; frequent urination, urgency, and even difficulty urinating may also develop. Physical examination may show bulging and tenderness of the rectal wall, as well as relaxation of the anal sphincter. Diagnosis can be confirmed using ultrasound, abdominal-pelvic CT, or diagnostic puncture.
14.2.3 Indications and Contraindications of Super Minimally Invasive Drainage
(1) Indications
In general, mediastinal abscesses and abdominal-pelvic abscesses are indications for super minimally invasive drainage via gastrointestinal endoscopy. For patients who, based on preoperative assessments, are expected to have difficulty achieving successful drainage through endoscopy, this procedure may be considered a relative indication for trial.
(2) Contraindications
For abscesses that are difficult to reach with current endoscopic equipment and techniques, such as those located under the right diaphragm, this should be considered a relative contraindication. If a patient’s general condition does not permit endoscopic intervention—such as in cases of severe anemia or when cardiopulmonary function cannot tolerate the procedure—it should be considered an absolute contraindication for endoscopic treatment.
14.2.4 Preoperative Preparation
Mediastinal and abdominal-pelvic abscesses, in addition to standard anti-infection treatment, often require abscess drainage. This section mainly discusses super minimally invasive drainage via gastrointestinal endoscopy for the treatment of mediastinal and abdominal-pelvic abscesses, excluding percutaneous drainage techniques, which are not within the scope of this discussion. Preoperative preparation for super minimally invasive drainage via gastrointestinal endoscopy is consistent with other endoscopic surgeries. For mediastinal abscess drainage through the esophagus, preparation should follow the standard procedures for gastroscopy or ultrasonic gastroscopy. For abdominal-pelvic abscesses drained via the upper gastrointestinal tract, gastroscopy preparation should also be followed. If pelvic abscesses are drained via the colon, preparation should align with colonoscopy procedures. For intestinal abscesses requiring small bowel endoscopy, preparation should follow the standard procedure for small bowel endoscopy.
14.2.5 Surgical Procedure
Mediastinal and pelvic abscesses can be classified into two categories for drainage based on whether they are connected to the lumen of the gastrointestinal tract: conventional endoscopic drainage and endoscopic ultrasound (EUS)-guided drainage. Abscesses that are connected to the gastrointestinal lumen can be treated with conventional endoscopic drainage, while abscesses that are not connected to the lumen can be treated with EUS-guided puncture drainage.
Endoscopic drainage is performed under direct visualization. If the abscess has a large fistula to the lumen, the drainage tube or stent can be placed directly under direct vision. If the fistula is smaller and the placement of a drainage tube or stent is difficult, dilation is required. Prior to drainage under direct endoscopic vision, lavage and aspiration of the abscess cavity using the endoscope can be performed to remove purulent fluid and reduce the infection source. The use of antibiotic lavage inside the abscess cavity remains a controversial topic.
The general steps of EUS-guided drainage involve using a 19G or 22G needle for fine-needle aspiration (EUS–FNA) under EUS guidance, which punctures the gastrointestinal tract wall to enter the abscess cavity. Once the puncture needle reaches the appropriate position within the abscess cavity, a guidewire is placed through the needle. The gastrointestinal tract wall is then dilated using a balloon under the guidance of the guidewire, and finally, a drainage tube or pig-tail stent is placed. The length of the drainage tube and stent is determined based on the distance between the abscess and the gastrointestinal lumen. If the abscess is located less than 1 cm from the gastrointestinal lumen, or if there is significant solid material within the abscess, a metal stent may be considered. The recommended dilation diameters are 6 mm for the esophagus, 6–8 mm for the colon and jejunum, 8–10 mm for the duodenum, and 8–15 mm for the stomach.
Currently, there is no consensus regarding the timing for drainage tube removal. It is generally believed that the tube can be removed once the infection is largely controlled, the abscess has significantly reduced in size, and the drainage volume remains minimal for 48–72 h.
14.2.6 Postoperative Management
Super minimally invasive drainage via gastrointestinal endoscopy is a minimally invasive procedure, and postoperative management primarily involves handling the drainage tube and administering infection control measures. Postoperative management includes regular replacement of drainage bags, monitoring drainage volume, and sending drainage fluid for bacterial culture and sensitivity testing. If the patient’s clinical symptoms do not improve or if they worsen, or if no drainage fluid is observed in the drainage bag, the drainage tube may be blocked or displaced, requiring repositioning to the appropriate location. Prior to receiving sensitivity results, empirical antibiotic therapy is applied for infection control. Once sensitivity results are available, antibiotics should be adjusted based on the findings.
14.2.7 Hot-Spot Issues and Discussion in the Consensus
In the treatment of mediastinal and pelvic abscesses, surgical debridement and percutaneous drainage guided by ultrasound or CT still dominate current treatment strategies. Endoscopic drainage of abscesses has been performed less frequently, and available data mostly consist of case reports, with no relevant guidelines or consensus available at present.
There is limited research comparing the efficacy of endoscopic drainage of abscesses with other main drainage methods. One retrospective cohort study compared EUS-guided drainage of upper abdominal abscesses with percutaneous puncture drainage. There was no significant difference in technical or clinical success rates between the two groups, and the incidence of adverse events was also similar: EUS-guided drainage had an adverse event rate of 22.2% (4/18), while percutaneous drainage had an adverse event rate of 21% (13/62). This study preliminarily indicated the effectiveness of endoscopic drainage, though its reliability is limited due to the small sample size of the EUS-guided group (16 cases). Future studies comparing endoscopic drainage with traditional drainage methods, focusing on technical success rates, drainage efficacy, hospitalization duration, and patient experiences, are needed.
Due to anatomical considerations and limitations of current instruments, endoscopic drainage is more commonly applied to pelvic abscesses. One study analyzed 17 patients who underwent EUS-guided drainage of pelvic abscesses, reporting a technical success rate of 100%. Among them, two cases (12%) required a second endoscopic intervention. All patients were followed up with CT scans, which showed complete abscess resolution, yielding a treatment success rate of 100%. The median hospital stay was five days, and no abscess recurrence was observed during a median follow-up of 39 months. Another study involving 37 patients with pelvic abscesses undergoing EUS-guided drainage found a 100% technical success rate, with 34 cases (91.9%) achieving clinical success. Five patients required secondary interventions within 14 days post-drainage. Early complications included perforation requiring surgery (n = 1), stent displacement (n = 1), and rectal discomfort (n = 1). Two patients had persistent abscesses; one required surgery and the other optimal supportive treatment. During a median follow-up of 64 months, two patients experienced abscess recurrence at three and twelve months, requiring surgical intervention. Among the 37 patients, 32 achieved long-term success (86.5%). These studies provide preliminary evidence supporting the safety and efficacy of EUS-guided pelvic abscess drainage, though larger, multicenter studies are needed to confirm these findings.
Moreover, there remains some debate on the optimal timing for pelvic abscess drainage. Traditionally, the duration of antibiotic treatment was used as a criterion, with drainage or surgical intervention considered if antibiotics failed after 48–72 h. Recent studies have indicated that larger abscesses are associated with higher antibiotic treatment failure rates and greater risks of complications. The 2020 French guidelines for pelvic inflammatory disease recommend drainage when the abscess diameter exceeds 3–4 cm. However, other guidelines have yet to provide clear recommendations, and further research is required to establish definitive criteria.
Endoscopic drainage of abscesses at other anatomical sites remains largely based on case reports, and there is a lack of large-scale studies. Further exploration and expansion of endoscopic treatment applications are warranted. With advances in gastrointestinal endoscopy and instrumentation, super minimally invasive drainage via endoscopy is expected to be increasingly applied in abscess treatment, and more related research is likely to emerge.
References
[1] Sharaiha R. Z., Shikora S., White K. P., et al. (2023) Summarizing consensus guidelines on obesity management: a joint, multidisciplinary venture of the International Federation for the Surgery of Obesity & Metabolic Disorders (IFSO) and World Gastroenterology Organisation (WGO), J. Clin. Gastroenterol. 57(10), 967–976.
[2] Blüher M. (2019) Obesity: global epidemiology and pathogenesis, Nat. Rev. Endocrinol. 15, 288–298.
[3] (NCD-RisC) NRFC. (2017) Worldwide trends in body-mass index, underweight, overweight, and obesity from 1975 to 2016: a pooled analysis of 2416 population-based measurement studies in 128·9 million children, adolescents, and adults, Lancet 390, 2627–2642.
[4] Abdulla M., Mohammed N., AlQamish J. (2023) Overview on the endoscopic treatment for obesity: a review, World J. Gastroenterol. 29, 5526–5542.
[5] Obesity Prevention and Control Branch of the Chinese Nutrition Society, Clinical Nutrition Branch of the Chinese Nutrition Society, Behavioral Health Branch of the Chinese Preventive Medicine Association, et al. (2022) Expert consensus on obesity prevention and treatment in China, Chin. J. Epidemiol. 43(5), 609–626.
[6] Digestive Endoscopy Terminology Examination Subcommittee. (2021) Terminology of digestive endoscopy. Science Press, Beijing.
[7] Bazerbachi F., Vargas E. J., Rizk M., et al. (2021) Intragastric balloon placement induces significant metabolic and histologic improvement in patients with nonalcoholic steatohepatitis, Clin Gastroenterol. Hepatol. 19, 146–154.e4.
[8] Abu Dayyeh B. K., Maselli D. B., Rapaka B., et al. (2021) Adjustable intragastric balloon for treatment of obesity: a multicentre, open-label, randomised clinical trial, Lancet 398, 1965–1973.
[9] Ribeiro I. B., Kotinda A., Sánchez-Luna S. A., et al. (2021) Adverse events and complications with intragastric balloons: a narrative review (with video), Obes. Surg. 31, 2743–2752.
[10] Muniraj T., Day L. W., Teigen L. M., et al. (2021) AGA clinical practice guidelines on intragastric balloons in the management of obesity, Gastroenterology 160(5), 1799–1808.
[11] Neto M. G., Silva L. B., Grecco E., et al. (2018) Brazilian Intragastric Balloon Consensus Statement (BIBC): practical guidelines based on experience of over 40,000 cases, Surg. Obes. Relat. Dis. 14, 151–159.
[12] Espinet Coll E., Del Pozo García A. J., Turró Arau R., et al. (2023) Spanish Intragastric Balloon Consensus Statement (SIBC): practical guidelines based on experience of over 20 000 cases, Rev. Esp. Enferm. Dig. 115, 22–34.
[13] Abdelhamid S. A., Kamel M. S. (2014) A prospective controlled study to assess the antiemetic effect of midazolam following intragastric balloon insertion, J. Anaesthesiol. Clin. Pharmacol. 30(3), 383–386.
[14] Van Hee R., Van Wiemeersch S., Lasters B., et al. (2003) Use of anti-emetics after intragastric balloon placement: experience with three different drug treatments, Obes. Surg. 13(6), 932–937.
[15] Ienca R., Giardiello C., Scozzarro A., et al. (2019) Improving nausea and vomiting post-elipse balloon: a novel single-dose regimen of 300 mg netupitant/0.5 mg palonosetron hydrochloride, Obes. Surg. 29(9), 2952–2956.
[16] Rossi A., Bersani G., Ricci G., et al. (2007) Intragastric balloon insertion increases the frequency of erosive esophagitis in obese patients, Obes. Surg. 17(10), 1346–1349.
[17] Yoo I. K., Chun H. J., Jeen Y. T. (2017) Gastric perforation caused by an intragastric balloon: endoscopic findings, Clin. Endosc. 50(6), 602–604.
Appendix
I. Published Videos of Classic Cases
| SMIS channel | Title | References |
| Natural orifice channel | (Video 2.1) A modified endoscopic resection of the entire thickness of a gastrointestinal stroma tumor: A new sealing technique based on the principles of super minimally invasive surgery(Video 2.2) Novel endoscopic papillectomy technique for reducing postoperative adverse events(Video 2.3) Successful endoscopic transgastric retrieval of a plastic stent that migrated into the abdominal cavity during pancreatic fistula drainage.(Video 2.4) Endoscopic retrieval of a migrated lumen-apposing metal stent into the abdominal cavity during transluminal drainage(Video 2.5) Incidental discovery of a pancreatic ductal adenocarcinoma during percutaneous cholangioscopy through a T-tube tract(Video 2.6) Three pancreatic duct lesions discovered at an early stage in a patient by cholangioscopy | Endoscopy 2023, 55(S 01):E561–E562.WJG 2020, 26(40):6250-6259.Endoscopy 2020, 52(8):E289–E290.AJG 2020, 115(4):504.Endoscopy, 2023, 55:E404–E405. Endoscopy, 2023, 55:E332–E333. |
| Tunnel channel | (Video 2.7) Autologous skin-grafting surgery for the prevention of esophageal stenosis after complete circular endoscopic submucosal tunnel dissection(Video 2.8) Challenging use of the digestive endoscopic tunnel technique to treat schwannoma originating in the trachea(Video 2.9) Endoscopic submucosal tunnel dissection with an elastic traction device for a circumferential superficial esophageal neoplasm(Video 2.10) Magnetic multi-directional anchoring-guided endoscopic submucosal tunnel dissection for large gastric lesions(Video 2.11) Simultaneous performance of one-tunnel per-oral endoscopic myotomy, submucosal tunnel endoscopic resection, and diverticulotomy(Video 2.12) Peroral endoscopic myotomy with simultaneous submucosal and muscle dissection for achalasia with severe interlayer adhesions(Video 2.13) Moving the knife’s tip on the thoracic aorta: high-risk submucosal tunneling endoscopic resection procedure for a puzzling submucosal tumor in the esophagus(Video 2.14) Prepyloric submucosal tunneling endoscopic resection for a case of inflammatory mass | Endoscopy 2024, 56(S 01): E290–E291.Am J Gastroenterol 2020, 115(5):650.Endoscopy 2023, 55(S 01):E388–E389.Endoscopy 2021, 53(10):E382–E383.Gastrointest Endosc 2016, 84(5):846–847.Gastrointest Endosc 2016, 83(3):651–652.Dig Endosc 2018, 30(3):397–398.Dig Endosc 2018, 30(4):546–547. |
| Puncture channel | (Video 2.15) Rendezvous-assisted endoscopic retrograde pancreatography in a patient with annular pancreas and coexisting pancreas divisum(Video 2.16) Novel treatment of pancreatic cystic neoplasms: EUS-guided radiofrequency ablation combined with lauromacrogol ablation(Video 2.17) Spyglass discovery of a mucinous cystic neoplasm by introducing an optical fiber into the cyst through a 19G needle during EUS examination | Endoscopy 2023, 55(S 01):E559–E560.Turk J Gastroenterol 2018, 29(1):101–104.Endosc Ultrasound 2019, 8(1):60–62. |
| Multi-space channel | (Video 2.18) Flexible choledochoscopy via the cystic duct combined with laparoscopic cholecystectomy for the treatment of cholelithiasis | Endoscopy 2023, 55(S 01): E659–E661. |
II. Super Minimally Invasive Surgery Video
| No. | Chapter Section | Title | SMIS Procedure |
| 1 | Section 8.1 | (Video 8.1) Peroral super minimally invasive resection for early esophageal carcinoma | Traction-assisted endoscopic submucosal dissection |
| 2 | Section 8.1 | (Video 8.2) Early esophageal carcinoma resection by super minimally invasive surgery through a tunnel channel | Tunnel method endoscopic submucosal dissection |
| 3 | Section 8.1 | (Video 8.3) Digestive endoscopic super minimally invasive esophageal skin grafting surgery-1 | Digestive endoscopic super minimally invasive esophageal skin grafting |
| 4 | Section 8.1 | (Video 8.4) Digestive endoscopic super minimally invasive esophageal skin grafting surgery-2 | Digestive endoscopic super minimally invasive esophageal skin grafting with autologous skin graft and medicated stent placement |
| 5 | Section 8.2 | (Video 8.5) Peroral super minimally invasive resection of esophageal subepithelial lesions through a tunnel channel | Tunnel method endoscopic submucosal tumor resection |
| 6 | Section 8.3 | (Video 8.6) Super minimally invasive radiofrequency ablation of low grade intraepithelial neoplasia | Radiofrequency ablation + APC treatment |
| 7 | Section 8.4 | (Video 8.7) Super minimally invasive prevention and treatment of peroral esophageal stenosis | Gastrointestinal stricture incision + endoscopic submucosal injection |
| 8 | Section 8.5 | (Video 8.8) Super minimally invasive cardiac constriction for gastroesophageal reflux disease | Cardia tightening procedure |
| 9 | Section 8.6 | (Video 8.9) Per-tunnel super minimally invasive myotomy for esophageal Zenker’s diverticulum | Endoscopic tunnel diverticular septum division |
| 10 | Section 8.6 | (Video 8.10) Per-tunnel super minimally invasive myotomy for lower esophageal diverticulum | Endoscopic tunnel diverticular septum division |
| 11 | Section 8.7 | (Video 8.11) Per-tunnel super minimally myotomy for achalasia-1 | Ac-iia type, peroral endoscopic myotomy |
| 12 | Section 8.7 | (Video 8.12) Per-tunnel super minimally myotomy for achalasia-2 | Post-Heller procedure, peroral endoscopic myotomy |
| 13 | Section 9.1 | (Video 9.1) Peroral super minimally invasive non-full-thickness resection for early cancer at the gastric cardia and fundus | Endoscopic submucosal dissection |
| 14 | Section 9.1 | (Video 9.2) Peroral super minimally invasive non-full-thickness resection for early cancer of the gastric body | Endoscopic submucosal dissection |
| 15 | Section 9.1 | (Video 9.3) Super minimally invasive non-full-thickness resection for early cancer of the gastric antrum | Endoscopic submucosal dissection |
| 16 | Section 9.1 | (Video 9.4) Super minimally invasive non-full-thickness resection for early gastric angular cancer | Endoscopic submucosal dissection |
| 17 | Section 9.1 | (Video 9.5) Super minimally invasive non-full-thickness resection for early cancer of the gastric antrum | Traction-assisted endoscopic submucosal dissection |
| 18 | Section 9.1 | (Video 9.6) Super minimally invasive full-thickness resection for early gastric cancer of the gastric body | Endoscopic full-thickness resection |
| 19 | Section 9.2 | (Video 9.7) Super minimally invasive radiofrequency ablation for gastric low-grade intraepithelial neoplasia | Endoscopic radiofrequency ablation |
| 20 | Section 9.3 | (Video 9.8) Super minimally invasive resection of a stromal tumor of the gastric fundus | Endoscopic submucosal excavation |
| 21 | Section 9.3 | (Video 9.9) Super minimally invasive peroral resection of subepithelial tumor of the cardia | Endoscopic submucosal excavation |
| 22 | Section 9.3 | (Video 9.10) Peroral super minimally invasive full-thickness resection of a subepithelial tumor in the gastric body | Endoscopic full-thickness resection + endoscopic suturing + assisted traction |
| 23 | Section 9.3 | (Video 9.11) Peroral super minimally invasive full-thickness resection of a tumor in the lesser sac of the gastric fundus | Endoscopic full-thickness resection + endoscopic suturing |
| 24 | Section 9.3 | (Video 9.12) Peroral super minimally invasive non-full-thickness resection of multiple gastric neuroendocrine tumors | Band ligation-assisted snare resection |
| 25 | Section 9.4 | (Video 9.13) Super minimally invasive peroral endoscopic myotomy for gastroparesis | Peroral pyloric sphincter myotomy |
| 26 | Section 10.1 | (Video 10.1) Peroral super minimally invasive non-full-thickness resection of duodenal LST | Endoscopic submucosal dissection + assisted traction |
| 27 | Section 10.2 | (Video 10.2) Peroral super minimally invasive resection of a duodenal ampullary adenoma | Snare resection + biliopancreatic duct stenting |
| 28 | Section 10.3 | (Video 10.3) Peroral super minimally invasive resection of a subepithelial tumor in the descending duodenum | Snare resection |
| 29 | Section 10.3 | (Video 10.4) Peroral super minimally invasive resection of a subepithelial tumor in the descending duodenum | Endoscopic submucosal dissection + endoscopic closure |
| 30 | Section 10.4 | (Video 10.5) Peroral super minimally invasive non-full-thickness resection of duodenal adenomas-1 | Snare resection |
| 31 | Section 10.4 | (Video 10.6) Peroral super minimally invasive non-full-thickness resection of duodenal polyps-2 | Nylon-loop-ligation-assisted resection |
| 32 | Section 11.1 | (Video 11.1) Super minimally invasive bile duct stone extraction via duodenal papillary bile duct endoscopy | Direct cholangioscopic super minimally invasive stone removal |
| 33 | Section 11.1 | 11.1 Super minimally invasive bile duct stone extraction via duodenal papillary bile duct endoscopy | Direct cholangioscopic super minimally invasive removal |
| 34 | Section 11.4 | (Video 11.2) Gallbladder stone removal via super minimally invasive stone fragmentation and extraction at the duodenal papilla | Direct cholangioscopic super minimally invasive irrigation and suction |
| 35 | Section 11.5 | (Video 11.3) Peroral super minimally invasive resection of biliary polyps | Direct cholangioscopic super minimally invasive resection |
| 36 | Section 12.1 | (Video 12.1) Super minimally invasive polidocanol ablation for pancreatic cystic tumors via a puncture channel | Super minimally invasive endoscopic lauromacrogol ablation |
| 37 | Section 12.4 | (Video 12.2) Super minimally invasive drainage of pancreatic walled-off necrosis via a gastric puncture pathway | Super minimally invasive drainage + direct visualization debridement |
| 38 | Section 13.1 | (Video 13.1) Peranal super minimally invasive full-thickness resection of laterally developing lesion in the colon | Traction-assisted endoscopic submucosal dissection + clip closure method |
| 39 | Section 13.1 | (Video 13.2) Peranal super minimally invasive full-thickness resection of early rectal cancer | Traction-assisted endoscopic submucosal dissection |
| 40 | Section 13.1 | 13.3 Peranal super minimally invasive full-thickness resection for early cecal cancer | Endoscopic submucosal full-thickness resection + assisted traction |
| 41 | Section 13.1 | (Video 13.4) Peranal super minimally invasive full-thickness resection for early ascending colon cancer | Endoscopic submucosal full-thickness resection + assisted traction |
| 42 | Section 13.1 | (Video 13.5) Rectal cancer post-chemoradiotherapy treated by peranal super minimally invasive full-thickness resection | Endoscopic submucosal full-thickness resection + assisted traction |
| 43 | Section 13.1 | (Video 13.6) Super minimally invasive resection of early rectal cancer through a tunnel channel | Endoscopic submucosal tunnel dissection |
| 44 | Section 13.2 | (Video 13.7) Peranal super minimally invasive non-full-thickness resection of rectal subepithelial tumors | Band-ligation-assisted endoscopic mucosal resection |
| 45 | Section 13.2 | (Video 13.8) Peranal super minimally invasive full-thickness resection of a lesion developing laterally in the colon | Endoscopic submucosal full-thickness resection |
| 46 | Section 13.3 | (Video 13.9) Super minimally invasive non-full-thickness resection for rectal tumor. | Pre-cutting endoscopic mucosal resection |
AI翻译
笔记
高亮
编辑
删除
0 / 500